Remote Monitoring To Achieve Self-Management of Type 2 Diabetes Mellitus: A Prospective Study
Hayat Mushcab1*, George Kernohan1, Suzanne Martin1 and Alan Nevill2
1Institute of Nursing and Health Research, Ulster University, United Kingdom
2Performing Arts and Leisure, University of Wolverhampton, United Kingdom
Submission: December 05, 2016; Published: December 14, 2016
*Corresponding author: Hayat Mushcab, Institute of Nursing and Health Research, Ulster University, United Kingdom, Tel:+447460314350; Email:hayat.mushcab@gmail.com
How to cite this article: Hayat M, George K, Suzanne M, Alan N. Remote Monitoring To Achieve Self-Management of Type 2 Diabetes Mellitus: A Prospective Study. Curr Trends Biomedical Eng & Biosci. 2016; 1(1): 555554. DOI: 10.19080/CTBEB.2016.01.555554
Abstract
Background: The world’s population is aging, and more people are living with chronic conditions including diabetes mellitus. Healthcare providers are moving towards the use of telemonitoring to identify patient self-management approaches and ensure the delivery of health care at their home.
Aim: to explore three telemonitoring technologies that intend to achieve self-management of Type 2 diabetes mellitus thereby improve HbA1c levels and quality of life.
Methods: Interrupted time series design to evaluate the impact of three different telemonitoring solutions provided by one large combined health and social care Trust with technology-naïve people, aiming to manage their Type 2 diabetes mellitus.
Sample: A total of 166 patients met the criteria at the diabetes clinic, with 29 patients consenting to take part in the study.
Results: participants’ baseline measurements were similar. The d-Nav solution exhibited significant improvement in HbA1c over the other telemonitoring solutions. Participants showed acceptability and significant satisfaction of using all three solutions and exhibited improved quality of life.
Conclusion: This exploratory study demonstrates the feasibility of using telemonitoring to self-manage Type 2 diabetes mellitus offering a line of communication between the patient and their clinical care team at a distance.
Keywords: Telemedicine; Telemonitoring; Diabetes mellitus; Interrupted time series; Self-management; T2DM; Type 2; Remote Monitoring; eHealth
Introduction
The prevalence of type-2 diabetes mellitus (T2DM) is increasing rapidly worldwide [1]. In 2000, the prevalence of T2DM was estimated to be 2.8 percent of the world’s population, which meant that 171 million people were living with diabetes mellitus around the world and it was projected that the number would increase to 366 million by 2030 (WHO 2016) [2]. However, in 2014, the World Health Organization estimated the prevalence of T2DM amongst adults to be 9 percent of the world’s population that is equivalent to 450 million people living with diabetes in addition to 179 million people living with undiagnosed diabetes [3].
Life expectancy around the world has increased due to the continued advances in science and technology [4]. Consequently, the world’s population is aging, and more people are living with chronic conditions including diabetes mellitus [4,5]. The mortality rate increases significantly for people with diabetes when associated with other critical health complications, such as heart disease, hypertension and strokes [4,6]. Close monitoring of blood glucose levels keeps the condition under control and reduces the long-term complications and therefore, it decreases morbidity rates [5,7].
Healthcare providers are moving towards the use of telemedicine and more specifically “telemonitoring” to identify patient self-management approaches and ensure the delivery of health care at their home [4,5]. Engaging the individual in managing their own condition can eventually result in fewer consultations and/or hospitalizations [5]. Telemonitoring is defined as utilizing information communication and technology to monitor patients over distance by transmitting biometric data between the patient and healthcare provider for decision-making [6,8,9]. It is a method that has been recently used to complement healthcare provision for many chronic diseases including diabetes [6]. The way telemonitoring operates starts with the individual using a monitoring device at home to monitor their vital signs such as blood glucose, blood pressure and body weight with a preset setting prepared by the clinician. The monitoring device then transmit the stored data to the clinician’s computer in real-time or whenever required [8]. Any abnormal reading would trigger an alarm to alert both the individual and healthcare team and help make a decision instantly [8]. Telemonitoring encompasses the use of different telecommunication technologies such as text, audio, video… etc. to monitor the individual’s status at a distance [6].
Telemonitoring for people living with T2DM is a promising approach to managing their condition with the possibility for improved communication between the individual and their healthcare provider [10]. It has the potential to enhance their compliance with self-care while at home, and therefore improve their quality of life [6]. Telemonitoring can support a diverse range of individuals living with their chronic conditions at home while giving their healthcare providers personal freedom to follow-up with them and give better attention to the severely ill [11].
With the increasing number of people living with T2DM worldwide, diabetes telemonitoring is continuously advancing to help achieve real-time data transmission and effective management of the disease [6]. In general, telemedicine is being used widely as a main tool for delivering healthcare and diabetes education to patients’ homes [12]. Many studies on diabetes telemonitoring concluded that telemonitoring of blood glucose whilst empowering and educating the individual on their condition is an effective approach to managing T2DM. However, few studies have investigated in detail the efficacy of telemonitoring at all levels (i.e. informational, clinical, behavioral, structural and economical levels). So far, the evidence on the impact of telemonitoring and its efficacy over longer periods of time is still contradictive and further investigation is required [6,10].
In this study, the aim is to explore three telemonitoring technologies that intend to achieve self-management and glycemic control for people with T2DM. We conducted an interrupted time series study to investigate telemonitoring solutions provided by one large Health & Social Care Trust in Northern Ireland to help develop a better understanding to the technologies, evaluate the systems’ usability, users acceptance and to analyze the impact of the technology on the individual’s clinical status and quality of life.
Methodology
Interrupted time series design for interventional clinical trial
There is a growing demand to deliver high-quality evidencebased medicine in clinical practice and improve the effectiveness and safety of patient care [13]. The World Health Organization (WHO) defines a clinical trial as: “any research study that prospectively assigns human participants or groups of humans to one or more health-related interventions to evaluate the effects on health outcomes.” Interventions in clinical trials include drugs, procedures, biological produces, devices, behavioral treatments, process and policy changes, etc. (WHO 2015).
Randomized controlled trial or RCT has been described as the cornerstone of evidence-based medicine [13], the gold standard, the ideal and the criterion standard methodology for assessing and evaluating the effectiveness of healthcare interventions [14-16]. However, generalizing results to “real world” settings can sustain systematic errors leading to biased estimates [15]. Circumstances might not permit all the conditions of a true experiment, randomization is not logistically feasible or when there are practical and/or ethical barriers; hence, the increasing interest in quasi-experimental design methodology [16-20].
Researchers aim to move the discipline of quality improvement and implementation forward with quasiexperimental designs, informed by extensive qualitative work about decision-making where researchers are able to demonstrate causality between an intervention and an outcome using observational methods [15,21,22]. In health informatics this design is mostly used to evaluate the efficacy of a specific intervention in a healthcare system [22]. Health informatics is defined as “The interdisciplinary study of the design, development, adoption and application of IT-based innovations in healthcare services delivery, management and planning” [23].
The most commonly used designs are: (1) uncontrolled pre-posttest design, (2) interrupted time series design, and (3) controlled pre-posttest design [16,24]. In an uncontrolled preposttest study, the researcher evaluates the performance of a provider before and after the introduction of an intervention and any changes are then inferred to have occurred due to the intervention [19,24]. Even though uncontrolled pre-posttest studies are superior to observational studies and are relatively simple to conduct, they are fundamentally weak evaluative designs, as they would be affected by secular trends or sudden changes and make it difficult to attribute observed changes to the intervention which could lead to overestimating the effectiveness of that particular intervention [17].
In a controlled pre-posttest study observations of two groups of participants who have received an intervention and a control group who have not, measuring the outcome variable both before and after the introduction of the intervention [25,26]. Data are collected in both groups simultaneously using the same methods before and after the intervention (Grimshaw, Campbell, Eccles, & Steen, 2000). If the researcher has control over the introduction of the intervention to the participants, then this design can be considered an experimental design [26]. However, even in wellmatched control and study groups, performance at baseline is often different causing a baseline imbalance between the study groups.
The interrupted time series (ITS) is a design that uses observations at several time points in time before and after the introduction of an intervention the “interruption” and a trend analysis can be conducted for the targeted outcomes. This design aims to detect whether the intervention has had a significant influence on the underlying trend over time in a defined population of individuals [21]. ITS design is arguably considered as the strongest quasi-experimental research design for evaluation of longitudinal effects of interventions or when the researcher has no control over the implementation of an intervention [27,28]. A simple interrupted time series design may be a study of repeated observations without an intervention or collecting data at several times before and after introducing the intervention to the participant employing a trend analysis [28,29]. Campbell and Stanley in 1963 described a simple ITS design as follows:
O1 O2 O3 O4 O5T O6 O7…
All the Os are observations taken before and after the T, which is the “interruption” where the intervention was introduced [30,31]. Segmented regression or regression discontinuity are variations of the ITS design and they all often yield excellent effect estimate [28].
Healthcare providers and clinical researchers sometimes want to monitor hospitalized patients or patients with chronic illnesses on a regular basis, thus, single subject designs can be useful where the individual variability of a human subject can be very overwhelming(Matowe, Leister, Crivera, & Korth- Bradly, 2003). More intensive examination across occasions over time of the same person before and after introducing the intervention may be needed design can be used, therefore, ITS design can be used to strengthen before-and-after design [29]. In an ITS design, each participant serves as their own control, and several dependent variables are being measured where some are expected to be influenced by the intervention and others not expected to change.
Besides evaluating trends before and after the introduction of an intervention, ITS can also predict patterns of events that occur over time [30]. Subsequently, valuable and informative data can be collected and analyzed by implementing protocols that measure essential variables at equally spaced intervals. Collecting several sets of data over time in an ITS design minimizes the weaknesses of single measurements in the preposttest design, such as regression to the mean and history as a threat to internal validity.
ITS design is mainly beneficial when the introduction of the intervention happens gradually or at several time points because it allows for statistical investigation of potential biases in the estimate of the intervention’s effect [15]. Potential bias includes autocorrelation, external time varying effects such as secular trend, cyclical or season effects, the duration of the intervention and the change of characteristics of study population over time. Using appropriate statistical methods and tools can help the researcher investigate such biases. Statistical methods to measure outcome include time series regression techniques, autoregressive integrated moving average models (ARIMA), sequential or piecewise regression methods that can identify both sudden and more gradual changes over time due to the intervention [32]. Using the segmented regression allows the researcher to measure the change in level and change in slope associated with the intervention [21].
ITS design evaluates changes in rates of an outcome and the analysis of data can be conducted with respect to population rates rather than at the individual level only [21]. These outcomes can be expressed as averages, proportions or rates and can be clearly and easily interpreted in graphic results, which is considered a strength that ITS design has over alternative statistical approaches. With ITS design, researchers can construct time series of the rates of other potentially negative population level events with the analysis of unintended consequences of the intervention. Researchers can also evaluate the differential impact of an intervention on subpopulations of individuals (e.g. by age, gender, race) by conducting stratified analyses.
However, with all the advantages ITS design has there are few statistical limitations. First of all, in order to have sufficient power to estimate the regression coefficient, ITS design requires a minimum of eight pre and post intervention observations. The proper specification of a time series typically requires a long time series - 100 observations, as a rule of thumb [33,34]. To estimate the impact of the “interruptions” independently, a sufficient number of time periods between them is needed and this is often infeasible and/ or undesirable. However, short time series needs more fundamental knowledge and stronger assumptions about the functional form to justify the results from a specific ITS design [34].
Moreover, there are some threats to validity in ITS design analysis. The most serious threat is the effect of history [28]. ITS analysis is considered valid only to the extent that the intervention was the only change at the “interruption” point in time and an observed change in the level, slope or both must be uniquely because of the intervention [34]. Other changes coexisting with the intervention that could influence the outcome variable are commonly referred to as “competing interventions”. These competing interventions would only occur and be considered immediately after the announcement, implementation, or becoming aware of the intervention [34]. They require thoughtful consideration and should be included in a discussion when reporting the results of an ITS analysis.
A second threat to validity in ITS design is the changes in the measurement framework and/ or the ability to measure the rate of the outcome of interest. Measurement instrumentation such as the reporting, measuring, or recording of the outcome of interest may change over time, and it would influence the outcome if it happened simultaneously with the intervention [34].
Two assumptions must be met to achieve unbiased estimates in an ITS design; 1) time is the sole variable determining implementation of intervention and 2) the functional form of the outcome over time must be correctly modeled. This leads to a third threat to the validity of ITS design which is selection bias, specifically if the composition of the study group changes at the same time as the intervention. Since groups are not randomly assigned, researchers must carefully examine the time series data to verify that the composition of the population under study did not change before and/ or after the intervention.
A forth threat to ITS design is when the intervention delivers delayed rather than immediate effects. This may be because the intervention was not immediately and completely implemented, slowly diffused through the study group, or both. Delayed effects occur frequently and very plausible are difficult to interpret and analyze if not justified theoretically to explain the observed delay [20]. The possibility of alternative interpretations to an outcome increases as the time period between the intervention and the first possible effects increases. Finally, if the time series are short and show high amount of unexplained error then weak effects will be difficult to prove and the study will face power issues [34].
ITS design is a simple yet powerful approach to intervention implementation. It is the strongest quasi-experimental design with a high value in quality improvement and intervention evaluation. Although it has its limitations, with the appropriate statistical tests ITS may be more useful than an RCT.
Study design
For this study, the UK Medical Research Council’s (MRC) framework for developing and evaluating complex interventions was adopted to provide guidance in structuring the study and recognizing the appropriate methods to meet our aim. There are four main stages (Figure 1) to the process from development through to implementation of a complex intervention, and it consists of a wide range of different forms and key functions at each stage [35].
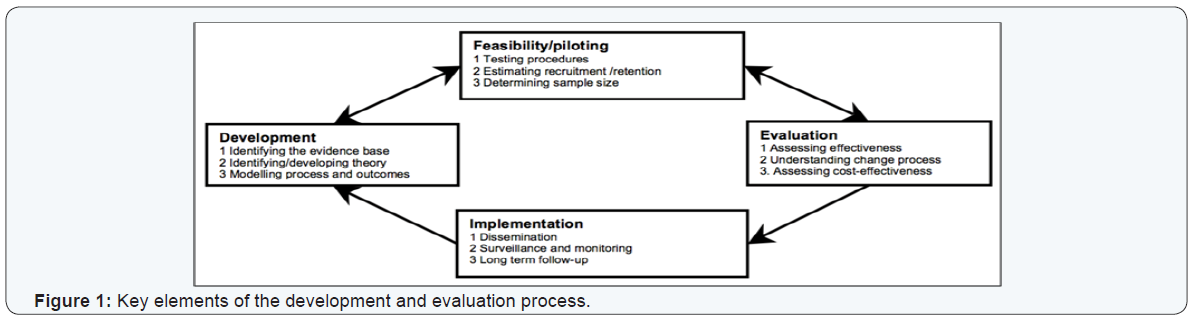
Developing a complex intervention
Identifying the evidence base: A comprehensive background of the evidence and a systematic review [8] were conducted to identify the relevant and existing evidence base on telemonitoring of T2DM as reported in chapters two and three.
Identifying/developing appropriate theory: The literature review has shown that more studies are needed to enrich the evidence-base on using telemonitoring to self-manage T2DM. For ethical purposes, the interrupted time series (ITS) design was proposed to evaluate the current healthcare plan provided at the diabetes clinic at one site (Ulster Hospital). Healthcare providers are often more comfortable with this design because it does not interfere with the care they have planned for the individual by avoiding the uncertainty associated with randomization. Based on the literature review, ITS design is useful within the context of monitoring patients on a routine basis [35,36] and it can identify key outcomes associated with an on-going treatment process [37].
This study targeted people that had no experience with telemonitoring and who are naïve to such technology. Data was collected at three different time points: 1) at baseline before introducing any of the technologies to the patient, 2) after twelve weeks of using the telemonitoring solution to self-manage their T2DM, and 3) after another twelve weeks of the previous follow-up. The existing clinical information verified the patient’s health status at baseline and a blood test was taken at weeks 12 and 24. A set of questionnaires was used to collect data about patients’ quality of life and satisfaction with the treatment at all three time points. Another questionnaire was used to evaluate the participants’ satisfaction with the technology they used at weeks 12 and 24. With repeatedly measuring relevant clinical and quality of life variables on an equally spaced time points, the pattern of change in each individual’s clinical outcome and the overall patient’s satisfaction were possible to investigate.
Modelling process and outcomes: Modelling a complex intervention prior to a full-scale study can provide important information about the design of both the intervention and the evaluation process. This study has the potential to help endocrinologists, general practitioners, family practice physicians, diabetes clinics and people living with diabetes to make informative decisions in the treatment plan to improve their health status and improve their overall quality of life.
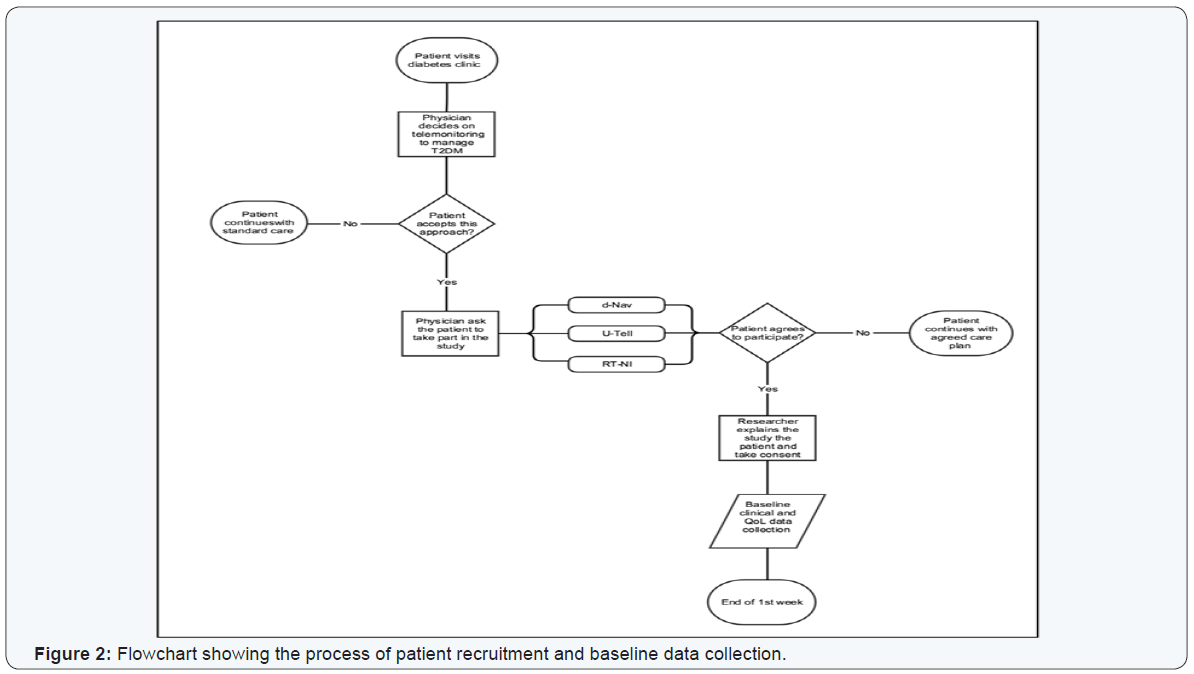
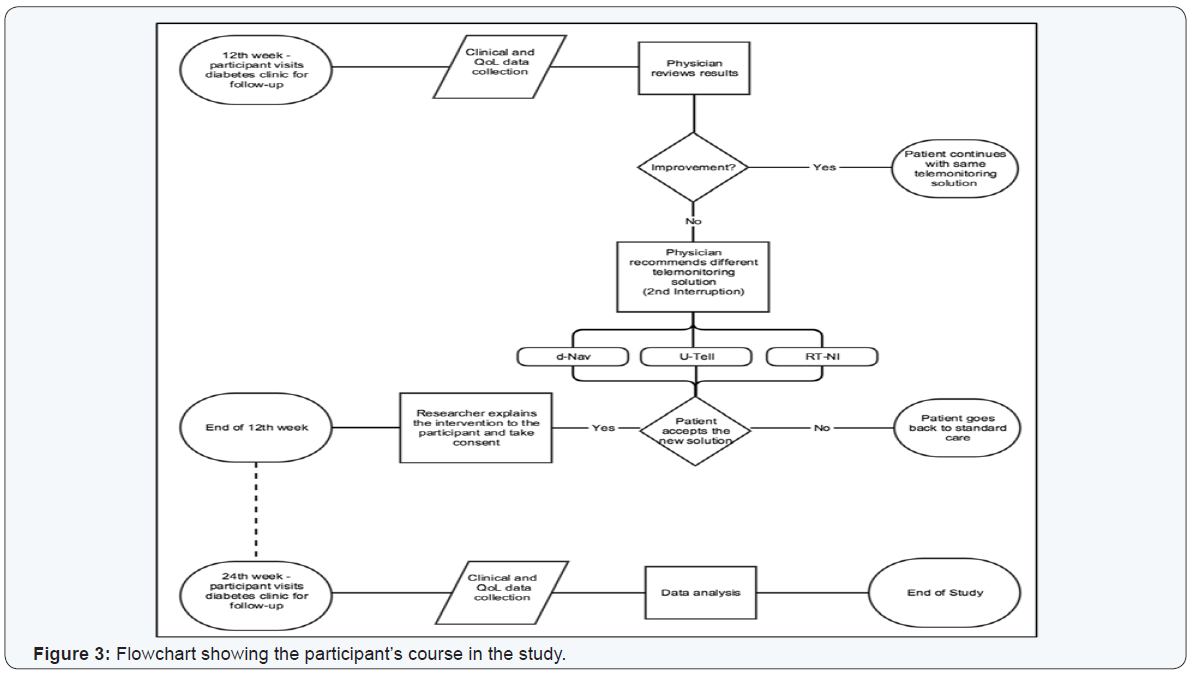
The physician at the clinic introduced the telemonitoring solution to the patient as part of the individualized care plan. After both the physician and the patient have agreed on the plan, the physician then consented them to take part in the study (Figure 2). It was anticipated that after introducing the telemonitoring technology to the participant, the clinical outcomes and quality of life measures were to improve within the first twelve weeks. At this time point, the physician would assess the participant’s health status, and either proceeds with the same plan for another 12 weeks or alter the original care plan to a different telemonitoring solution or take them off telemonitoring and back to standard care (Figure 3).
Using remote monitoring for people with diabetes mellitus was an established technology as healthcare service intervention at the hospital. The clinical teams were already using it to help their patients self-manage their diabetes. However, there is no evidence that these telemonitoring solutions are achieving that goal and that improvements are actually met. Therefore, this study aims to explore clinical outcomes, patients’ satisfaction with their treatment and patients’ quality of life levels after introducing the technology to them at this one site and hopefully expand this evaluation in a long-term and multiple sites implementation plan.
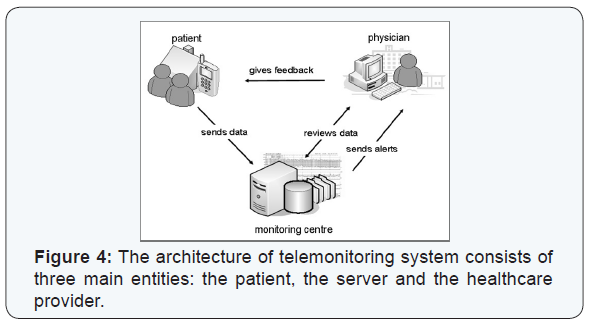
It is vital to understand the mechanism of each telemonitoring solution provided, know the difference between each one and to understand the reason behind a physician’s choice of one solution to a certain patient rather than the other. The three telemonitoring solutions used are: 1) Hygieia - Diabetes Insulin Guidance System (d-Nav), 2) U-Tell: Diabetes and 3) Remote Telemonitoring Northern Ireland (RT-NI). The general mechanism behind telemonitoring starts with a wireless device used by the participant to transmit their vital signs including blood pressure, blood glucose and body weight to a secured server. The server sends alerts to the healthcare provider to review the data remotely and send feedback to the participant regarding titration of insulin dosage and any other recommendations needed to manage their condition (Figure 4).
Each telemonitoring solution used in this study is described as follows:
I.Hygieia - Diabetes Insulin Guidance System (d-Nav):
D-Nav is a technology that enables automatic regular insulin dosages adjustment for people with diabetes to enhance and simplify the insulin therapy [38]. The d-Nav technology was developed by Hygieia; a diabetes healthcare company in the United States. It is a navigation tool for insulin therapy, thereby, the “d” stands for diabetes and the “Nav” stands for navigation. With smart cloud-based technology and the healthcare team, d-Nav is inclined to provide an efficient service and improved health to people with diabetes.
d-Nav provides personalized insulin adjustments as needed, using each individual’s insulin therapy regimen and glucose pattern [38,39]. It is a small device, very similar to a glucometer that uses a special d-Nav strip for blood glucose (BG) measurements. However, after displaying the BG reading for few seconds, the device then calculates the insulin intake based on the BG levels and the pre-programmed settings by the physician and then displays the dosage to the individual. d-Nav aims to maximize insulin effectiveness for both the individual and the physician prescribing it.
After the first meeting with the participants recruited to use d-Nav to self-manage their T2DM received a full educational session and a leaflet to take home on how to operate the device and to measure their BG levels. For the following twelve weeks, the participants were completely independent on self-managing their condition. On their follow-up appointment, the healthcare team would review the data stored in the device for the past period and assess the individual’s health status. A decision is made after that whether to proceed using the same technology or to change it to one of the other available options for another twelve weeks.
II.U-Tell: Diabetes:
U-Tell solution was originally developed to remotely monitor patients on Warfarin (an anticoagulant medicine that stops blood from clotting). It was a collaborative development between Ulster University and the South Eastern Health and Social Care Trust and it became the first telemonitoring system that allows people on Warfarin to upload their readings via voice recognition technology, a telephone keypad or a web-based interface (Executive, 2014).
The technology included patients that needed glycaemic control, weight management or blood pressure control so their health could be managed more effectively. After uploading their readings onto the U-Tell system, it alerts the healthcare provider to the patients who require urgent attention every time they log into the system and provide them tailored feedback and instructions on what action they need to take either via telephone or message through the internet. The patient then can adjust the insulin dosages according to these instructions.
Each participant had a secured login portal into U-Tell: Diabetes. In addition to uploading the readings into the system, it allows them to view their results, providing them with charts and highlighted results for any abnormal readings. Study participants enrolled on using U-Tell received a one-on-one educational session with the diabetes nurse on how to use the technology as well as a Patient Information Leaflet.
III.Remote Telemonitoring Northern Ireland (RT-NI)
RT-NI is a large-scale, clinically led managed monitoring service that was launched in Northern Ireland in October 2011 to improve health status of people with chronic diseases including diabetes. The Health Service in Northern Ireland introduced the innovative telemonitoring service aiming to enable patients to control their diabetes and reduce the risk of serious complications through better engagement with the healthcare team [40].
RT-NI is a system designed by a group of companies with complimentary expertise known as TF3¹ [41]. Each participant is provided with a computerised hub - a small unit installed in their home along with linked ancillary equipment - a wireless scale, a blood pressure monitor and a glucometer for self-monitoring blood glucose. A technician would visit the participant at their home to install the device and explain to them how to operate it. RT-NI enables the measurement of multiple variables such as weight, blood pressure, blood glucose, temperature and oxygen levels in the body using the provided equipment. The unit is programed to ask a set of “yes or no” questions determined by the healthcare team. These measurements are checked regularly and if the readings are abnormal, the diabetes nurse would receive an alert on the system and contact the participant for more information. The nurse would assess the situation and provide them with the suitable instructions. If necessary, the physician would be consulted on the matter.
Assessing Feasibility and Piloting Methods
This stage of the study includes testing the interventions for their acceptability, estimating the likely rates of recruitment and retention of subjects and the calculation of ideal sample size.
Inclusion criteria
- Participant diagnosed with T2DM and newly referred to the diabetes clinic for an endocrinologist consultation on managing the condition.
- Participants’ age group (≥ 18 years) as T2DM is commonly found in adults.
- Participants on Insulin-Treatment for over four weeks on stable regimen whether it was a simple regimen involving a single insulin injection per day or more complex regimen with insulin treatment combined with oral agents. The decision as to which treatment regimen to be established is made by the physician tailored to the individual’s health status and needs.
Exclusion criteria
- Participants with complex or multiple morbidity conditions were excluded, to avoid confounding effects,
- Participants with cognitive impairment were excluded. To avoid collecting unreliable and invalid data, these individuals would not be able to self-manage or operate the devices correctly.
- Pregnant women with gestational diabetes mellitus were excluded; to avoid confounding effects.
- Participants were involved in other research or were recently involved in any research prior to recruitment were excluded; to avoid undue burden.
Participant recruitment process
The literature review indicated that a total of 60-80 participants wouldresult in a study within 80-90 percent confidence level. The target was a sample size of (n= 80) to ensure a powerful study with a 25 percent possibility of dropouts or lack of consent.A consultation from an expert statistician was pursued and a sample size calculator was used [42].
An average of 45 people with diabetes visit the diabetes clinic held at the site where this study was conducted. In 13 months of recruitment, the researcher attended 39 clinics; a total of 166 patients met the criteria in an average of 4.3 patients per clinic. The average age of the screened patients was 63 (±10.6) years and had T2DM for nearly 13.5 years. 17 percent of the screened patients consented to take part in the study. 11 percent had their physician decide that they are not ready to use the technology to manage their condition for several reasons, such as patient did not show intentions for commitment, patient was too unstable or they suffer from other health complications. Equally, 11 percent of the patients were not interested in using technology: 7 percent had good management of their T2DM and were content with the standard care they are receiving. 4 percent did not need any telemonitoring solutions because they recently stopped taking insulin treatments. Another 3 percent needed a carer to help them in their daily life. 18 percent of the patients gave no answer or final decision whether to proceed with the study or to decline. However, 29 percent of the patients used -or currently using - telemonitoring solutions to manage their T2DM, therefore, they were excluded from the study.
Evaluating a Complex Intervention
Assessing effectiveness
The interventions in question were already in use within the diabetes clinic and randomization was an impractical study design to pursue. Therefore, the ITS design was an appropriate quasi-experimental alternative.
Understanding processes
The provision of insight and reasonable explanation an intervention’s outcome, whether it failed unexpectedly or worked successfully was essential. A rigorous knowledge of outcomes is crucial to interpret the results of any study [35]. The results of a given trial can be used to assess the quality of implementation, clarify casual mechanisms and identify related factors associated with any change might occur in the outcome [2].
Data collection, extraction, and processing
Data was collected at three time points: Week 0 (Baseline), Week 12 (2nd interruption), and week 24 (end of study). Two types of data were collected at each point: clinical data - extracted from the hospital integrated system and participant’s satisfaction and quality of life data -gathered using standardized questionnaires. The goal of using the telemonitoring devices was to improve the glycated haemoglobin (HbA1c) while maintaining a healthy quality life. So, the following data was collected to understand the reality of using these telemonitoring solutions:
- Clinical Data - extracted at weeks 0, 12, and 24 - includes: HbA1c, blood pressure, blood glucose, bodyweight, BMI and cholesterol levels. Satisfaction Quality of Life data:
- Audit of Diabetes Dependent Quality of Life (AddQoL) questionnaire: collected at all time points - weeks 0, 12, and 24. This is a standard questionnaire that is routinely used by the diabetes clinical team to evaluate the basic daily tasks and quality of life in general for people with T2DM [43].
- Diabetes Treatment Satisfaction Questionnaire (DTSQs)i: collected at all time points - weeks 0, 12, and 24. This questionnaire gathers data on individual’s satisfaction and experience with their diabetes mellitus treatment including: insulin treatment, oral agent, diet, standard care, and /or telemonitoring.
- Diabetes Treatment Satisfaction Questionnaire-change (DTSQc)i : collected at the end of the study only to evaluate the individual’s satisfaction after experiencing the change in their treatment plan - after taking part in the study.
- Quebec User Evaluation of Satisfaction with Assistive Technology (QUEST 2.0): collected at weeks 12 and 24 - after the 1st and 2nd interruptions and the introduction of the telemonitoring device to the participant. Use to evaluate the individual’s satisfaction with the technology and their experience with the services provided.
Hypothesis and Data analysis: The question we aimed to answer was: How do three telemonitoring solutions differ in improving HbA1c levels and the quality of life of people with T2DM?
Data analysis and the selection of the right statistical test largely depend on the nature of both independent and dependent variables. Variables can be divided to two classes: Categorical such as gender and ethnicity and Continuous such as weight and blood glucose levels [44]. In this study, both classes of variables were collected and several statistical tests were performed to ensure accurate results.
All the quality of life and satisfaction variables were analysed using the following two tests:
(sometimes called the one way ANOVA or ranks) [45]: is a nonparametric test used to compare ordinal or non-normal variables for more than two groups [46]. This test is used when the assumptions of Analysis of Variance (ANOVA) are not met. Both these tests are used to assess the significant differences on continuous dependent variables by grouping independent variables. In the ANOVA the distribution of each group is assumed to be normal and the variance is approximately equal for each group [47]. However, the Kruskal-Wallis is considered the alternative test when these assumptions are not available [46].
Chi-Square Test: is a test used compare two categorical variables from a single population according to a specific hypothesis [49,50]. For this study, the grouping variable was the three different points of time (Week) where the data collection took place.
Paired Sample T-Test: used to compare the means of two correlated samples. This test is mostly used in the “pre-posttest” studies. Using the paired sample t-test allows the researcher to investigate whether the telemonitoring technology improved the clinical and quality of life outcomes or not [50].
For the clinical data analysis the following statistical tests were used:
- Descriptive Analysis: used to describe the basic features of the data gathered in the study summarising the sample and in a meaningful way [51]. Descriptive statistics along with graphics analysis allow simpler interpretation of the data and form the basis of nearly every quantitative analysis.
- Mixed Model Analysis: this statistical method is used in repeated measures designs where the outcome is continuously measured at fixed time points [52]. Mixed Model analysis was used in this study to measure the significant difference within the subjects themselves and between the subjects as a group. It differs from Repeated Measures ANOVA is that the latter had limitations with missing observations (data) and that would cause dismissing the whole participant’s data.
Results
Of 29 initially recruited participants, 25 opted to complete the study. Three of the remaining four participants consented but did not use the technology to manage their T2DM and one participant withdrew from the study before starting. Recruitment started in May 2014 and it was planned to finish in 4 months but the process was extended until August 2015 because of the poor number of participants. Results were processed using statistical analysis software and double-checked using different statistical software.
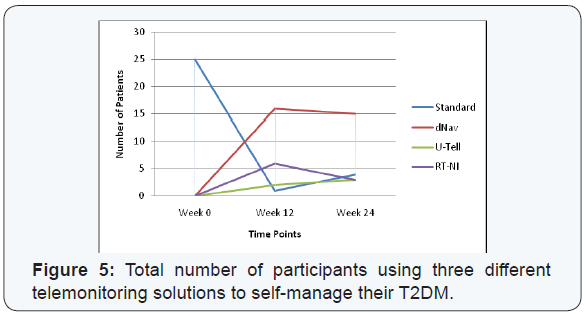
At the beginning of the study, all participants were on standard care using a glucometer and following the instructions given to them every three-six months. The majority of the participants (n=16) were assigned to use d-Nav at their first interruption to monitor and self-manage their T2DM, RT-NI came in second with (n=6) and finally U-Tell with only two users (n=2). At the second time point, for the second interruption, the majority were still using d-Nav (n=15) while RT-NI and U-Tell had equal number of users of (n=3). However, out of the 25 participants two withdrew half way through the study and managed with standard care, one did not need telemonitoring to manage their condition and went back to standard care as well and one participant passed away within few weeks from recruitment (Figure 5).
All the participants felt that diabetes mellitus is a burden on their family and social life and it has slightly improved after using telemonitoring to manage their T2DM. Their worries and hopes towards the future improved significantly at weeks 12 and 24 with (p= 0.044). Most of the participants were still working and felt that their work life and their financial status would improve slightly without diabetes mellitus.
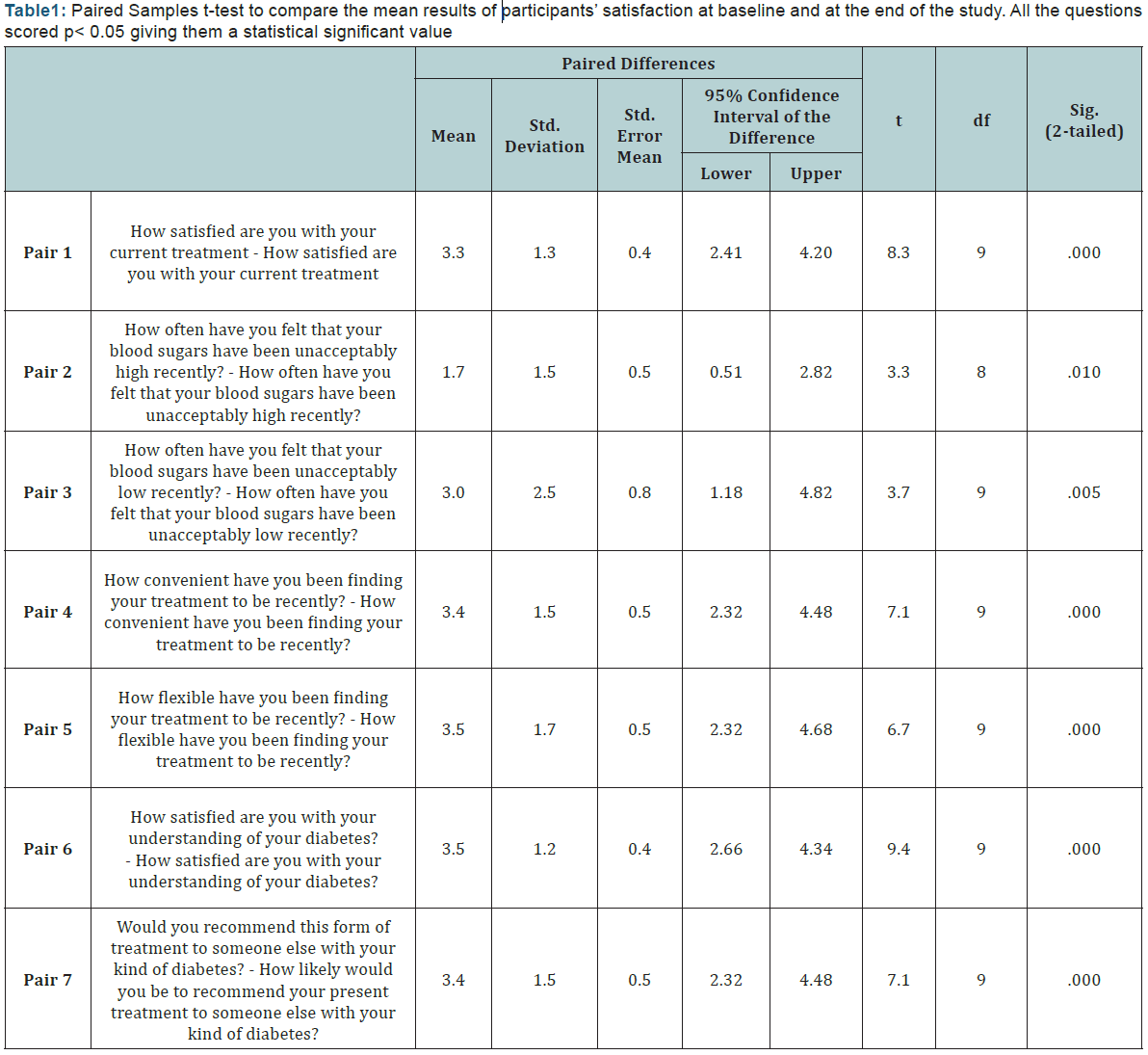
Participants felt that their high blood glucose levels have significantly improved after using telemonitoring at weeks 12 and 24 with (p=0.006). In addition, participants satisfaction with the flexibility of the new treatment plan and their overall satisfaction with their diabetes care have significantly improved after using telemonitoring with (p= 0.41) and (p=0.42), respectively. A paired sample t-test was performed to compare the means of participant’s satisfaction with the treatment plan at baseline and at the end of the study (Table 1). All the questions included had statistically significant improvement with (p<0.05).
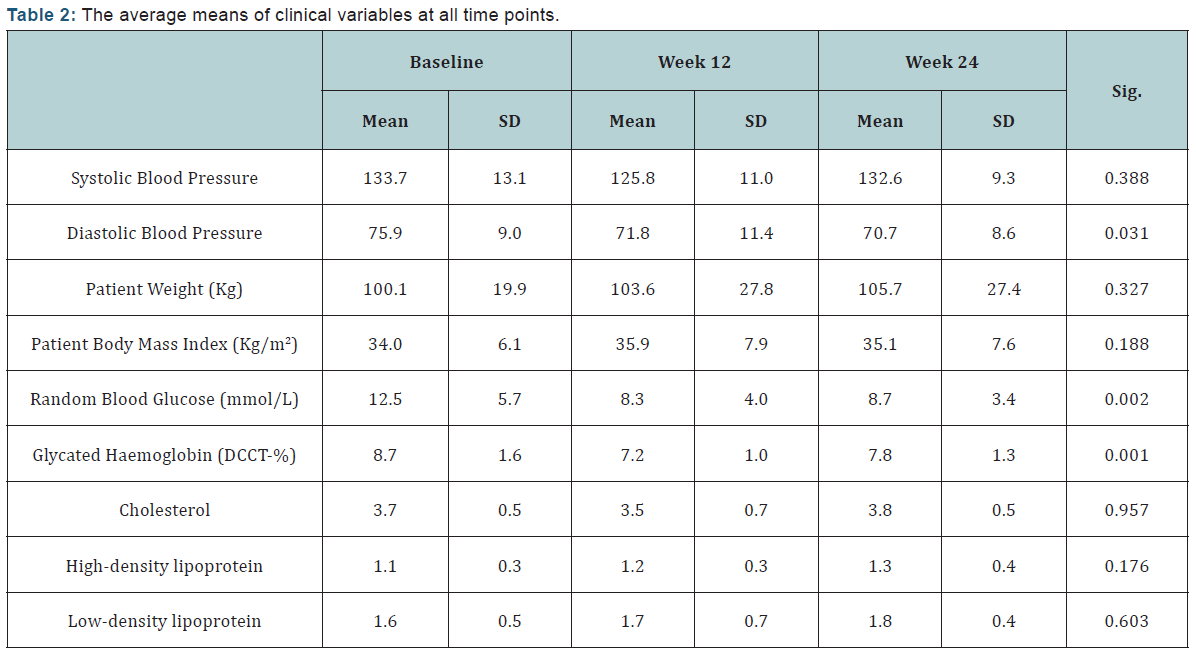
Using telemonitoring technology has helped improve the participants’ diastolic blood pressure, blood glucose and most importantly HbA1c levels significantly with (p<0.05) (Table 2). However, participants had a slight increase in their body weight and cholesterol levels including both high-density lipoprotein (HDL) and low-density lipoprotein (LDL).
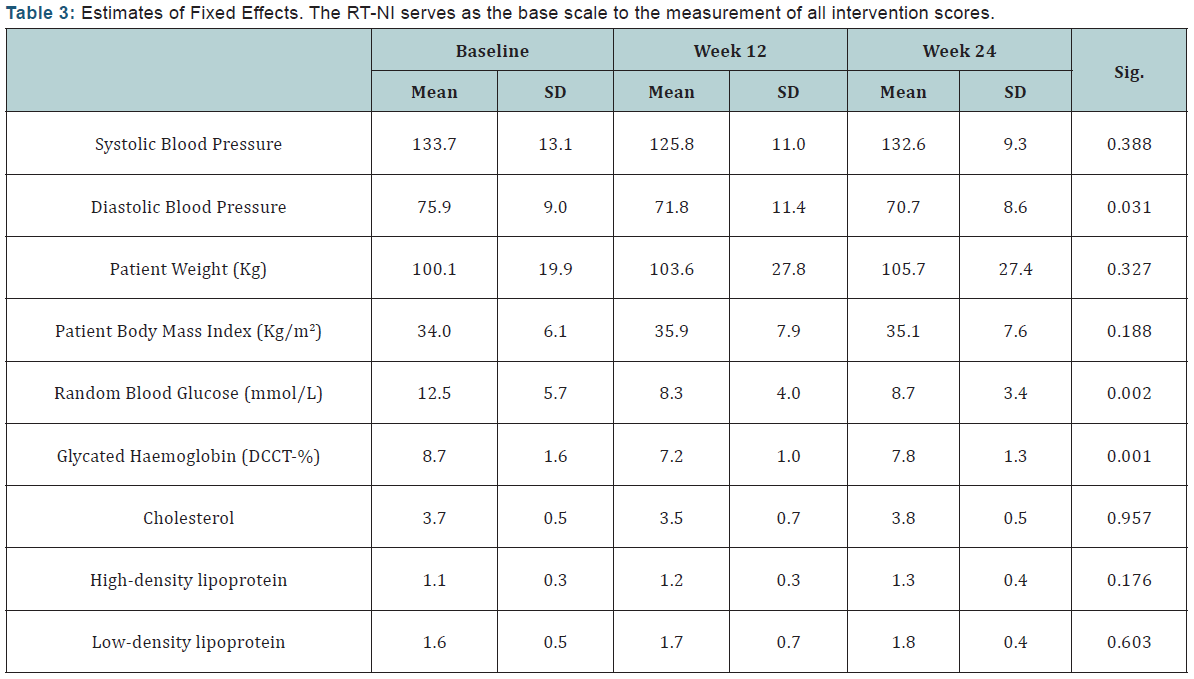
The parameter estimates given in the fixed effects table (Table 3) are estimate of mean parameters that were set to zero. RT-NI was the “baseline” as it was set as the last category. Comparing the intercept mean for all telemonitoring solutions including the standard care provided before taking part in the study, the d-Nav users have shown better results than the standard care as well as the other two solutions.
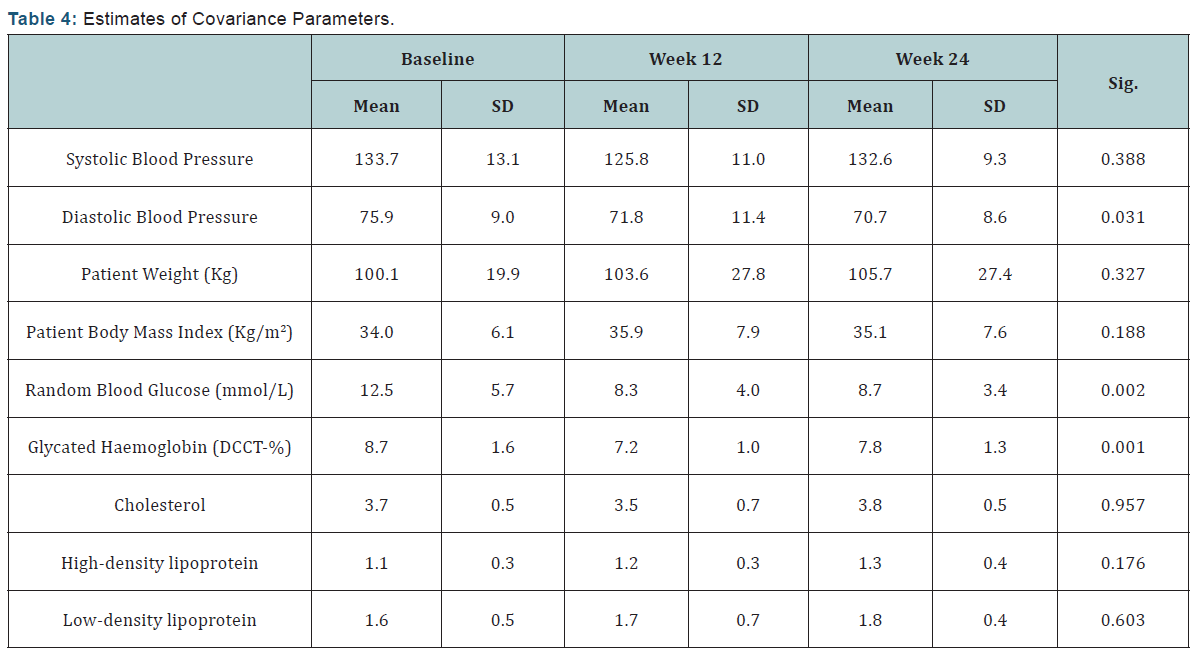
The estimate of residual (personal) variance and the intercept variance between subjects is presented in the Estimates of Covariance Parameters table (Table 4). This test shows the intercept variance “between subjects” is estimated as 25.54 for systolic blood pressure, 54.51 for diastolic blood pressure, 439.25 for body weight, 43.34 for body mass index, 10.11 for random blood glucose, and an estimate of 1.03 for HbA1c levels. This indicates that each individual subject will have “personal” intercepts of ±5.05, ±7.38, ±20.9, ±6.58, ±3.17 and ±1.01, respectively. Moreover, the variation “within subjects” is equal to 55.36, 45.72, 266.46, 26.28, 10.18 and 0.88 respectively. The following formula is used to calculate the significance value:
Coefficient Estimate > 2 x Standard Error
Therefore, a significant difference was shown in all the dependent variables except for systolic blood pressure and random blood glucose.
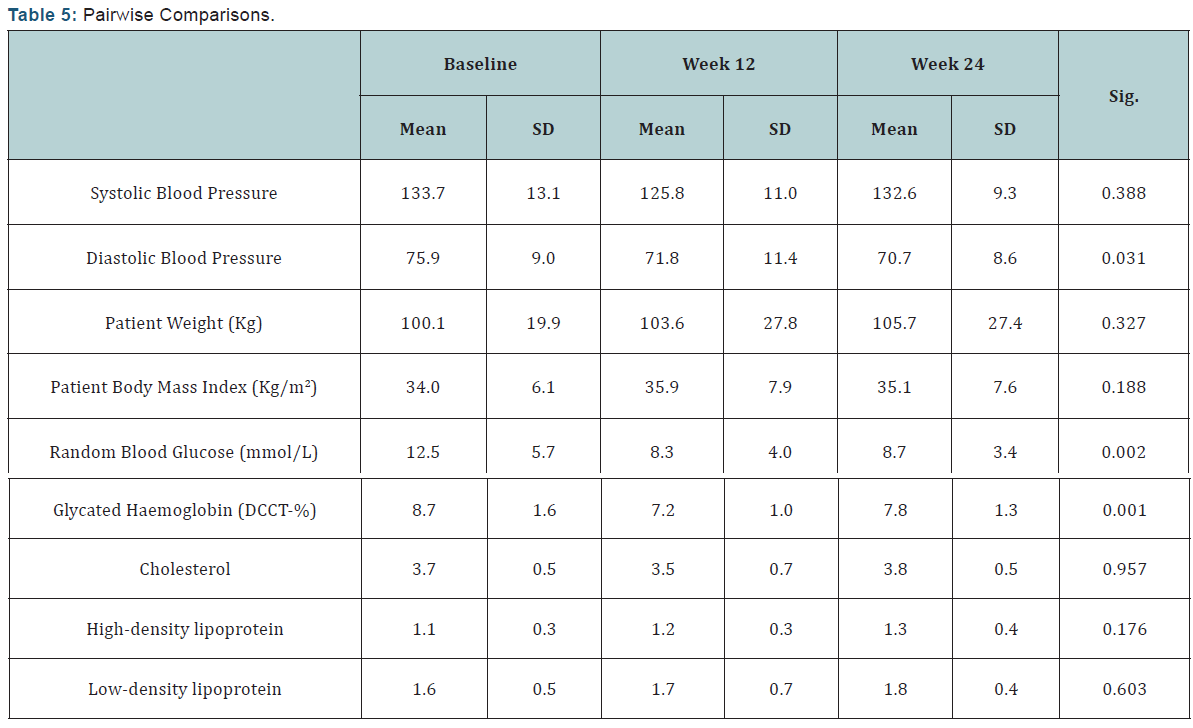
The Pairwise Comparison test compares the means of all three telemonitoring solutions to the previously administered standard care, the d-Nav showed the most significant improvement in most of the clinical variables with (p<0.05) for the diastolic blood pressure, random blood glucose, HDL and HbA1c levels. RT-NI users also showed significant improvement in their weight (p=0.07), therefore their BMI also improved with (p=0.04), as well as their HDL levels (p=0.03); while U-Tell: Diabetes users did not show any significant improvement at all (Table 5).
Discussion
A most recent qualitative study published in 2016 by Hanley et al. praised the convenience of home monitoring of blood glucose and blood pressure to the patient as well as the healthcare provider. The data transmitted by the telemonitoring technology supported self-management and medical treatment decisions [53]. Similarly, the findings of this study show that individuals are able to self-manage their T2DM knowing that their healthcare provider is remotely monitoring their condition. Transmitting BG data with other relative data such as BP and weight using telemonitoring solutions gave the individuals the confidence and motivation they needed to self-manage their diabetes while supporting the healthcare provider make clinical decisions and continuous follow-up.
The three different telemonitoring devices had different levels of clinician’s involvement in managing T2DM as an attempt to shift the control over to the individual to self-manage their conditions. The most popular device amongst them was the d-Nav, giving the individual more of that control over their condition and giving the clinical team assurance that the device would provide their patients the correct dosage of insulin needed at the time. D-Nav was the only telemonitoring solution that exhibited significant improvement in HbA1c and RBG levels. However, RT-NI showed significant improvement in the RBG levels as well. On the other hand, individuals using RT-NI showed significant improvement in their body weight and therefore BMI levels. That was an advantage of providing them with a wireless scale as well to continuously monitor their weight. A similar study conducted by [54] to assess the effect of home based telemonitoring interventions on the use of secondary healthcare and mortality for 3230 people with different chronic diseases including diabetes mellitus. The study resulted in lower hospitalization and mortality rates in the intervention group than in the control. Concluding that telemonitoring helped people with chronic diseases manage their own condition better and therefore reduce the incidence of acute deterioration in their condition that needs emergency admissions.
Another recent study was conducted in New Zealand published in 2015 assessing the effect of home monitoring commercially-available technology showed no significant improvement in the quality of life, self-efficacy and diseasespecific measures. However, the telemonitoring solution led to patients and their families taking more responsibility and being more active in self-management [55]. This study aimed to measure the quality of life and participants’ self-management and satisfaction with telemonitoring. All participants were overwhelmed with certain concerns and worries that affected their management of the condition. Some of the concerns they exhibited were following a healthy diet, exercising more often and determining the insulin dosage they need. Each participant took part in the study was looking for a solution that helps them manage their condition and overcome their worries. Participants’ satisfaction, quality of life, and acceptance to all three solutions was significant by the end of the study making telemonitoring conveniently feasible. The telemonitoring solutions provided telemedicine procedure asynchronously that allowed the healthcare provider to make a decision from a distance. During the study, none of the provided devices have faced any error or technical problems and reliable and accurate measures were consistently transmitted to and from the server. The increasing advancement in technology and telecommunication offers reliable data transmission, ensuring timely availability of quality data as those collected in a face-to-face examination. Giving the healthcare provider better opportunities for clinical decisionmaking wherever they were and whenever they are needed is an indication of a successful process and a goal achieved.
This study was limited to one site and to insulin-treated T2DM only and the effects may be smaller or more variable and response rates lower when the intervention is implemented across a wider range of settings. As part as of MRC’s framework of implementing a complex intervention is assessing costeffectiveness of the telemonitoring solutions could enhance this study and make it more useful for-decision-makers. Further work on economic evaluation is to estimate the cost rather than testing a hypothesis and handle the uncertainty of cost and address the effects of differences appropriately during development work. Stone et al. also suggested that consideration should be given to the intensity and duration of telemonitoring interventions and that improvements in HbA1c levels can be achieved using telemonitoring can be sustained for at least additional six months after the clinical trial [56].
Implementation
To understand barriers to participations and for better estimation of response rates, a mixture of qualitative and quantitative methods was likely to be more appropriate. And depending on the results, a series of similar studies in different sites with different types of diabetes mellitus might be required to progressively refine the design of this exploratory study before embarking on a larger full-scale evaluation [57].
Getting evidence into practice: A successful implementation of an intervention depends on adjusting or changing certain behaviour of a wide range of people, this includes patients, physicians and decision-makers [58]. Therefore, it is essential to have a scientific understanding of these behaviours, the factors maintaining them, the barriers to changing them, and the skills and experience needed to develop strategies that facilitate the process of changing these behaviours.
Translating the results of this study and making them available for these key people will help them make the change in the routine practice or policy change. Thus, the information this study provides are made available and accessible to the Health & Social Care South Eastern Trust and submitted for publication in an international journal for wider spread of knowledge [59].
Surveillance, monitoring and long-term outcomes: It is unlikely for a pilot study like this to provide a comprehensive and extensive account of the effectiveness of the three interventions. Unexpected consequences may begin to occur and effects are likely to be smaller and more variable when implementing the intervention to a bigger population and more sites [35]. To determine whether the interventions’ short-term changes continue and remain, a long-term follow-up like approaching the study participants could be helpful.
Conclusion
The number of people with T2DM is greatly increasing and the need of new strategies to control the disease and its complications is well needed. Remote monitoring is an innovative solution to manage chronic diseases and specially diabetes mellitus. Telemonitoring can give people the chance to take control over their T2DM with a better quality of life. This study carried out three solutions has shown the feasibility of using telemonitoring in clinical routine and its potential benefits for diabetes care. RT-NI and U-Tell: Diabetes faced some reluctance from few healthcare providers because of their concerns about the potential workload they might add to them and preferred the face-to-face standard care.
More evidence of the valuable effects of telemonitoring on people’s self-management motivation may help to encourage healthcare providers to adopt these solutions in their practice. Further research is required to validate the results suggested by this exploratory study refining telemedicine delivery models and specifically improvements in telemonitoring systems. The next step would be to extend the study to a wider population and including people with different types of diabetes mellitus (such as Type 1 diabetes mellitus and gestational diabetes mellitus). Additional research is also needed to reinforce cultural and behavioural change on both patients and clinicians sides [60].
Today, the advancement in information technology and telecommunication has a great impact on delivering a better quality of life and care by integrating disease management, quality assessment, cost-effectiveness and knowledge through telemonitoring.
Acknowledgment
The authors would like to thank Dr Roy Harper and his team for their clinical input and support during the recruitment and data collection process.
Disclosure
The authors declare that there is no conflict of interest regarding the publication of this paper.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
References
- van Dieren S, Beulens JW, van der Schouw YT, Grobbee DE, Neal B (2010) The global burden of diabetes and its complications: an emerging pandemic. Eur J Cardiovasc Prev Rehabil 17(Suppl 1): S3-S8.
- Bonell C, Oakley A, Hargreaves J, Strange V, Ree R (2006) Assessment of generalisability in trials of health interventions: suggested framework and systematic review. BMJ 333(7563): 346-349.
- Diabetes UK (2015) Diabetes: Facts and Stats. Diabetes UK, UK.
- Jaana M, Pare G (2007) Home telemonitoring of patients with diabetes: a systematic assessment of observed effects. Journal of Evaluation in Clinical Practice 13(2): 242-253.
- Hanley J, Fairbrother P, McClouughan L, Pagliari C, Paterson M, et al. (2015). Qualitative study of telemonitoring of blood glucose and blood pressure in type 2 diabetes. BMJ Open 5(12).
- The bmj (2016) 13. Study design and choosing a statistical test.
- Holman RR, Paul SK, Bethel A, Matthews DR, Neil HW (2008) 10-year follow-up of intensive glucose control in type 2 diabetes. The new England Journal of Medicine 359(15): 1577-1589.
- Mushcab H, Kernohan G, Wallace J, Martin S (2015) Web-Based Remote Monitoring Systems for Self-Managing Type 2 Diabetes: A Systematic Review. Diabetes Technol Ther 17(7): 498-509.
- Meystre S (2005) The current state of telemonitoring: a comment on the literature. Telemed J E Health 11(1): 63-69.
- Karhula T, Vuorinen AL, Rääpysjärvi K, Pakanen M, Itkonen P, et al. (2015) Telemonitoring and Mobile Phone-Based Health Coaching Among Finnish Diabetic and Heart Disease Patients: Randomized Controlled Trial. J Med Internet Res 17(6): e153.
- Department of Health (2005) Building Telecare in England. Department of Health, London, England, p.16.
- Gomez E, Hernando M, Garcia A, Del Pozo F, Cermeno J, et al. (2002) Telemedicine as a tool for intensive management of diabetes: the DIABTel experience. Computer Methods and Programs in Biomedicine 69(2): 163-177.
- Kao LS, Tyson JE, Blakely ML, Lally KP (2008) Clinical Research Methodology I: Introduction to Randomized. J Am Coll Surg 206(2): 361-369.
- Matowe L, Leister CA, Crivera C, Korth-Bradly JM (2003) Interrupted Time Series Analysis in Clinical Research. Ann Pharmacother 37(7-8): 1110-1116.
- Kontopantelis E, Doran T, Springate D, Buchan I, Reeves D (2015) Regression based quasi-experimental approach when randomisation is not an option: interrupted time series analysis. BMJ 350.
- Eccles M, Grimshaw J, Campbell M, Ramsay C (2003) Research designs for studies evaluating the effectiveness of change and improvement strategies. Qual Saf Health Care 12: 47-52.
- Kitiş Y, EmVrog˘lu N (2006) the effects of home monitoring by public health nurse on individuals’ diabetes control. Applied Nursing Research, 19(3): 134-143.
- Laerd Statistics (2013) Kruskal-Wallis H Test using SPSS Statistics..
- Ramsay C, Matowe L, Grilli R, Grimshaw J, Thomas RE (2003) Interrupted time series designs in health technology assessment: lessons from two systematic reviews of behavior change strategies. Int J Technol Assess Health Care 19(4): 613-623.
- Glass GV (1997) Interrupted Time Series Quasi-Experiments. In: RM Jaeger (Ed.), Complementary methods for research in education, (2nd edn), American Educational Research Association, Washington, USA, pp. 589-608.
- Penfold RB, Zhang F (2013) Use of interrupted time series analysis in evaluating health care quality improvements. Acad Pediatr 13(6S) S38-S44.
- Harris AD, MCGregor JC, Perencevich EN, Furuno JP, Zhu J, et al. (2006) The Use and Interpretation of Quasi-Experimental Studies in Medical Informatics. Journal of the American Medical Informatics Association 13(1): 16-23.
- Procter R (2009) Health Informatics.
- Grimshaw J, Campbell M, Eccles M, Steen N (2000) Experimental and quasi-experimental designs for evaluating guideline implementation strategies. Familty Practice 17(Suppl. 1): S11-S16.
- http://childhoodcancer.cochrane.org/non-randomised-controlledstudy- nrs-designs
- Deeks J, Dinnes J, D’Amico R, Sowden A, Sakarovitch C, et al. (2003) Evaluating non-randomised intervention studies. Health Technol Assess 7(27): 1-173.
- Wagner AK, Soumerai SB, Zhang F, Ross-Degnan D (2002) Segmented regression analysis of interrupted time series studies in medication use research. J Clin Pharm Ther 27: 299-309.
- Shadish WR, Cook, TD, Campbell DT (2001) Experimental and Quasi- Experimental Designs for Generalized Causal Inference (2nd edn). Wadsworth Publishing, Boston, USA.
- http://www.northernireland.gov.uk/index/media-centre/newsdepartments/ news-dhssps/news-dhssps-june-2014/news-dhssps- 120614-embracing-technology-has.htm
- Goodwin J (2009) Research in Psychology: Methods and Design. (6th edn), John Wiley & Sons Inc., Danvers, Massachusetts.
- Heppner PP, Wampold E, Owen J, Wang KT, Thompson MN (2015) Research Design in Counseling. (4th edn), Cengage Learning, Boston, Massachusetts, USA.
- Michielutte R, Shelton B, Paskett E, Tatum CM, Velez R (2000) Use of an interrupted time-series design to evaluate a cancer screening program. Health Education Research 15(5): 615-623.
- Velicer WF, Harrop J (1983) The Reliability and Accuracy of Time Series Model Identification. Evaluation Review 7(4): 551-560.
- Ludford PJ (2016) An Overview: Choosing the Correct Statistical Test.
- Craig P, Dieppe P, Macintyre S, Michie S, Nazareth I (2006) Developing and evaluating complex interventions: new guidance. Medical Research Council, UK, p.1-39.
- Hebl M (2016) Descriptive Statistics.
- Biglan A, Ary D, Wagenaar AC (2000) The Value of Interrupted Time- Series Experiments for Community Intervention Research. Prev Sci 1(1): 31-49.
- http://www.diabetes.co.uk/news/2014/jun/d-nav-diabetes-insulinguidance- system-trialled-in-northern-ireland-97432524.html
- http://hygieia.com/
- http://www.northerntrust.hscni.net/about/2559.htm
- NI T (2015) Partners (Telemonitoring NI).
- http://homepage.stat.uiowa.edu/~rlenth/Power/
- Snoek FJ, Pouwer F, Welch GW, Polonsky WH (2000) Diabetes-related emotional distress in Dutch and US. diabetic patients: cross-cultural validity of the problem areas in diabetes scale. Diabetes Care 23(9): 1305-1309.
- NCSS, LLC (2016) Mixed Models - Repeated Measures.
- Laerd Statistics (2013) Descriptive and Inferential Statistics.
- http://www.graphpad.com/guides/prism/6/statistics/index. htm?how_the_kruskalwallis_test_works.htm
- Solutions S (2016) Kruskal-Wallis Test.
- Penn State Lehigh Valley (2016) Chi-Square Test.
- Stat Trek Teach (2016) Chi-Square Test for Independence.
- Statistics Solutions (2016) Paired Sample T-Test.
- Based RM (2006) Descriptive Statistics.
- NCSS, LLC (2016) Mixed Models - Repeated Measures.
- http://clinfowiki.org/wiki/index.php/Main_Page
- Steventon A, Bardsley M, Billings J, Dixon J, Doll H, et al. (2012) Effect of tele health on use of secondary care and mortality: findings from the whole system demonstrator cluster randomised trial. BMJ 344: e3874.
- Kenealy TW, Parsons MJ, Rouse AB, Doughty RN, Sheridan NF, et al. (2015) Telecare for Diabetes, CHF or COPD: Effect on Quality of Life, Hospital Use and Costs. A Randomised Controlled Trial and Qualitative Evaluation. Plos One 10(3): e0116188.
- Stone R, Sevick M, Rao R, Macpherson D, Cheng C, et al. (2012) The Diabetes Telemonitoring Study Extension: an exploratory randomized comparison of alternative interventions to maintain glycemic control after withdrawal of diabetes home telemonitoring. J Am Med Inform Assoc 19(6): 973-979.
- WHO (2015) Clinical Trials. World Health Organisation, Geneva, Switzerland.
- WHO (2006) Definition and Diagnosis of Diabetes Mellitus and Intermediate Hyperglycaemia. World Health Organization and International Diabetes Federation. World Health Organization, Geneva, Switzerland.
- WHO media Center (2015) Diabetes.
- Stone R, Sevick M, Rao R, Macpherson D, Cheng C, et al. (2012) The Diabetes Telemonitoring Study Extension: an exploratory randomized comparison of alternative interventions to maintain glycemic control after withdrawal of diabetes home telemonitoring. J Am Med Inform Assoc 19 (6): 973-979.






























