CMV Seroconversion Associated with an high Avidity in a Previously Seronegative Pregnant Woman during her first Trimester of Pregnancy
Marie-Luce Delforge, Jean-Michel Cirriez, Patricia Schatt, Clinicalpathologist, Laurence Auger, Patrick Vankerkhoven, Patrick Vankerkhoven, Alexandre Grimmelprez and Quentin Delefortrie*
1ClinicalPathologist, Clinique Notre Dame de Grâce, Belgium
Submission: December 06, 2016; Published: December 07, 2016
*Corresponding author: Quentin Delefortrie, intern in clinicalpathology, Clinique Notre Dame de Grâce, Chaussée de Nivelles 212, 6041 Gosselies, Belgium, Email:quentin.delefortrie@uclouvain.be
How to cite this article: Marie-Luce D, Jean-Michel C, Patricia S, Clinicalpathologist, Laurence A et al. CMV Seroconversion Associated with an high Avidity in a Previously Seronegative Pregnant 002 Woman during her first Trimester of Pregnancy. Curr Trends Biomedical Eng & Biosci. 2016; 1(1): 555552. DOI: 10.19080/CTBEB.2016.01.555552
Dear Editor
Recently, we observed in our laboratory a CMV seroconversion with the appearance of CMV-specific IgGtogether with an IgM response in a previously seronegative pregnant woman during her first trimester (week 14) of pregnancy (kit used: Liaison® CMV IgGII and IgMII kits,Diasorin®, Italy). Surprisingly, the latest sample displayed high avidity using the Liaison® IgG Avidity II kit (Diasorin®, Italy). Similar results were observed with those samples using the Vidas® CMV IgG, IgM and Avidity kits (biomérieux®, France) (Table 1).
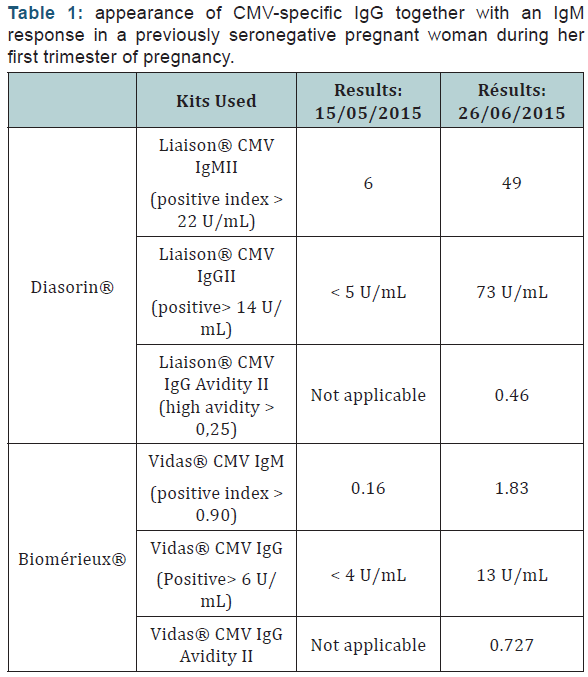
Seroconversion was also confirmed based on former (present in our serobank) and new sera requested to the patient. In addition, a western blot was conducted with the RecomLine CMV IgG and Avidity (Mikrogen®, Germany) and based on that, the infection was estimated within 14 weeks.Seroconversion together with high avidity has been described in the literature in transplants by passive immunization or in studies for which sera of pregnant women were acquired more than 12 weeks post-primary CMV [1,2]. However, Sellier et al. [3] reported CMV seroconversion together with a high avidity in three pregnant women, using the Liaison® IgG Avidity II kit (Diasorin®, Italy), with a fetal transmission in two of them [3].
As the universal screening of pregnant woman is the most reliable way to identify primary CMV infection [4] and as primary rather than non-primary maternal CMV infection was shown to be associated with higher risk of congenital infection disease (> 20 times more likely to result in placental transmission)[5], our patient was managed as a primary CMV infection. CMV-PCR amniocentesis performed at week 26 showed negative results. Fetal abnormalities were not detected using ultrasound either. Finally, at birth, the newbornwasasymptomatic, CMV culture and molecular detection by PCR obtained from the infant’s urine werefound negative. Routine hearing assessment was also normal. All those results reasonablyexclude the possibility of an intrauterine CMV transmission and congenital infection in our case.
Without the known serological status of the patient, these obvious falsely high avidity resultswould have led to miss a case of primary CMV infection. Apart from passively acquired immunization and bleed timing issues as aforementioned, this phenomenon is relatively rare and hardly described in the literature. Recently, Berth et al. highlighted the importance of integrating low CMV IgG levels for interpreting avidity results [6]. However, in contrast to our observation, they only observed erroneously low avidity in immune patients. Moreover, IgG levels of our patients could not be considered as low (>2 times cut-offs levels for both kits). Nevertheless, Sellier et al. made the same observation with a IgG level close to the one observed for our patient using the same Diasorin® kit (73 U/mL vs 50 U/mL for [2]) In conclusion, based on our observation and on the literature, caution must be brought to the interpretation of avidity values and improvement of the serological diagnosis of CMV is still needed.
Funding
Clinique Notre Dame de Grâce, Chaussée de Nivelles 212, 6041 Gosselies, Belgium (companynumber : 0401.690.559)
Ethical Approval
This study was cleared by the Ethics committee of the Hospital: “Clinique Notre Dame de Grâce”(company number: 0401.690.559)
In addition, the patient signed an informed consent.
References
- Bodéus M, Feyder S, Goubau P (1998) Avidity of IgG antibodies distinghishes primary from non-primary cytomegalovirus infection in pregnant women. Clin Diagn Virol 9(1): 9-16.
- Lazzarotto T, Spezzacatena P, Pradelli P, Abate DA, Varani S et al. (1997) Avidity of Immunoglobulins G Directed Against Human Cytomegalovirus During Primary and Secondary Infections in Immunocompetent and Immunocompromised Subjects, Clin Diagn Lab Immunol 4(4): 469-473.
- Sellier Y, Guilleminot T, Ville Y, Leruez-Ville M (2015) Comparison of The Liaison® CMV IgG Avidity II and the Vidas® CMV IgG Avidity II assays for the diagnosis of primary infection in pregnant wome. J Clin Virol 72: 46-48.
- Naing ZW, Scott GM, Shand A, Hamilton ST, van Zuylen WJ, et al. (2015) Congenital cytomegalovirus infection in pregnancy: a review of prevalence, clinical features, diagnosis and prevention, Aust N Z J Obstet Gynaeco 56(1): 9-18.
- Revello MG, Genini E, Gorini G, Klersy C, Piralla A, et al. (2010) Comparative evaluation of eight commercial human cytomegalovirus avidity assays. J Clin Virol 48(4): 255-259.
- Berth M, Grangeot-Keros L, Heskia F, Dugua JM, Vauloup-Fellous C (2014) Analytical issues possibly affecting the performance of commercial human cytomegalovirus IgG avidity assays. Eur J Clin Microbiol Infect Dis 33(9): 1574-1584.






























