Persistent Hyperglycemia Induced Demyelination and Fibrosis in Sensory Ganglia of Albino Rats
Muhamed Faizal and Aijaz Ahmed Khan*
Department of Anatomy, Aligarh Muslim University, India
Submission: March 03, 2018; Published: May 25, 2018
*Corresponding author: Aijaz Ahmed Khan, Department of Anatomy, Jawaharlal Nehru Medical College, Aligarh Muslim University, Aligarh, India, Email: aijazahmedkhan7@live.com
How to cite this article: Muhamed Faizal, Aijaz Ahmed Khan. Persistent Hyperglycemia Induced Demyelination and Fibrosis in Sensory Ganglia of Albino Rats. Anatomy Physiol Biochem Int J: 2018; 5(1): 555655. DOI: 10.19080/APBIJ.2018.05.555655.
Abstract
Hyperglycemia-induced neurotoxicity is believed to affect both neurons and glial cells. This study was aimed to assess the histopathological alteration in the Trigeminal Ganglion (TrG) of STZ-induced diabetic adult male albino rats. A total number of 12 adult albino rats of either sex were divided into age-matched control and six month diabetic group. After six month animals of both groups were euthanized with ether anesthesia and perfuse with Karnovsky fixative. TrG from both sides was dissected, processed and 5 μm thick sections were stained for collagen. Light microscopy showed that long standing hyperglycemia was associated with increasing occurrence of dark and dead neurons and excess quantity and more thickened collagen fibers in the peri-ganglionic as well as peri-neuronal capsule and along the nerve fibers. It was concluded that glucose toxicity not only induces neuronal death and demyelination but also excessive deposition of collagen which appear to constitute the contributing factors in the progression of diabetic peripheral neuropathy.
Introduction
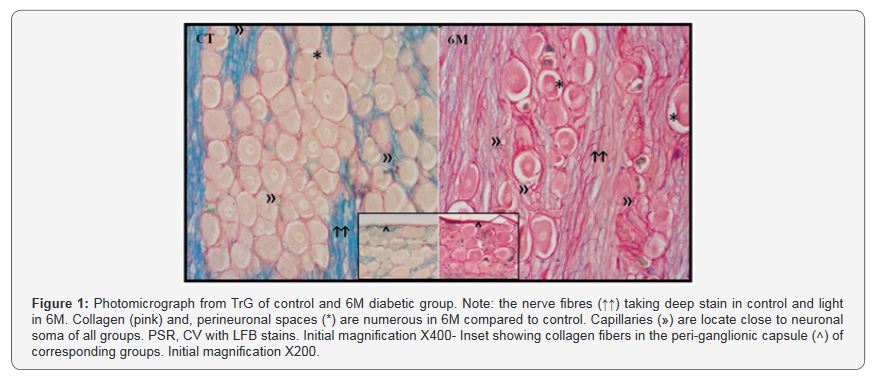
Glucose toxicity cause functional and structural disorders in both central and peripheral nervous systems. Lack of blood-brain and blood-nerve barriers, sensory neurons in the TrG are highly sensitive to oxidative stress in diabetes [1]. Mitochondrial dysfunction may leads to neuronal apoptosis. In addition, micro environmental alterations in the sensory ganglia may cause transformation of neuronal phenotype, ion channel alterations, and abnormal growth factor signaling [2]. Its clinical manifestations include sensory alterations in the orofacial complex like trigeminal neuralgia [3]. The aim of present study was to assess the effect of longstanding hyperglycemic state on sensory neurons, nerve fibres and supporting tissue of TrG which may possibly explain the clinical symptoms following STZ induced hyperglycemia (Figure 1).
Material and Method
Total numbers of 12 adult albino rats of either sex (Weight ~250gm) from central animal house, Aligarh Muslim University, Aligarh were used in the present study (IAEC- D:No-9025/2014). After one week rats were divided into two groups
(I) Non-diabetic healthy control, age-matched;
(II) 6M diabetic experimental group. Experimental diabetic model was induced by streptozotocin (STZ) (60mg/kg, aqueous sol., I. P., single dose) whereas age-matched control group did not receive any active compound.
Diabetic rats maintained blood sugar level more than 250 mg/dl throughout the experiment. After six month, rats were euthanized with ether and perfusion fixed with Karnovsky fixative. Both fixed TrG was dissected out then process for paraffin embedding. Five μm thick sections were stained with Cresyle Violet (CV) and Picro Sirus with Luxol Fast Blue (PSR with LFB) and observed under the light microscope.
Observation
In high power view, TrG of control group showed normal distribution of well defined neurons and few collagen fibres around the neurons, along the nerve fibers and capsule of TrG. Approximately similar features were also found in 6M diabetic group but the number and thickness of collagen fibers were markedly increased around the neurons, along the nerve fibers and between clusters of neurons and capsule of the ganglia. In addition increased number of dark neurons with degenerative changes was also noticeable. Myelin showed poor staining and perineuronal spaces were more frequently associated with neurons of 6M diabetic group.
Discussion
Histopathological observations in the current study were suggestive of neurotoxic and degenerative effects of glucose on the sensory ganglion. Some researchers have shown myelin sheath defect and perineuronal spaces in chronic hyperglycemia [4]. I was suggested that changes may be apoptotic due to progression of hyperglycemia. Similar results were also found in the 6M diabetic group of the current study. One study observed that type I, II and III collagen are the fibrillar - interstitial collagens and are responsible for fibrosis. One related study observed that p38 mitogen-activated protein kinase, PKC-β, AGE and RAGE interaction to be responsible for the increased expression of TGF-β which contributes to the development of fibrosis [5,6]. In the present study 6M hyperglycemic group, exhibit thickening of collagen around the neurons and along the nerve fibers and ganglionic capsule. In addition fibrosis was also seen around the intra-ganglionic vessels. These histopathological findings indicated that the prolonged hyperglycemia seems to promote both vascular pathology and neurotoxicity which is correlated very well with previous observations regarding link between hyperglycemia and sensory neurons and collagen connective tissues in sensory ganglion [4,7].
Conclusion
Prolonged hyperglycemia induces neuronal death, demyelination and increase fibrosis in the trigeminal ganglion which appears to be an important causative factor in the development of diabetic neuropathy.
References
- Zochodne DW, Verge VKM, Cheng C, Sun H, Johnston J (2001) Does diabetes target ganglion neurones? Progressive sensory neurone involvement in long-term experimental diabetes. Brain 124(11): 2319- 2334.
- Nones CFM, Reis RC, Jesus CHA, Veronez DAL, Cunha JM, et al. (2013) Orofacial sensory changes after streptozotocin-induced diabetes in rats. Brain research 1501: 56-67.
- Krastev D, Paloff A, Krastev N, Apostolov A, Ovtscharoff W (2008) Ultrastructure of trigeminal ganglion in human. Journal of IMAB 1: 36-39.
- Malak HW, Saleh SI, Salah El Din RA, Abdul Hamid HF (2015) Histological and immunohistochemical study on the consequences of acute glycemic level alteration on the dorsal root ganglia and sciatic nerve integrity in neonatal albino rats. Egyptian Journal of Histology 38: 332-345.
- Olubunmi A Adebiyi, Oluwafeyisetan O Adebiyi, Peter M O Owira (2016) Naringin Reduces Hyperglycemia-Induced Cardiac Fibrosis by Relieving Oxidative Stress. Plos One 11: 1-15.
- De Vriese AS, Flyvbjerg A, Mortier S, Tilton RG, Lameire NH (2003) Inhibition of the interaction of AGE-RAGE prevents hyperglycemiainduced fibrosis of the peritoneal membrane. J Am Soc Nephrol 14(8): 2109-2118.
- Faizal M, Khan AA (2017) Effect of streptozotocin-induced diabetes on the autonomic ganglia of albino rats. Anatomy 11: 51-60.






























