Gender-linked Variation in the Removal of Damaged Erythrocytes by Hepatic and Splenic Macrophages - Review Article
Latif Ahmad1,2*, Safdar Ali2, Imdad Hussain Leghari3, Meher-un-Nisa Memon3, Rajesh Kumar Oad1, Khalid Hussain Memon1, Tariq Afzal2, NajeebUllah Lail1, Hamid Ali Memon1, Athar Ali Khan4, Muahmmad Asif Raza1, Noreen Mehmood1, Faiza Rubab3, Tanzeela Farooq1, Safia Kandhro1, Mashhood Ahmed1, Viram Kumar1 and Waseem Ali Vistro5*
1Baqai College of Veterinary Sciences, Baqai Medical University, Pakistan
2Sargodha Medical College, University of Sargodha, Pakistan
3Department of Poultry Husbandry, Pakistan
4Department of Animal Nutrition, Sindh Agriculture University, Pakistan
5Nanjing Agricultural University, China
Submission: April 12, 2018; Published: May 24, 2018
*Corresponding author: Latif Ahmad, Baqai College of Veterinary Sciences, Baqai Medical University, Karachi, Pakistan, Email: latifahmaddr@gmail.com
How to cite this article: Latif Ahmad, Safdar Ali, Imdad Hussain Leghari, Meher-un-Nisa Memon, et al. Gender-linked Variation in the Removal of Damaged Erythrocytes by Hepatic and Splenic Macrophages - Review Article. Anatomy Physiol Biochem Int J: 2018; 5(1): 555654. DOI: 10.19080/APBIJ.2018.05.555654.
Abstract
Gender-specific differences in innate and acquired immunity have been well-documented in mammals, avian and other species, coining the terms male-specific diseases and female specific diseases. Various workers have reported differences in biological, social and behavioral drivers to be cause of such sex disparity. But the sex of individual cells, independently of above-mentioned factors, also plays a critical role in disease. This study is multidisciplinary multi-professional joint venture to focus on gender variation in hepatic and splenic macrophages to remove auto antigens. The macrophages of female birds express IFN-responsive genes constitutively. The female birds compensate for the reduced dose of inducible IFN with higher basal set point of IFN-responsive genes. Gender specific difference about enzyme mechanism is noted e.g., DNaseII is a lysosomal enzyme responsible for the degradation of phagocytosis-encountered DNA in macrophages. In humans, the homozygotic presence of a single-nucleotide polymorphism in the 5’ regulatory region of the DNaseII gene confers an increased risk for RA in male patients.
Introduction
The male sex is obviously stronger, bigger and muscular, but to a pathologist, who deals with diseases, male sex is weaker than the female sex, as males are more prone to diseases. The worldwide opinion, both in technologically advanced republics and in unindustrialized nations, suggests a longevity gap in males and females in favor of females [1]. Gender-specific differences in innate and acquired immunity have been well-documented in mammals, avian and other species. So male-specific diseases and female specific diseases have been recognized including various infections, metabolic and nutritional diseases and diseases linked to cardiovascular, reproductive, neuro-endocrine, pulmonary, hepatic and other systems. Various workers have reported differences in hormones, chromosomes, genes, basal set-points, reproductive anatomy, metabolism etc. as biological drivers of such sex disparity. Various social and behavioral factors have also been considered culprit of such sex disparity. But the sex of individual cells, independently of above-mentioned factors, also plays a critical role in disease.
Various auto antigens are removed from the body to keep the body healthy. Damaged erythrocytes are one of those auto antigens. The immunologists in the previous six decades had worked out physiological analysis of body iron regulations and during the recent decade its molecular considerations have been momentously focused [2,3]. On the other hand, in vivo and in vitro studies have demonstrated that the macrophages (MΦs) motivate the formation of erythrocytes or Red Blood Cells (RBCs) under homeostatic as well as deviated situations. After being fully-fledged, the RBCs persist in blood for almost 4 months in cattle and humans and 1½ month in mice [4]. Generally speaking, RBCs of small animals have shorter lifespan as compared to those of large animals; it fluctuates from 70-160 days in domestic animals although the life span is often shorter in immature animals than mature ones.
At the end of their lifespan, RBCs are cleared by MΦs residing in the spleen and the liver. Apoptosis and other death modes of cells in the animal body are continuous processes and are more frequent in the events of growth, reaction to wounds and repair of the damaged parts. Hence, fast and proficient clearance of dead cells is a compulsory criterion for homeostatic well-being. Any inadequacy in getting rid of dead and defected cells causes gathering of auto antigens in tissues that nurture diseases including chronic inflammations, autoimmunity and developmental anomalies [5]. Present study tried to search the gender dissimilarity in eliminating spoiled RBCs by MΦs of liver and spleen
Presentation Of Experimental And Clinical Studies
In a number of studies, certain factors linked to gender-based adaptable controlling of MΦ working. For example, differences in gene expression in MΦs grown from the bone marrow of male and female chickens have been identified [6]. It may be due to such variable functioning of MΦs that a number of diseases have gender-specific risks e.g. various animal models of autoimmune disorders (such as systemic lupus erythematosus) have been described in which female animals are significantly more susceptible to the disease. The sex steroids have been linked with immunity and self-recognition [7]. Also, sex hormones increase the high-affinity auto-reactive B cells, the latter have tremendous importance in elementary understanding for the mechanism of immune dis-regulation and the diseases so produced.
The endo toxin-induced MΦ stimulation have been reported to produce De-oxyriboNucleic Acid (DNA) damage via restricting nuclear factor kappa-B (NFκB) stimulation [8]. Contrary to the general rule of more disease to males, epidemiological studies show exceptionally high (¾) risk of autoimmune diseases in women [5]. Limited sex chromosome dosage compensation in birds occurs on a gene-by-gene, rather than a chromosome-wide basis, as appears to be the case for random allelic inactivation on the autosomes in mammals. The RBCs may get unfurnished from blood flow in response to modification in their volume, configuration or deformability. Such response is controlled by the precise arrangement of the Inter-Endothelial Slit (IES) in the spleen. The spleen performs specifically in allocating size and contour of normal RBCs [9].
The MΦs firmly regulate the manufacture and unfurnishing of RBCs [4]. The splenic red pulp has the capacity to recognize aged RBCs and early reticulocytes through filtering out rigid cells; both aged RBCs and early reticulocytes lack deformability [10]. The spleen is also a lymphoid organ with cellular diversity and compartmentalized microenvironments, suitable for the definition of immunologic processes that may influence the outcome of infections. There tends to be species variation e.g., contrary to what is observed in mice, LPS did not induce thymidine incorporation by monkey or rabbit spleen cells. A macrophage engulfs another cell or foreign particle in an adhesive process that often activates myosin-II, unless the macrophage also engages “marker of self” CD47 that inhibits myosin. Rigid but rounded RBC stomatocytes signal self-better than rigid RBC discocytes, highlighting the effects of shape on CD47 inhibition.
CD47 interacts with the macrophage self-recognition receptor Signal Regulatory Protein Alpha (SIRPA). Physical properties of phagocytic targets thus regulate self signaling, as is relevant to erythropoiesis, to clearance of rigid RBCs after blood storage, clearance of rigid pathological cells such as thalassemic or sickle cells, and even to interactions of soft/stiff cancer cells with macrophages. Micro-anatomically, the spleen is divided into the white pulp and the Red Pulp (Rp), separated by the Marginal Zone (MZ). Rp and MZ have a complex MΦ network with distinct origins and functions in the immune response to infections. Rp MΦs form a vast network inside the Rp and are characterized in mice by expression of F4/80highCD68+CD1 1blow/− and intense autofluorescence [11]. In turn, inside the MZ, two populations of MΦs can be discerned. Marginal Metallophilic Macrophages (MMMΦs) and Marginal Zone Macrophages (MZMΦs) are cells with great ability to internalize blood-borne pathogens such as virus or bacteria.
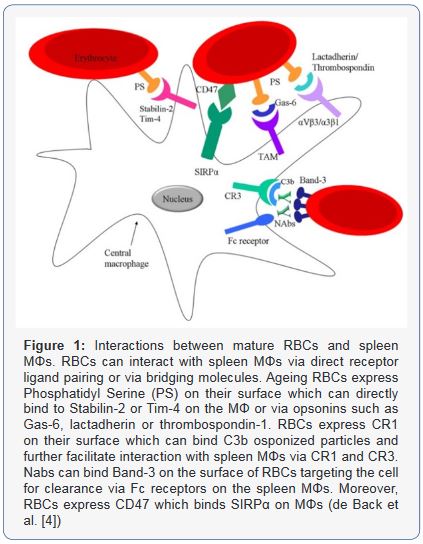
The MZMΦs typically express in their surface the C-type lectin SIGN-related 1 (SIGNR1) and a type I scavenger receptor called MΦ Receptor with Collagenous Structure (MARCO), which recognize non-opsonized molecules, mainly blood-borne antigens. Furthermore, Marginal Metallophilic MΦs (MMMΦs) are defined, among other molecules, by the expression of Sialic acid-binding Ig-like Lectin-1 (Siglec-1, Sialoadhesin, CD169) and MOMA-1 [12] (Figure 1).
Ahmad et al. [13] described cypermethrin-induced anemia in male rabbits only and mentioned that female rabbits were spared to develop anemia with the same treatment. Another study on rabbits described cypermethrin-treated rabbit bucks/ males to develop anemia, leukopenia, neutropenia, monocytosis, eosinophilia and basocytopenia. In contrast, rabbit does/females showed transient but significant leukocytosis, neutropenia, lymphopenia, monocytosis, eosinophilia and basophilia. Moreover, spleen histology revealed congestion, depletion of white pulp with increased red pulp and hemosiderin deposition in CY treated rabbit bucks, but not rabbit does [1].
One of the most important paradigm shifts in MΦ biology during recent years referred to the origin of tissue MΦs, including MΦs in the liver. Previously tissue MΦs were thought to originate from circulating adult blood monocytes. A recent scientific opinion is that many resident tissue MΦs are established during embryonic development and persist independent from blood monocytes in homeostasis. This dichotomy of MΦs is prototypically found in the liver. The liver harbors about 80% of all MΦs of the body and is furthermore patrolled by blood monocytes. Hepatic MΦs consist of Kupffer cells (KCs), which are originated from the fetal yolk-sack, and infiltrated bone marrow-derived monocytes/MΦs [14]. Hepatic MΦs, the KCs are involved in Hepatocellular Carcinoma (HCC). The HCC is the most common liver cancer, occurring mainly in men. Similar gender disparity is seen in mice given a chemical carcinogen, Diethylnitrosamine (DEN). DEN administration caused greater increases in serum Interleukin-6 (IL-6) concentration in males than it did in females. Furthermore, ablation of IL-6 abolished the gender differences in hepatocarcinogenesis in mice. DEN exposure promoted production of IL-6 in KCs in a manner dependent on the Toll-like receptor adaptor protein MyD88, ablation of which also protected male mice from DEN-induced hepatocarcinogenesis. Estrogen inhibited secretion of IL-6 from KCs exposed to necrotic hepatocytes and reduced circulating concentrations of IL-6 in DEN-treated male mice. Estrogenmediated inhibition of IL-6 production by KCs may reduce liver cancer risk in females, and these findings may be used to prevent HCC in males [15]. IL-6 is an important cytokine. Cytokines have low molecular weight and are pleiotropic signaling glyco-proteins. They are involved extensively in inter-cellular communications, including earliest immune and inflammatory reactions to noxious stimuli and in the later adaptive (specific) immune responses to microbes.
IL-6 coordinates other cytokines and creates an association between non-specific and adaptive immune systems. IL-6 is among causative factors in insulin resistance, diabetes and pulmonary damage in allergic asthma [8]. So all of these conditions may also have gender-linked variation. Amongst the schemes employed in drug delivery, RBCs were found to have extraordinary prospective as drug carriers. The techniques bringing the opening of nanopores outwardly letting the access of molecules have enabled such demonstrations. Being able to adjust surfaces of cellular membranes through stimulating the creation of MΦs-accepted antigenic sites, such cells can accept pharmaceutical molecules. MΦs can also produce and adjust MΦs-accepted antigenic sites favoring the transport of pharmaceutical molecules to such cells.
On the other hand, the RBCs reformed through lipophilic antibodies can specifically contact MΦs. The role of RBCs bearing pharmaceutical molecules in removing MΦs has been demonstrated in in vitro and in vivo research [16]. In addition to role of RBCs in toxicity to counteract oxidative stress, they also maintain immunological homeostasis. Upon intense creation of reactive species, RBCs can also behave in a pro-oxidant manner. Oxidized, senescent, or stored RBCs employ a compromised modulatory action on all types of immune cells; the latter free their representative physical and physiological properties. Such activity is due to modifications in surface antigens and discharge of inflammatory or any endo-biotic or xeno-biotic molecule inducing oxidative stress [17].
In a pilot study on 56 human patients, some RBC parameters in samples with metabolic syndrome and subclinical atherosclerosis, but without any sign of coronary artery disease were analyzed to determine new peripheral gender-associated bioindicators of possible diagnostic or prognostic value. In particular, three different “indicators” of RBC injury and aging were evaluated: glycophorin A, CD47, and phosphatidylserine externalization. Interestingly, all those determinants appeared significantly modified and displayed gender differences [18]. All of the above findings could provide novel and useful hints in the research for gender-based real-time bioindicators in the progression of various metabolic and immune-inflammatory diseases. Further, more extensive studies are, however, necessary in order to validate such findings.
Conclusion
Upon thorough research literature study, a gender specific difference about enzyme mechanism is noted e.g., DNaseII is a lysosomal enzyme responsible for the degradation of phagocytosis-encountered DNA in macrophages. In humans, the homozygotic presence of a single-nucleotide polymorphism in the 5’ regulatory region of the DNaseII gene confers an increased risk for RA in male patients. A better understanding of the cellular communications can enable us utilizing teamwork of RBCs and immune cells in clinical settings according to gender specificity.
References
- Ahmad L, Khan A, Rind MI, Vistro WA, Raza MA, et al. (2017) Cypermethrin-induced Repeated-dose Immuno-toxicity in Oryctolagus Cuniculus: An Investigation into Pathology of Spleen and Blood. Int Arch BioMed Clin Res 3(2): 9-14.
- De Domenico, I, E Nemeth, JM Nelson, JD Phillips, RS Ajioka, et al. (2008) The hepcidin-binding site on ferroportin is evolutionarily conserved, Cell Metab 8(2): 146-156.
- Preza GC, P Ruchala, R Pinon, E Ramos, B Qiao, et al. (2011) Minihepcidins are rationally designed small peptides that mimic hepcidin activity in mice and may be useful for the treatment of iron overload. J Clin Invest 121(12): 4880-4888.
- De Back DZ, EB Kostova, M van Kraaij, TK van den Berg, R van Bruggen (2014) Of macrophages and red blood cells: a complex love story. Frontiers Physiol 5(9): 11.
- Whitacre CC (2001) Sex differences in autoimmune disease. Nat Immunol 2: 777-780.
- Garcia-Morales C, S Nandi, D Zhao, KA Sauter, L Vervelde, et al. (2015) Cell-autonomous sex differences in gene expression in chicken bone marrow-derived macrophages. J Immunol 194(5): 2338-2344.
- Sundburg CR, JM Belanger, DL Bannasch, TR Famula, AM Oberbauer (2016) Gonadectomy effects on the risk of immune disorders in the dog: a retrospective study. BMC Vet Res 12: 278-287.
- Ahmad L, Dogar MZH (2016) Insecticidal exposure may be the cause of progression of immune disorders and inflammatory diseases through dys-regulation of cytokines. Int Arch BioMed Clin Res (IABCR) 2(3): 18-27.
- Pivkin IV, Z Peng, GE Karniadakis, PA Buffet, M Daom, et al. (2016) Biomechanics of red blood cells in human spleen and consequences for physiology and disease. Proc Natl Acad Sci USA 113(28): 7804-7809.
- Klei TRL, SM Meinderts, TK van den Berg, R van Bruggen (2017) From the Cradle to the Grave: The Role of Macrophages in erythropoiesis and erythrophagocytosis. Frontiers Immunol 8: 73.
- Epelman S, KJ Lavine, GJ Randolph (2014) Origin and Functions of Tissue Macrophages. Immunity 41(1): 21-35.
- Biesen, R, C Demir, F Barkhudarova, JR Grun, M Steinbrich-Zollner, et al. (2008) Sialic acid-binding Ig-like lectin 1 expression in inflammatory and resident monocytes is a potential biomarker for monitoring disease activity and success of therapy in systemic lupus erythematosus. Arthritis Rheum 58(4): 1136-1145.
- Ahmad L, A Khan, MZ Khan, I Hussain (2009) Cypermethrin induced anaemia in male rabbits. Pakistan Vet J 29(4): 191-195.
- Ju C, F Tacke (2016) Hepatic macrophages in homeostasis and liver diseases: from pathogenesis to novel therapeutic strategies. Cellular and Molecular Immunology 13(3): 316-327.
- Naugler WE, T Sakurai, S Kim, S Maeda, K Kim, (2007) Gender Disparity in Liver Cancer Due to Sex Differences in MyD88-Dependent IL-6 Production. Science 317(6): 121-124.
- Sabatino R, A Antonelli, S Battistelli, R Schwendener, M Magnani, et al. (2014) Macrophage depletion by free bisphosphonates and zoledronate-loaded red blood cells. Plos One 9(6).
- Buttari, B, E Profumo, R Rigano (2015) Crosstalk between Red Blood Cells and the Immune System and Its Impact on Atherosclerosis. Bio Med Res Int.
- Straface E, L Gambardella, A Mattatelli, E Canali, F Boccalini, et al. (2011) The Red Blood Cell as a Gender-Associated Biomarker in Metabolic Syndrome: A Pilot Study Int J Cell Biol p. 7.






























