Biochemical Aspects of Vestibular Stimulation in Neuroplasticity
Devi NP and Mukkadan JK*
Department of Physiology, Little Flower Medical Research Centre, India
Submission: March 05, 2018; Published: April 24, 2018
*Corresponding author: Mukkadan JK, Department of Physiology, Little Flower Medical Research Centre, Angamaly, Kerala, India, 683572, Email: drmukkadan@gmail.com
How to cite this article: Devi NP, Mukkadan JK. Biochemical Aspects of Vestibular Stimulation in Neuroplasticity. Anatomy Physiol Biochem Int J: 2018; 5(1): 555652. DOI: 10.19080/APBIJ.2018.05.555652.
Abstract
Objective:This article reviews the potential uses of vestibular stimulation to augment neuroplasticity and cognition in rats through suppressing the AChE activity.
Methods:Rotatory vestibular stimulation was provided in a rotatory vestibular stimulating apparatus at a rate of 50 rpm for 5 minutes, for 30 days for rats. 0.3mg/kg of physostigmine also administered to rats of another group as a standard drug. No rotatory vestibular stimulation or physostigmine is given to the control rats. Biochemical studies were done after vestibular stimulation.
Results:The activity of AChE decreased significantly in rats subjected to vestibular stimulation by rotation when compared with the control.
Conclusion: Rotatory vestibular stimulation enhances neuroplasticity via inhibiting acetyl cholinesterase activity in rats.
Keywords:Cognition; Neuroplasticity; Neurotransmitter
Abbrevations: LTP: Long Term Potentiation; RVS: Rotatory Vestibular Stimulated; AChE: Acetyl Cholinesterase; Acetyl CoA: Acetyl Coenzyme A; AD: Alzheimers Disease; HD: Hippocampal Cells; DTNB: 5,5’-Dithiobis-(2-Nitrobenzoic Acid)
Introduction
People endowed with a prodigious memory generally admit that they use various strategies to improve their cognition. But even if you’re not a memory genius, these common sense strategies can help you forget less often. Various neurological disorders like AD and dementia is related with synaptic loss. In a hypothetical point of view, subjects with AD are able to reacquire knowledge, and pharmacological modulation of acetylcholine, norepinephrine and dopamine can influence the maintenance and reformation of neuronal networks. Manipulation of certain neurotransmitters with vestibular stimulation therapy has the potential to promote the maintenance and restoration of the functional connectivity within the semantic networks that are the basis of neuroplasticity [1]. How the release of neurotransmitters occurs is briefly explained in Figure 1.
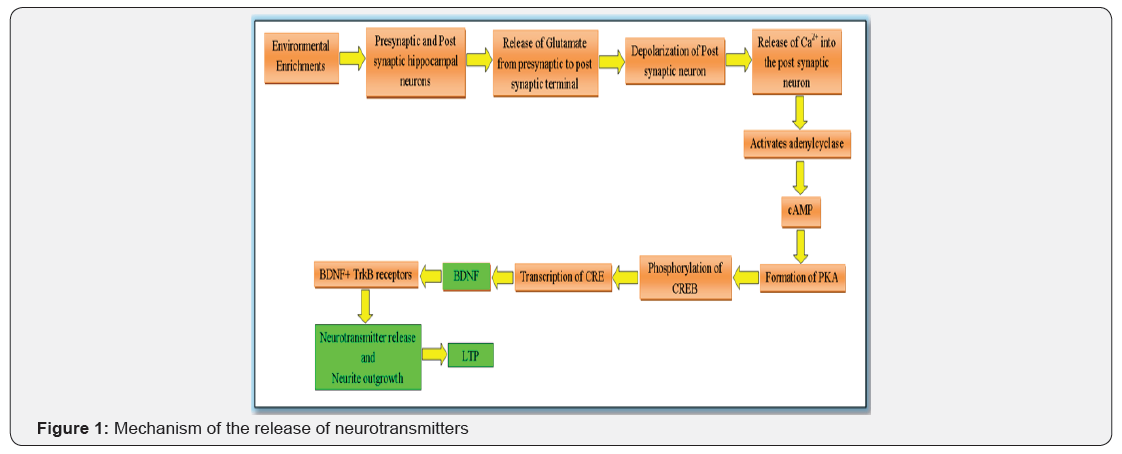
As an outcome of living in an enriched environment, the brain of animals undergoes molecular and morphological changes leading to improvements in learning and memory. These improvements associate well with intensification in neurogenesis, synaptic density, or neurotropic factors [2]. Neural plasticity is continuing in the brain during aging and also plastic changes occur in young and aged animals as a result of living in an enriched environment [3]. We highlight the results from our own laboratory on the dynamics of neurotransmitter release in the area of the brain confined to cognition. Specifically, we reviewed the effects of an enriched environment (Vestibular Stimulation) on the changes in neurons, dendrites and in the activity of AChE in hippocampus [1]. Acetylcholinesterase (AChE) or acetyl hydrolase, is the primary cholinesterase in the body with a very high catalytic activity. It is an enzyme catalyzes the breakdown of acetylcholine and of some other cholinesters that function as neurotransmitters.
AChE is found mainly at neuromuscular junctions and in chemical synapses of the cholinergic type, where its activity serves to terminate synaptic transmission. Each molecule of AChE degrades about 25000 molecules of acetylcholine (ACh) per second, approaching the limit allowed by diffusion of the substrate [4,5]. Throughout neurotransmission, ACh is released from the presynaptic neuron into the synaptic cleft and binds to ACh receptors on the post synaptic membrane, relaying the signal from the nerve. AChE, also located on the post synaptic membrane, terminates the signal transmission by hydrolyzing ACh. The released choline is taken up again by the pre-synaptic neuron and ACh is synthesized by combining with acetyl CoA through the action of choline acetyl transferase. A cholinomimetic drug disrupts this process by acting as a cholinergic neurotransmitter that is impervious to acetylcholinesterase’s lysing action [6,7].
Vestibular stimulation acts as an environmental enriching factor and it helps the increased release of neurotransmitters and also enhances the performance of neurotransmitter like acetylcholine by inhibiting the activity of AChE, as this is the enzyme which hydrolyze acetylcholine. Thereby it accelerates the transmission of action potential through the increased synaptic connections. The hippocampus is the major center for learning and memory and is furnished with neurogenesis throughout the life and firing of two types of hippocampal neurons (place cells and HD cells) crucial for spatial behavior [8]. Figure 2 shows the Hippocampal HD cells and Place cells.
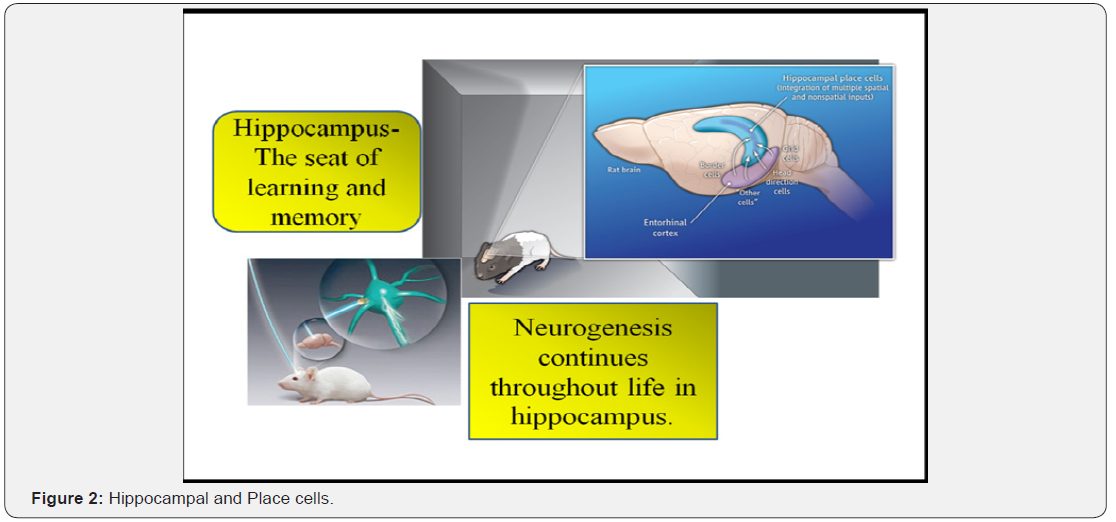
Neuroplasticity - brains power to modify in response to experience, practice or training
Learning a behavioral task or something complicated with excess physical activity causing the animal to shift away from homeostasis. This results in consumption of more energy leads to physiological stress. This in turn leads to the production of free radicals which will lead to oxidative stress. This can be harmful for proteins or DNA involved in dendritic growth. This will get reflected in learning and memory performance also. Enriched environments and soothing physical activities can be a good remedy for memory problems and also to enhance learning and memory via enhancing the production of the required neurotransmitters.
Materials and Methods
Animals used for the study
The present investigation was conducted in our laboratory using Male Wistar albino rats of 30 days old weighing about 120±30 gm.
Rotatory Vestibular Stimulating Apparatus
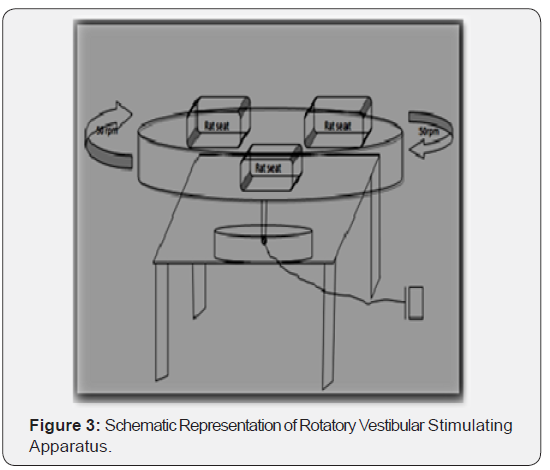
In our previous papers we have clearly explained about the vestibular stimulation apparatus which we designed and used in our lab [1]. Schematic Representation of Rotatory Vestibular Stimulating Apparatus is given in Figure 3.
Experimental Design
Rats were randomly divided into different groups comprising 18 rats. Physostigmine (Standard drug) and Rotatory Vestibular Stimulation were administered to group B and C respectively for 30 days before the beginning of the behavioral task and also 15 minutes prior to the start of acquisition phase as well as each retention test.
Group A: Control group (neither Vestibular Stimulation, nor the drug was administered).
Group B: Standard drug Physostigmine Treated Group (Rats of Group B were administered with the standard drug Physostigmine, 0.3mg/kg intraperitoneally)
Group C: Rotatory Vestibular Stimulated Group (RVS).
Experimental design for behavioral analysis and administration of rotatory vestibular stimulation and physostigmine
After 30 days of vestibular stimulation, the rats were subjected for Behavioral studies in 8 arm Radial arm Maze (Figure 4). The details of the behavioral experiments were also published in our earlier papers [1]. For the assessment of learning and memory the no. of trails taken for attaining the task were recorded and the we recorded the Long Term Potentiation (LTP), by subjecting the experimental rats for a retention test which includes various sessions [9]. Control rats (Group A) were under gone the same procedure of behavioral task without providing any drug or vestibular stimulation. The rats of Group B were administered with the drug physostigmine (0.3mg/kg, orally) for 30 days without any vestibular stimulation and kept as standard drug group [10,11].

Acetylcholinesterase activity
AChE activity was done by using UV Spectrophotometer by Ellman et al. [12] method. Immediately after last retention test, the rats were sacrificed and the hippocampus was dissected out in an ice cold 0.1M phosphate buffer saline (pH 7.2). The hippocampus (10mg/ml) was homogenized in ice cold 0.1M phosphate buffer saline (pH 7.2) using Teflon homogenizer. The homogenate was centrifuged at 5000 rpm for 10 min at 40C. The supernatant (0.4ml) mixed with 2.6ml of phosphate buffer (0.1M, pH 8) and 0.1ml of DTNB (0.01M). After that 0.1ml of acetylthiocholine iodide added, the absorbance was measured every one minute for 10 minutes at 412nm using Spectrophotometer.
Results
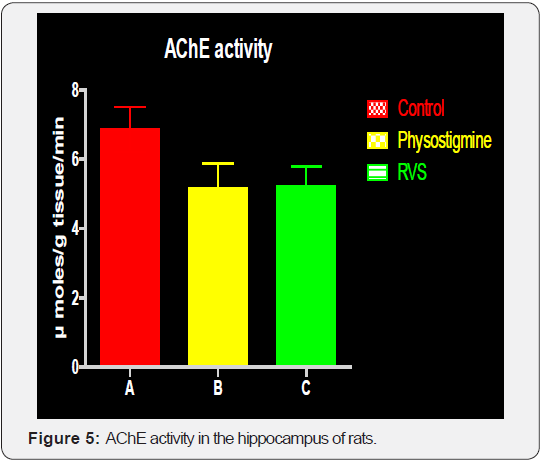
Rate of Acetyl cholinesterase of Group B (5.17±0.71) and C (5.24±0.55) is significantly decreased when compare with the Group A (6.87±0.65, p< 0.001). There is no significant difference between group B and C. From the result it is clear that rate of AChE activity is reduced in treated groups of rats and this in turn results in an improved learning and memory. Results shown in Figure 5.
Discussion
Rotatory Vestibular Stimulation enhances long term potentiation (LTP). Vestibular nuclei and hippocampus have anatomical connections thus improves cognition because of the firing of two types of hippocampal neuronal cells (place cells and HD cells) crucial for spatial behavior. Vestibular stimulation activates the hippocampal formation and increases neuronal activity and synaptic connections via increasing dendritic arborization also. The horizontal semicircular canals activated by rotation which leads to improved learning and memory by enhancing the bustle of hippocampus and also by increasing the level of acetylcholine indirectly by inhibiting acetylcholinesterase. This can be considered as the root of neural basis for the improved cognitive functions in the treated rats..
References
- Devi NP and Mukkadan JK (2017) Impact of rotatory Vestibular Stimulationin Memory Boosting, MOJ Anat and Physiol 4 (4): 00143.
- Gregorio Segovia, Alberto del Arco, Francisco Mora (2009) Environmental enrichment, prefrontal cortex, stress, and aging of the brain. J Neural Transm (Vienna) 116(8): 1007-1016.
- Francisco Mora, Gregorio Segovia, Alberto del Arco (2007) Aging, plasticity and environmental enrichment: Structural changes and neurotransmitter dynamics in several areas of the brain. Brain Research Reviews 55(1): 78-88.
- Quinn DM (1987) Acetylcholinesterase: enzyme structure, reaction dynamics, and virtual transition states. Chemical Reviews 87(5): 955- 979.
- Taylor P, Radic Z (1994) The cholinesterases: from genes to proteins. Annu Rev Pharmacol Toxicol 34: 281-320.
- Whittaker VP (1990) The Contribution of Drugs and Toxins to Understanding of Cholinergic Function, Trends Pharmacol Sci 11(1): 8-13.
- Purves D, Augustine GJ, Fitzpatrick D (2008) Neuroscience (4th edn), Sinauer Associates, Massachusetts, USA, pp. 121-122.
- Robert W Stackman, Ann S Clark, Jeffrey S Taube (2002) Hippocampal Spatial Representations Require Vestibular Input. Hippocampus 12(3): 291-303.
- Silviya R J (2010) Enhancement of memory in rats with Centella asiatica. Biomedical research 21(4): 429-432.
- Haroutunian V, Baenes E and Davis KL (1985) Cholinergic modulation of memory in rats. Psychopharmacology 87(3): 266-271.
- Haroutunian V, Baenes E, Davis KL (1985) Pharmacological alleviation of cholinergic lesion induced memory deficits in rats. Life Sci 37(10): 945-952.
- Ellman GL, Courtney KD, Andres V, Feather Stone RM (1961) A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol 7: 88-95.






























