The Preliminary Study on the Histopathological Effect of Aqueous Extract of Zingiber officinale on Lead Acetate Induced Toxicity of Adult Sprague- Dawley Rats.
Ajayi AJ 1,2, Adebajo AO1,2* and Isah KP1
1Department of Anatomy, College of Medicine, University of Lagos, Nigeria
2Department of Anatomy, Ladoke Akintola University of Technology, Nigeria
Submission: August 23, 2017; Published: September 11, 2017
*Corresponding author: Adebajo AO, Department of Anatomy, College of Medicine, University of Lagos, Lagos State, Nigeria, Email: adebajoseye@gmail.com
How to cite this article: Ajayi AJ, Adebajo AO, Isah KP. The Preliminary Study on the Histopathological Effect of Aqueous Extract of Zingiber officinale on Lead Acetate Induced Toxicity of Adult Sprague-Dawley Rats. 2017; 3(3): 555611. DOI:10.19080/APBIJ.2017.03.555611.
Abstract
Lead Acetate is one of the environmental pollutants that can threaten the life of living creatures in many ways. The use of plant derived herbal compounds in herbal preparations as alternative sources of medication has continued to play major roles in the general wellness of people all over the world Farombi 2003; Rajesh et al. [1], Ekor [2]. However the treatment with the extract of Zingiber officinale modulates the toxicity caused by lead acetate administration. The aim of this study is to investigate the ameliorative properties of ginger on lead acetate induced on kidney induced toxicity. A total of forty adult rats of weight 150 ± 20 g were used for this research.
The animals were grouped into four (A, B, C and D) of 10 animals each. Group A-C was the treatment groups while group D served as the control. Group a received 0.4 g of Lead Acetate, Group B received 0.4 g of Lead Acetate and then 0.1g of Ginger, Group C received 0.1 g of Ginger and Group D received 1 ml of distilled water. The mode of administration was oral and the study was for 25 days after a 2 week acclimatization period. At the end of the experiment, the animals were sacrificed using ketamine and an anterior abdominal incision was performed and the kidney was excised. The organs were fixed in 10% formal Saline and processed for histology. The histological findings revealed cytoarchitectural distortion in the podocytes, dilation in the capsular space and cells with pyknotic nuclei in the distal and proximal convoluted tubules in the Group A and to lesser extent in Group B compared to normal cyto architectural photomicrographs obtained in the Group C and D. These findings thus underlined the protective effect of Zingiber officinale in Lead acetate induced toxicity as it affects ultrafiltration as well as tubular fluid reabsorption properties of the kidney.
Keywords: Zingiber officinale; Lead Acetate; Sprague Dawley; Histology
Introduction
Man has persistently increased the toxicity suffered from heavy metal exposures due to increased environmental pollution. Generally, heavy metals produce their toxicity by forming complexes or ligands with organic compounds thereby affecting the function of biological molecules, inactivate some biochemical enzymes and affect protein structures. Pirkle et al. [3]. Lead (Pb) is a dangerous heavy metal and harmful even in small amounts. Nevertheless, humans get exposed to lead through their environment and diet Gidlow [4]. Lead has no known function in the body as it has diverse and deleterious effects on man and animal health Suradkar et al. [5].
The effects of Lead toxicity are systemic and multiple organs are involved. It damages the cellular material and alters cellular genetics and produces oxidative damage leading to increased production of free radical and decrease availability of antioxidant reserves to respond to the resultant damage Sujatha and Srilatha [6]. It also affects the hematopoietic system where it causes anemia Sohler et al. [7]; Papionnou et al [8] ; Anetor [9], in cardiovascular system causes hypertension Landis and Flegal [10]; Schwarts [11], in nervous system it causes neurotoxicity Shannon [12] including cognitive deficits Weisskopf et al. [13], in the gastrointestinal system it causes gastric ulcer Olaleye et al. [14] and in renal system causes renal damage Kimber et al. [15], in the hepatic system it causes hepatitis and hepatic failure Beattie et al. [16]. Lead induces wide range of physiological, biochemical and behavioral dysfunction Suradkar et al. [5].
Lead poisoning is, and for centuries has been, one of the most significant preventable causes of neurological morbidity from an environmental toxin. The manifestation of its poisoning in humans are nonspecific as it can include weight loss, anemia, Waldron [17]; Khalil-Manesh et al. [18] memory loss, Hopkins [19] nephropathy, infertility, liver, testis and heart damages Patocka et al [20]; Gurer-Orhan et al [21]. Sources of lead exposure may include air, water supplies, food, soil and industry such as lead-based paint, leaded gasoline, battery manufacture and reclamation, pottery/ceramics and eye cosmetics Mage. The toxic effects of lead are treated by chelating therapy which also depletes the body store of essential cations Ruff et al [22], therefore there is need to look for an alternative solution to lead poisoning.
Today, it is estimated that about 80% of the world population rely on medicinal plants as they play important roles in pharmacology and medicine for many years to meet health needs Ogbera et al [23]. Ginger (Zingiber officinale) which belongs to the family Zingiberaceae is an example of plants that is rapidly gaining popularity among modern physicians and its underground rhizomes are medicinally useful part Mascolo et al. [24]. Zingiber officinale is a strong anti-oxidant substance and may either mitigate or prevent generation of free radicals. The pharmacological actions of ginger and compounds isolated include immuno-modulatory, antitumorigenic, antiinflammatory, anti-apoptotic, anti hyperglycemic, anti-lipidemic actions Amir and Arash [25]. Among the pharmacological effects demonstrated are anti-platelet, anti-oxidant, anti-tumour, antirhinoviral, anti-hepatotoxicity and anti-arthritic effect Fisher et al; Sharma et al; Kamtchouing et al [26]. Zingiber officinale was found to have hypocholesterolaemic effect and cause decrease in body weight, glucose in blood, serum total cholesterol and serum alkaline phosphatase in adult male rats Bhandari et al. [27].
Ginger extracts have been extensively studied for a broad range of biological activities, especially antioxidant activities Miller et al ; Ahmed et al. [28] which showed that Ginger significantly lowered lipid per-oxidation by maintaining the activities of the antioxidant enzymes such as super oxide dismutase, catalase and glutathione peroxides in rats. For decades, the cellular effects of Lead Acetate and Ginger have been discussed extensively but the effect on the cyto-architecture of the kidney has not studied when administered simultaneously. Lead acetate has a deleterious effect on the organs and systems in the body but the level of its damage when administered with ginger on the kidney still need to be further investigated, hence the birth of this study.
Materials and Methods
Plant Materials
The plant Zingiber officinale was purchased from General market in Ogbomosho, Oyo State, Nigeria, they were identified and authenticated in the Department of Botany, Faculty of Pure and Applied Science, Ladoke Akintola University ofTechnology, Ogbomosho, Oyo State, Nigeria.
Preparation of Extract
The aqueous extract was prepared using cold extraction techniques. Forty grams of ginger powder were placed in 160 ml of sterile distilled water and left at room temperature for 24 hours with continuous mixing using magnetic stirrer. Then mixture was filtered and after filtration it was dried using incubator at 40°C. The liquid has evaporated, and the precipitated extract was left at the base of the baker. Five mls of distilled water was added to 1 g of this extract powder to produce 20% (w/v) as standard stock solution.
Experimental Animals
Animals
Forty healthy Sprague-Dawley rats between 150 ± 20 g were used for this study. They were housed in well standard ventilated wire mesh plastic cages in the animal house of the Department of Anatomy, Faculty of Basic Medical Sciences of Ladoke Akintola University of Technology under standard room temperature ranging between 26oC-28oC; relative humidity 50-55% and were exposed to twelve hours light and twelve hours dark cycle. They were allowed unrestricted access to water and commercial rat chow ad libitum. They were left to acclimatize for a period of two weeks before the commencement of the experiment. The animals were identified by different ear tags. All experimental procedures and techniques were approved by the departmental committee on the use and care of animals and tissue collection. The weights of the animals were taken every week.
Experimental Design
- Group A: received oral dose of 0.4 g of Lead Acetate daily
- Group A: received oral dose of 0.4 g of Lead Acetate daily
- Group C: received oral dose of 0.1 g of Zingiber officinale daily
- Group C: received oral dose of 0.1 g of Zingiber officinale daily
At the end of the experiment of 25 days, the animals were sacrificed using ketamine and an anterior abdominal incision was performed and the kidneys were excised for histology.
Histological Procedures
The excised organs were fixed in 10% formal saline for 72 hours. The tissues were processed for microscopic examination using a standard protocol and 5 μm thick paraffin sections were made. Slides were stained with routine haematoxylin and eosin stains and photomicrographs were made at a magnification of 100 and 400 using Olympus and Leica microscopes.
Statistical Analysis
The data obtained were analysed statistically by students’ T-test and one-way Analysis of Variance (ANOVA) test. The level of significance was at P<0.05. The data were expressed as mean ± SEM.
Results
No mortality was observed during the duration of the experimentation. All animals in the groups appeared healthy showed normal behaviour throughout the study.
Effect On Body Weight
At the end of the study, there was decrease in body weight of the treatment groups (A and B) when compared to the Control, there was increase in the group C that was administered with extract alone (Table 1).
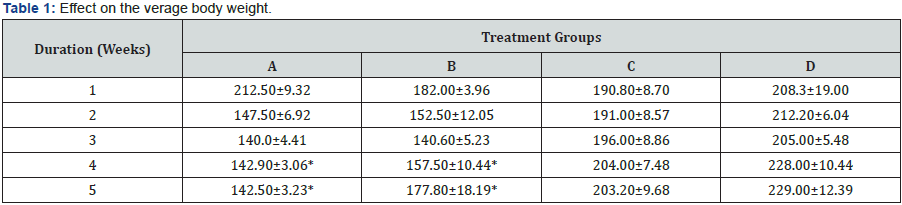
Values are mean ± standard error of mean; n=10,*p<0.05 compared to control (student’s test)
Effect On Kidney Histology
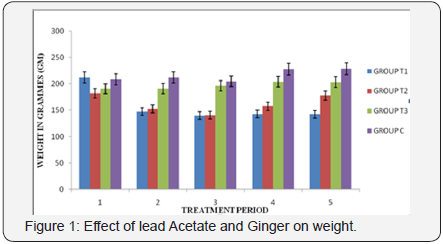
The section from Group A treated rat showed an increased luminar diameter of the renal tubules particularly the proximal convoluted tubule. The morphological features appeared to be distorted with dilation in the capsular space and cytoplasmic vacuolated cells with pyknotic nuclei were found. The glomeruli also showed adhesive glomerulitis, with damage varying from single adhesions to complete obliteration of the capsular space (plate B). The section from group B demonstrates marked reduction of the histological features of renal injury, consisting of more focal and mild tubular necrosis. Also there is attenuation of the histological lesions observed in this section compared with A(plate C). Photomicrograph of Group C showing normal morphology of the renal corpuscle, proximal tubules (P), the capsular space (CS) and the glomeruli (G) were seen to be prominent and morphologically normal (plate D). The photomicrograph from control (Group D) rat showed a normal renal corpuscle. The bowman capsule and the glomeruli appeared to be prominent and normal. The podocytes and luminar diameter were found to be normal, with no distortion and there were no cytoplasmic vacuolations (Figure 1).
Section of Group A showing an increased luminar diameter of the renal tubules particularly the proximal convoluted, dilation in the capsular space and vacuolation with pyknotic nuclei were found (H&E, X 400) (Figure 2).
At GD21, the lung of control group showed the formation of secondary crests and appearance of air saccules. They were separated by intersaccular septa and lined with flat epithelium (Figure 7). The PCNA immunohistochemical staining showed mild positive immunoreactions in the bronchiolar epithelium and the adjacent stroma (Figure 8).
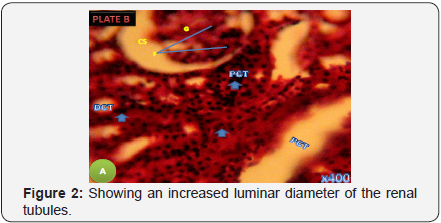
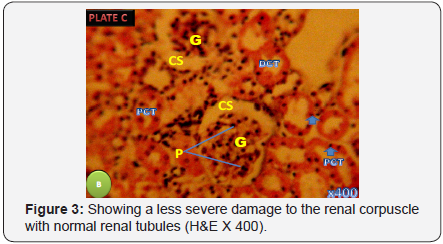
Photomicrograph of the longitudinal section of the kidney in the treatment Group B Showing a less severe damage to the renal corpuscle with normal renal tubules (H&E X 400) (Figure 3).
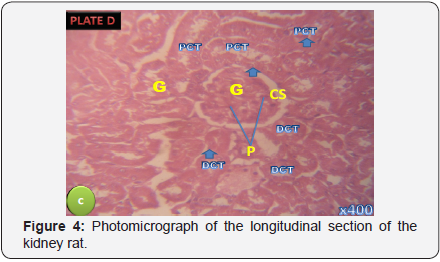
Photomicrograph of the longitudinal section of the kidney rat in treatment Group C presenting the normal morphology of the renal corpuscle. Proximal tubules (P), the capsular space (CS) and the glomeruli (G) were seen to be prominent and morphologically normal (H&E X 400) (Figure 4).
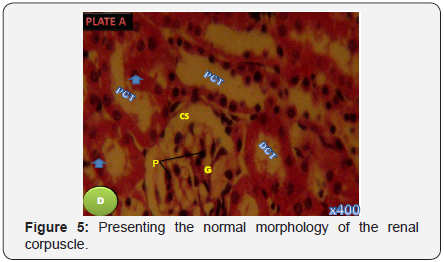
Section of Group D group presenting the normal morphology of the renal corpuscle. Proximal tubules (P), the capsular space (CS) and the glomeruli (G) were seen to be prominent and morphologically normal (H&E, X 400) (Figure 5).
Discussion
The study demonstrates the adverse effect of Lead Acetate on the kidney of the Sprague-Dawley rats. The morphometric result showed a significant decrease in the body weight of the rats in the treatment Group A receiving lead acetate which agrees with Harvey et al. [29] that the significant decrease in weight is as a result of loss of appetite and gastrointestinal disturbances via lead effects on the satiety set-up studies Pentenusci, [30]; Hamilton [31]; Djebli et al. [32]. Also, there was decrease in the body weight of treatment Group B that received Lead Acetate and the extract of Zingiber officinale, although, it was quite better than the group A, which can be as a result of the extract when compared with Group C that had increase in body weight.
Histological examination of the kidney of the control group revealed entirely normal structures of the renal cortex which comprised renal corpuscles, proximal and distal convoluted tubule, which quite similar to section of Group C that was administered with the extract of Zingiber officinale alone. However, the Group A that was administered with Lead Acetate alone showed a complete deviation from the control, the section showed dilation in the capsular space and cytoplasmic vacuolated cells with pyknotic nuclei. Also there was an increased luminar diameter of the renal tubules particularly the proximal convoluted tubule as compared with renal tubules in group D. The glomeruli also showed adhesive glomerulitis, with damage varying from single adhesions to complete obliteration of the capsular space. The less severe damage of the renal corpuscle with normal renal tubules observed in the photomicrograph of the section of Group B that received both Lead Acetate and the extract of Zingiber officinale show an improvement when compared to Group A. This shows the ameliorative property of the extract on the Lead Acetate toxicity which agrees with the antioxidant effects as documented by Jagetia et al. [33] ; Haksar et al. [34,35].
Conclusion
In conclusion, the present body showed that the extract of Zingiber officinale possess protective potentials on Lead Acetate induced renal toxicity. This study therefore suggests that the extract of Zingiber officinale may be a useful preventive agent against the adverse effect of Lead Acetate and other heavy metals that possess threat to the kidneys.
References
- Rajesh P, Latha S, Selvamani P, Rajesh Kannan V (2010) Phytochemical Screening and Toxicity Studies on the Leaves of Capparis sepiaria Linn (Capparidaceae). J b clin pharm 1(1): 41-46.
- Ekor M (2013) The growing use of herbal medicines: issues relating to adverse reactions and challenges in monitoring safety. Front Pharmacol 4: 177.
- Pirkle JL, Schwartz J, Harlan WR (1985) Relationship between blood lead level and blood pressure and its cardiovascular risk implications. Am J Epidemol 121(2): 246-258.
- Gidlow D (2004) Lead Toxicity. Occup Med (Lond) 54(2): 76-81.
- Suradkar SG, Vihol PD, Patel JH, Ghodasara DJ, Joshi BP et al. (2010) Patho-morphological changes in tissues of Wistar rats by exposure of Lead acetate. Veterinary World 3(2): 82-84.
- Sujatha K, Srilatha C, Anjaneyulu Y, Amaravathi P (2011) Lead Acetate Induced Nephrotoxicity in Wister Albino Rats. A pathological immunohistochemical and Ultra-structural Studies. International Journal of Pharma and Biosciences 2: 2-11.
- Sohler A, Krues M, Pfeiffer CC (1977) Blood lead level in psychiatric outpatients reduced by vitamin C. J Othomole. Psychiatr 6: 272-276.
- Papanouannou R, Sholer A, Pfafer C (1978) Reduction of blood lead level in battery works by zinc andvitamin C. J Othomole Psychiatr 7: 94-106.
- Anetor JI (2002) Observation of Hematopoietic system in tropical lead poisoning. Nig J of Physiol Science 17(1-2): 9-15.
- Landis JR, Flegal KM (1988) A generalized mantel-Haenszel analysis of the regression of blood pressure on blood lead using NHANES II data. Environ Health Prosp 78: 35-41.
- Schwartz J (1991) Lead, blood pressure and cardiovascular disease in men and women. Environmental Health Perspectives 91: 71-75.
- Shannon M, Graef J, Love joy FH (1988) Efficacy and toxicity of d-penicillamine in low-level lead poisoning. J Pediatr 112(5): 799-804.
- Weisskopf MG, Hu H, Mulkern RV, White R, Aro A, et al. (2004) Cognitive deficits and magnetic resonance spectroscopy in adult monozygotic twins with lead poisoning. Environ Health Perspect 112(5): 620-625.
- Olaleye SB, Omotosho IO, Adewoye EO, Olaifa AK, Onwuka SK, et al. (2002) Nig J of Physiol Science 17(1-2): 73.
- Kimber I, Stoard MD, Cerilow DA (1986) Influence of chronic lowlevel exposure to lead on plasma immunoglobulin concentration and cellular immune function in man. Int’l. Arch Occup Environ Health 57(2): 117-125.
- Beattie AD, Mullin PJ, Baxter Rl, Moore MR (1979) acute lead poisoning: an unusual cause of hepatitis. Scott Med J 24(4): 318-332.
- Waldron HA (1966) The Anaemia of Lead Poisoning: A Review. Brit J Ind Med 23(2): 83-100.
- Khalil-Manesh F, Tartaglia-Erler J, Gonick HC (1994) Experimental model of lead nephropathy. Correlation between renal functional changes and hematological indices of lead toxicity. J Trace Elem Electrolytes Health Dis 8(1): 13-19.
- Hopkins A (1970) Experimental lead poisoning in the baboon. Brit J Ind Med 27(2): 130-140.
- Patocka J, Cerný K (2003) Inorganic lead toxicology. Acta Medica (Hradec Kralove) 46(2): 65-72.
- Gurer Orhan H, Sabir HU, Ozgunes H (2004) Correlation between clinical indicators of lead poisoning and oxidative stress parameters in control and lead-exposed workers. Toxicology 195: 147-154.
- Ruff HA, Markowitz ME, Bijur PE (1996) Relationship. Environ Health perspect 104(2): 180-185
- Ogbera AO, Dada O, Adeleye F, Jewo PI (2010) Complementary and alternative medicine use in diabetes mellitus. West African journal of medicine 29(3): 158-162.
- Mascolo N, Jain R, Tain SC, Capasso FJ (1989) Ethnopharmacologic investigation of ginger (Zingiber officinale). J Ethno pharmacol 27(1- 2): 129-140.
- Amir AK, Arash K (2010) Antioxidant effect of ginger to prevents lead-induced liver tissue apoptosis in rat. Journal of medicinal plants research 4(14): 1492-1495.
- Kamtchouing P, Mbongue GYF, Dimo T, Watcho P, Jatsa HB, et al. (2002) Effects of Aframomum melegueta and Piper guineense on sexual behaviour of male rat. Behav Pharmacol 13(3): 243-247.
- Bhandari N, Mazumder S, Bahl R, Martines J, Black RE, et al. (2004) An educational intervention to promote appropriate complementary feeding practices and physical growth in infants and young children in rural Haryana, India, The Journal of Nutrition 134(9): 2342-2348.
- Ahmed F, Wyckoff J, Lin E, Wang W, Wang Y, et al. (2002) GFP expression in the mammary gland for imaging of mammary tumor cells in transgenic mice Cancer. Res 62(24): 7166-7169.
- Harvey SC (1970) Heavy metals. In: Goodman, LS and Gilman A (Eds). The Pharmaceutical Basis of Therapeutics, London.
- Pentenusci SO, Favaretto AL, Lopes RA, Azoubek R (1988) Functional disturbance of rat sexual accessory glands in an early phase of lead intoxication. Gegenbaus morphol Jahrb 134(5): 791-798.
- Hamilton JD, O Flaherty EJ (1994) Effects of lead exposure on skeletal development in rats. Fundam Appl Toxicol 22(4): 594-604.
- Djebli N, Slimani M, Aoues A (2004) Effect of lead exposure on dopaminergic transmission in the rat brain. Toxicol 207(3): 363-368.
- Jagetia GC, Baliga MS, Venkatesh P, Ulloor JN (2003) Influence of ginger rhizome (Zingiber officinale Rosc.) on survival, glutathione and lipid peroxidation in mice after whole-body exposure to gamma radiation. Radiation Research 160(5): 584-592.
- Haksar A, Sharma A, Chawla R, Kumar R, Arora R, et al. (2006) Zingiber officinale exhibits behavioral radioprotection against radiation. Pharmacology Biochemistry Behaviour (84): 179-188.
- Weisskoff MG, Howard M, Mulkern RV, White R, Olivera S, et al. (2004) Cognitive deficits and magnetic resonance spectroscopy in adult monozygotic twin with lead poisoning. Environ health perspect 112(5): 620-625.






























