Effect of Gestational Exposure to Monosodium Glutamate on the Structure of Fetal Rat Lung
Abdelmonem Awad Hegazy1*, Ibrahim Hassan Ibrahim2, Rasha Mohammed Sabry3 and Eman Sabry Abass4
Department of Anatomy and Embryology, Zagazig University, Egypt
Submission: July 28, 2017; Published: August 10, 2017
*Corresponding author: Abdelmonem Awad Hegazy, Anatomy and Embryology Department, Faculty of Medicine, Zagazig University, Zagazig 44519, Egypt, Tel: +201110504321; E-mail: dr.abdelmonemgazy@yahoo.com
How to cite this article: Hegazy A A, Ibrahim H I, Sabry R M, Abass E S. Effect of Gestational Exposure to Monosodium Glutamate on the Structure of Fetal Rat Lung. Anatomy Physiol Biochem Int J. 2017; 3(2): 555609. DOI:10.19080/APBIJ.2017.03.555609.
Abstract
The aim of this work was to elucidate the possible developmental changes in the offspring lungs induced by administration of monosodium glutamate (MSG) to the pregnant albino rats. Forty pregnant albino rats were used in this study, equally divided into two groups. The first group was the control one, administered distilled water by gastric tube; the second was instead given MSG dissolved in distilled water. The animals were sacrificed at different four days of gestations; 14th (GD14), 16th (GD16), 19th (GD19) and 21st (GD21). Fetal lung sections were stained with Haematoxyline and Eosin (H&E) and with proliferating cell nuclear antigen (PCNA) reactions. The obtained data were statistically analyzed. At GD14, the bronchial tubes started to branch to form small simple primitive bronchiolar tubules. They were lined with pseudo stratified epithelium. At GD16, the bronchial tubes were lined with columnar epithelium with cytoplasmic vacuolization giving a glandular appearance. At GD19, the lining of tubular air spaces was cuboidal epithelium. GD21 showed the formation of secondary crests and appearance of air saccules lined with flat epithelium. Treated animals showed the epithelium of these tubules with desquamation and necrotic cells. Also, there was marked growth retardation in comparison to the control one. The PCNA immune histochemical staining showed high positive immunoreactions in the bronchiolar epithelium and the adjacent stroma. This is more evident in the treated subgroups than in the control ones. It has been concluded that administration of MSG to mothers during pregnancy may exert some growth retardation and histopathological changes in the fetal lungs.
Keywords: Lung Development, Rat, Monosodium glutamate, PCNA.
Introduction
Lung diseases represent one of the leading causes of worldwide morbidity and mortality. The problem is expected to be worse in the future [1]. This necessitates investigating the possible environmental factors, habits and food types that might be engaged in pathogenesis of pulmonary diseases, particularly during the fetal life. This of particular importance as many drugs and substances taken by (or given to) the pregnant mothers can cross the placental barrier to affect their fetuses [2]. One of such substances is monosodium glutamate (MSG) that might affect the lungs of fetal mice after exposure of their mothers to it [3]. Various developmental changes in the animals’ offspring tissues might occur following MSG treatment [4]. Moreover, the administration of MSG in high concentrations for long period of time triggers the production of reactive oxygen species (ROS) coupled with impaired oxidant/ antioxidant balance leading to a state of oxidative stress [5]. The lung is more susceptible to oxidative injury than any other organ in the body because of constant exposure to air that might contain toxic particles or oxidant gases such as nitrogen oxide or ozone [6].
MSG is a well-known flavor enhancer used by both Western and Eastern populations. It is responsible for the delicious taste; and it is incorporated into a large number of solid and liquid foods. Experimental studies have established that the added MSG in foods influences palatability and preference [7]. Nowadays, MSG is commonly sold in the Egyptian supermarkets. Many Egyptians consume MSG in both ready and home-made foods, almost daily [8].
MSG contains glutamic acid (78%), sodium (22%) and water [9]. Glutamate receptors are present in the central nervous system as the major mediators of excitatory neurotransmission. Neural injury associated with trauma, stroke, epilepsy and many neurodegenerative diseases might be mediated by excessive activation of such receptors. Several subtypes of glutamate receptors are demonstrated in the rat and monkey heart, with preferential distribution within the conducting system. The receptors are also present in the kidney, liver, lung, spleen and testis [10]. Juvenile obesity, reduced general activity and learning disability have been reported in rat and mouse offspring that received MSG through their mothers during pregnancy [11]. Moreover glutamate in high doses produces neuroendocrine abnormalities and neuronal degeneration [12]. It also might be engaged in oxidative damage of many organs such as rat thymus and kidney [13-15].
Many questions have been raised about the role of MSG in provocation of asthma. It has been observed that MSG induces signs and symptoms of asthma [16]. Previous studies reported cases of asthma following MSG ingestion through food. The reaction to MSG was noticed to be a dose dependent; and it might be delayed up to 12 hours, making its recognition difficult for both patient and physician [17,18]. PCNA is a 36 kd protein involved in protein synthesis; and can be considered as an early indication of deviations to normal functioning [19].
The aim of this work was to elucidate the changes that might take place in the developing lung after oral administration of MSG to the pregnant mothers of the albino rats using routine histological and PCNA immunohistochemical investigations.
Materials and Methods
Animals
This study was carried out on forty adult female and twenty adult male albino rats of 200-250 gm body weight (BW). They were reared in the animal house, Faculty of Medicine, Zagazig University. The experiment was performed according to the norms of the ethical committee of the University.
Adult females were caged with adult males for mating, at ratio of 2:1 respectively in the cage. Vaginal smear was done for each female rat at the morning. The day in which the smear was sperm positive was considered as the zero day of pregnancy. The females with negative vaginal smear were isolated and re-kept again with males. The process was repeated until all females became pregnant.
Pregnant females were equally divided into two groups; each contains 20 animals. The first group was the control one, administered distilled water by gastric tube; the second was instead given MSG in a dose of 2mg/kg BW, dissolved in distilled water. The animals were sacrificed using ether inhalation, at different four days of gestations; 14th (GD14), 16th (GD16), 19th (GD19) and 21 (GD21).
Tissue Specimens
Their embryos were extracted; and lung tissue specimens were retrieved and processed for light microscopic examination [20]. Sections were stained with H&E for study of the histological changes; and with PCNA for immunohistochemical examination.
Morphometric and Statistical Analysis
The data were obtained using Leica Qwin 500 image analyzer computer system (England) at the Histology Department, Qasr Eleini Faculty of Medicine. The image analyzer consisted of colored camera, monitor and hard disc of IBM personal computer connected to the microscope, and controlled by Leica Qwin 500 software. The image analyzer was first calibrated automatically to convert the measurement units (pixels) produced by the image analyzer program into actual micrometer units.
The optical density of immunoreactions of PCNA positive cells was measured in intersaccular and interstitial cells. The optical density was expressed in the form of maximum, minimum grey as well as sum of grey and mean grey parameters. Data obtained about the optical density of PCNA in the epithelial and interstitial cells for the control and treated groups were analyzed statistically using ANOVA Test. The statistical analysis was done using SPSS software (version 19.0; SPSS, Chicago, IL). The p ≤ 0.05 was considered to indicate a statistically significant difference.
Results
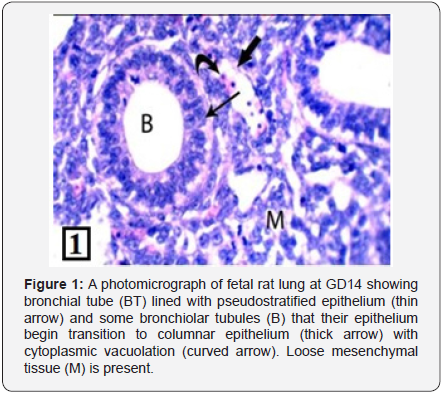
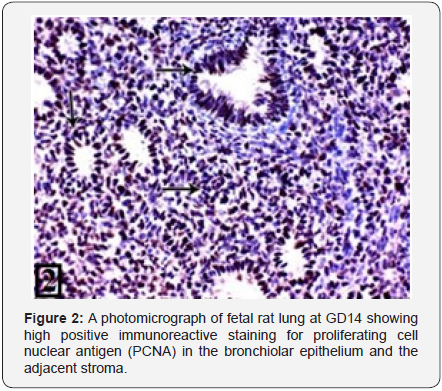

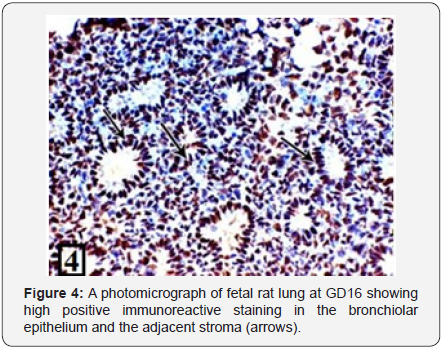
Light microscopic examination of the rat lung of control group at GD14 showed the bronchial tubes start to branch forming simple primitive bronchiolar tubules. They were lined with pseudostratified epithelium (Figure 1). The PCNA immunohistochemical staining showed high positive immunoreactions in the bronchiolar epithelium and the adjacent stroma (Figure 2). The rat lung of control group at GD16 showed that the bronchial tubes were lined with columnar epithelium with cytoplasmic vacuolization giving them the glandular appearance (Figure 3). The PCNA immunohistochemical staining showed high positive immunoreactions in the bronchiolar epithelium and the adjacent stroma (Figure 4).
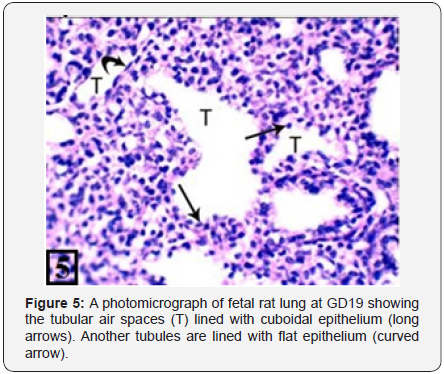
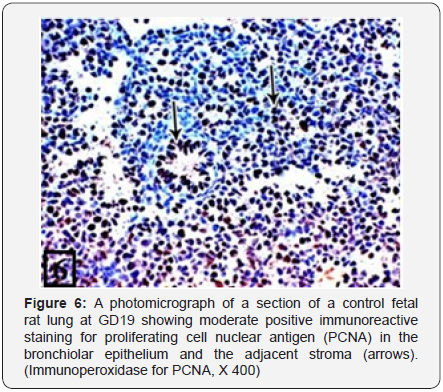
The rat lung of control group at GD19 showed the appearance of the tubular air spaces lined with cuboidal epithelium (Figure 5). The PCNA immunohistochemical staining showed moderate positive immunoreactions in the bronchiolar epithelium and the adjacent stroma (Figure 6).

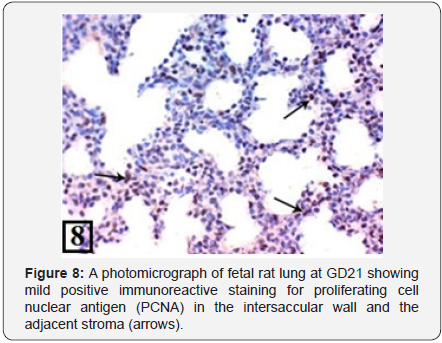
At GD21, the lung of control group showed the formation of secondary crests and appearance of air saccules. They were separated by intersaccular septa and lined with flat epithelium (Figure 7). The PCNA immunohistochemical staining showed mild positive immunoreactions in the bronchiolar epithelium and the adjacent stroma (Figure 8).
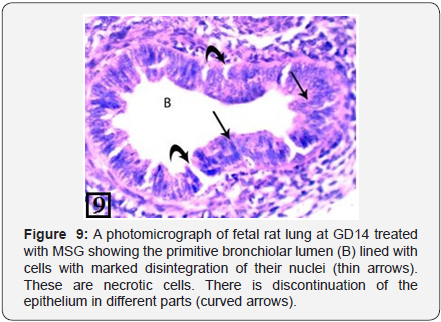

At GD14, the lung of treated group showed simple primitive bronchiolar tubules; and the epithelium of these tubules showed desquamation and necrotic cells (Figure 9). The PCNA immunohistochemical staining showed high positive immunoreactions in the bronchiolar epithelium and the adjacent stroma (Figure 10).
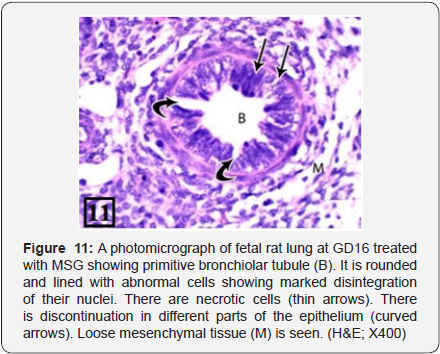

The rat lung of treated group at GD16 showed bronchiolar tubules lined with pseudostratified epithelium with abnormal cells showing marked disintegration of their nuclei. There were necrotic cells. There was discontinuation in different parts of the epithelium; and picnotic dark stained nuclei were seen (Figure 11). The PCNA immunohistochemical staining showed high positive immunoreactions (higher than the previous group) in the bronchiolar epithelium and the adjacent stroma (Figure 12).
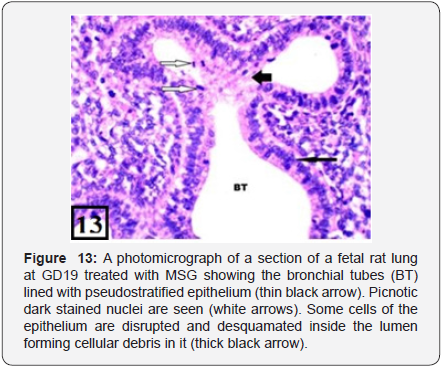
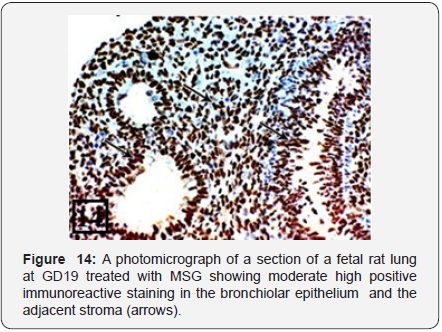
The lung of treated group at GD19 showed marked growth retardation with delayed development of the lung in comparison with the control one. Only bronchial tubes and small number of the primitive bronchiolar tubules were developed and lined with pseudostratified epithelium with marked desquamation to their epithelium (Figure 13). The PCNA immunohistochemical staining showed high positive immunoreactions (higher than the previous group) in the bronchiolar epithelium and the adjacent stroma (Figure 14).
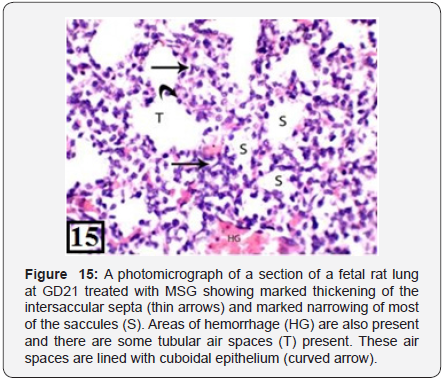
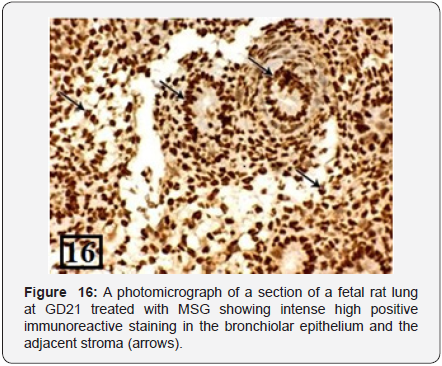
The lung of treated group at GD21 showed marked congestion and dilatation of the blood vessels and areas of hemorrhage were seen. There was marked thickening of the intersaccular septa and narrowing of most of the saccules. These saccules were lined with cuboidal epithelium not flat as the control group of the same age and no secondary crests and no capillaries were seen in comparison with the control group of the same age (Figure 15). The PCNA immunohistochemical staining showed intense high positive immunoreactions (higher than the previous group) in the bronchiolar epithelium and the adjacent stroma (Figure 16).
Morphometrical and Statistical Analysis
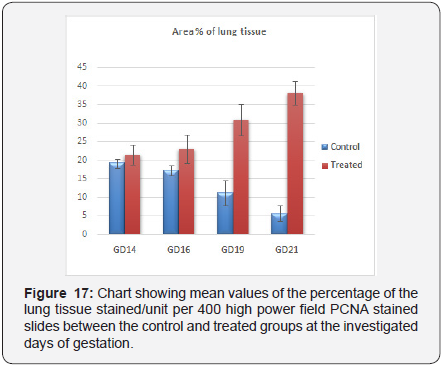
There was marked increase in the PCNA positivity in all treated subgroups with the increase at the rat embryo age. It reached to the highest positivity at GD21. The percentage of tissues stained per 400 high power field in PCNA stained slides between the control and treated subgroups showed highly statistical significant differences at the GD19 and the GD21 (p <0.001) and there was statistically significant difference at the GD16 (p <0.01), but that there was no statistical difference between the control and treated subgroups at the GD14 in area % of lung tissue (p =0.11) (Table 1); (Figure 17).
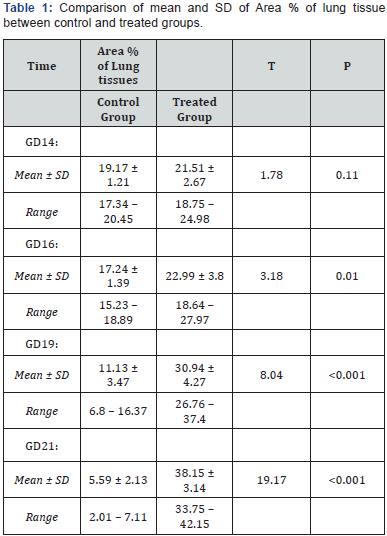
SD: Standard Deviation; T: ANOVA Test.
Discussion
In this work, albino rat was used as an experimental model as it is easy to be bred, operated and injected. Moreover, the animal model makes it possible to follow the changes from initial diagnosis to the first sign of organ impairment [21].
In the present study, the fetal rat lung treated with MSG at GD14 showed a decrease in the branching of the bronchial tubes into simple primitive bronchiolar tubules in comparison with the control group of the same age; and they were also situated within abundant loose mesenchyme. The bronchial tube was lined with pseudostratified epithelium, but some cells showed cytoplasmic vacuolation. Also, there were some cells desquamated inside the bronchial lumen forming cellular debris. These findings are in agreement with Lieberthal and Levine [22] who attributed the vacuolation of the cytoplasm to the cellular necrosis that causes impaired membrane transport activity and results in influx of sodium and water into the cell. This results in cell swelling, a characteristic early marker of necrosis, as well as loss of functional and structural integrity of the plasma membrane.
In the present study, the histological examination of the fetal rat lung treated with MSG at GD16 showed in comparison with the control group of the same age a decrease in the progression of the bronchial tubes into primitive bronchiolar tubules. The bronchial tube was lined with pseudostratified epithelium in some parts, but there were some epithelial cells contained deeply stained nuclei with no cytoplasmic vacuolation which is characteristic to this stage of development as in the control group. There were some epithelial cells that were markedly desquamated inside the bronchial lumen. This delay in the lung development could be a result of MSG given to the rat mothers during pregnancy. The cause of growth suppression after MSG treatment is thought to be brought about by the impaired production of growth hormone releasing factor, which accompanies the necrosis of nerve cells in the arcuate nucleus [23].
Another bronchial tube showed that its epithelium was consisted of abnormal cells showing marked disintegration of their nuclei (necrotic cells) and picnotic dark stained nuclei were seen (a marker for apoptosis). Pavlovic et al. [13] said that one of the mechanisms involved in MSG-induced apoptosis was the down-regulation of Bcl-2 protein expression. MSG-induced apoptosis and altered level of Bcl-2 protein are also related with oxidative stress. Namely, the treatment of animals with MSG resulted in an increase in the oxidative stress within the kidneys, liver, brain and lung; and this presented a possible mechanism of cell toxicity.
The fetal rat lung treated with MSG at GD19 showed a retardation of development of the lung in comparison with the control one. Only bronchial tubes and small number of the primitive bronchiolar tubules were developed among abundant loose mesenchymal tissue. No branching of the bronchiolar tubules occurred and no tubular air spaces were developed as in the control group of the same age. This delay in the lung development is in general agreement with Millard et al. [23]. The bronchial tube was lined with pseudostratified epithelium and picnotic dark stained nuclei were seen (apoptosis). Some cells of the epithelium were disrupted and desquamated inside the lumen forming cellular debris. This is in agreement with Pavlovic et al. [13].
The primitive bronchiole in this group was lined with pseudostratified epithelium not cuboidal epithelium as the control group of the same age; and there were necrotic cells showing disintegration of their nuclei in the lining epithelium and discontinuation of the epithelium was seen. So, the lung of this group was still in the pseudoglandular stage not the canalicular stage as the control group of the same age. Also, Hildeman et al. [24]. Mentioned that the excessive generation of reactive oxygen species (ROS) in the cells is known to damage DNA, lipids and proteins. Lipid peroxidation in cellular membranes damages the polyunsaturated fatty acids especially in lymphoid cells, and sensitizes T cells to apoptosis by decreasing the expression of Bcl-2 protein.
In the present study, the histological examination by H&E of the fetal rat lung treated with MSG at GD21 showed marked congestion and dilatation of the blood vessels and they were lined with a layer of flat endothelial cells. Many areas of hemorrhage were also present. Oda et al. [25] (said that this hemorrhage indicates separation of endothelial cell junction. However, Martin said that this hemorrhage is due to increase in the number of neutrophils in the blood capillaries that damage the epithelium by secreting many injurious substances such as oxygen derived free radicals and lysosomal enzymes [26]. Also, there was marked edema around the congested blood vessels and in the interstitium. Doohan [27] reported that the thickening and expansion of the interstitium was due to the interstitial edema resulting from histamine release from the immune cells which led to increase the infiltration rate and protein leak out from the blood capillaries into the interstitium. Also, there was marked thickening of the intersaccular septa and marked narrowing and collapse of most of the saccules. This thickening in the intersaccular septa was due to marked cellular infiltration. Other authors reported similar findings; and added that the inflammatory cellular infiltration was related to the oxidative stress resulting in generation of mediators such as IL-8 and cytokine-induced neutrophil chemoattractant that attract the inflammatory cells into the pulmonary microcirculation and then to the air spaces due to destruction of the endothelial cells and the capillary membrane by the free radicals [28].
Some of these collapsed saccules were lined with cuboidal epithelium only with no transition to flat epithelium as in the control group of the same age and there was discontinuity of the epithelial lining of some of these saccules, also the bronchioles were lined with epithelial cells showing marked disintegration of their nuclei (necrotic cells) and there was marked disruption and discontinuity of its epithelium with cytoplasmic vacuolation. The increase of lipid peroxidation following MSG treatment is an important factor causing endothelial cell damage as the increase in lipid peroxidation leads to loss of membrane integrity and cell degeneration [29]. On the other hand, another study attributed the endothelial cell damage to the direct effect of the circulating toxins or to the release of vasoactive cytokines from macrophages and platelets or from the release of lysosomal enzymes [30].
Also, these results are in agreement with others who stated the hypertrophy of the epithelial cells lining the bronchioles which might be distended by mucus [16]. The increase in mucus and the hypertrophy of mucus secreting cells represent an important cause of airflow obstruction, decreased alveolar ventilation or hypoxia and consequently asthma. This observation might explain the state of asthma which was reported by many authors following MSG treatment.
In the present study, the immunohistochemical staining using PCNA stain showed that in the control group at both GD14 and GD16, there was high positive immunoreactions for PCNA in the bronchiolar epithelium and the adjacent stoma, but at GD19 there was moderate positive immunoreactions for PCNA in the bronchiolar epithelium and the adjacent stoma and finally at GD21 there was mild positive immunoreactions for PCNA in the bronchiolar epithelium and the adjacent stoma as the cell proliferation of the lung is increased during the peudoglandular stage and begin to decrease in the canalicular stage then markedly decreased during the saccular stage also found that total cell proliferation of fetal rat lung declined during gestation [31]. They added that there was a relative decrease of epithelial proliferation during the latter stages of gestation, whereas endothelial cell proliferation increased during this time. Differentiation of type II alveolar epithelial cells to type I alveolar epithelial cells was associated with decreased epithelial cell proliferation.
In the group treated with MSG, there were high positive immunoreactions for PCNA at all age subgroups in comparison with the control ones of the same ages. This marked increase in PCNA reactions in all treated group might be due to the marked cell degeneration, damage and delayed maturation that occurred in these groups, so these cells try to proliferate more to compensate this degeneration and damage. Similar findings were reported by who said that increased cell proliferation, particularly alveolar type II epithelial cells, is characteristic of several acute and chronic pulmonary injuries [32]. The results of the present study were confirmed by statistical analysis of the morphometric study. The percentage of PCNA stained tissue/unit area showed that there was marked increase in all treated groups compared with the control one of the same ages. This could be an indication of delayed maturation and/or degeneration [19].
Conclusion
Administration of MSG during pregnancy and the period of organogenesis might exert histopathological changes in the fetal lungs. Therefore, it might be suggested to minimize or even prohibit eating foods containing MSG especially during pregnancy to avoid the occurrence of such possible hazards. Future studies are recommended using larger numbers of animals with different doses to define at which one the hazard effect could be inevitable.
Acknowledgement
The Authors would like to thank to Prof. Hayam Rashed for her assistance in immunohistochemical preparation.
References
- Anto JM, Vermeire P, Vestbo J, Sunyer J (2001) Epidemiology of chronic obstructive pulmonary disease. Eur Respir J 17(5): 982-994.
- Hegazy A (2014) Clinical Embryology for medical students and postgraduate doctors. Lambert Academic Publishing, Berlin, Germany.
- Sakr AA (2004) Light and electron microscopic studies on lung and myocardium of the mice fetuses maternally treated with monosodium glutamate and role of vitamin E against toxicity. Egypt J Hospital Med 15: 95-117
- Yu T, Zhao Y, Shi W, Ma R, Yu L (1997) Effects of maternal oral administration of monosodium glutamate at a late stage of pregnancy on developing mouse fetal brain. Brain Res 747(2): 195-206.
- Xu L, Sun J, Lu R, Ji Q, Xu JG (2005) Effect of glutamate on inflammatory responses of intestine and brain after focal cerebral ischemia. World J Gastroenterol 11(5): 733-736.
- Gottschling BC, Maronpot RR, Hailey JR, Peddada S, Moomaw CR, et al. (2001) The role of oxidative stress in Indium Phosphide-Induced Lung Carcinogenesis in Rats. Toxicol Sci 64(1): 28-40
- Bellisle F (1998) Effects of monosodium glutamate on human food palatability. Ann N Y Acad Sci 855: 438-441.
- Swelim HH (2004) Monosodium glutamate (MSG) induced retinopathy in adult and neonate CD-1 mice. Egypt J Med Lab Sci 13: 45-71.
- Samuels A (1999) The toxicity/safety of processed free glutamic acid (MSG): a study in suppression of information. Account Res 6(4): 259- 310.
- Gill SS, Mueller RW, McGuire PF, Pulido OM (2000) Potential target sites in peripheral tissues for excitatory neurotransmission and excitotoxicity. Toxicol Pathol 28(2): 277-284.
- Frieder B, Grimm VE (1984) Prenatal monosodium glutamate (MSG) treatment given through the mother’s diet causes behavioral deficits in rat offspring. Int J Neurosci 23(2): 117-126 .
- Moreno G, Perelló M, Gaillard RC, Spinedi E (2005) Orexin a stimulates hypothalamic- pituitv- adrenal (HPA) axis function, but not food intake in the absence of full hypothalamic NPY- ergic activity. Endocrine 26(2): 99-106.
- Pavlovic V, Pavlovic D, Kocic G, Sokolovic D, Sarac M, et al. (2009) Ascorbic acid modulates monosodium glutamate induced cytotoxicity in rat thymus. Bratisl Lek Listy 110(4): 205-209.
- Khalaf HA, Arafat EA (2015) Effect of different doses of monosodium glutamate on the thyroid follicular cells of adult male albino rats: a histological study. Int J Clin Exp Pathol 8(12): 15498-15510.
- Sharma A (2015) Monosodium glutamate-induced oxidative kidney damage and possible mechanisms: a mini review. J Biomed Sci 22:93.
- Stevenson DD (2000) Monosodium glutamate and asthma. J Nutr 130(4S): 1067S-1073S.
- Allen DH, Delohery J, Barker G (1987) Monosodium L-glutamateinduced asthma. J Allergy Clin Immunol 80(4): 530-537.
- Woessner KM, Simon RA, Stevenson D (1999) Monosodium glutamate sensitivity in asthma. J Allergy Clin Immunol 104(2 Pt 1): 305-310.
- Dezfuli BS, Giari L, Lui A, Squerzanti S, Castaldelli G, et al. (2012) Proliferative cell nuclear antigen (PCNA) expression in the intestine of Salmo trutta trutta naturally infected with an acanthocephalan. Parasites & Vectors 5: 198.
- Hegazy R, Hegazy A (2015) Simplified Method of Tissue Processing (Consuming Less Time and Chemicals). Ann Int Med Den Res 1(2): 57- 61.
- Koo HP, Santarosa RP, Buttyan R, Shabsigh R, Olsson CA, et al. (1993) Early molecular changes associated with streptozin-induced diabetes bladder hypertrophy in the rat. Urol Res 21(6): 375-381.
- Lieberthal W, Levine JS (1996) Mechanisms of apoptosis and its potential role in renal tubular epithelial cell injury. Am J Physiol 271(3 Pt 2): F477-88.
- Millard WJ, Martin JB, Audet J, Sagar SM, Martin JB (1982) Evidence that reduced growth hormone secretion observed in monosodium glutamate treated rats is the result of deficiency in growth hormone releasing factor. Endocrinology 110(2): 540-550.
- Hildeman DA, Mitchell T, Aronow B, Wojciechowski S, Kappler J, et al. (2003) Control of Bcl –2 expression by reactive oxygen species. Proc Natl Acad Sci USA 100(25): 15035-15040.
- Oda H, Arakawa M, Kambara K, Nakahara K, Segawa T, et al. (1993) Ultrastructure substrates for increased lung water content in experimental pulmonary edema. Histol Histopathol 8: 73-82.
- Martin TR (2002) Neutrophils and lung injury: Getting it right. J Clin Invest 110(11): 1603–1605.
- Doohan J (2000) Cardiac output and blood pressure. Human physiology. McGraw-Hill Companies, New York, USA.
- Rahman I, MacNee W (2000) Oxidative stress and regulation of glutathione in lung inflammation. Eur Respir J 16(3): 534-554.
- Tsou TC, Yeh SC, Tsai FY, Chang LW (2004) The protective role of intracellular GSH status in the arsenite-induced vascular endothelial dysfunction. Chem Res Toxicol 17(2): 208-217.
- Rubin E (1995) Essential pathology (3rd Edn). Lippincott. Williams and Wilkins, Philadelphia, USA.
- Adamson IY, King GM (1984) Sex differences in development of fetal rat lung. I. Auto radiographic and biochemical studies. Lab Invest 50(4): 456-60.
- Maniscalco WM, Watkins RH, O’Reilly MA, Shea CP (2002) Increased epithelial cell proliferation in very premature baboons with chronic lung disease. Am J Physiol Lung Cell Mol Physiol 283(5): L991-L1001.






























