Synergistic Effect of Thyrotropin Releasing Hormone (TRH) and onNorepinephrine (NE) on the Metabolic Rate of Warm-Acclimated Rats
Ateegh Al-Arabi*
Department of Public Health, South University, USA
Submission: March 30, 2017; Published: April 24, 2017
*Corresponding author: Ateegh Al-Arabi, Department of Public Health, South University, West Palm Beach, Florida 33411, USA, Email: ateeghallarabi@gmail.com
How to cite this article: Ateegh Al-A. Synergistic Effect of Thyrotropin Releasing Hormone (TRH) and on Norepinephrine (NE) on the Metabolic Rate of Warm-Acclimated Rats. Anatomy Physiol Biochem Int J. 2017; 2(3): 555588. DOI:10.19080/APBIJ.MS.ID.555588.
Abstract
We have studied the effects of chronic peripheral infusion of thyrotropin releasing hormone (TRH) and norepinephrine (NE) alone and together. Do these hormones additively affect energy balance and body weight similarly in lean and obese animals? 20 each lean Wistar and obese (fa/fa) Zucker strain rats were studied. Food intake, body weight and resting metabolic rate (RMR) were measured over a 14 day period. We confirmed that in the obese animals, separately the hormones act as in the lean: NE stimulated RMR, but with parallel stimulation of appetite so that weight gain continued; TRH stimulated RMR but inhibited food intake so that rate of weight gain was reduced. TRH and NE together acted to further enhance RMR, but the inhibitory and stimulatory effects on food intake cancelled out. Thus a negative energy balance resulted in both groups; however, resultant weight loss was profound in lean but only moderate in obese. We suggest that the relative insensitivity of the obese to TRH and NE together reflects the inability of their brown adipose tissue to respond to thermogenic agents, even when chronically administered.
Keywords: Norepinephrine; TRH; Metabolism; Obesity
Abbreviations: TRH: Thyrotropin Releasing Hormone; NE: Norepinephrine; RMR: Resting Metabolic Rate; TSH: Thyroid Stimulating Hormone
Introduction
There have been suggestions that the genetically obese rat has abnormal thyroid function [1,2]. It is also known that while the obese rat shows various physiological and metabolic abnormalities normally associated with hypothyroidism, it also has other defects that are not generally found in hypothyroid animals. Like the hypothyroid rat, which shows decreased oxygen consumption in adipose tissue in vitro [3], the obese Zucker rat has decreased oxygen consumption in vivo [4] and a β-oxidation of lipids [5]. Obese Zucker rats also show cold intolerance [6-8] that is unresponsive to catecholamine infusion [8]. Preliminary study of our own showed that norepinephrine enhanced food intake and resting metabolic rate in lean rats [9]. It has been demonstrated that peripheral administration of thyrotropin releasing hormone (TRH) has both an inhibitory effect on food intake and acts, probably through the thyroid axis, to stimulate whole body metabolism [10], TRH administration causes an increased release of thyroid stimulating hormone (TSH) [11] which in turn causes the release of triiodothyronin [12] which causes weight loss in the obese Zucker rat [1]. Thyroid hormone administration is known to potentiate the metabolic response to noradrenaline in lean and obese rats [13]. Cold acclimation is known to cause increased thermogenic activity of brown adipose tissue [14]. We therefore treated warm-acclimated, obese Zucker and lean Wistar rats with a combined and separate infusions of TRH and NE to investigate the influence of TRH on the whole-body metabolic effect of NE.
Materials And Methods
Adult male rats were studied: 20 lean animals of Wistar strain, initial body weight range 200-300g (source: Trinity College Dublin, animal house colony) and 20 Zucker obese (fa/ fa) rats had an intital body weight range of 350-500 (source: Olac 1976 Ltd. Oxon, UK). Animals were individually housed on a 12-h light-dark cycle and were acclimated to 28°C for at least two weeks prior to study, with food ad libitium (rat diet: Redmills Co. Goresbridge, Ireland)1. Each group was randomly divided into five subgroups: 1) untreated controls; 2) carrier treated; 3) TRH treated; 4) NE treated; and 5) TRH+NE treated. On day 1 Alzet (TM) Osmotic Minipumps, model 2002 (Alza, California, USA) were subcutaneously implanted in the dorsal interscapular region (subgroups: 2 - 5). Surgery was performed under halothane anaesthesia (May & Baker Ltd. Dagenham, England). Pumps were filled with either carrier alone, 0.1M L-ascorbic acid (Sigma, USA) and 0.02M 4,5-dihydroxy-1,3 benzene sulphonic acid (Tiron, Sigma, USA) (subgroup 2); or carrier plus 0.3M TRH (CRB, UK) (subgroup 3) or carrier plus 0.3M (-) arterenol bitartrate (NE) (Sigma, USA) (subgroup 4), or carrier plus TRH and NE (subgroup 5). TRH delivery rate was estimated as 5 ug/hr. and NE as 20 ug/hr. extending over a period of 14 days. Commencing on day two, food intake and body weight were determined every two days, and the resting metabolic rate (RMR) was measured on days 2, 8 and 14 (indirectly as minimal oxygen consumption), in a multi-channel closed-circuit system (local construction). To avoid any stress effects on metabolic rate during measurements of minimal oxygen consumption, animals were pre-exposed to the system at least twice during the acclimation period, and to avoid circadian rhythm effects measurements were done at the same time of day (10:00-13:00h). Results were compared using the two-tailed paired Student's t-test [15], n=4 per treatment except in the case of the untreated obese where one animal was found to be sick and excluded were n=3. Results were expressed as the mean (SD).
Results
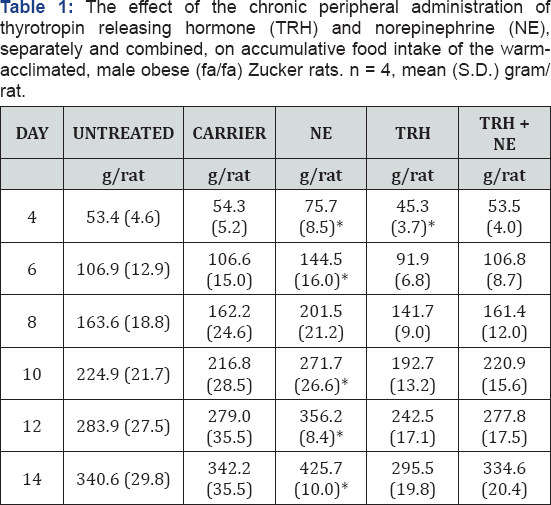
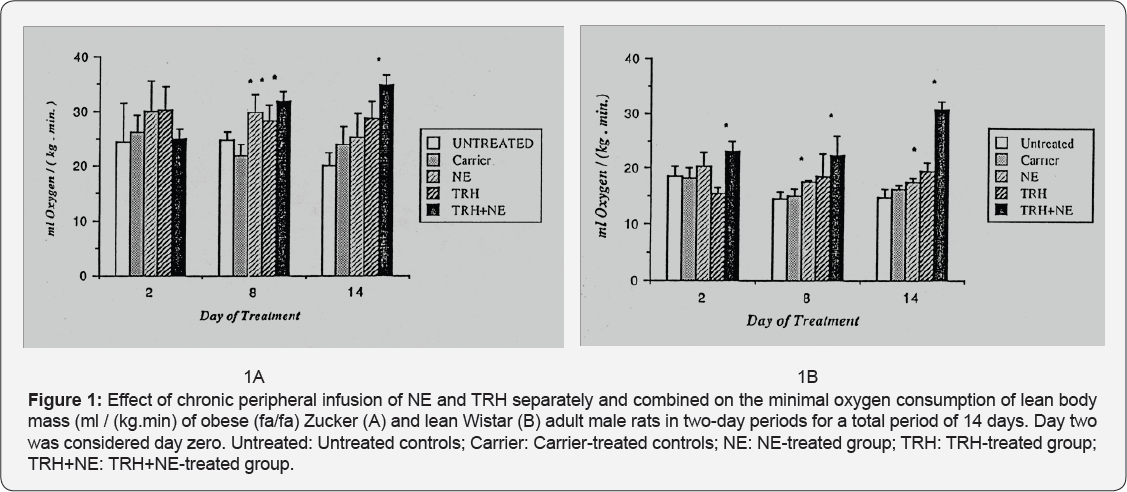
Oxygen Consumption: (Figures 1A & 1B) Results are expressed as minimal oxygen consumption per kg of lean body mass. This was computed from whole body oxygen consumption assuming that 45.2% and 10.5%, respectively, of the total body weight represents nonmetabolizing fat in the obese and lean rats [16]. The results show that the resting metabolic rate (RMR) (per unit of fat-free weight) of the obese animals is higher than that of the lean. They also show that norepinephrine (NE) caused an increase in the RMR of both groups. Carrier (vehicle) showed no effect on the RMR of both groups. TRH alone did not initially affect the RMR of the obese rats in the first period of treatment; to the contrary it caused reduction of the RMR of the lean group. Significant increase in the RMR of both groups appeared during the second half of TRH-treatment period. Combined treatment with TRH and NE caused significant increase in RMR of both groups throughout the experiment except on day two in the case of the Zucker group.
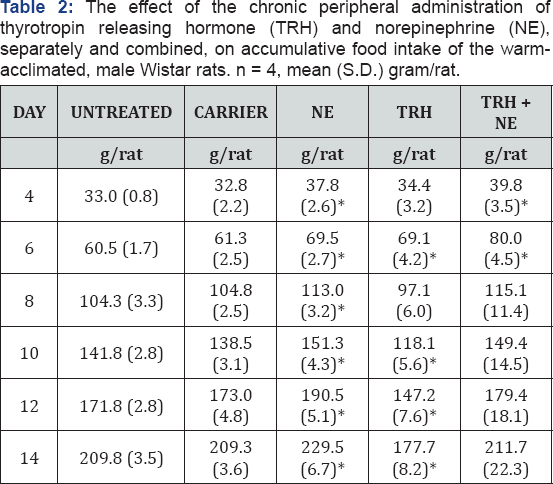
Food Consumption: (Tables 1 & 2) Data are expressed as accumulative food intake after every two-day period. The carrier treated and the untreated animals showed almost identical food consumption in both groups, with higher food intake by the obese animals. NE treatment did enhance the food intake of both groups. TRH treatment showed the opposite effect in both groups, significantly so during the second week of treatment in the lean animals. Combined treatment with TRH and NE showed insignificant difference in food intake from the carrier treated animals.
*indicates the significance from controls at P< 0.05.
*indicates the significance from controls at P < 0.05
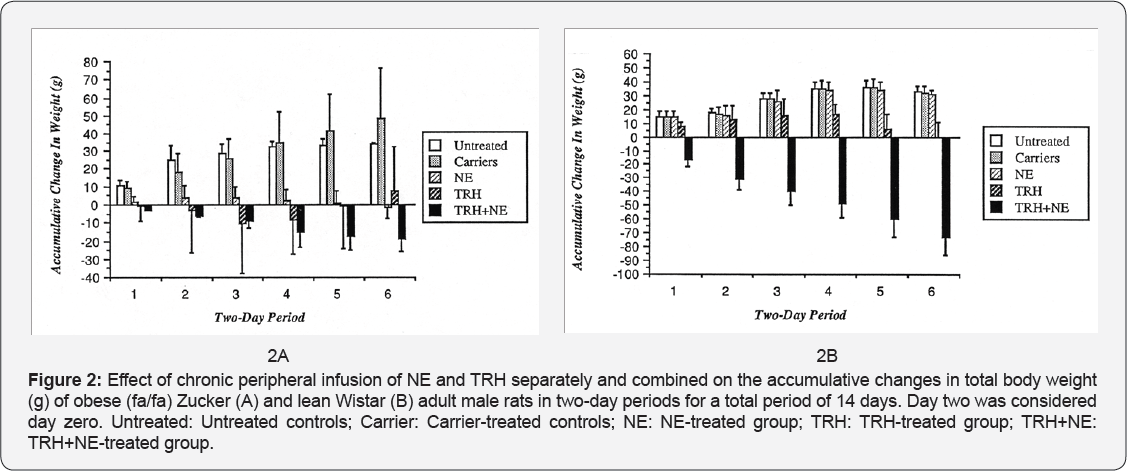
Change in Body Weight: (Figures 2A & 2B) Data was expressed as accumulative change in total body weight starting from day 2. The carrier treated controls of the lean group showed changes in body weight similar to those of the untreated controls of the same group, while in the obese there were small statistically insignificant differences. NE treatment also showed no effect on the weight gain by the lean animals while the NE-treated obese group almost maintained their initial body weight indicating a significant difference from the controls. TRH treatment mainly affected the lean animals causing retardation in their weight gain during the second week of the treatment. The obese animals when treated with TRH showed great variability in their response, however, the mean response was not significant from the controls. The combined treatment with TRH and NE did cause a persistent significant effect causing both groups of animals to lose significant amount of weight, with higher response from the lean rats (Figures 1A & 1B) (Tables 1 & 2) (Figures 2A & 2B).
Discussion
Oxygen consumption results (Figures 1A & 1B) show that the RMR per lean body mass of the obese group is generally higher than that of the lean. This has been observed previously in obese mice [17]. The authors suggested that the increased RMR reflects the much increased oxygen costs associated with the processing of enhanced food intake, particularly the energy costs of storing the surplus as fat. Tables 1 and 2 confirm that whole body energy intake was increased by 61.9% and 62.9% respectively in untreated and carrier treated obese rats compared with the similar lean groups. Large variability of response on day two could be due to delay of some pumps to achieve sustained rate of release, or due to slow recovery from surgery by some animals. NE is known to enhance RMR in lean animals [9]. Obese animals are known to have defective thermogenesis [4] which is not responsive to catecholamine treatment [8]. However, most of these studies were done using acute administration of NE. Results of this work (Figure 1A & 2) using chronic NE treatment caused a slight increase of RMR in both lean and obese groups (Figure 1). This may mean that NE plays a role not only in acute stimulation of the thermogenic system on cold exposure but also chronically activates resting thermogenic organs such as brown adipose tissue. TRH caused an increased RMR in lean animals.This effect could be explained as: (a) direct action of TRH on thermogenesis; or (b) indirect action by causing release of TSH [11] followed by increased release of triiodothyronine [12] which in turn increases RMR [13]. The results also show that TRH caused increased RMR separately without administration of exogenous NE in both groups. This can be referred to the defective thyroid function in the obese animals [1,2] which might have been corrected by the release of TSH; and to the increased release of the thyroid hormones in the lean animals. Combined treatment of NE and TRH demonstrated the highest stimulation in both groups. Thus when correct function of the parasympathetic system and the thyroid axis are met, the two hormones interact in their metabolic action.
Central, acute and chronic, administration of exogenous norepinepherine has been demonstrated to rapidly elicit a robust feeding response and to potentiate daily food intake [18]. The peripheral infusion of NE, applied in this study, caused the same effect (Tables 1 & 2) with different degrees in the different groups of animals. In contrast, TRH administration is known to cause a satiety effect [19]. Results of this work support these findings. The two opposite effects of both hormones disappeared when the two hormones were infused simultaneously. This phenomenon can be explained as the result of a summation of the negative and positive responses to the hormones.
As shown in Tables 1 and 2 the only treatment that caused chronic loss of weight in both groups is the combined infusion of both hormones. This obviously is due to the increased metabolic rate caused by this treatment, which was not compensated for by an increase in food intake. It must be noted that though this treatment did produce a fall in body weight in obese animals the effect was much less than in the lean, only 20g being lost versus 70g. At the end of two weeks treatment the obese were still obese. Finally, it can be concluded that the results of this work confirm earlier findings. They suggest that TRH has a direct inhibitory effect on food intake. It has also a direct enhancing effect (potentiated when administered with NE) on the minimal oxygen consumption in obese rats. Loss in body weight caused by the combined treatment by TRH and NE is the result of the significant increase in metabolic rate, which has not been compensated by an increase in food intake.
References
- Autissier N, Dumas P, Loireau A, Michel R (1980) Thyroid status and effects of 3,5,3' triiodothyroacetic acid and fenoproporex in genetically lean and obese female rats. Biochem Parmacol 29(11): 1612-1615.
- Bray GA, York DA (1971) Thyroid function of genetically obese rats Endocrinology 88(4): 1095-1099.
- Bray GA, Goodman HM (1968) Metabolism of adipose tissue from normal and hypothyroid rats. Endocrinology 82(4): 860-864.
- Bray GA (1969) Oxygen consumption of genetically obese rats. Experientia 25(10): 1100-1101.
- Triscarri J, Sullivan AC (1978) Defects in hepatic lipid metabolism in obese Zucker rats. Int J Obesity 2: 371-372.
- Godbole V, York DA, Bloxham DP (1978) Developmental changes in the fatty (fa/fa) rat: evidence for defective thermogenesis preceding the hypolipogenesis and hyperinsulinaemia. Diabetologia 15: 41-44.
- Levin BE, Comai K, Sullivan AC (1981) Metabolic and sympathoadrenal abnormalities in the obese Zucker rat: effect of chronic phenoxybenzamine treatment. Pharmacol Biochem Behav 14(4): 517525.
- Levin BE, Triscari J, Sullivan AC (1980) Abnormal sympathoadrenal function and plasma catecholamines in obese Zucker rats. Pharmacol Biochem Behav 13: 107-113.
- Al-Arabi A, Andrews JF (1987) Action of chronic peripheral administration of noradrenaline on food intake, body weight and metabolic rate of rats. Irish J Med Sci 156: 76.
- Al-Arabi A, Andrews JF (1986) Effect of the chronic periferal administration of thyrotropin releasing hormone (TRH) and noradrenaline (NA), separately and combined, on food intake, body weight, metabolic rate of warm acclimated rats. Irish J Food Sci Tech 10: 190-191.
- Strbak V, Angyal R, Jorcovicova J, Randuscova A (1986) Role of the thyrotropin releasing hormone in thyroid-stimulating hormone and growth hormone regulation during postnatal maturation in female Wistar rats. Biol Neonate 50(2): 91-96.
- Martin JB (1974) Regulation of the pituitary-thyroid axis. In: MTP International Review of Science. Physiology Series One. (Edn 5th), Butterworths, University Park Press, Baltimore, London, UK, pp. 67108.
- Levin BE, Triscari J, Sullivan AC (1982) Sympathetic activity in thyroid- treated Zucker rats. Am J Physiol 243(1): R170-R178.
- Kuroshima A, Yahata T (1985) Effect of food restriction on cold adaptability of rats. Can J Physiol Pharmacol 63(1): 68-71.
- Sendecor GW, Cochran W (1971) Statistical Methods Sixth edition. The Iowa State University Press, Ames, Iowa, USA, pp. 59-60.
- Seelbach JD, Etherton TD, Kris-Etherton PM (1985) The effect of vigorous treadmill exercise on adipose tissue development in Zucker rats. Int J Obes 9(1): 11-19.
- Mercer SW, Trayhurn P (1983) Developmental changes in fatty acid synthesis in interscapular brown adipose tissue of lean and genetically obese (ob/ob) mice. Biochem J 212: 393-398.
- Leibowitz SF, Shor-Posner G (1986) Hypothalamic monoamine system for control of food intake: analysis of meal patterns and macronutrient selection. Raven Press, New York, USA, pp. 29-49.
- Morley JE, Levin AS (1980) Thyrotropin releasing hormone (TRH) suppresses stress induced eating. Life Sci 27(3): 269-274.






























