Evaluation of the Metabolic Indicators in Obese Diabetic Patients: A Case-Control Study
Tarig H Merghani1* and Azza O Alawad2
1Associate Professor, Physiology Department, University of Khartoum, Africa
2Assistant Professor, AI-Neelain University, Africa
Submission : March 31, 2017; Published : April 24, 2017
*Corresponding author : Tarig H Merghani, MD, PhD., Associate Professor, Department of Physiology, Faculty of Medicine, University of Khartoum, PO Box 102, Khartoum- Sudan, Africa, Email: tarighm@gmail.com
How to cite this article : Tarig H M,Azza O A. Evaluation of the Metabolic Indicators in Obese Diabetic Patients: A Case-Control Study. Anatomy Physiol Biochem Int J. 2017; 2(3): 555587. DOI : 10.19080/APBIJ.2017.02.555587.
Abstract
Background: The rate of energy expenditure in the obese type II diabetic patients is influenced by the abnormal metabolic reactions in skeletal muscle, liver and adipose tissue that are caused by insulin resistance. The aim of this study is to evaluate the resting oxygen consumption (VO2), the carbon dioxide production (VCO2), and the respiratory exchange ratio (RER) in obese type II diabetic patients in comparison to healthy obese subjects, and to assess the relation of these indicators to diabetes control and duration.
Methods: We conducted a case-control study in the Specialized Diabetes Center, Ministry of Health, Khartoum- Sudan. Two groups of participants, 40 obese diabetics and 40 obese non-diabetics, were matched according to age, gender, residence, and body size. Indirect calorimetry was carried out using the Power Lab 8/35 metabolic kit (AD Instruments, Castle Hill Australia) for analysis of a resting expired air of each participant. The "NycoCardHaemoglobinA1c test” (Axis -Shield/ Norway) and the one touch® glucometer (LifeScan Canada Ltd) were used for measurement of hemoglobin A1C (HbA1C) and fasting blood sugar (FBS), respectively. The student's t-test and the chi-square test were used for analysis of the variables. Statistical significance was accepted at p < 0.05.
Results: Both the VO2 (mean ± SD) and the VCO2 were higher in the obese diabetic patients compared to the healthy ones (209.9 ± 42.7 Vs 192.4 ± 28.1; p= 0.034) and (191.4 ± 35 Vs 178.3 ± 22.5; p= 0.05), respectively. The RER was almost equal in the two groups (0.93 ± 0.16 Vs 0.93 ± 0.11; p= 0.868). Both VO2&VCO2 were significantly higher in those with high HbA1C compared to those with normal HbA1C values (212.6 ± 42 Vs 192.6 ± 31; p= 0.015) and (193.0 ± 37 Vs 178.2 ± 22.0; p= 0.021), respectively. The relations with FBS and diabetes duration were statistically insignificant.
Conclusion: The poorly controlled diabetes mellitus causes a marked increase in VO2, VCO2, but not the RER.
Keywords : Metabolic; Oxygen Consumption; Carbon Dioxide Production; Respiratory Exchange Ratio; Diabetes; Obesity
Abbreviations : VCO2: Carbon Dioxide Production; VO2: Resting Oxygen Consumption; RER: Respiratory Exchange Ratio; HbA1C: Hemoglobin A1C; FBS: Fasting Blood Sugar; RQ: Respiratory Quotient; RMR: Resting Metabolic Rate; BMI: Body Mass Index; DD: Diabetes Duration
Introduction
Measurement of the energy expenditure in man is a key step in the calculation of the energy requirements that are needed for clinical nutritional assessment and body weight management. Obese diabetic patients suffer from variable metabolic abnormalities that are associated with insulin resistance. The respiratory quotient (RQ) is an important metabolic indicator. It is the ratio of carbon dioxide produced to oxygen consumed at the cellular level; however, when it is calculated from expired gasses, it is referred to as the respiratory exchange ratio (RER). Under steady conditions, the RER provides a precise estimate of the RQ. On a mixed diet, part of the total CO2 produced and O2 consumed by the cells comes from metabolism of carbohydrates (RQ= 1.0), and proteins (RQ= 0.82) in addition to fats (RQ= 0.71). In general, the RQ varies between 0.7 to 1.1 depending on the proportion of substrates being metabolized and the rate of ventilation [1]. When the value of the RER is high, oxygen consumption is lesser than required, and anaerobic metabolism is taking place, e.g. during high-intensity exercise. It is worth noting that, RER excludes protein metabolism, which during rest is assumed to be very minimal. Many factors affect the value of the RQ and accordingly cause changes in the metabolic rate. Previous studies showed ethnic variation with the Africans having higher RQ and lower resting metabolic rate (RMR) compared to the European subjects [2,3]. The high RQ indicates that the carbohydrate is utilized as the primary source of energy and this is a recognized cause of weight gain.On the other hand, a low RQ indicates a high-fat utilization, which is reported in healthy overweight and obese subjects [4]. Obesity is a predisposing factor to insulin resistance and type II diabetes mellitus. The effect of diabetes mellitus on RQ depends on the state of management. A higher RQ has been found in diabetic patients treated with insulin or hypoglycemic agents when compared to the non-treated patients, thus indicating weight gain [5]. Therefore, the combined effect of both obesity and diabetes on the RQ and energy expenditure is a complex one. The aim of this study is to evaluate the resting metabolic indicators (RER, VO2, and VCO2) in obese type II diabetic patients in comparison to healthy obese subjects, and to assess their relations to the level of hemoglobin A1C (HbA1C) and the fasting blood sugar (FBS) in the two groups.
Methods
Study design and participants
A case-control study was conducted in the Specialized Diabetes Center, Ministry of Health, Khartoum- Sudan. A total of 80 obese subjects, 40 diabetic patients (cases) and 40 nondiabetic subjects (controls), participated in this study. The cases were randomly selected from those attending for consultation, and the controls from their healthy relatives. Inclusion criteria for all participants were adult subject, age 35 to 50 years old, obese (defined as body mass index ≥ 30), not a smoker, afebrile, and showing no symptoms or signs of an acute or a chronic infection. The selected cases didn't receive insulin or hypoglycemic medications for at least two days before presentation. Matching of the two groups was considered for the age, gender, BMI, and area of residence.
Measurements
The body mass index (BMI) was calculated as weight (in kilograms)/ height (in meters squared). A standardized height and weight scales were used for measurement of the height and weight of each participant. Five microlitres of blood were drawn from each participant for HaemoglobinA1c (HbA1C) measurement. The "NycoCardHaemoglobinA1c test” (Axis -Shield/ Norway); was used for HbA1C measurements. Results were classified into two groups, normal HbA1C (< 6.5%), and high HbAlC (≥ 6.5%) [6]. Fasting blood sugar (FBS) was measured using one touch® glucometer (LifeScan Canada Ltd). Results were grouped into normal FBS (< 126 mg/dl), and high FBS (≥ 126 mg/dl). The PowerLab 8/35 (AD Instruments, Castle Hill Australia) data acquisition system [7,8], comprising hardware and software, was used with bio-amplifier, thermistor, spirometer, gas analyzer, gas mains chamber, and computer windows seven program for metabolic indicators measurements. The system allowed measurement of resting oxygen consumption (VO2), carbon dioxide production (VCO2), and respiratory exchange ratio (RER). All participants took light dinner and fasted overnight before the presentation. They were allowed to rest in a thermally neutral room for 30 minutes before the measurements. Each test took about 20 min, with at least 5 min steady state. The steady state was defined as the minimal variation (< 10%) in VO2, VCO2 VE, and RER.
Ethical considerations
Our study conformed to the ethical principles of medical research as developed by the World Medical Association Declaration of Helsinki. Ethical clearance was given by the Institutional Research Committee (Faculty of Medicine/ University of Al-Neelain- Sudan), and approval was obtained from the Ministry of Health. Permission letters were sent to the director of the Diabetes Center. Written consents were obtained from all participant before participation.
Statistical analysis
Data was reported as mean ± SD. Statistical analysis was carried out with the Statistical Package for the Social Sciences, version 20 (SPSS Inc., Chicago, IL, USA). The student's t-test was used to compare the continuous variables (means of the VO2, VCO2, and RER) between the two groups. The chi-square test was used for analysis of the categorical variables. Statistical significance was accepted at p < 0.05.
Results
Table 1 describes the demographics of the participants. The age ranged from 35 to 50 years old with a mean (SD) of 45.0 (5.1) and 44.3 (5.7) for the cases and the controls, respectively (p= 0.564). Male: female ratio was 2:3 in each group (p= 0.590). The mean (SD) body mass index was 32.0 (2.1) for both cases and controls (p= 0.633) (Table 1).
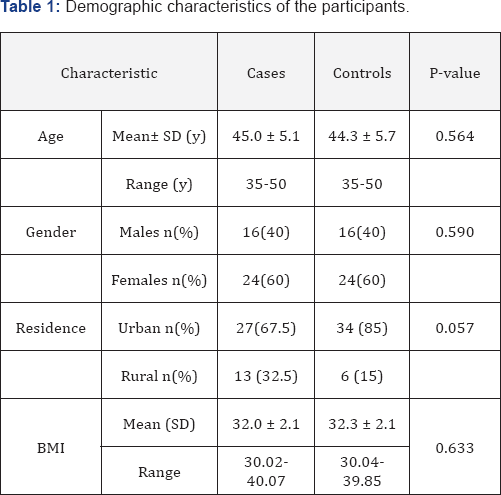
Table 2 shows a comparison between cases and controls in the resting oxygen consumption (VO2), carbon dioxide production (VCO2), and respiratory exchange ratio (RER). VO2 (mean ± SD)was significantly higher in the obese diabetic patients compared to the healthy ones (209.9 ± 42.7 Vs 192.4 ± 28.1; p= 0.034). The VCO2 was similarly higher in the first group (191.4 ± 35 Vs 178.3 ± 22.5; p= 0.05); however, the RER was almost equal in the two groups (0.93 ± 0.16 Vs 0.93 ± 0.11; p= 0.868) (Table 2).
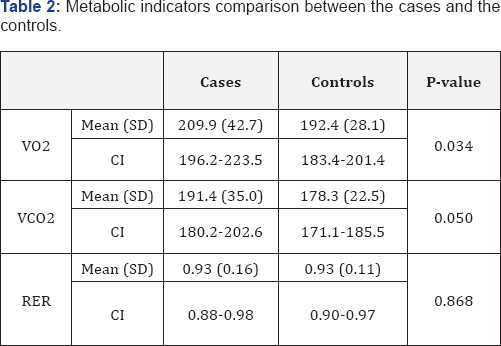
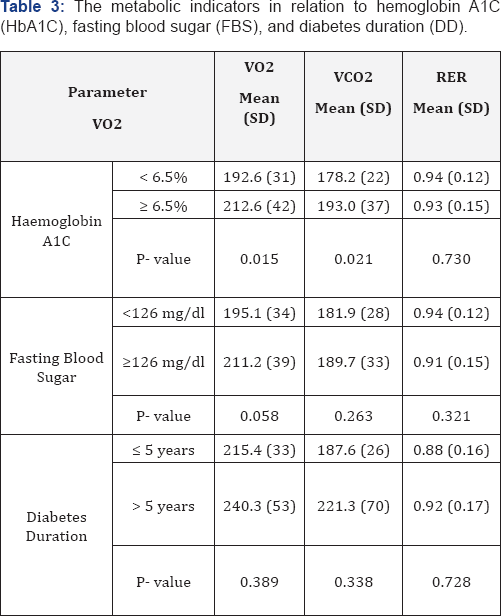
Table 3 shows the relation between different parameters of diabetes (hemoglobin A1C (HbA1C), fasting blood sugar (FBS), and diabetes duration (DD) with VO2, VCO2, and RER. Both VO2&VCO2 were significantly higher in those with abnormally high HbA1C compared to those with normal HbA1C values (212.6 ± 42Vs192.6 ± 31; p= 0.015) and (193.0 ± 37 Vs 178.2 ± 22.0; p= 0.021), respectively. The relations with FBS and DD were statistically insignificant (Table 3).
Discussion
The metabolic rate measurements are recommended for the calculation of energy requirements during health and disease. The gold-standard method is the indirect calorimetry where VO2 and VCO2 are measured directly from the expired air for calculation of the caloric burning rate. Historically, instruments used in the past for indirect calorimetry were often referred to as the "metabolic cart” because they were large, complex, and difficult to transport. On the contrary, the Power Lab instruments, used recently for metabolic rate measurements [7,8], are easier to mobilize. Its high-performance data acquisition system is suitable for a broad range of research applications, including the metabolic rate measurements. The calibration of oxygen and carbon dioxide sensors, which is needed before each test session, is much easier and less complicated with the Power Lab than the older methods.
In this study, the diabetic patients and their controls were matched according to age, sex, residence, and BMI. Age is a cause of physiological variation in energy expenditure [9]. We opted to conduct this study in a narrow range of age (35 to 50 years old), during which the change in the rate of metabolism is very slow [10]. Gender is another determinant of energy expenditure, with men having a greater VO2 than women [11]. Matching of the residence is equally important. Hayter and Henry previously reported that variation in geographical location between people is a source of a significant difference in the rate of energy expenditure [12]. The influence of body size on the metabolic rate had been confirmed long time ago [13]. In this study, the range and the mean body mass index (BMI) were similar among the cases and the controls. Since all the above factors, including the measurement site, the equipment used, and the environmental factors were all the same for the two groups, our findings were unlikely to be explained by variation in these factors.
The major finding is this study is the higher resting VO2&VCO2 in the diabetic patients compared to their controls. However, the RER was almost equal in the two groups. Our results were similar to those reported by Huang et al. [14]. The high fasting RER value of 9.3 was previously reported among obese subjects with positive history of parental obesity whereas a low fasting RQ was found in those without a positive family history of obesity, suggesting that hereditary factors might influence the ability to oxidize fat in obese subjects [15].
Many mechanisms could explain the rise in VO2 and therefore the higher energy expenditure among the diabetic patients. One of the mechanisms is the increase in gluconeogenesis and the associated high plasma levels of free fatty acids caused by insulin resistance [16]. In support with that, previous studies reported a reduction in resting energy expenditure following improvement in glycemic control [17]. Another supporting finding from our study is the significant association between poor diabetes control, as indicated by HbA1C values ≥ 6.5%, and higher VO2, but not the RER. However, we found an insignificant relation with the FBS. Conversely, Weyer and his colleagues in 1999 found that the energy expenditure correlates positively with the deterioration in glycemic control [18]. A new predictive equation that uses diabetes mellitus as a variable for calculation of the resting metabolic rate has been proposed recently [14]. In another equation, the fasting blood sugar has been suggested [19]. Other mechanisms that might explain the higher VO2 among the obese diabetic participants could be an abnormal protein metabolism [20], increased sympathetic activity [21], and hyperglucagonemia [22]. All these abnormalities are connected with type II diabetes mellitus.
Our study has many limitations. We did not measure the skeletal muscle or the free fat mass for the participants, which might be different between the two groups. The core body temperature that represents the state of thermoregulation for each participant was not determined. The intra-individual variation of energy expenditure in the same individual from time to time within the same day was not considered. However, matching of the cases and controls was assured, and therefore the effects of these factors are expected to be small.
Conclusion
This study showed that the resting VO2 was significantly higher among obese diabetic patients compared to obese nondiabetics whereas the RER is almost equal in the two groups. A significant relation was found between the poorly controlled diabetes mellitus, as indicated with HbA1C ≥ 6.5%, and the VO2.
References
- Stipanuk H (2000) Biochemical and physiological aspects of human nutrition. W.B. Saunders Company, USA.
- Adzika Nastimba PA, Pathak K, Soares MJ (2016) Ethnic differences in resting metabolic rate, respiratory quotient and body temperature: a comparison of Africans and European Australian. Eur J Nutr 55(5): 1831-1838.
- Shook RP, Hand GA, Wang X, Paluch AE, Moran R, et al. (2014) Low fitness partially explains resting metabolic rate differences between African American and White women. Am J Med 127(5): 436-442.
- Pujia A, Gazzaruso C, Ferro Y, Mazza E, Maurotti S, et al. (2016) Individuals with Metabolically Healthy Overweight/Obesity Have Higher Fat Utilization than Metabolically Unhealthy Individuals. Nutrients 8(1): 2.
- Gonzales C, Fagour C, Maury E, Cherifi B, Salandini S, et al. (2014) Early changes in respiratory quotient and resting energy expenditure predict later weight changes in patients treated for poorly controlled type 2 diabetes. Diabetes Metab 40(4): 299-304.
- American Diabetes, Association (2014) Diagnosis and classification of diabetes mellitus. Diabetes Care 37(1): S81-90.
- Alawad AO, Merghani TH, Ballal MA (2013) Resting metabolic rate in obese diabetic and obese non-diabetic subjects and its relation to glycaemic control. BMC Res Notes 6: 382.
- AD Instruments (2017) Metabolic Add-on for Lab Chart.
- Wilson MM, Morley JE (2003) Invited review: Aging and energy balance. J Appl Physiol 95(4): 1728-1736.
- Henry CJ (2000) Mechanisms of changes in basal metabolism during aging. Eur J Clin Nutr 54: 77-91.
- Lazzer S, Bedogni G, Lafortuna CL, Marazzi N, Busti C, et al. (2010) Relationship Between Basal Metabolic Rate, Gender, Age, and Body Composition in 8,780 White Obese Subjects. Obesity 18(1): 71-78.
- Hayter JE, Henry CJ (1994) A re-examination of basal metabolic rate predictive equations: the importance of geographic origin of subjects in sample selection. Eur J Clin Nutr 48(10): 702-707.
- Frankenfield DC, Muth ER, Rowe WA (1998) The Harris-Benedict studies of human basal metabolism: history and limitations. J Am Diet Assoc 98(4): 439-445.
- Huang KC, Kormas N, Steinbeck K, Loughnan G, Caterson ID (2004) Resting Metabolic Rate in Severely Obese Diabetic and Nondiabetic Subjects. Obesity Research 53(11): 1395-1398.
- Hainer V, Kunesova M, Pari'zkova J, Stich V, Mikulova R, et al. (2000) Respiratory quotient in obesity: its association with an ability to retain weight loss and with parental obesity. Sb Lek 101(1): 99-104.
- Felig P, Wahren J, Hendler R (1978) Influence of maturity-onset diabetes on splanchnic glucose balance after oral glucose ingestion. Diabetes 27(2): 121-126.
- Franssila-Kallunki A, Groop L (1992) Factors associated with the basal metabolic rate in patients with Type2 (non-insulin-dependent) diabetes mellitus. Diabetologia 35(10): 962-966.
- Weyer C, Bogardus C, Pratley RE (1999) Metabolic Factors Contributing to Increased Resting Metabolic Rate and Decreased Insulin-Induced Thermogenesis during the Development of Type 2 Diabetes. Diabetes 48(8): 1607-1614.
- Gougeon R, Lamarche M, Yale JF, Venuta T (2002) The Prediction of resting energy expenditure in type 2 diabetes mellitus is improved by factoring for glycemia. Int J Obes Relat Metab Disord 26(12): 15471552.
- Gougeon R, PencharzPB, Marliss EB (1994) Effect of NIDDM on the kinetics of whole-body protein metabolism. Diabetes 43(2): 318-328.
- Huggett RJ, Scott EM, GilbeySG, Stoker JB, Mackintosh AF, et al. (2003) Impact of type 2 diabetes mellitus on sympathetic neural mechanisms in hypertension. Circulation 108(25): 3097-3101.
- Charlton MR, Nair KS (1998) Role of hyperglucagonemia in catabolism associated with type 1 diabetes: effects on leucine metabolism and the resting metabolic rate. Diabetes 47(11): 1748-1756.






























