A Comprehensive Case Report of OPHN1 X-Linked Intellectual Disability and a Hazardous Association with Familial Mediterranean Fever Heterozygote Mutation
Hadi Fakih M1* and Abdallah Dbouk2
1Pediatrics, Faculty of Medical Sciences, Lebanese University, Beirut, LBN, Pediatrics, NICU Division, Sheikh Ragheb Harb University Hospital, Toul, Lebanon
2Pediatrics, Faculty of Medical Sciences, Lebanese University, Beirut, LBN, Pediatrics, Sheikh Ragheb Harb University Hospital, Toul, Lebanon
Submission: June 13, 2024;Published: June 20, 2024
*Corresponding author: Hadi Fakih M, Pediatrics, Faculty of Medical Sciences, Lebanese University, Beirut, LBN, Pediatrics, NICU Division, Sheikh Ragheb Harb University Hospital, Toul, Lebanon
How to cite this article: Hadi Fakih M* and Abdallah Dbouk. A Comprehensive Case Report of OPHN1 X-Linked Intellectual Disability and a Hazardous Association with Familial Mediterranean Fever Heterozygote Mutation. Acad J Ped Neonatol 2024; 14(2): 555936. 10.19080/AJPN.2024.14.555936
Abstract
OPHN1 X-linked intellectual disability (XLID) is a rare genetic disorder characterized by moderate intellectual disability and various neurological and developmental manifestations. This report describes a 4-year-old male with multiple clinical features consisting of dysmorphic facial features, external ear protuberance, failure to thrive, short stature, hypothyroidism, cerebellar and periventricular gliosis and wide based gait with delayed milestones development The case highlights the importance of considering a broad differential diagnosis and the utility of genetic testing in patients with complex multisystem presentations. Early diagnosis and intervention can significantly impact the management and quality of life for affected individuals and for the appropriate genetic counseling for the family.
Keywords: OPHN1; Intellectual; Disability; Cerebellar; Gliosis
Abbreviations: XLID: X-linked Intellectual Disability; PICU: Pediatric Intensive Care Unit; FMF: Familial Mediterranean Fever; MRI: Magnetic Resonance Imaging
Introduction
OPHN1 encodes oligophrenin 1, a Rho-GTPase activating protein involved in synaptic morphogenesis and functions through the regulation of the G protein cycle [1], OPHN1 is related to syndromic X-linked intellectual disability (XLID), it contains 25 exons, located in the Xq12 region, encoding an 802 amino acid Rho GAPs domain [2]. This protein is observed both in glial cells and neurons where it coexists with F-actin, especially at the tip of rising dendrites and at both parts of the synapse [3]. The recognizable phenotype includes intellectual disability, a distinctive facial appearance with vision problem, brain anomalies such as cerebral ventricular augmentation and cerebellar hypoplasia preponderant in the vermis, ataxia, seizures, and endocrine disorders [4]. We report the case of a 4,5 years-old boy diagnosed as having OPHN1 mutation with full clinical and diagnostic investigations that directed us to the final diagnosis.
Case Presentation
The case was a 4,5 YO boy, born on 2019, by an elective cesarean section from no -consanguineous parents with normal first male sibling, he was admitted on March 2024 to the pediatric intensive care unit (PICU), for severe sepsis and pneumonia, with a history of multiple hospitalization due to recurrent fever and infections. given his special features and clinical findings that consist of (Table 1) & (Figure 1). Giving the complex clinical disorders that involved multiple organs and system like the brain, endocrine and the immune system which correlated with positive findings on the MRI imaging of the brain that showed periventricular and cerebellar chronic gliosis (Figure 2).
Associated with a severe bone age delay and resistant hypothyroidism, despite an appropriate hormonal replacement therapy, we decided to perform a comprehensive genetic test that could lead to the likely syndrome or genetic disorder that combines the above cited various features. The whole exome sequencing (WES) confirmed the clinical diagnosis and explained the periodicity of fever that the patient complained of (Figure 3).
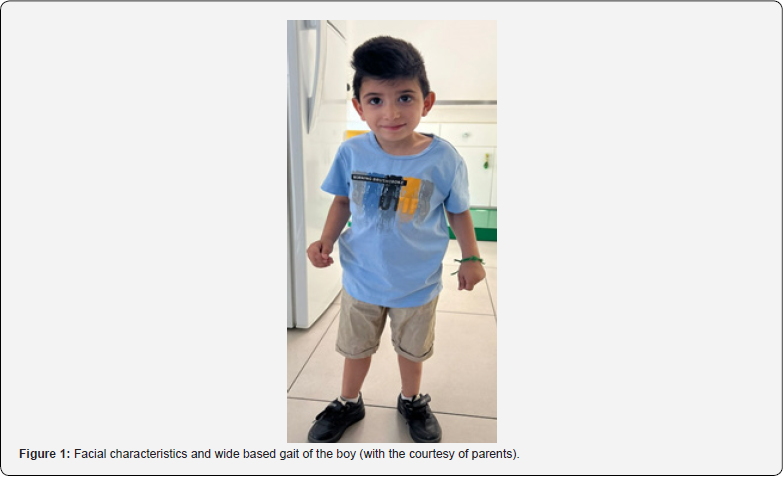
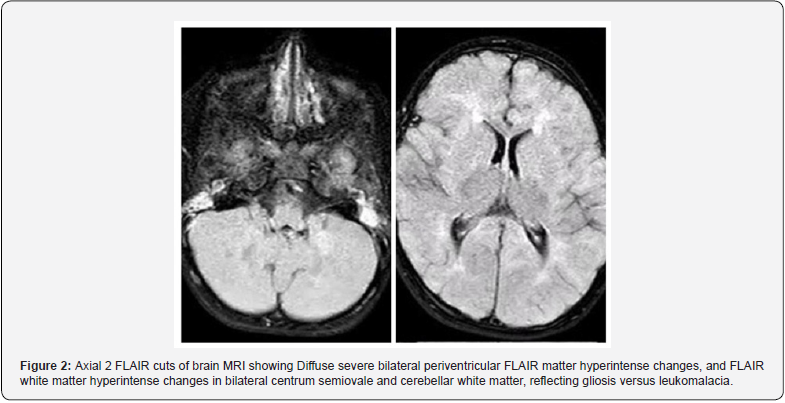
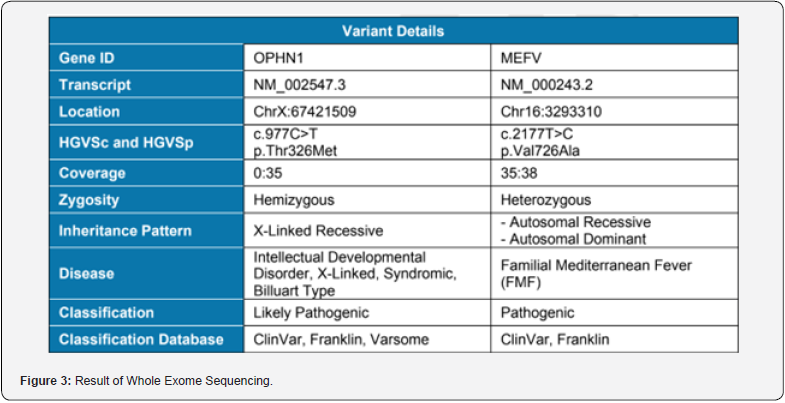
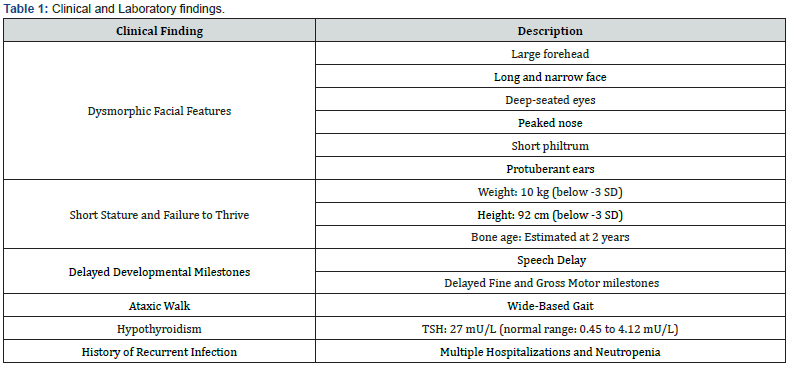
Figure 3 findings:
i. A hemizygous missense variant (c.977C>T, p. Thr326Met) has been detected in OPHN1 gene located at exon 11 out of 25 total exons. This variant has been classified as likely pathogenic variant in Clin Var by single submitter in 2023 (1-star) as it is not observed at significant frequency in large population cohorts (gnom AD); In silico analysis supports that this missense variant has a deleterious effect on protein structure/function and has not been previously published as pathogenic or benign to our knowledge. Franklin database classified this variant as a likely pathogenic variant using the following ACMG criteria (PS4, PM2, and PP5). Varosme also calls this variant a likely pathogenic variant using (PM2, and PP5) ACMG criteria.
ii. A heterozygous missense variant has been found in MEFV gene located at exon 10 out of 10 total exons that is associated with Familial Mediterranean Fever (FMF). This variant (c.2177T>C, p. Val726Ala) is classified as pathogenic/likely pathogenic variant in Clin Var by multiple submitters with no conflicts (2-stars).
Further investigations performed included an abdominal ultrasonography that showed a mild left hydronephrosis, a normal echocardiography and a normal hearing screen test performed at birth.
Discussion
OPHN1 syndrome or X-linked intellectual disability-cerebellar hypoplasia syndrome, is a rare disorder that was discovered in 1998 [5], it is characterized by intellectual disability and changes in the part of the brain and the cerebellum, The syndrome mainly affects males. Signs and symptoms may include intellectual disability, hypotonia, developmental and cognitive delay, earlyonset seizures, abnormal behavior, small or underdeveloped genitals, characteristic facial features that comprises long face, prominent forehead, eye creases, deep set eyes, and protuberant ears, The facial dysmorphism seen in almost all affected patients could be explained by the expression of OPHN1 in the craniofacial bones [6].
Strabismus and ataxic gait and movement are explained by the cerebellum function that is involved in oculomotor coordination, which may contribute to nystagmus and strabismus [7]. The oculomotor problems which seem to appear due to hypoplasia of the cerebellum are many times remarked on brain imaging studies [8]. Additionally, OPHN1 mutations have also been reported in individuals with autism or childhood onset schizophrenia, so OPHN1-associated clinical phenotypes are variable [9]. The most common Magnetic resonance imaging (MRI) brain scan anomalies reported in different studies were cerebellar hypoplasia, especially of the lower vermis, enlarged cisterna magna, or retrocerebellar cysts, the anterior vermis may be disorganized and also hippocampal alteration [10].
The determination of a genetic diagnosis for the patient described in this report significantly altered his care. Prior to the diagnostic finding on WES, with presence of idiopathic short stature, failure to thrive and intellectual disability, his parents seek multiple medical and specialties advices and consultation with large number of tests and investigations performed and therapeutic recommendations. Subsequent to the diagnosis, with an improved understanding of the underlying cause of his clinical condition, the parents are convinced about his final diagnosis and prognosis and focused mainly on performing musculoskeletal, speech and behavioral therapy.
Conclusion
As in this case, a thorough diagnostic evaluation and the performance of appropriate diagnostic test such as Whole Exome Sequencing WES of individuals considered to have intellectual disability, dysmorphism and short stature may provide significant benefit to families and patients via improved understanding of prognosis, therapy, and recurrence risk and solicit the family to perform an early genetic screening for the coming pregnancy.
References
- Nadif Kasri N, Van Aelst L (2008) Rho-linked genes and neurological disorders. Pflugers Arch 455(5): 787-797.
- Mandel JL, Chelly J (2004) Monogenic X-linked mental retardation: is it as frequent as currently estimated? The paradox of the ARX (Aristaless X) mutations. Eur J Hum Genet 12(9): 689-693.
- Zamboni V, Jones R, Umbach A, Alessandra A, Maria P, et al. (2018) Rho gtpases in intellectual disability: from genetics to therapeutic opportunities. Int J Mol Sci 19(6): E1821.
- Santos-Reboucas CB, Belet S, Guedes de Almeida L, et al. (2014) A novel in-frame deletion affecting the BAR domain of OPHN1 in a family with intellectual disability and hippocampal alterations. Eur J Hum Genet 22(5): 644-651.
- Billuart P, Bienvenu T, Ronce N, des Portes V, Vinet MC, et al. (1998) Oligophrenin-1 encodes a rhoGAP protein involved in X-linked mental retardation. Nature 392(6679): 923-926.
- Zanni G, Saillour Y, Nagara M, Billuart P, Castelnau L, et al. (2005) Oligophrenin 1 mutations frequently cause X-linked mental retardation with cerebellar hypoplasia. Neurology 65(9): 1364-1369.
- Menten B, Buysse K, Vermeulen S, Valerie M, Vandesompele J, et al. (2007) Report of a female patient with mental retardation and tall stature due to a chromosomal rearrangement disrupting the OPHN1 gene on Xq12. Eur J Med Genet 50(6): 446-454.
- Ophn1-syndrome (2024) National Organization for Rara Disorders.
- Piton A, Gauthier J, Hamdan FF, Lafrenière RG, Yang Y, et al. (2011) Systematic resequencing of X-chromosome synaptic genes in autism spectrum disorder and schizopherinia. Mol Psychiatry 16(8): 867-880.
- Tzschach A, Grasshoff U, Beck-Woedl S, Claudia D, Claudia B, et al. (2015) Next-generation sequencing in X-linked intellectual disability. Eur J Hum Genet 23(11): 1513-1518.






























