Neonatal Jaundice, Risk Factors and Implication of Direct Coombs Test as a Model of Severity. A Retrospective Study from One Single Center in South Lebanon
Haidar Houmani1, Taghrid Chaaban2,3, Ghassan Ghssein4,5 and Hadi Fakih1,6*
1Department of Pediatrics, Faculty of Medical Sciences, Lebanese University, Beirut, Lebanon
2Nursing Sciences Department, Faculty of Public Health, Islamic University of Lebanon,
3Nursing Sciences Research Chair, Laboratory Educations and Health Practices (LEPS), (EA 3412), UFR SMBH, University Paris 13, Sorbonne Paris Cite, F-93017 Bobigny, France
4Laboratory Sciences Department, Faculty of Public Health, Islamic University of Lebanon (IUL), Khalde P.O. Box 30014, Lebanon
5Faculty of Sciences V, Lebanese University, Nabatieh 1700, Lebanon
6Neonatal Intensive Care Unit, Sheikh Ragheb Harb University Hospital, Toul, Nabatieh District, Lebanon
Submission: May 09, 2024;Published: May 22, 2024
*Corresponding author: Hadi Fakih, Department of Pediatrics, Faculty of Medical Sciences, Lebanese University and Intensive Care Unit, Sheikh Ragheb Harb University Hospital, Toul, Nabatieh District, Beirut, Lebanon
How to cite this article: Haidar Houmani, Taghrid Chaaban, Ghassan Ghssein and Hadi Fakih. Neonatal Jaundice, Risk Factors and Implication of Direct Coombs Test as a Model of Severity. A Retrospective Study from One Single Center in South Lebanon. Acad J Ped Neonatol 2024; 14(1): 555933. 10.19080/AJPN.2024.14.555933
Abstract
Background: Neonatal indirect hyperbilirubinemia is a common disorder in newborns of multifactorial etiologies with different presenting symptoms. Severe jaundice may cause kernicterus, and multiple metabolic disturbances. Objective: To understand the importance of direct coombs test (DCT) and other risk factors in determining the severity and the prognosis of jaundice.
Methods: This is a retrospective study over a 1-year period extending from July 2018 till June 2019. All newborns admitted to the neonatal intensive care unit at a tertiary healthcare center in South Lebanon; with the diagnosis of neonatal hyperbilirubinemia were recruited in the study. Demographic data, laboratory results and the clinical courses of participant’s newborns were recorded. SPSS was used to carry out statistical analyses.
Results: 119 newborns were successfully enrolled in the present study, ABO incompatibility especially O-A, was the most common cause for indirect hyperbilirubinemia (31%). It was also associated with the highest encounter of positive coombs test (31%) results compared to other types of ABO incompatibility (20%). Cases of rhesus incompatibility were associated with higher total and indirect bilirubin peak level; longer hospital stay and longer duration of phototherapy (150.00 ± 55.68 Vs 88.17 ± 37.37 hours). Jaundice with NDCT was associated with urinary tract infection.
Conclusion: Neonatal jaundice should be well investigated to determine the exact etiology and its underlying pathology that will clarify the prognosis, mode of treatment and its economic burden. Urinary tract infection or sepsis remain possible causes of neonatal jaundice when an unidentified cause for the jaundice is identified.
Keywords: Hyperbilirubinemia; Direct Coombs Test; Jaundice; UTI; Sepsis; Phototherapy; Lebanon
Abbreviations: AAP: American Academy of Pediatrics; BG: Blood Group; BWT: Birth Weight; CRP: C-Reactive Protein; DCT: Direct Coombs Test; PDCT: Positive Direct Coombs Test; NDCT: Negative Direct Coombs Test; GA: Gestational Age; HB: Hemoglobin; HDN: Hemolytic Disease of Newborn; ICN: Intensive Care of Neonates; IUGR: Intrauterine Growth Retardation; UTI: Urinary Tract Infection; WBC: White Cell Count
Introduction
One of the most prevalent clinical manifestations in newborns is neonatal jaundice. In infants, jaundice manifests as a yellow coloring of the skin and sclera, signifying an elevated blood bilirubin level that causes bilirubin to accumulate in the tissues (hyperbilirubinemia), encompassing the skin and mucous membranes [1]. Babies with pale skin tones are expected to exhibit jaundice at bilirubin levels of approximately 90 μmol/L [1]. Jaundice affects 60% of term babies and 80% of preterm babies during the first week of life. At one month of age, 10% of breastfed babies still have jaundice [2]. There are two different types of neonatal hyperbilirubinemia; the unconjugated hyperbilirubinemia (UHB) and the conjugated hyperbilirubinemia (CHB). Although high levels of unconjugated bilirubin can infrequently result in kernicterus, which is lifelong brain damage, newborn jaundice is usually not harmful [3]. Additionally, jaundice may indicate a more serious liver problem, such as biliary atresia, for which treatment before the patient is six weeks’ old improves the prognosis [4]. The primary obstacle is to distinguish between the majority of babies whose jaundice will be harmless and the rare infant whose severe jaundice could result in bilirubin encephalopathy and kernicterus [5]. To enhance result, it is also critical to diagnose biliary atresia in newborns with conjugated hyperbilirubinemia as soon as possible. When biliary atresia is surgically corrected in infants before 6 weeks of age, the prognosis is significantly better than when the condition is diagnosed later [5].
In the absence of associated risk factors such as: hemolysis, sepsis, birth trauma or prematurity, it usually resolves within 3-5 days without significant complications, and this is referred to as physiologic neonatal jaundice [6,7]. However, epidemiological evidence suggests that severe neonatal jaundice results in substantial morbidity and mortality [8]. It has been recognized as a significant cause of long-term neurocognitive and other severe sequelae such as: cerebral palsy, auditory neuropathy, deafness and learning difficulties [9]. Treatment of any underlying illness and effective use of phototherapy, which can safely lower bilirubin amounts in most cases, depend on the early detection of jaundice [9].
The role of positive Direct Coombs Test (DCT) in prediction of severity of hemolysis in cases of neonatal jaundice is not very well established. While the American Academy of Pediatrics (AAP) in its 2004 Clinical Practice Guideline considers positive DCT as a major risk factor for severe hyperbilirubinemia and bilirubin neurotoxicity [10,11], other publications regard it as weakly predictive of hyperbilirubinemia [12,13].
The aim of this study is to investigate the role of positive DCT in the assessment of full-term newborns with indirect hyperbilirubinemia and its implication in case management, duration of hospital stays and prognosis. This would be translated practically by early detection and tracking of babies at risk, and by taking extreme precautions especially for the primary care physician to know which newborn will be predisposed to develop an early onset jaundice with neonatal hemolytic anemia that could be a life-threatening complication. So, a close clinical follow-up with determination of serum bilirubin level before and or after his hospital discharge will be encouraged to prevent its adverse effects. On the other hand, the validation of international results with the Lebanese result is a requirement to confirm the use of different risk factors to develop severe neonatal jaundice, and to issue the Lebanese guidelines.
Materials and Methods
Population and Study design
We retrospectively enrolled and studied newborns admitted to the neonatal intensive care unit of a tertiary care teaching hospital, Sheikh Ragheb Harb University Hospital (SRHUH), Toul, Lebanon; with the diagnosis of Neonatal Hyperbilirubinemia treated with phototherapy during a 1-year period extending from July 2018 till June 2019.
Inclusion criteria were full term newborns ≥36 weeks’ gestational age and less than 10 days old whose birth weight (BWT) ≥ 2500g with indirect hyperbilirubinemia requiring phototherapy. Exclusion criteria were newborn babies less than 36 weeks’ gestational age, older than 10 days, less than 2500 grams BWT, syndromic newborns or with multiple malformations, or those with cholestatic jaundice.
Demographic data included gestational age, gender, birth weight, feeding method, mode of delivery and family history of jaundice were collected. In addition, laboratory data including blood group and Rh type of mother and neonate, serum Total, direct and indirect bilirubin levels and its maximum level (peak), DCT, reticulocyte count, C-Reactive Protein (CRP) were collected and considered to determine the etiology of the jaundice and its risk factors. The effect on total days of hospitalization and number of hours of phototherapy needed was recorded.
Ethics Statement
The present study was given approval by the Sheikh Ragheb Harb University Hospital’s (SRHUH) Institutional Review Board (IRB): IRB23RP29. To conduct the study, prior informed consent from the care giver was waived due to the analytical nature of the study. Patients’ complete anonymity was protected, and strict confidentiality was adhered to.
Statistical Analysis
The statistical analyses were performed using the SPSS (IBM Corp., Released 2013, SPSS Statistics for Windows Version 22.0, Armonk, NY, USA). This software was used as well for data management and cleaning. Quantitative variables were tested for normality distribution using the Kolmogorov–Smirnov test. Categorical and normally distributed continuous variables were expressed as frequencies and mean ± standard deviation, respectively. Patients’ demographic and clinical characteristics among the two studied groups were tabulated. Baseline comparisons between groups were performed using the Mann- Whitney U test. T-tests were used if data were normally distributed. The Chi-square test assessed any significant difference between the categorical variables. The significance level was set at P < 0.05 for all statistical analyses.
Results
The total number of neonates admitted to the neonatal intensive care unit for jaundice during the one-year period was 170 cases. After applying the inclusion and exclusion criteria, 51 patients didn’t meet the inclusion criteria. 119 cases remained.
Out of these 119 patients 46.2% (n=55) were males, and 53.8% were females. 19.3% (n=23) of the participant’s newborns were positive for DCT. Considering maternal history, 61.3% (n=73) of mothers were multiparous with 42.9% (n = 51) of newborns were born by vaginal delivery and 57.1% (n=68) by cesarean section. Only 8.4% (n = 10) of newborns in this study were breastfed. According to Table 1, newborns females were tested positive more than newborns males (P < 0.001). Also, babies of multiparous mothers were tested positive more than babies of primiparous mothers (P = 0.020), while the mode of delivery and feeding method had no statistical association (Table 1).
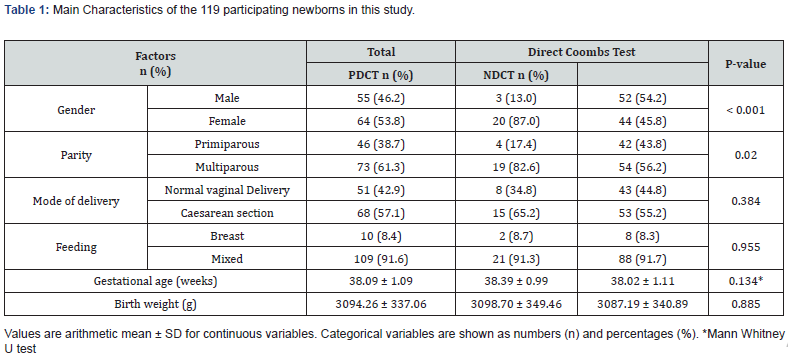

When focusing on maternal and newborn blood groups, the most frequent one in mothers was O positive (76.5%, n = 91) and A positive (45.4%, n = 54) in newborns (Table 2). Overall, 61% (73/119) of cases had ABO incompatibility, with 89% (65/73) of these attributable to maternal blood group O positive and newborn blood group A positive or B positive. ABO blood group incompatibility developed a positive Coombs test result in a total of (19/73) cases. In 27.7% (18/65) of ABO incompatibility patients’ mother was O and newborn A, or B and they were with PDCT, while 12.5% (1/8) was that of other ABO blood group incompatibilities as shown in (Table 2).
In Table 3, according to the Mann Whitney U test, the duration of phototherapy and the peak of bilirubin were higher in O-A newborns who were tested positive than in their O-B counterparts. For babies who were tested Coombs direct positive, the duration of phototherapy and the peak of bilirubin were significantly higher in newborns with Rh incompatibility than babies with ABO incompatibility (P = 0.022 and P = 0.022, respectively).


According to Figure 1, the readmission age and the peak of bilirubin levels were significantly higher in babies with Negative Direct Coombs test (NDCT), in orange, than babies with Positive Direct Coombs test (PDCT), in blue, (P < 0.001). In contrast, the duration of phototherapy and the days of stay were significantly higher in babies with PDCT (in blue) than babies with NDCT (in orange) (P < 0.001).
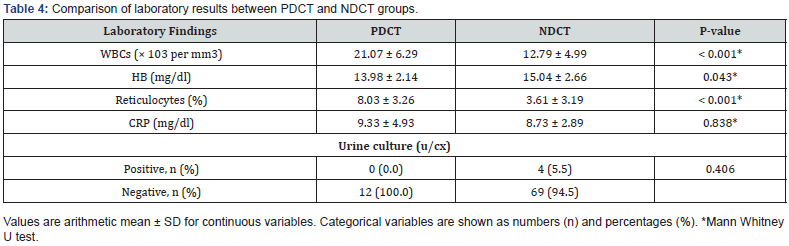
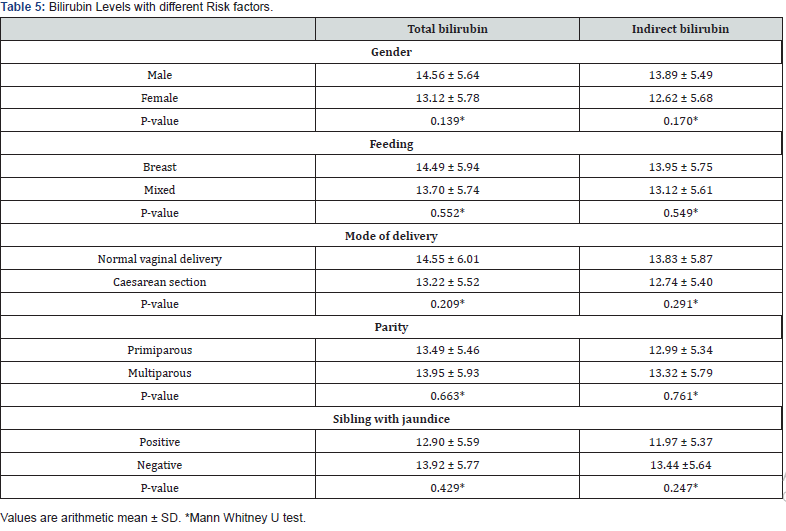
According to Table 4, White Blood Cells (WBCs) count and reticulocytes counts were higher in newborns with PDCT than newborns with NDCT (P < 0.001 and P < 0.001, respectively). However, babies who tested negative had significantly higher hemoglobin (HB) than those who tested positive (P = 0.043). According to Table 5, no significant difference was found in the mean of total and indirect bilirubin according to gender, feeding, and urine culture. No significant difference was also found in the mean of total and indirect bilirubin according to the mode of delivery, parity, or family history with jaundice.
Discussion
Due to the early newborn’s hospital discharges, usually before 48 hours after in-hospital delivery, it is often difficult for the primary care physician to evaluate all newborn infants for the development of jaundice within 48-72 hours of delivery, as is recommended by the American Academy of Pediatrics [10]. Also, there is no recommendation to do the serum bilirubin testing for newborns before hospital discharge except when indicated by the visual assessment of any color changes. However, visual inspection of neonatal jaundice is proven to be inaccurate in detecting hyperbilirubinemia [14], Guidelines in several high-income countries advise universal screening for jaundice in newborns using the noninvasive method of transcutaneous bilirubin (TcB) measurement [15]. The determination of Coombs test is one of the keystones for the diagnosis of neonatal hemolysis. The indirect Coombs test determines anti-D antibodies in the mother’s serum whereas the direct Coombs is used as a screening test for the presence of antibodies on the surface of Red Blood Cells. However, it has low positive predictive value in identifying newborns who will require phototherapy as treatment for neonatal hemolytic disease which depends on the total bilirubin level [16].
Therefore, this study was conducted to investigate possible predictors associated with the development of early neonatal jaundice that would require admission to the neonatal care unit for management. We retrospectively analyzed the blood group and rhesus incompatibility of all full-term newborns admitted to the neonatal intensive care unit with a diagnosis of hemolytic jaundice and their corresponding results of combs tests, as well as other laboratory findings.
Of the 119 newborns enrolled in our study with early indirect hyperbilirubinemia, ABO incompatibility was encountered in 61% (73/119) of cases. Positive DCT due to ABO incompatibility was encountered in 26% (19/73) of cases which is higher than the incidence of 15 % reported by Kaplan and his colleagues [17]. O-A incompatibility was the most frequent association with a higher percentage of PDCT (31%) in comparison with O-B cases where PDCT found in 20% of cases (Table 2). This finding goes along with what was reported in previous studies, which found that hemolysis due to anti‐A is more common than that to anti‐B and was usually associated with a positive direct combs test [18]. Of all ABO blood group incompatibility due to maternal O blood group the mean percentage of PDCT was 27.7%, which is higher than the percentage of 21% reported by Kaplan et al., [18], in comparison to 12.5% when the maternal blood group was either A or B a percentage that is double of (6.9%) reported by Ozolek et al. [19].
Our study revealed that jaundiced babies born to multiparous mothers have higher chance of to have PDCT in comparison to primiparous mothers (P = 0.02). This may be explained by the fact that in case of primiparous mother, the maternal Rh Anti-D antibodies are of the IgM type that cannot cross the placental barrier. In the following pregnancies, a repeat contact with the Rh-D antigen of the baby, induces the rapid synthesis IgG Anti-D antibodies that cross the placenta directly to the fetal circulation where it adheres to the Rh-D antigens of the fetal RBCs leading to PDCT and destroying the antibody coated RBC’s with subsequent severe hemolysis [20].
As for duration of phototherapy, newborns with O-A incompatibility had longer duration of phototherapy and higher peak of bilirubin level, but with insignificant P value, than O-B incompatibility cases in contrast to what was reported before, that an infant whose blood group was A was as likely to be affected by ABO hemolytic disease as a blood group B infant [21].
Newborns with Rh incompatibility had higher peak of bilirubin level and required twice the duration of phototherapy compared to ABO incompatibility, because the fetal RBCs express less of the ABO antigens on their surface compared to the adult RBCs. In addition, the ABO antigens are expressed on variety of fetal tissues, reducing the chances of anti-A and anti-B binding their target antigens on the fetal RBCs [20]. Thus, the clinical presentation of ABO incompatibility with positive antibodies, include hemolysis, and hyperbilirubinemia without significant neonatal anemia. Its successful management is usually achieved by phototherapy only [22].
Our study revealed that newborns with PDCT were younger in age on admission to the neonatal intensive care unit, had higher level of reticulocyte count and white blood cell count, and a lower level of hemoglobin which are consistent with active hemolysis that is characterized by an early onset of jaundice within 24 hours of life, presence of pallor with or without hydrops, presence of hepatosplenomegaly, evidence of hemolysis on the peripheral blood smear, increased reticulocyte count (>8%), rapid rise of bilirubin (>5 mg/dl in 24 h or >0.5 mg/dl/hr) and presence of positive family history of neonatal jaundice [23].
Newborns with NDCT, had higher prevalence of urinary tract infections in comparison to PDCT cases. As it was previously shown, Urinary tract infection (UTI) can cause jaundice in newborns [24]. Also, Francisco and his colleagues in his study, published in 2002, studies the relation between UTI in the newborns and jaundice [23]. Our study confirms that finding in newborns patients with jaundice, mainly those with NDCT.
We found no effect of gender, mode of delivery, parity, family history or types of feeding of the newborns on the level and severity of neonatal jaundice.
Limitations
Small sample size, Single center recruitment, short time period
Conclusion
Neonatal jaundice (OR indirect hyperbilirubinemia) is a common cause of hospitalization in the neonatal period. The presence of positive direct combs test, mild hemolytic anemia, elevated reticulocyte count in a multiparous mother are suggestive of ABO incompatibility. Early recognition and management with phototherapy are associated with good prognosis and reduced morbidity. In the absence of hemolysis, investigation for other causes of jaundice should be started particularly UTI and sepsis if the CRP is elevated. Limitations to our study include the following: Small sample size, Single center recruitment and Short time period.
Acknowledgment
We would like to thank all the parents and their children for their approval to be enrolled in our study, and to thank SRHUH, its general manager and medical direction department, for approving and facilitating our data collection. A special appreciation for the endless effort of all participants who helped make this work finalized, whether persons, nurses or physicians who assisted in turning it to a real medical and scientific achievement.
References
- Battersby C, Michaelides S, Upton M, Rennie JM (2017) Term admissions to neonatal units in England: a role for transitional care? A retrospective cohort study. BMJ Open 7(5): e016050.
- Livesey E, Cortina Borja M, Sharif K, Alizai N, McClean P, et al. (2009) Epidemiology of biliary atresia in England and Wales (1999-2006). Arch Dis Child Fetal Neonatal Ed 94(6): F451-F455.
- McKiernan PJ, Baker AJ, Lloyd C, Mieli-Vergani G, Kelly DA (2009) British paediatric surveillance unit study of biliary atresia: outcome at 13 years. J Pediatr Gastroenterol Nutr 48(1): 78-84.
- Hannam S, McDonnell M, Rennie JM (2007) Investigation of prolonged neonatal jaundice. Acta Paediatrica 89(6): 694-697.
- Mitra S, Rennie J (2017) Neonatal jaundice: aetiology, diagnosis and treatment. Br J Hospital Med 78(12): 699-704.
- (2012) A summary of selected new evidence relevant to NICE clinical guideline 98 ‘Neonatal jaundice’ (2010). National Institute for Health and Care Excellence (NICE), London, England.
- Slusher TM, Zipursky A, Bhutani VK (2011) A global need for affordable neonatal jaundice technologies. Semin Perinatol 35(3): 185-191.
- Bhutani VK, Zipursky A, Blencowe H, Rajesh K, Michael S, et al. (2013) Neonatal hyperbilirubinemia and rhesus disease of the newborn: incidence and impairment estimates for 2010 at regional and global levels. Pediatr Res 74(Suppl 1): 86-100.
- Mwaniki MK, Atieno M, Lawn JE, Charles RJC Newton (2012) Long-term neurodevelopmental outcomes after intrauterine and neonatal insults: a systematic review. Lancet 379(9814): 445-452.
- (2004) American Academy of Pediatrics Subcommittee on: Hyperbilirubinemia. Management of hyperbilirubinemia in the newborn infant 35 or more weeks of gestation. Pediatrics 114(1): 297-316.
- Maisels MJ, Bhutani VK, Bogen D, Newman TB, Stark AR, Watchko JF (2009) Hyperbilirubinemia in the newborn infant. or =35 weeks' gestation: an update with clarifications. Pediatrics 124: 1193-1198.
- Ozolek JA, Watchko JF, Mimouni F (1994) Prevalence and lack of clinical significance of blood group incompatibility in mothers with blood type A or B. J Pediatr 125(1): 87-91.
- Meberg A, Johansen KB (1998) Screening for neonatal hyperbilirubinaemia and ABO alloimmunization at the time of testing for phenylketonuria and congenital hypothyreosis. Acta Paediatr 87: 1269-1274.
- Barrington KJ, Sankaran K (2007) Fetus and Newborn Committee: Guidelines for detection, management and prevention of hyperbilirubinemia in term and late preterm newborn infants. Paediatr Child Health 12(5): 401-418.
- Lease M, Whalen B (2010) Assessing jaundice in infants of 35-week gestation and greater. Curr Opin Pediatr 22(3): 352-365.
- Murray NA, Roberts IA (2007) Haemolytic disease of the newborn. Arch Dis Child Fetal Neonatal Ed 92(2): F83-F88.
- Kaplan M, Na'amad M, Kenan A, Bernard R, Cathy H, et al. (2009) Failure to predict hemolysis and hyperbilirubinemia by IgG subclass in blood group A or B infants born to group O mothers. Pediatrics 123(1): 132-137.
- Kaplan M, Hammerman C, Vreman HJ, Wong RJ, Stevenson DK (2010) Hemolysis and hyperbilirubinemia in antiglobulin positive, direct ABO blood group heterospecific neonates. J Pediatr 157(5): 772-777.
- Chan-Shu SY, Blair O (1979) ABO hemolytic disease of the newborn. Am J ClinPathol 71(6): 677-679.
- Dean L (2005) Blood Groups and Red Cell Antigens [Internet]. Bethesda (MD): National Center for Biotechnology Information (US) Chapter 4, Hemolytic disease of the newborn.
- Ziprin J H, Payne E, Hamidi L, Roberts I, Regan F (2005) ABO incompatibility due to immunoglobulin G anti‐B antibodies presenting with severe fetal anaemia. Transfus Med 15(1): 57-60.
- Frank JE (2005) Diagnosis and management of G6PD deficiency. Am Fam Physician 72(7): 1277-1282.
- Francisco JG, Alan L Nager (2002) Jaundice as an Early Diagnostic Sign of Urinary Tract Infection in Infancy. Pediatrics 109(5): 846-851.
- Harb A, Yassine V, Ghssein G, Salami A, Fakih H (2023) Prevalence and Clinical Significance of Urinary Tract Infection among Neonates Presenting with Unexplained Hyperbilirubinemia in Lebanon: A Retrospective Study. Infect Chemother 55(2): 194-203.






























