Abdominal Compartment Syndrome in Children: Perioperative Concerns and Considerations ACS in Children
David Fanelli1, Carolyn Barbieri1,2, Shannon Grap1,2, Sanjib Adhikary1 and Uma R Parekh1,2*
1Penn State Health Milton S Hershey Medical Center, Hershey, PA, USA
2Penn State Health Children’s Hospital, Hershey, PA, USA
Submission:December 10, 2023;Published: May 02, 2024
*Corresponding author: Uma R Parekh, Penn State Health Milton S Hershey Medical Center, Penn State Health Children’s Hospital, Hershey, PA, USA
How to cite this article: David F, Carolyn B, Shannon G, Sanjib A, Uma R P. Abdominal Compartment Syndrome in Children: Perioperative Concerns 002 and Considerations ACS in Children. Acad J Ped Neonatol 2024; 14(1): 555932. 10.19080/AJPN.2024.14.555932
Abstract
Abdominal compartment syndrome describes new organ dysfunction associated with an acute increase in intra-abdominal pressure. While this condition is well described in adults, this syndrome is underappreciated in children and carries a high mortality rate. The pathophysiology of abdominal compartment syndrome results in deleterious effects on multiple organs systems. However, monitoring for the development of abdominal compartment syndrome remains controversial. Optimal treatment is predicated on prompt recognition of this syndrome and emergent abdominal decompression. Specific considerations of the pathophysiology of abdominal compartment syndrome have important clinical implications for anesthesia providers in the pre, intra and post-operative periods. In this article, we present a review of current literature on this important topic with practical considerations for anesthetic management of these complex patients.
Keywords: Abdominal Compartment Syndrome; Pediatric Anesthesiology Perioperative Optimization; Intrabdominal Hypertension
Abbreviations: ACS: Abdominal Compartment Syndrome; APP: Abdominal Perfusion Pressure; CVP: Central Venous Pressure; CVPEE: End-Expiratory CVP; IAH: Intra-Abdominal Hypertension; IAP: Intra-Abdominal Pressure; MAP: Mean Arterial Pressure; PPLAT: Airway Plateau Pressure; CVPTM: Transmural Central Venous Pressure; WSACS: World Society on Abdominal Compartment Syndrome
Introduction
Abdominal compartment syndrome (ACS) describes new organ dysfunction associated with an acute increase in intra-abdominal pressure (IAP) [1]. While this syndrome is well described in adults, it is often underappreciated in children and carries a high mortality rate [2]. Anesthesiologists often encounter these children when they present for emergent exploratory laparotomy for abdominal decompression. The authors recently managed a 5-year-old child who was presented for exploratory laparotomy for increasing abdominal distension. She had a persistent metabolic acidosis and concern for bowel ischemia based on distended bowel loops with portal venous gas in the liver on abdominal x-ray. IAP was not measured and thus the progression to abdominal compartment syndrome was not appreciated. The girl suffered cardiac arrest soon after the induction of anesthesia. Subsequent abdominal decompression allowed successful resuscitation of the patient. The preoperative diagnosis of ACS is crucial as the induction and maintenance of anesthesia can exacerbate its pathophysiologic effect and lead to adverse outcomes.
Discussion
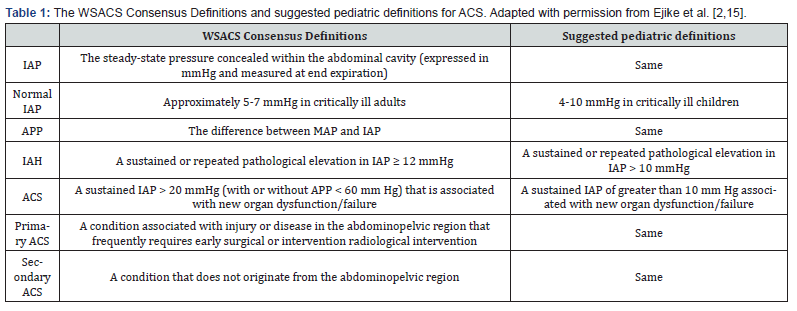
The consequences of increased intra-abdominal hypertension (IAH), specifically its effects on the cardiovascular, pulmonary and renal systems are well described [3,4]. In pediatric populations, the detrimental effect of increased IAP following repair of omphalocele and gastroschisis are well known [5]. Despite this, emergent abdominal decompression for ACS in children was not described until 2000 [6-8]. In 2004, the World Society on Abdominal Compartment Syndrome (WSACS) was founded and published consensus definitions in 2006 [9]. These definitions were last updated in 2013 and include clinical practice guidelines and recommendations for both the adult and pediatric population [10] (Table 1).
Incidence and Risk Factors for Intraabdominal Hypertension and Abdominal Compartment Syndrome in Children
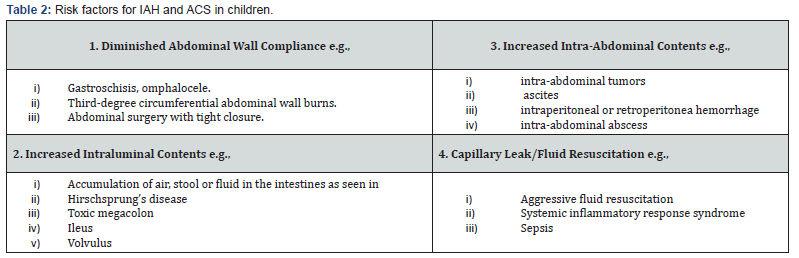
In adults the reported incidence of IAH varies from 18% to 81% and that of ACS is between 1% and 60% [11,12]. In children, the incidences of IAH and ACS are lower at 9% and 4% respectively [13]. This lower incidence may be explained by the greater distensibility of the abdominal wall in children [14]. Additionally, ACS is still likely underreported in children despite a high mortality range of 40-60%.13 WSACS has categorized risk factors in adults for developing ACS which have been modified to reflect common pathology for pediatric patients (Table 2) [2,10]. Additional parameters including abdominal distension and a ventilation plateau pressure of more than 30 mmHg were also found to be independent predictors of IAH [14].
Pathophysiology of Abdominal Compartment Syndrome
ACS is due to an increase in IAP either from an increase in abdominal contents, a decrease in abdominal wall compliance, or both. Although data is limited, Ejike et al, have demonstrated that IAP in children during critical illness is between 4- and 10-mm Hg [15]. Based on this, WSCAS defined IAH in children as a pathological elevation in IAP > 10 mmHg [9,10]. Progressive increases in IAP have deleterious effects on nearly every abdominal and extra-abdominal end organ function. An increase in IAP decreases splanchnic, hepatic and renal perfusion. Animal experiments using radioactive microspheres to measure intra-abdominal organ blood flow demonstrate a decrease in blood flow to all intra-abdominal organs (except adrenal glands) when IAP is increased to > 20 mmHg [16]. Mesenteric arterial and mucosal blood flow decreases significantly at an IAP of 20 mmHg despite minimal changes in cardiac output in anesthetized pigs [17]. In patients undergoing laparoscopy, gastric mucosal oxygen saturation decreases with moderate increases in IAP to 8 mmHg [18]. The resulting intestinal ischemia may cause bacterial translocation across the gut mucosa into the systemic circulation leading to the release of pro-inflammatory cytokines and a systemic inflammatory response [19]. Glucose metabolism, lactate clearance, cytochrome p450 activity and mitochondrial function are also deranged leading to metabolic acidosis [2].
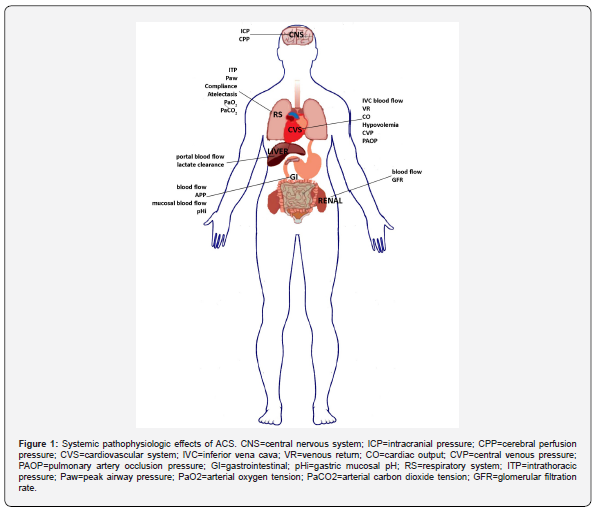
ACS is associated with a decrease in renal blood flow and glomerular filtration rate due to direct compression of the kidneys, reduced cardiac output and an increase in renal vascular resistance. This results in reduced urine output and worsening fluid retention [20,21]. Oliguria occurs between 15 and 20 mmHg of IAP which can progress to anuria when IAP exceeds 30 mmHg [21]. Lung function may also be affected, as rising IAP causes cephalad displacement of the diaphragm leading to increased intra-thoracic and pleural pressures which results in a progressive reduction in lung and chest wall compliance and atelectasis [22]. This leads to increased ventilatory-perfusion mismatch and pulmonary dead space resulting hypoxia and hypercapnia. Collectively, this may precipitate the need for mechanical ventilation. Hemodynamics alterations are observed when IAP increases > 20 mmHg through changes in preload, afterload and intrathoracic pressure. Direct compression of the inferior vena cava and portal vein decreases preload and consequently cardiac output. Increased intra-thoracic pressure from elevation of the diaphragm leads to compression of the heart, decreasing ventricular end-diastolic volume and consequently cardiac output [23]. Increased systemic vascular resistance due to direct compression of abdominal vascular beds results in an initial increase in mean arterial pressure. However, a subsequent decline in arterial pressure occurs as the left ventricular compliance decreases from increasing afterload resulting in decreased cardiac output. The lower cardiac output contributes to poor perfusion and accelerates multi-organ failure. Finally, IAH may elevate intracranial pressure especially in patients with concomitant head injuries due to functional obstruction of cerebral venous flow [24]. Because of these multisystem effects, untreated ACS can result in multi-organ failure and death [2].
Measurement of Intra-abdominal Pressure
Clinical examination is an inaccurate predictor of IAP and should not be substituted for IAP measurement [25]. The most accurate method of determining IAP is direct transcutaneous measurement by needle or catheter placement in the peritoneal space with pressure wave transduction [26]. Complications of this approach include bowel perforation and peritoneal contamination [17]. Indirect methods of measurement include intravesical, gastric, rectal, uterine, inferior vena cava and airway pressure measurements. Of these, intra-vesicular measurement, has become the gold standard [9]. This method is performed via a urethral catheter placed in the bladder after instilling 1 ml/kg (minimum of 3 ml to a maximum of 25 ml) of saline [18]. The level of the pubic symphysis serves as a reference point allowing transduction of the pressure gradient between the bladder and the transducer. Common sources of error with the intravesical method of IAP measurement include air bubbles in the system, incorrect positioning of the pressure transducer and catheter blockage [26]. The WSACS recommends measuring IAP when any known risk factors for IAH/ACS are present in critically ill or injured patients. If IAH is detected, IAP monitoring should be used for detection and management of ACS [9]. In spite of these recommendations, intra-vesicular measurements are not done frequently as they are time consuming, discontinuous, observer dependent and increase risk of urinary tract infections. Recently, an air-capsule-based measurement of intra-gastric pressure has been described which is continuous, fully automated, and operator-independent. In this method, IAP is monitored via a customized nasogastric tube and agrees favorably with IVP [27].
Prevention and Management of Abdominal Compartment Syndrome
Conservative management of IAH is predicated on halting the progressive rise of IAP. Medical options to reduce IAP include improving abdominal wall compliance (e.g. use of neuromuscular blockade, or analgesia), evacuation of intra-luminal contents via gastric or rectal tube, evacuation of abdominal fluid collections via paracentesis and optimization of fluid balance [10,28]. Specific to pediatric patients, WSACS recommends maintaining a high degree of clinical suspicion for IAH/ACS. This includes measuring IAP in the presence of known risk factors, utilization of protocolized monitoring in critically ill patients, percutaneous catheter drainage when technically feasible, negative pressure wound therapy for open abdominal wounds and avoidance of excessive fluid resuscitation [1]. The endpoint of resuscitative therapy remains controversial and was not specifically addressed for pediatric patients in the 2013 WSACS guidelines.10 However, abdominal perfusion pressure (APP) defined as mean arterial pressure (MAP) minus IAP has been suggested to be a superior predictor of end organ damage than IAP alone [25,29]. APPs of 50-60 mmHg may be a better resuscitation endpoint compared to arterial pH, base deficit, arterial lactate or hourly urine output in adult surgical patients [19]. In children, due to a wider range of age-appropriate MAPs, a single value of IAP or APP is impractical. Pediatric studies defining criteria for decompression and adequate IAPs are warranted. When IAH progresses to ACS despite medical measures, decompressive laparotomy should be undertaken [28]. The goal of laparotomy is to reverse the effects of end organ dysfunction and improve cardiac preload, decrease airway pressures, lower oxygen requirements and restore hemodynamic stability [30].
Anesthetic Implications in Children with Abdominal Compartment Syndrome
ACS in pediatric patients creates challenges for anesthesia providers in the peri, intra and post-operative periods. Perioperatively, patients presenting with ACS to the operating room often do so emergently and consequently may not have time for adequate perioperative optimization. These patients often have a tenuous volume status and clinical evaluation is challenging. A positive fluid balance has been associated with organ dysfunction, and thus colloid and hypertonic fluid may be preferred to crystalloids for resuscitation [31]. However, overly aggressive fluid resuscitation may contribute to further development of ACS as noted by Pearson et al in children where large volume resuscitation was required.30 Pediatric patients with ACS also typically present with a degree of renal dysfunction that may affect drug metabolism and volume of distribution [21]. Venous access in the upper extremities is preferred as blood flow to the lower extremities may be compromised from compression of the IVC [32]. Intraoperatively, anesthetic induction in these children may result in further hemodynamic deterioration. As previously discussed, ACS is associated with decreased cardiac preload, output and pulmonary reserve. Induction with propofol or methohexital should be titrated cautiously because of their depressive effect on the cardiovascular system [33,34]. Alternatively, ketamine may be utilized with less myocardial depression.34 While etomidate maintains cardiovascular stability, it is now associated with greater 30-day mortality, cardiovascular morbidity and increased length of hospital stay in adults [35]. The effects of etomidate on adrenocorticoid suppression are still under investigation in pediatric patients [36]. Regardless of strategy, all induction agents should be titrated carefully to clinical effect. Neuromuscular blockers should be utilized to facilitate laryngoscopy and improve abdominal wall compliance [10]. Due to the tenuous clinical condition of these patients, the attending surgeon should be present for induction of anesthesia and be prepared to perform a decompressive laparotomy in the event of acute cardiovascular collapse.
Intraoperative ventilation and oxygenation should be optimized with appropriate alveolar recruitment measures and PEEP. Providers should be aware that excessive PEEP or tidal volume can further reduce preload and cardiac output [37]. Above an IAP of 5 mmHg, 50% of the IAP is transmitted to airway plateau pressure (PPLAT) at a constant tidal volume. Hence, airway pressure can best be interpreted as PPLAT = Plateau - 0.5 * IAP.10, [38]. Therefore, anesthesia providers should focus on controlling tidal volume instead of restricting PPLAT to ensure adequate ventilation [39]. The IAP should be monitored intraoperatively to aid in optimization of ventilation prior to decompressive laparotomy, during abdominal closure and postoperatively. In patients undergoing a procedure other than abdominal surgery, abdominal pressures should be carefully monitored throughout the period of anesthesia management. Resuscitation in these critically ill children should be further guided by invasive monitoring including central venous pressure, arterial pressure and arterial blood gas analysis as indicated. However, anesthesia providers need to be mindful with interpretation invasive monitor data. Transmission of IAP to intrathoracic pressure (ITP) can lead to erroneously elevated intracardiac filling pressures that are not reflective of true intravascular volume status.39 Therefore, measuring transmural central venous pressure (CVPTM) is recommended and can be calculated by end-expiratory CVP (CVPEE) - ITP. Assuming 50% of IAP is transmitted to the thorax, CVPTM can be calculated as: CVPTM = CVPEE - IAP*0.5 [32,40].
Following successful decompression, there may be immediate circulation of vasoactive mediators like effect seen in limb compartment syndrome [28,41]. Anesthesia providers should be prepared to treat resulting hypotension and/or arrythmias [41]. Following successful decompression, the decision to remain intubated is nuanced and dependent on multiple factors including patient age, success of abdominal closure, hemodynamic status, and fluid status. The abdomen may be left open postoperatively in selected patients to prevent ACS [42]. Respiratory mechanics during abdominal wall closure should be closely monitored to ensure adequate ventilation. However, peak airway pressures do not accurately reflect IAP. Therefore, airway pressures as a surrogate for IAP should be used with caution [43]. Patients may return to the operating room over several days for a staged closure.
Conclusion
Although ACS is associated with a high mortality there is little knowledge about the optimal method to manage this condition. A survey of pediatric intensive care physicians established that 24% were unaware of the utility of bladder pressure measurement while 33% would not consider use of decompressive laparotomy to treat ACS [44]. Increased awareness of the implications of ACS and tools for management may improve outcomes in pediatric patients. This has been shown in adults where evidence-based management of IAH and ACS, including prophylactic use of an open abdomen, resulted in increased survival and a decrease in resource utilization [45]. A similar approach in pediatric patients may be warranted. Pearson et al suggest that early decompressive laparotomy in patients with elevated ventilatory pressures, persistent oliguria, serum lactate greater than 3mg/dL, or elevated intravesical pressures.30 Extracorporeal Membrane Oxygenation may be part of a strategy to manage these complex patients with open or closed abdomens [46]. Additional research is needed to establish the role of IAP measurements in improving outcomes in pediatric patients as well as more discrete criteria for management of this potentially fatal condition.
References
- Kron IL, Harman PK, Nolan SP (1984) The measurement of intra-abdominal pressure as a criterion for abdominal re-exploration. Ann Surg 199(1): 28-30.
- Ejike JC, Mathur M, Moores DC (2011) Abdominal compartment syndrome: focus on the children. Am Surg 77(Suppl 1): S72-S77.
- Burch JM, Moore EE, Moore FA, Franciose R (1996) The abdominal compartment syndrome. Surg Clin North Am 76(4): 833-842.
- Cheatham ML, Malbrain MLNG (2007) Cardiovascular implications of abdominal compartment syndrome. Acta Clin Belg 62(Suppl 1): 98-112.
- Lacey SR, Carris LA, Beyer AJ, Azizkhan RG (1993) Bladder pressure monitoring significantly enhances care of infants with abdominal wall defects: A prospective clinical study. J Pediatr Surg 28(10): 1370-1375.
- Gross RE (1948) A new method for surgical treatment of large omphaloceles. Surgery 24(2): 277-292.
- Neville HL, Lally KP, Cox CS (2000) Emergent abdominal decompression with patch abdominoplasty in the pediatric patient. J Pediatr Surg 35(5): 705-708.
- Decou JM, Abrams RS, Miller RS, Gauderer MWL (2000) Abdominal compartment syndrome in children: Experience with three cases. J Pediatr Surg 35(6): 840-842.
- Malbrain MLNG, Cheatham ML, Kirkpatrick A, Michael S, Michael P, et al. (2006) Results from the International Conference of Experts on Intra-abdominal Hypertension and Abdominal Compartment Syndrome. I. Definitions. Intensive Care Med 32(11): 1722-1732.
- Kirkpatrick AW, Roberts DJ, De Waele J, Roman J, Manu LNG Malbrain, et al. (2013) Intra-abdominal hypertension and the abdominal compartment syndrome: Updated consensus definitions and clinical practice guidelines from the World Society of the Abdominal Compartment Syndrome. Intensive Care Med 39(7):1190-1206.
- Kuteesa J, Kituuka O, Namuguzi D, Cynthia N, Samuel K, et al. (2015) Intra-abdominal hypertension; prevalence, incidence and outcomes in a low resource setting; a prospective observational study. World J Emerg Surg 10(1).
- Malbrain MLNG, Chiumello D, Pelosi P, David B, Richard I, et al. (2005) Incidence and prognosis of intraabdominal hypertension in a mixed population of critically ill patients: A multiple-center epidemiological study. Crit Care Med 33(2): 315-322.
- Divarci E, Karapinar B, Yalaz M, Ergun O, Celik A (2016) Incidence and prognosis of intraabdominal hypertension and abdominal compartment syndrome in children. J Pediatr Surg 51(3): 503-507.
- Thabet FC, Bougmiza IM, Chehab MS, Bafaqih HA, Almohaimeed SA, et al. (2016) Incidence, Risk Factors, and Prognosis of Intra-Abdominal Hypertension in Critically Ill Children: A Prospective Epidemiological Study. J Intensive Care Med 31(6): 403-408.
- Ejike JC, Bahjri K, Mathur M (2008) What is the normal intra-abdominal pressure in critically ill children and how should we measure it? Crit Care Med 36(7): 2157-2162.
- Caldwell CB, Ricotta JJ (1987) Changes in visceral blood flow with elevated intraabdominal pressure. J Surg Res 43(1): 14-20.
- Diebel LN, Dulchavsky SA, Wilson RF (1992) Effect of increased intra-abdominal pressure on mesenteric arterial and intestinal mucosal blood flow. J Trauma - Inj Infect Crit Care 33(1): 45-49.
- Schwarte LA, Scheeren TWL, Lorenz C, De Bruyne F, Fournell A (2004) Moderate Increase in Intraabdominal Pressure Attenuates Gastric Mucosal Oxygen Saturation in Patients Undergoing Laparoscopy. Anesthesiology 100(5): 1081-1087.
- Rezende-Neto JB, Moore EE, De Andrade MVM, Mauro Martins T, Felipe Asis L, et al. (2002) Systemic inflammatory response secondary to abdominal compartment syndrome: Stage for multiple organ failure. J Trauma - Inj Infect Crit Care 53(6): 1121-1128.
- Bradley SE, Bradley GP (1947) The Effect of Increased Intra-Abdominal Pressure on Renal Function in Man. J Clin Invest 26(5): 1010-1022.
- Harman PK, Kron IL, McLachlan HD, Freedlender AE, Nolan SP (1982) Elevated intra-abdominal pressure and renal function. Ann Surg 196(5): 594-597.
- Ridings PC, Bloomfield GL, Blocher CR, Sugerman HJ (1995) Cardiopulmonary effects of raised intra-abdominal pressure before and after intravascular volume expansion. J Trauma - Inj Infect Crit Care 39(6): 1071-1075.
- Silveira LGT, Brocca IC, Moraes ES, Brandão MB, Nogueira RJN, et al. (2021) Hemodynamic effects of increased intra-abdominal pressure in critically ill children. J Pediatr (Rio J) 97(5): 564-570.
- Citerio G, Vascotto E, Villa F, Celotti S, Pesenti A (2001) Induced abdominal compartment syndrome increases intracranial pressure in neurotrauma patients: A prospective study. Crit Care Med 29(7):1466-1471.
- Sugrue M, Bauman A, Jones F, Gillian Bishop, Arthas F, et al. (2002) Clinical examination is an inaccurate predictor of intraabdominal pressure. World J Surg 26(12): 1428-1431.
- Malbrain MLNG (2004) Different techniques to measure intra-abdominal pressura (IAP): Time for a critical re-appraisal. Intensive Care Med 30(3): 357-371.
- Kaussen T, Gutting M, Lasch F, Boethig D, Von Gise A, et al. (2021) Continuous intra-gastral monitoring of intra-abdominal pressure in critically ill children: a validation study. Intensive Care Med Exp 9(1): 24.
- Gottlieb M, Davenport DV, Adams S, Chien N (2019) Current Approach to the Evaluation and Management of Abdominal Compartment Syndrome in Pediatric Patients. Pediatr Emerg Care 35(12): 874-878.
- Horoz OO, Yildizdas D, Sari Y, Unal I, Ekinci F, et al. (2019) The relationship of abdominal perfusion pressure with mortality in critically ill pediatric patients. J Pediatr Surg 54(9): 1731-1735.
- Pearson EG, Rollins MD, Vogler SA, Megan K Mills, Elizabeth L Lehman, et al. (2010) Decompressive laparotomy for abdominal compartment syndrome in children: before it is too late. J Pediatr Surg 45(6): 1324-1329.
- Oda J, Ueyama M, Yamashita K, Takuya I, Mitsuhiro N, et al. (2006) Hypertonic lactated saline resuscitation reduces the risk of abdominal compartment syndrome in severely burned patients. J Trauma - Inj Infect Crit Care 60(1): 64-71.
- Malbrain MLNG, De Waele JJ, De Keulenaer BL (2015) What every ICU clinician needs to know about the cardiovascular effects caused by abdominal hypertension. Anaesthesiol Intensive Ther 47(4): 388-399.
- Zausig YA, Busse H, Lunz D, Sinner B, Zink W, et al. (2009) Cardiac effects of induction agents in the septic rat heart. Crit Care 13(5): R144.
- Appel E, Dudziak R, Palm D, Wnuk A (1979) Sympathoneuronal and sympathoadrenal activation during ketamine anesthesia. Eur J Clin Pharmacol 16(2): 91-95.
- Komatsu R, You J, Mascha EJ, Sessler DI, Kasuya Y, et al. (2013) Anesthetic induction with etomidate, rather than propofol, is associated with increased 30-day mortality and cardiovascular morbidity after noncardiac surgery. Anesth Analg 117(6): 1329-1337.
- Sokolove PE, Price DD, Okada P (2000) The safety of etomidate for emergency rapid sequence intubation of pediatric patients. Pediatr Emerg Care 16(1): 18-21.
- Regli A, Mahendran R, Fysh ET, Brigit R, Bart L De Keulenaer, et al. (2012) Matching positive end-expiratory pressure to intra-abdominal pressure improves oxygenation in a porcine sick lung model of intra-abdominal hypertension. Crit Care 16(5).
- Wauters J, Wilmer A, Valenza F (2007) Abdomino-thoracic transmission during ACS: Facts and figures. Acta Clin Belg 62(Suppl 1): 200-205.
- Cortes-Puentes GA, Gard KE, Adams AB, Katherine AF, Christopher PA, et al. (2013) Value and limitations of transpulmonary pressure calculations during intra-abdominal hypertension. Crit Care Med 41(8): 1870-1877.
- De Laet I, Malbrain MLNG (2007) ICU management of the patient with intra-abdominal hypertension: What to do, when and to whom? Acta Clin Belg 62(Suppl1): 190-199.
- Moore AFK, Hargest R, Martin M, Delicata RJ (2004) Intra-abdominal hypertension and abdominal compartment syndrome. Br J Surg 91(9): 1102-1110.
- Einav S, Zimmerman FS, Tankel J, Leone M (2021) Management of the patient with the open abdomen. Curr Opin Crit Care 27(6): 726-732.
- Bunnell A, Cheatham ML (2015) Airway pressures as surrogate estimates of intra-abdominal pressure. Am Surg 81(1): 81-85.
- Kimball EJ, Rollins MD, Mone MC, Hiedi JH, Gabriele KB, et al. (2006) Survey of intensive care physicians on the recognition and management of intra-abdominal hypertension and abdominal compartment syndrome. Crit Care Med 34(9): 2340-2348.
- Cheatham ML, Safcsak K (2010) Is the evolving management of intra-abdominal hypertension and abdominal compartment syndrome improving survival? Crit Care Med 38(2): 402-407.
- Brown J, Warnock B, Turk E, Hobson MJ, Friedman ML, et al. (2022) Open abdomen during extracorporeal membrane oxygenation is a safe and effective treatment for abdominal compartment syndrome. J Pediatr Surg 57(9): 216-222.






























