Plasmatic Concentrations of Vitamin D in Pregnant Women During the Last Trimester and in Umbilical Cord Blood
Mery A Muñoz Percy1, Ruxandra Simona Bacanu2 and Mauricio Moreno López2
1Santa Bárbara Hospital Ciudad Real, Spain
2Tomelloso General Hospital Ciudad Real, Spain
Submission:August 31, 2023;Published: December 15, 2023
*Corresponding author: Mauricio Moreno López, Tomelloso General Hospital Ciudad Real, Vereda de Socuéllamos, s/n, 13700. Tomelloso Ciudad Real, Spain
How to cite this article: Mery A Muñoz P, Ruxandra Simona B, Mauricio Moreno L. Plasmatic Concentrations of Vitamin D in Pregnant Women During the Last Trimester and in Umbilical Cord Blood. Acad J Ped Neonatol 2023; 13(3): 555917. 10.19080/AJPN.2023.13.555917
Abstract
Fundaments: a raise in vitamin D deficiency has been reported in Spain in recent years, being the health of both mother and newborn also affected by said deficit. The goal of the present work was to evaluate the behavior of vitamin D in the third trimester of pregnancy and in the umbilical cord blood, as well as establish a relation with associated factors in a group of pregnant women in Tomelloso General Hospital, Spain. Methods: through descriptive and correlational investigation, 346 mother-newborn pairs were evaluated from May 2022 to April 2023. Clinical history data were extracted; mother age, pregnancy age, method of birth, polyvitamin intake, ethnicity, skin phenotype, style of clothing worn, anthropometrical characteristics and gender of newborn. Biochemical variables were determined on the third trimester, and the sample for the umbilical cord was gotten after the pinching, defining deficiency of 25 (OH) D3 as every value inferior to 20 ng/ml. Intake of calcium and vitamin D was evaluated, as well as solar exposure and skin phototypes. Descriptive and non-parametrical statistics and correlations between variables and groups were applied. Results: a high percentage of deficiency (72% between severe and moderate deficit) was observed, with adequate vitamin D intake in the majority. In the blood of the umbilical cord was reported a 61,3% deficiency, with a tendency to severe deficit in the winter. There were significances between the levels of vitamin D according to the type of clothing worn, with a higher range average in women who wore regular clothes as opposed to those who covered their head and neck, being the group of pregnant Gypsy and Moroccan women the ones that showed lower values of Vitamin D, despite belonging to countries where solar radiation is very high. There was also correlation between vitamin D and solar exposition, levels of parathormone, and cord blood vitamin D. Conclusion: there was a high deficiency of vitamin D both in the mother and in the cord blood, and therefore in the newborns. The necessity to make effective programs from early gestation to prevent severe problems in the mother-newborn duo in public health is proposed.
Keywords: Vitamin D; Parathormone; Gestation; Cord Blood; Solar Exposition
Introduction
Currently, vitamin D, besides being an essential micronutrient related to calcium and phosphorus homeostasis [1], has also been considered a prohormone involved in a complex endocrine system that regulates mineral homeostasis, protects musculoskeletal integrity, modulates growth, and cell differentiation in a wide variety of tissues [1]. It is synthesized in the skin through the effects of ultraviolet radiation, accounting for 80% of its production (the primary source for humans), with the rest coming from a complete and healthy diet. The primary natural sources of vitamin D are fatty fish, cod liver oil, beef liver, and egg yolk [2]. Unfortunately, the consumption of these foods does not meet the requirements for the entire population (less than 10% of vitamin D comes from dietary sources), and combined with limited vitamin D supplementation, vitamin D deficiency has become significant in the region [1,2]. In Spain, epidemiological studies in the general population have reported an increase in vitamin D deficiency in recent years [3,4,5]. The impact of this deficiency has also affected the health of both mothers and newborns, which is why its study has gained special interest in recent years [1,6,7]. Adequate vitamin D levels in pregnant women have been described as important for both their own health and that of their offspring, with maternal stores being the only way to ensure an adequate level of vitamin D at birth [8]. Vitamin D is transferred from the mother to the fetus through the placenta, with the fetus being entirely dependent on maternal vitamin D. Since the half-life of 25(OH) D3 is relatively short, after birth, newborns can only have adequate levels for up to 8 weeks, which exposes them to a higher risk of deficiency during the neonatal period, potentially leading to the development of hypocalcemia and rickets during this time [9,10,11,12].
Studies indicate a positive correlation between maternal blood concentrations of 25(OH) D3 and umbilical cord concentrations; umbilical cord 25(OH) D3 levels are between 50% and 80% of maternal values, and in newborns, they are around 60% to 75% of maternal levels [13,14,15]. Consequently, newborns of mothers with vitamin D deficiency could also be deficient at birth. After birth, in exclusively breast-fed infants, the source of vitamin D depends on sunlight exposure and the intake of vitamin D contained in breast milk [16,17]. The milk of healthy women contains relatively small amounts of vitamin D, which is considered insufficient to prevent vitamin D deficiency in infants without adequate sunlight exposure [8,9]. This could have significant implications for population health, as vitamin D deficiency is associated with various pathologies such as cardiovascular diseases, type I and II diabetes, asthma, inflammatory bowel disease, certain types of cancer, and certain neurological diseases, collectively contributing to a higher burden of morbidity and mortality worldwide [18,19]. Sun exposure enables cutaneous vitamin D synthesis, with the effective UV radiation dose for this synthesis being calculated considering that 25% of the minimal UV radiation dose is necessary on 20% of the body surface (hands, arms, and face). The required irradiation dose for minimal vitamin D production in the skin also depends on the skin phototype; the darker the skin, the greater the required sunlight dose, as melanin competes with active photons for vitamin D production [20,21]. However, it has recently been described that during pregnancy, cutaneous synthesis does not seem sufficient to meet vitamin D needs, which could have implications for neonatal public health [22,23]. Considering these pieces of evidence, the objective of this study has been to evaluate the behavior of serum vitamin D during the third trimester of gestation and in umbilical cord blood, as well as its relationship with associated factors in a group of adult pregnant women at the General Hospital of Tomelloso (Ciudad Real), Spain.
Materials and Methods
Descriptive, Cross-Sectional Correlational Research. The population consisted of 346 mother-newborn pairs, attended at the General Hospital of Tomelloso (Ciudad Real) from May 2022 to April 2023, with prior informed consent. The study was approved by the Ethics Committee for Clinical Research of Hospital La Mancha Centro, adhering to the principles of the Declaration of Helsinki. The inclusion criterion was being pregnant in the last trimester and giving birth at the General Hospital of Tomelloso (Ciudad Real), in addition to signing the corresponding consent to participate in the research. Pregnant individuals who declined to participate in the study were excluded, as well as those who experienced labor onset before 35 weeks of gestation and those with any medical emergency endangering the life of the mother or fetus.
Data Collection
Epidemiological Assessment:
For data collection, information was extracted from the medical records, including maternal age, gestational age, type of delivery, intake of multivitamins, weight gain during pregnancy, smoking habits, ethnicity, skin phenotype, clothing style worn, amniotic fluid characteristics, anthropometric characteristics, newborn’s gender, and APGAR score.
Maternal and Umbilical Cord Biochemical Assessment:
Maternal biochemical variables were determined during routine third-trimester analysis (prenatal control), and for umbilical cord blood, samples were collected immediately after clamping. The samples were centrifuged and stored at -18°C, protected from light in the laboratory freezer at the Hospital, until they were sent to the Hospital Valld’Hebron laboratory for subsequent analysis. Levels of 25(OH) D (LIAISON® 25 OH Vitamin D Total Assay) and Parathormone (LIAISON® N – TACT® PTH II Assay) were determined, considering the following criteria: PTH: Normal range of values between 15-65 pg/ml. The levels of 25(OH) D were defined as: Severe deficiency: levels below 10 ng/ml Moderate deficiency: levels between 10 and 20 ng/ml Insufficiency: levels between 21 and 29 ng/ml Sufficiency: levels above 30 ng/ml. For the evaluation of biochemical parameters and phosphocalcic metabolism, the following normal values were considered: Calcium: range of values between 8.6 and 10.2 mg/dl. Phosphorus: range of values between 2.7 and 4.5 mg/dl.
Magnesium:
Range of values between 1.5 and 2.8 mg/dl. Determination was carried out by photometry on the COBAS 6000 autoanalyzer, module C501 (ROCHE). Substrate method using 5-nitro-5’-methyl- BAPTA (NM-BAPTA) and colorimetric technique.
Vitamin D and Calcium Intake and Supplementation Assessment:
Daily intake of calcium and vitamin D was assessed through a previously validated survey, considering the following categories for vitamin D: Low (<80 IU/day) Normal (80-200 IU/day) High ( >200 IU/day) And for calcium: Low (400 mg/day) Normal (400 to 1200 mg/day) High ( >1200 mg/day) Solar Exposure Assessment: For solar exposure, the following categories were considered: Insufficient exposure for values below 4 Moderate exposure for values between 4 and 7 Satisfactory exposure for all values >7 Skin Phototype Assessment: For different skin phototypes, the Fitzpatrick classification was used: Levels I to VI.
Statistical Analysis:
The statistical approach employed was descriptive, correlational, and included group comparisons. Descriptive statistics involved frequency distribution, means, and standard deviation. In certain numerical variables, transformations to categorical and ordinal variables were performed. To assess the normality of numerical variables, the Kolmogorov-Smirnov test was applied, revealing that none of them exhibited a normal distribution. As a result, non-parametric statistics were employed, specifically for associations and correlations: chisquare, Spearman’s correlation coefficient (rho), and coefficient of determination (rho2). Group comparisons were conducted using the Mann-Whitney U test and the Kruskal-Wallis test. The statistical software used was SPSS.
Results
Table 1 describes the characteristics of the 346 studied mothers. The presented data highlights that there is an average age at childbirth of 30.75±5.69 years, and three-quarters of the mothers were multiparous. Among them, 68.2% were of European descent. Skin types III and IV were observed in 84.9% of the pregnant women. Nearly 20% of mothers had gestational diabetes, and 13.6% had hypothyroidism. The most frequent type of delivery was eutocic (61%), with clear amniotic fluid (77.5%) and an average gestational age of 39.28±1.42. The risk of infection was present in 17% of the patients. Regarding general characteristics for the newborns (Table 2), it’s notable that they had an average weight of 3223 ± 493 grams, a birth length of 49.06 ± 1.93 cm, and a head circumference of 34.75 ± 1.42 cm. Additionally, fetal distress was observed in 15.6% of cases. Regarding vitamin and mineral values in pregnant women (Table 3), it’s noteworthy that only 0.6% of mothers reported low vitamin D intake, but only 7.8% of mothers had adequate blood values. Regarding parathormone, 5.5% of pregnant women had low values, with the remaining 94.5% within the normal to high range. Calcium intake also reported only normal to high values, but blood calcium levels showed 19.1% with low values. Concerning magnesium and phosphorus levels in pregnant women, magnesium deficiencies were found in only 5.8% of mothers. Of these, 26.6% reported taking multivitamins, but upon survey, it was found that they didn’t meet the minimum vitamin D requirement.
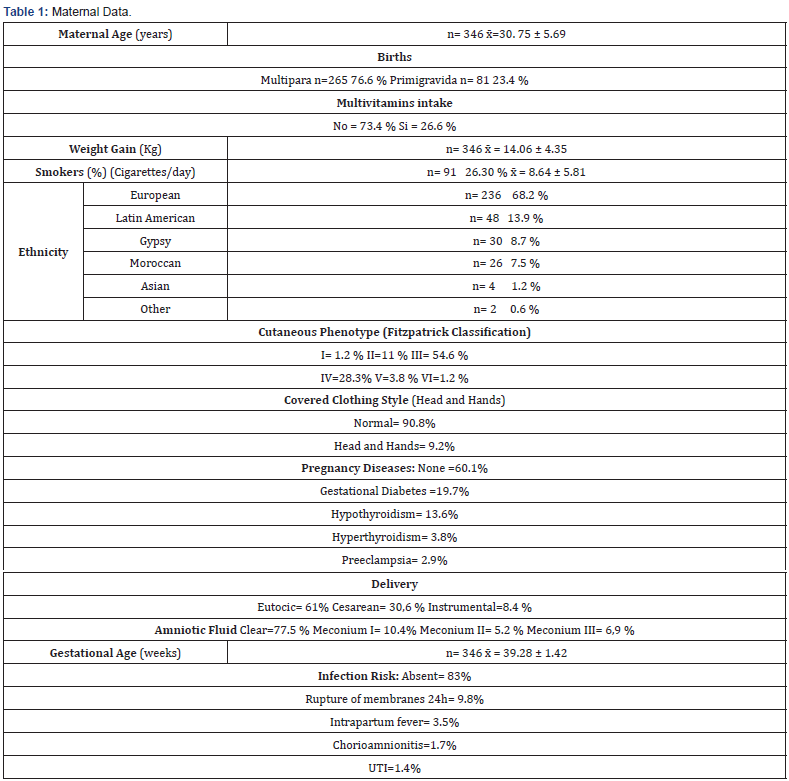
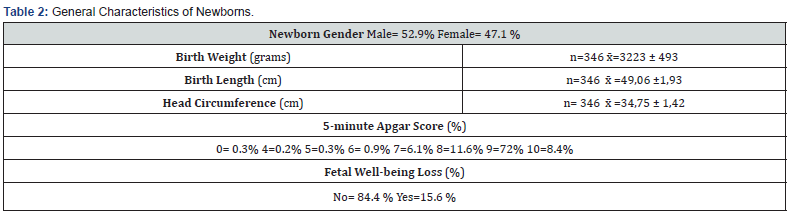
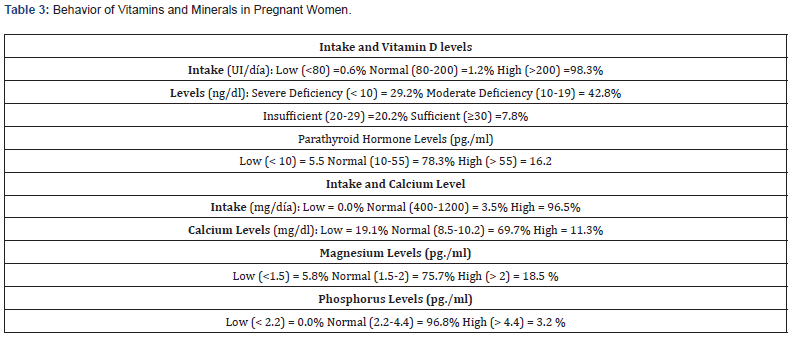
Results of vitamin D levels in umbilical cord blood of infants (Table 4) indicated sufficient levels in only 16.5% and moderate to severe deficiency in 72% of cases. Magnesium and phosphorus didn’t show significant low values, as only 7.5% of infants had low magnesium levels. To understand the relationship between maternal vitamin D levels and the season of childbirth, the chisquare test was applied (Table 5). It’s observable that the variables were related, implying, among other things, a tendency to present severe vitamin D deficiency in the winter season. Not meeting the requirements to demonstrate association with this test were maternal vitamin D levels with clothing style worn, phototype, and ethnicity.
Comparing vitamin D levels using the Mann-Whitney U test within groups based on clothing style worn showed significant differences (p<0.05), with a higher average range of vitamin D among those wearing regular clothing compared to those consistently covering their head and neck. This test did not yield significance (p ≤ 0.05) when comparing clothing style worn with parathormone and calcium levels in mothers. To compare vitamin D levels among different ethnicities, those with a frequency greater than 5 were selected: European, Latin American, Romani (Gypsy), and Moroccan. Applying the Kruskal-Wallis test found significant differences (p<0.05) among these four ethnicities, with the average ranks presented as follows: Latin American=187.02, European=175.66, Romani=139.67, and Moroccan=128.73 (Table 6). Table 7 shows the correlation values (rho) between vitamin D levels and some variables of interest. Significant correlations (p<0.01) were found between maternal vitamin D levels and sunlight exposure (rho=0.174), parathormone levels (rho=- 0.274), and fetal vitamin D (rho=0.391).
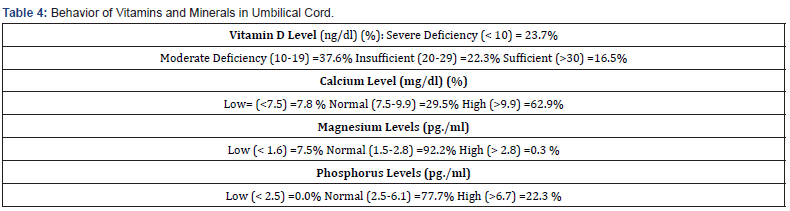
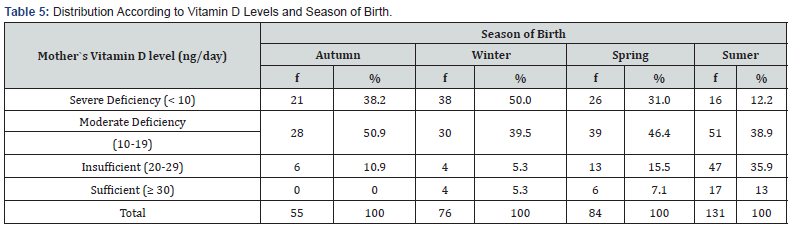
Chi2= 64.6 9gl significance=0.000.
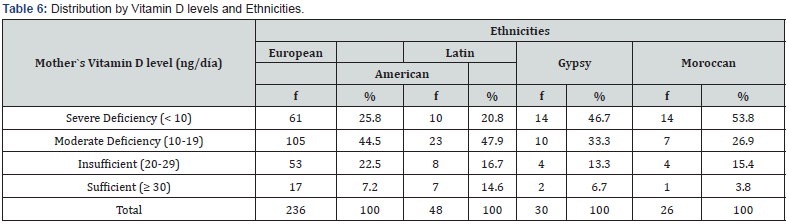
Chi2= 64.6 9gl significance=0.000
Kruskal Wallis Test Chi2= 10.860 3gl significance=0.013
Average Ranks: European = 175.56 Latin American= 187.02 Gypsy= 139.67 Moroccan= 128.73.
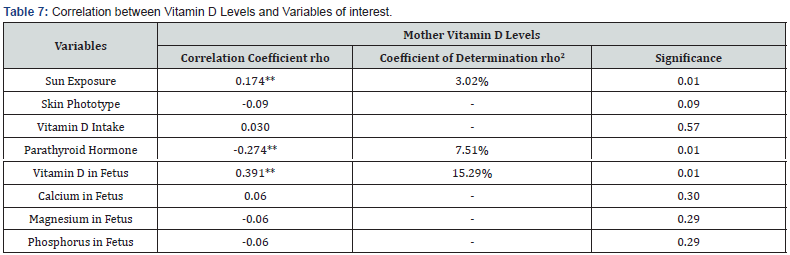
**Statistically significant at p < 0.01
Discussion
Vitamin D is one of the key hormones involved in bone metabolism and calcium homeostasis. The presence of vitamin D receptors in a wide variety of cells suggests its significant role in various physiological processes. It exerts its effects through binding to a receptor, which is not only found in tissues defining its “classical” actions, such as bone, the digestive system, and the kidney, but is also widely distributed throughout the body. Enzymes capable of producing vitamin D are found in these tissues, potentially turning them into target tissues that modulate its “non-classical” actions [28]. These “non-classical” actions can be grouped into three types: hormonal secretion regulation, immune response regulation, and cell differentiation regulation. In general, these aspects have led to its deficiency being associated with the onset of metabolic syndrome, type 2 diabetes, autoimmune diseases, and, at the maternal-fetal level, an increased risk of gestational diabetes, preeclampsia, as well as the presence of neonatal rickets and hypocalcemia, reduced bone mass in infants, and intrauterine growth retardation [28,29].
Although there has been some controversy regarding optimal vitamin D levels during pregnancy, most experts accept the definition of deficiency for any value below 20 ng/ml (1,10). In this context, a high percentage of serum deficiency (72%, ranging from severe to moderate) was observed in the studied group, which coincides with data published in other countries that have also shown a high percentage of suboptimal vitamin D levels in pregnant women and their newborns. Similarly, in the early 2000s, it was reported that approximately 2 out of every 3 pregnant women in the United States had deficient or insufficient vitamin D levels [30], and the same was happening in Canada [31]. Similar information was reported in other European countries such as the Netherlands [32], as well as in Asian countries like India, Iran, and Turkey, where prevalence reached around 90% [33-35]. At a regional level, information from the INMA Project (Childhood and Environment) reports high prevalence of deficiency (62.7%) in a group of pregnant women evaluated in the Asturias cohort at the University of Oviedo [1]. Similarly, high prevalence of insufficiency or deficiency of this nutrient has been reported in the general Spanish population; for instance, in a group of poor fertile-age women in Barcelona evaluated in 2005, 47.9% showed insufficiency and 37.2% deficiency [36]. In the Madrid area, a deficiency prevalence of 70% was described among the elderly population and 35% among postmenopausal women [37]. Similar situations were observed in young people from Madrid, with 25.6% having vitamin D deficiency and 56% showing insufficiency [3]. A deficiency of 32.6% and insufficiency of 28.6% were reported in medical students from the Canary Islands [4]. These figures align with what was found in the evaluated mother-newborn pairs in this study.
A positive correlation between maternal and umbilical cord blood 25(OH) D3 levels has been described in the mothernewborn pair, indicating that the latter’s concentrations directly depend on the mother’s vitamin status. Thus, cord blood levels are approximately 80% of those in maternal blood [6,7,16,38]. In this study, 61.3% deficiency of vitamin D in umbilical cord blood and 72% deficiency in maternal blood were observed. Maternal vitamin D level variation explained 15.29% of the variation in cord blood level, with significant correlations between both plasma values. Comparing these data with published information in Spain, the deficiency prevalence in cord blood in this study is lower than what was found in a group of mother-newborn pairs evaluated in 2013 at the Hospital del Mar in Barcelona, where deficiency was at 94% [6]. This prevalence also matches the data published by Manzano (2017), who reported an 83% deficiency of vitamin D in cord blood [7]. Among maternal factors associated with vitamin D deficiency, inadequate calcium and/or vitamin D consumption, insufficient sun exposure, covered clothing style, inadequate nutritional supplementation, and phototype are described [6,9]. In this study, vitamin D and calcium consumption in the maternal diet was adequate in almost the entire sample (99.4% and 100%, respectively). However, a very high deficiency of 25(OH) D3 was detected, especially during the winter season. This highlights that cutaneous synthesis of vitamin D is the main source of this nutrient for the body. There was no reported calcium deficiency in the diet, but 19.1% of the sample had low values, impacting cord calcium levels. The coefficient of determination showed that maternal calcium levels explained the variation in cord calcium levels.
Regarding sun exposure, a correlation between vitamin D levels and exposure was observed in this study. Moderate sun exposure is described as the main source of vitamin D for most humans, making insufficient sun exposure the primary cause of its deficiency. Additionally, a seasonal variation in 25(OH) D3 levels has been documented, which aligns with the findings of this study. A tendency to have severe vitamin D deficiency in the winter season was observed. Comparing vitamin D levels based on clothing style worn, significant differences and higher vitamin D levels were found in those who wore regular clothing compared to those who consistently covered their head and neck. Increased clothing coverage diminishes effective sun exposure, as does greater use of sun protection creams. To ensure effective exposure, it’s recommended to spend 10 to 15 minutes in the sun without protection daily [1]. However, recent information suggests that cutaneous synthesis may not be sufficient to meet vitamin D needs during pregnancy [23].
In terms of ethnicity, this study reflects the observed heterogeneity in the sampled area. Significant differences were found in relation to 25(OH) D3 plasma concentrations among different ethnicities. The group of Romani and Moroccan pregnant women presented lower vitamin D values compared to Latin American and European pregnant women. This aligns with the work of Moreno, who evaluated pregnant women in 2017 in the Murcia region and found that Arab pregnant women had lower vitamin D values than Caucasian and South American pregnant women. This effect is attributed to increased melanin offering sun protection, which impacts vitamin D production, as well as limited cutaneous synthesis due to skin color [39]. Furthermore, these findings align with the work of Bodnar [40], who evaluated pregnant women in the northeastern United States. The African American group had much lower average 25(OH) D3 levels than the Caucasian group, both in winter and summer months. Pérez López similarly reported lower vitamin D levels in Arab pregnant women living on the Mediterranean coast in Spain compared to Caucasian mothers in the same area [41]. Thus, belonging to certain darkerskinned ethnicities, which produce six times less vitamin D than lighter skin, negatively affects 25(OH) D3 plasma concentrations. This has been demonstrated by McAree’s multicenter study, where vitamin D deficiency prevalence was high among pregnant women overall but significantly more severe in those with darker skin tones [42]. Similar conclusions were reached by Bowyer et al. [43], who observed that deficiency prevalence was higher among pregnant women with darker skin and even higher among those who covered their face and parts of their body due to cultural or religious reasons.
In recent years, with technological advances and molecular techniques, various studies have been conducted to evaluate the effect of the Black race and its influence on different comorbidities. Reeves evaluated the risk of preeclampsia-like morbidities with low vitamin D levels in ethnic minorities, finding that lower levels of this vitamin were also present in the African American pregnant group, who also had a lower dietary intake of vitamin D [44]. This is because efficient sun exposure is the main source of vitamin D production. In Arab or Muslim pregnant women, who cover their bodies year-round, cutaneous vitamin D synthesis is hindered, leading to moderate and severe deficiencies throughout the year, despite living in areas with high solar radiation. Regarding nutrient supplementation, it’s noteworthy that only 26.6% reported using vitamins and minerals during gestation, and these were insufficient in terms of vitamin D content. It has been suggested that the high prevalence of vitamin D deficiency in pregnant women and cord blood, and consequently in newborns, necessitates initiating prophylactic supplementation [20,45,46]. However, this recommendation is not being effectively carried out, even though studies on vitamin D kinetics suggest that supplements of over 1,000 IU/day are necessary to achieve desired plasma concentrations. Effective quantities of 4,000 IU/day are needed to provide sufficient levels during pregnancy without risking toxicity. This would also help achieve adequate vitamin D levels in breast milk without supplementing the newborn [23,47,48]. More information is needed on the exact content of nutritional supplements used and/or recommended, dosages, forms of presentation, and whether there was medical prescription, as well as patient compliance with the recommendation.
Among the “non-classical” actions attributed to vitamin D, its role in hormonal secretion regulation has been described, particularly its inhibition of parathyroid hormone (PTH) synthesis and secretion. In this study, an inverse correlation between both plasma values was found, and 5.5% deficiency of PTH was observed [14]. This aligns with studies conducted on mother-newborn pairs, as well as the general adult population in the United States, which analyzed 3.8 million laboratory results from men and women in various regions. Similar findings were reported in a group of 9,890 Turkish women and 2,723 men evaluated between 2009 and 2015 [49,50]. Regarding other hormones, vitamin D deficiency affects insulin, stimulating its secretion while decreasing beta cell apoptosis mediated by cytokines. These aspects have been associated with the development of gestational diabetes [28]. In this study, gestational diabetes was present in 19% of cases, but further research is needed to clarify these implications and establish a relationship.
Based On the Findings, the Following Conclusions
High prevalence of vitamin D deficiency in both maternal and umbilical cord blood, consequently affecting newborns, despite reported adequate consumption of the vitamin. Directly proportional correlation between maternal serum vitamin D concentrations and cord blood concentrations, as well as a weak to moderate inverse correlation between parathyroid hormone and maternal vitamin D levels. Association with inadequate sun exposure and clothing style worn, with a higher average range of vitamin D in those wearing regular clothing compared to those who consistently cover their head and neck. Regarding ethnicity, this study reflects the observed heterogeneity in the sampled area, with significant differences in 25(OH) D3 plasma concentrations among different ethnicities. The Romani and Moroccan pregnant women presented lower vitamin D values. It is necessary to implement effective programs from early gestation to prevent severe public health issues in the mother-newborn pair. Promoting healthy lifestyles with outdoor activity and adequate vitamin D intake, particularly in pregnant women with associated factors, is recommended. Additionally, individual assessment of supplementation schemes is important.
Acknowledgments
The authors would like to express their gratitude to the following individuals and organizations for their contributions to this research: We are grateful to Servicio de Salud de Castilla La Mancha for providing the necessary facilities and resources that facilitated the execution of this study. Special thanks to The Hospital Valld’Hebron laboratory for their technical support and guidance throughout the research process.
References
- Rodríguez A, Riano I, Fernandez A, Navarrete E, Espada M, et al. (2015) Prevalencia de Deficiencia e insuficiência de vitamina D y factores asociados en mujeres embarazadas del norte de Españ Nutr Hosp 31(4): 1633-1640.
- Martinez V, Monrro J, Dalman J (2012) Recomendaciones de ingesta de cálcio y vitamina D: posicionamiento del Comité de Nutrición de la Asociación Española de Pediatrí An Pediatr 77: 57-58.
- Calatayud M, Joda E, Sánchez R, Guadalex S, Hawkins F (2009) Prevalencia de concentraciones deficientes e insuficientes de vitamina D en una población joven y sana. Endocrinol Nutr 56(4): 164-169.
- González E, Soria A, García S, Mirallave A, Groba M (2011) Elevada prevalencia de hipovitaminosis D en los estudiantes de medicina de Gran Canaria, Islas Canarias (España). Endocrinol Nutr 58(6): 267-273.
- Ramírez D, de la Torre M, Llorente F, Pérez J, Gil M (2012) Evaluación de la exposición solar, ingesta y actividad física en relación con el estado sérico de vitamina D en niños prepuberes españ Nutr Hosp 2: 1993-1998.
- Ortigoza S, García O, Mur A, Roser F, Carrascosa A, et al. (2015) Concentraciones plasmáticas de 25-OH vitamina D y Paratohormona en sangre de cordón umbilical. Rev Esp Salud Pública 89: 75-83.
- Manzano C, García O, Mur A, Roser F, Carrascosa A, et al. (2017) Concentraciones plasmáticas de 25-OH vitamina D en sangre de cordón umbilical tras los meses de verano. Rev Esp Salud Pública 91: 1-8.
- De-Regil L, Palácios C, Lombardo L, Peña-Rosas J (2016) Vitamin D supplementation for women during pregnancy. Cochrane Database of Systematic Rewiews 7(7): CD008873.
- Thandrayen K, Pettifor J (2012) Maternal vitamin D status: implications for the development of infantile nutritional rickets. Rheum Dis Clin North Am 38(1): 61-79.
- Holick M, Binkley N, Bischoff H, Gordon C, Hanley D, et al. (2011) Evaluation, treatment, and prevention of vitamin D deficiency: an endocrine society clinical practice guideline. J Clin Endocrinol Metab 96(7):1911-1930.
- Vio S, Moller K, Chen T, Mathieu J, Holick M (2007) Vitamin D deficiency in a healthy group of mothers and newborn infants. Clin Pediatr (Phila) 46(1): 42-44.
- Misra M, Pacaud D, Petryk A, Collell P, Kappy M (2008) Drug and therapeutics committee of the Lawson Wilkins Pediatric Endocrine Society. Vitamin D deficiency in children and its management: review of current knowledge and recommendations. Pediatrics 122(2): 398-417.
- Salle B, Delvin E, Lapillonne A, Bishop N, Glorieux T (2000) Perinatal metabolism of vitamin D. Am J Clin Nutr17(Supp 5) :1317-1324.
- Nicolaidou P, Hatzistamatiou Z, Papadopoulou A, Kaleyias J, Floropoulou E, et al. (2005) Low vitamin D status in the mother-newborn pairs in Greece. Calcif Tissue Int 78(6): 337-342.
- Karras S, Shah T, Petroczi A, Goulis D, Papadopoulou F (2013) An observational study reveals that neonatal vitamin D is primarily determined by maternal contributions: implications of a new assay on the roles of vitamin D forms. J Nutr 12: 77-89.
- Masvidal R, Ortigoza S, García O (2012) Vitamina D: fisiopatología y aplicabilidad clínica en pediatrí Ann Pediatr 77: 279.
- Cabezuelo G, Vidal A, Abeledo A, Frontera P (2007) Niveles d 25-hidroxivitamina D en lactantes. Relación con la lactancia materna. An Pediatr 66(5): 491-495.
- Morales E, Romieu I, Gierra S, Ribagliato M, Viogue J, et al. (2012) Maternal vitamin D status in pregnancy and risk of lower respiratory tract infections, wheezing, and asthma in offspring. Epidemiol 23(1): 64-71.
- Rosen C, Adams J, Demary M, Manson J (2012) The nonskeletal effects of vitamin D on an endocrine society statement. Endocr Rev 33(3): 456-472.
- Hobel C, Steer P (2015) Vitamin D supplementation should be routine in pregnancy. BJOG 122(7): 1021.
- Saraff V, Shaw N (2016) Sunshine and vitamin D. Arch Dis Chil 101: 190-192.
- Cuhaci C, Demirel F (2014) Effects of seasonal variation and maternal clothing style on vitamin D level of mothers and their infants. Turk J Pediatr 56: 475-481.
- Pratumvinit V, Wongkrajang P, Watagrana T, Nimmamnit A (2015) Maternal vitamin D status and its related factors in pregnant women in Thailand. PLoS One 10(7): 1311-1326.
- Hernández R, Fernández Collado C, Baptista Lucio P (2010) Metodología de la investigació 5a ed. México: Mcgraw-Hill.
- Padrón D (2012) Epidemiología Bá 1a ed. Venezuela: Dirección de Medios y Publicaciones de la Universidad de Carabobo.
- Garabédian M, Mens S, Nguyen T, Ruiz J, Callens A, et al. (1999) Prevention de la carence en vitamin d chez lenfant et ladolescent. Proposition et argumentarie pour lutisitaion de un abaque decisionnel. Arch Pediatr 6: 990-1000.
- Freedberg I, Eisen A, Wolff K, Austen K, Goldsmith L, et al. (1999) Dermatology in general Medicine. Fifth Edition. New York: Mc Graw Hill: 1999.
- Calle A, Torrejón M (2012) La vitamina D y sus efectos “no clásicos”. Rev Esp Salud Pública 86: 454-459.
- Winzenberg T, Jones G (2016) Vitamin D deficiency: Who needs supplementation? Rev Paul Pediatr 34(1): 3-4.
- Looker A, Pfeiffer C, Lacher D, Schleicher R, Piciano M, et al. (2008) Serum 25-hydroxyvitamin D status of the US population: 1988-1994 compared with 2000-2004. Am J Clin Nutr 88(6): 1519-1527.
- Li W, Green T, Invis S, Whiting S, Shard A (2011) Suboptimal vitamin D level in pregnant women despite supplement use. Can J Public Health 102(4): 308-312.
- Van der Meer I, Karamali N, Voeke A, Lips P, Middelkoop J, et al. (2006) High Prevalence of Vitamin D Deficiency in Pregnant Non-Western Women in The Hague, Netherlands. Am J Clin Nutr 84(2): 350-353.
- Sachan A, Gupta R, Das V, Agarwal A, Awasthi P, Vhatia V (2005) High prevalence of vitamin D deficiency among pregnant women and their newborns in northern India. Am J Clin Nutr 81(5): 1060-1064.
- Magbooli Z, Hossein A, Shafaei A, Karimi F, Madani F, et al. (2007) Vitamin D status in mothers and their newborns in Iran. BMC Pregnancy Childbirth 7(1):1-4.
- Halicioglu O, Aksit S, Koc F, Akman S, Albudak E, et al. (2012) Vitamin D deficiency in pregnant women and their neonates in springtime in western Turkey. Paediatr Perinat Epidemiol 26(1): 53-60.
- González M, Pérez A, Zavaleta E, Gudiña N, Pozo C, et al. (2008) Deficiencia de vitamina D en mujeres de edad fértil. Aten Primaria 40: 393-399.
- Aguado P (2000) Alta prevalencia de deficiencia de vitamina D en mujeres postmenopáusicas de una consulta reumatológica en Madrid. Evaluación de dos pautas de prescripción de vitamina D. Med Clin 114: 326-330.
- Wolfgang H. Complications of vitamin D deficiency from the foetus to the infant: one cause one prevention, but who´s responsibility? Best Pract Res Clin Endocrinol Metab 29(3): 385-398.
- Moreno Fuentes A. Asociación de los niveles maternos de 25 hidroxi vitamina D con la aparición de eventos adversos durante el embarazo, parto y en el neonato. Tesis Doctoral. Universidad Católica de Murcia 2017.
- Bodnar L, Simban H, Power R, Frank M, Roberts J (2007) High prevalence of vitamin D insufficiency in black and white pregnant women residing in the northern United States and their neonates. J Nutr137(2): 447-452.
- Pérez López F, Fernández A, Ferrado N, Gonzalez M, Dionis E, et al. (2011) First trimester serum 25 hydroxyvitamin D status and factors related to lower level in gravid living in the Spanish Mediterranean coast. Reprod Sci 18(8): 730-736.
- McAree T, Jabobs P, Manickavasagar T, Sivalokanathan S (2013) Vitamin D Deficiency in pregnancy still a public health issue. Matern Child Nutr 9(1): 23-30.
- Bowyer L, Catlin C, Diamont T, Horner C, Davis G, et al. (2009) Vitamin D, PTH and calcium levels in pregnant women an their neonates. Clin Endocrinolol 70(3): 327-330.
- Reeves I, Bamji Z, Rosario G, Lewis K, Young M, et al. (2014) Vitamin D deficiency in pregnant women of ethnic minority: a potential contribution to preeclampsia. J Perinatol 34(10): 767-773.
- Whithouse A, Hold V, Serralha M, Holt P, Kusel M, et al. (2012) Maternal serum vitamin D level during pregnancy and offspring neurocognitive development. Pediatrics 29(3): 485-493.
- Harvey N, Holroyd C, Ntani G, Jevaido K, Cooper P, et al. (2014) Vitamin D supplementation in pregnancy: A systematic review. Health Technol Assess 18(45): 1-190.
- Marshall L, Mehta R, Petrova A (2013) Vitamin D in the maternal-Feta-neonatal interface: clinical implications and requirements for supplementation. J Matern Fetal Neonatal Med 26(7): 633-638.
- Hollis B, Johnson D, Hulsey T, Eteling M, Wagner C (2011) Vitamin D supplementation during pregnancy: double blind, randomized clinical trial of safety and effectiveness. J Bone Miner Res 26(10): 2341-2357.
- Kroll M, Bi C, Garber C, Kaufman H, Caston-Balderrama A, et al. (2015) Temporal relationship between Vitamin D status and parathyroid hormone in the United States. PLoS One 10(3): 1-13.
- Serdar M, Bater C, Kilercik M, Durer Z, Aksungai F (2017). Analysis of changes in parathyroid hormone and 25(OH) Vitamin D levels with respect to age, gender and season: a data mining study. J Med Biochem 36(1): 73-83.






























