An Analysis of Neonatal Bowel Resections in the NICU and OR
Lauren Howser1, Cassandra Anderson1, Carly Goehring1, Eamaan Turk1, Sarah Fisher1, Yan Han2 and Brian Gray*
1Section of Pediatric Surgery, Riley Hospital for Children, Department of Surgery, Indiana University School of Medicine, Riley Hospital Drive, Indianapolis, USA
2Department of Biostatistics and Health Data Science, Indiana University, 410 W Tenth Street Suite 3000, Indianapolis, USA
Submission:October 23, 2023;Published:November 13, 2023
*Corresponding author: Brian W Gray, Assistant Professor of Pediatric Surgery, Riley Hospital for Children, 705 Riley Hospital Drive, Indianapolis, USA
How to cite this article:Lauren H, Cassandra A, Carly G, Eamaan T, Sarah F, et al. An Analysis of Neonatal Bowel Resections in the NICU and OR. Acad J Ped Neonatol 2023; 13(1): 555910. 10.19080/AJPN.2023.13.555910
Abstract
Background: Neonatal operations frequently occur in the neonatal intensive care unit (NICU) rather than the operating room (OR). Our goal was to identify factors associated with having a bowel resection procedure in the NICU as opposed to the OR, describe outcomes, and analyze modifiable factors that might improve survival.
Methods: We reviewed 139 neonates who underwent bowel resection from 2014-2018. Statistical analysis included bivariate, multivariable, and Kaplan-Meier analysis.
Results: Of the 139 operations, 33 occurred in the NICU and 106 in the OR. A diagnosis of necrotizing enterocolitis (NEC) and spontaneous intestinal perforation (SIP) and vasoactive medication requirement were most predictive of NICU operative location. Survival was lower in the NICU group (60.6% vs 94.3% survived 30 days, p<0.001), with a hazard ratio for mortality 7.9 times greater than the OR group (p<0.001). In NEC and SIP patients, those who underwent surgery in the NICU also had worse survival, but they had similar intra-operative complications and blood loss as NEC/SIP OR patients. Usage of heating devices, intra-operative temperature monitoring, and lower blood product transfusion were all positively correlated with survival in all patients.
Conclusion: Patients undergoing NICU bowel resections have more markers of illness severity, particular diagnosis patterns, and worse survival. Modifiable factors can be studied to further improve survival in both locations.
Keywords: Bowel Resection; Neonate; NICU; Survival; Bedside Procedure; NEC
Abbreviations: NEC: Necrotizing Enterocolitis; SIP: Spontaneous Intestinal Perforation; OR: Operating Room; NICU: Neonatal Intensive Care Unit; FFP: Fresh Frozen Plasma; EGA: Estimated Gestational Age; UOP: Urine Output; PRBC: Packed Red Blood Cells
Introduction
Neonatal operations often must take place in the neonatal intensive care unit (NICU) due to risk factors inherent in some disease processes and general patient instability [1]. The practice of performing surgical procedures in the NICU has been widely accepted since 1982, when the first study confirming the effectiveness of patent ductus arteriosus ligations performed in the NICU was published. This confirmed a safe practice with no increased risk of complications [2,3]. The practice has been expanding to more surgical procedures, including congenital diaphragmatic hernia repair, bowel resection with and without ostomy formation, ostomy closure, gastroschisis closure, silo placement, omphalocele repair, and central line placement, among others.
Risks have been identified for both surgical procedures in the operating room (OR) and NICU and are weighed by physicians when deciding on the location for each procedure. Risk factors of performing an operation in the NICU potentially include higher risk of nosocomial infection and complications [4]. Risks of performing surgery in the OR include transportation risks, such as discontinuation of monitoring, dislocation of artificial airways, dislocation of vascular access, and hypothermia [5]. Another common reason for an operation to occur in the NICU is that the patient requires advanced ventilatory strategies, such as high frequency oscillatory ventilation or jet ventilation, which cannot typically be performed in the OR in many institutions. Our institution specifically makes these decisions through a multidisciplinary process involving surgery, anesthesiology, and neonatology. Stability, respiratory support, and surgeon preference play the largest roles in these decisions. Anesthesia in both the NICU and the OR is managed by certified anesthesiologists, but type of anesthesia may differ, including a predilection towards the use of intravenous medications in the NICU versus inhalational in the OR.
A common pediatric general surgery procedure occurring in both the NICU and OR is a bowel resection. The most common primary diagnoses requiring bowel resection in neonates are necrotizing enterocolitis (NEC), spontaneous intestinal perforation, intestinal atresia, small bowel obstruction, and midgut volvulus [6-8]. Previous studies have analyzed operations occurring in the NICU and OR for other procedures, such as patent ductus arteriosus and congenital diaphragmatic hernia and both proved to be equally safe in the NICU and OR [9-11]. However, there are no studies that describe the outcomes of bowel resections occurring in the NICU and OR. The objective of this study was to identify factors associated with neonatal bowel resection procedure in the NICU as opposed to the OR, describe outcomes in both locations, and analyze modifiable factors that might improve survival. The null hypothesis for this study would show that there are no differences in outcomes between the two groups. However, we thought that the alternative hypothesis in which there was a stark difference would likely be more accurate. We hypothesized that bowel resections in the NICU would be performed on more unstable patients and would be associated with worse outcomes than operations in the OR.
Materials and Methods
After Institutional Review Board approval (#1905838491) with a waiver for informed consent was obtained, neonates (<30 days old) who underwent a bowel resection in either the NICU or OR at Riley Hospital for Children at Indiana University Health from 2014-2018 were identified from the electronic medical record. NICU operations all occurred in the same 60 bed level IV NICU. Inclusion criteria for the study population was a neonate defined as less than 30 days of life undergoing bowel resection with anastomosis, bowel resection with ostomy formation, or intestinal atresia repair. Exclusion criteria included operations performed in utero or while attached to placental support (EXIT) and neonates undergoing multiple simultaneous operations. This study was retrospective in nature and no randomization took place.
Outcomes studied included survival, post-operative factors, and modifiable factors intraoperatively such as temperature monitoring, heating device usage, and blood product administration. The data collected included: gestational age at birth, birth weight, weight at time of operation, birth weight z-score, weight at time of operation z-score, race, insurance status, diagnoses, number of operations, comorbidities, APGAR score, antibiotic administration, temperature reports, reports of bleeding, infections, hypotension, hat usage during procedure, blood product administration, anesthetic used, vasoactive medication usage, and perioperative disease severity markers.
The two study groups (NICU and OR) were first analyzed to determine a descriptive picture of patients undergoing bowel resections in each location. This was done utilizing two-sample t-tests, Chi-square tests, and Fisher’s exact tests. Risk factors for operative location were further assessed using bivariate logistic regression. Several factors, including operative location and blood loss, were also assessed as predictors of 30-day survival using Chisquare tests and a backward stepwise multivariable regression. This analysis was repeated exclusively for necrotizing enterocolitis and spontaneous intestinal perforation patients in an effort to create a better comparison of this specific group of patients who may be expected to have higher incidence of morbidity and mortality. We felt that this might remove some of the bias towards better outcomes seen in patients with other diagnoses that did not include a major inflammatory component potentially leading to worse outcomes.
A comparison of outcomes between the two groups was completed using Chi-square analyses and independent samples t-tests. To further analyze survival, a Kaplan-Meier estimator was used. The log-rank test was used to compare the two survival curves between the NICU and OR. A Cox regression survival analysis was also used to determine a hazard ratio for survival.
Finally, several “modifiable factors” were identified. These were defined as potentially controllable factors that could play a role in survival. The modifiable factors we chose to examine included temperature monitoring during surgery, usage of heating device during surgery, blood administration during surgery, and platelet/fresh frozen plasma (FFP) administration during the first 24 hours post-op. The patient cohort was used to analyze the relationship between these factors and 30-day survival. Chisquared and nonparametric tests were used. For all tests utilized, an α-level of 0.05 was used to determine statistical significance.
Results
Risk Factors for Surgery in the NICU
A total of 139 patients met inclusion criteria, and only the index operation for each patient was reviewed. There were 33 patients in the NICU group and 106 patients in the OR group. There was a statistically significant difference in 30-day survival between groups (60.6% vs 94.3%, p<0.001). The descriptive statistics of NICU vs OR patients showed two very different populations. The NICU group had lower mean estimated gestational age (EGA) at birth, lower weight at surgery, higher pre-op FiO2, lower 1 minute and 5 minute APGAR, lower serum pH pre-op, and more hemodynamic support (Table 1). Patients undergoing bowel resections in the NICU were more likely to have the operative diagnosis of spontaneous intestinal perforation (SIP) or necrotizing enterocolitis (NEC) compared to OR patients (Table 1). The methods of ventilation for patients with an operation in the NICU were conventional vent (n=8, 24%), high frequency oscillatory vent (n=24, 73%), and regular or high flow nasal cannula (n=1, 3%). The methods of ventilation for patients with an operation in the OR included conventional vent (n=51, 48%), CPAP/SiPAP (n=2, 2%), regular or high flow nasal cannula (n=9, 8%), and room air (n=44, 42%).
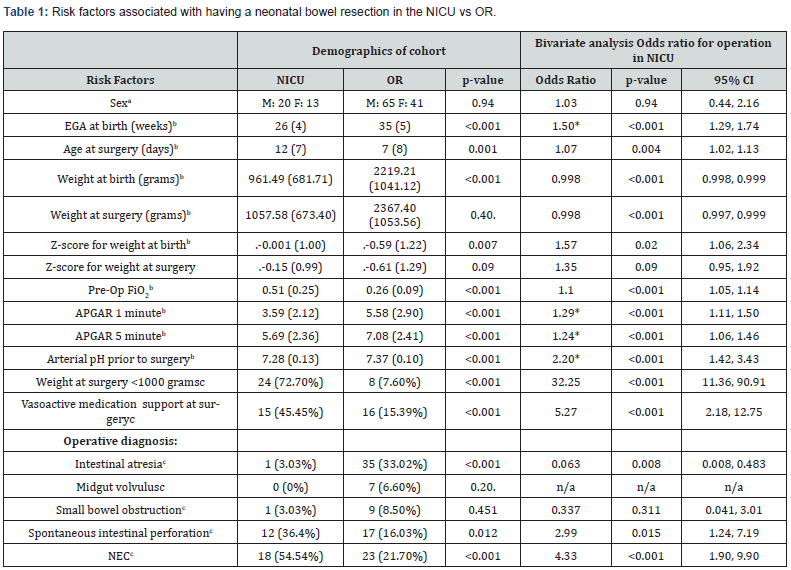
a Indicates frequency only, b indicates mean (SD), c indicates data presented as frequency (percentage)
* Indicates Odds Ratio inverted to represent higher probability of OR procedure with increasing numerical value.
Unadjusted odds ratios were included in this analysis.
Bivariate analysis determined odds ratios demonstrated that diagnosis of NEC (OR 4.33, p<0.001), spontaneous intestinal perforation (OR 2.99, p=0.015), increased pre-op respiratory FiO2 (OR 1.10, p<0.001), increased hemodynamic support (any vasoactive medication) at surgery (OR 5.27, p<0.001), and decreased z-score for weight at birth (OR=1.57, p=0.02) were significantly associated with operative location in the NICU. Inverse odds ratios were utilized to show lower 1 minute APGAR (OR 1.29, p<0.001), lower 5-minute APGAR (OR 1.24, p<0.001), lower arterial pH prior to surgery (OR 2.20, p<0.001), and lower EGA at birth (OR 1.50, p=<0.001) were also significant predictors of an operation in the NICU.
NICU vs OR Outcomes
Table 2 compares outcomes in the NICU vs OR groups. The NICU group had lower urine output (UOP) for 24 hours after surgery, more vasoactive hemodynamic support after surgery, lower total paralytic dose, lower arterial pH post-op, increased base deficit post-op, increased intra-operative complications, increased blood loss >10mL/kg, decreased survival to 30 days, decreased survival to discharge, and increased length of hospital stay. A Kaplan-Meier survival analysis curve (Figure 1) shows the survival for the entire cohort of NICU and OR operations (p<0.001). The hazard ratio for mortality of patients with surgery in the NICU was 7.9 times the hazard for patients in the OR group (p<0.001).
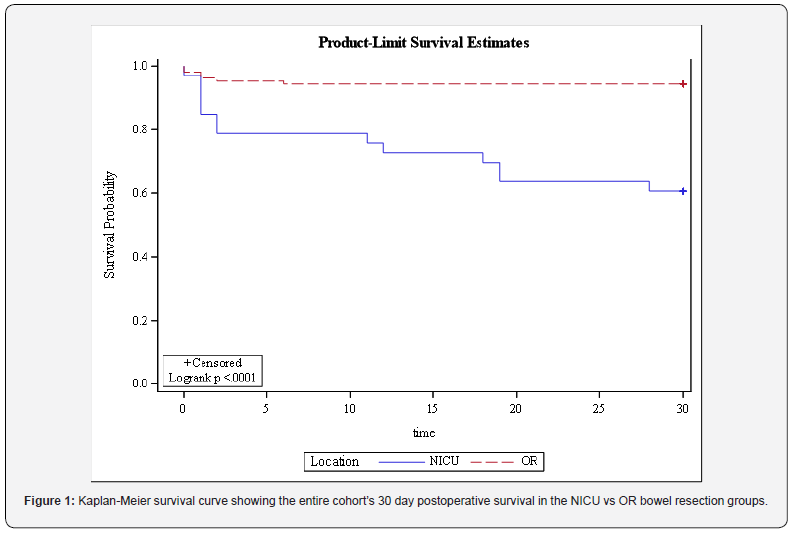
Survival Analysis
In the total cohort, 5.66% of patients in the OR group (6 patients) and 39.39% of patients in the NICU group (13 patients) died before 30 days post-op. Analysis of patients who died showed that the majority of deaths had a primary cause related to the operative diagnosis. Of the NICU patients who died, 56.25% (9 of 16) had a cause of death related to the operative diagnosis. Of the OR patients who died, 62.5% (5 of 8, one unknown cause of death) had a death related to the operative diagnosis. Among all patients, those with operative blood loss >10 mL/kg had significantly lower 30-day survival (61.1% vs 90.1%, p=0.001).
The causes of death in the NICU group related to the operative diagnosis included NEC (4), sepsis (3), ischemic bowel injury (1), multisystem organ failure (1), cardiorespiratory failure (1) and abdominal bleeding (1). Unrelated causes of death to the operative diagnosis in the NICU group included hypoxic respiratory failure (2), severe periventricular leukomalacia and renal failure (1), acute shunt thrombosis (1), and group B strep meningitis (1). The causes of death in the OR group related to the operative diagnosis included NEC (2) and NEC totalis (3). The causes of death in the OR group not related to the operative diagnosis included multiple congenital anomalies (1), multisystem organ failure (1), and irreversible brain damage (1).
Eleven variables were assessed as predictors of survival using a backward stepwise multivariable logistic regression: operative location, EGA at birth, Z-score for weight at birth, pre-op FiO2, preop pH, vasoactive support at surgery, operative blood loss >10mL/ kg, and operative diagnoses of NEC, SIP, intestinal atresia, small bowel obstruction, and midgut volvulus. Following eight steps of multivariable logistic regression, pre-op FiO2, diagnosis of NEC, and vasoactive medication at surgery were revealed as the most important factors contributing to 30-day survival. Higher pre-op FiO2 (OR=1.04, p=0.037), NEC diagnosis (OR=13.3, p=0.011), and vasoactive medication (OR=14.4, p=0.003) were most predictive of mortality (Table 3).
Analysis of NEC/SIP Patients
To remove any outcome bias from atresia and malrotation patients, we completed an analysis similar to what was previously reported in Tables 1 & 2 in patients who only had a diagnosis of NEC or SIP. The NICU group was still smaller in size and younger in estimated gestational age (Table 4). However, markers of illness severity and overall health such as the 1-minute APGAR, 5 minute APGAR, and arterial pH prior to surgery were not significantly different between the groups. Table 5 shows the outcomes comparison between the NICU and OR groups in the NEC/SIP patients. There are slight differences from the previous analysis. Survival was still worse in the NICU group, but early post-op infection was lower. In the NEC/SIP patients, there was no difference in intra-operative complications between NICU and OR.
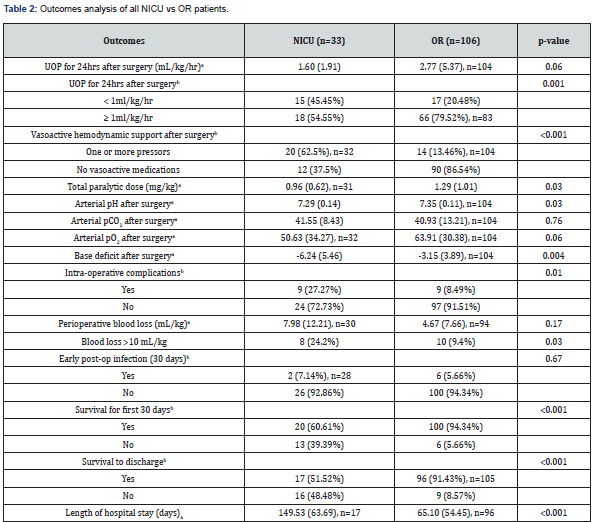
Values and p-values calculated for entire cohort, equal variances not assumed for NICU and OR groups
a indicates data presented as mean (SD), b indicates data presented as frequency (percentage).

Reference group is survived.
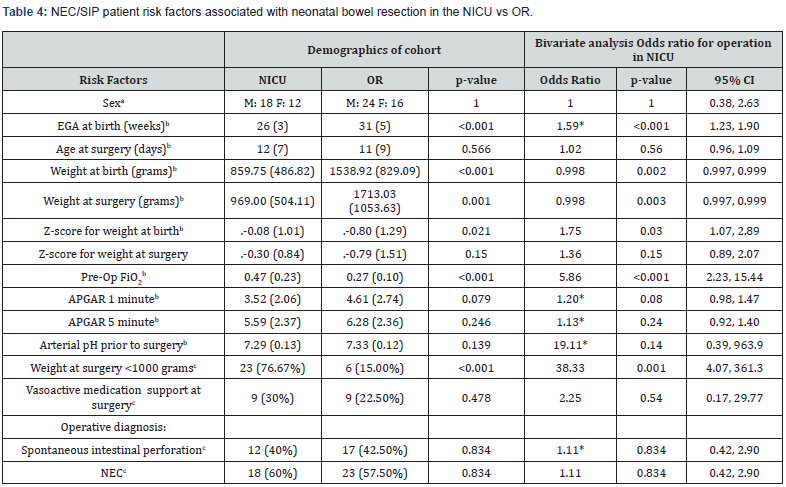
a indicates frequency only, b indicates mean (SD), c indicates data presented as frequency (percentage)
* Indicates Odds Ratio inverted to represent higher probability of OR procedure with increasing numerical value.
Unadjusted odds ratios were included in this analysis.
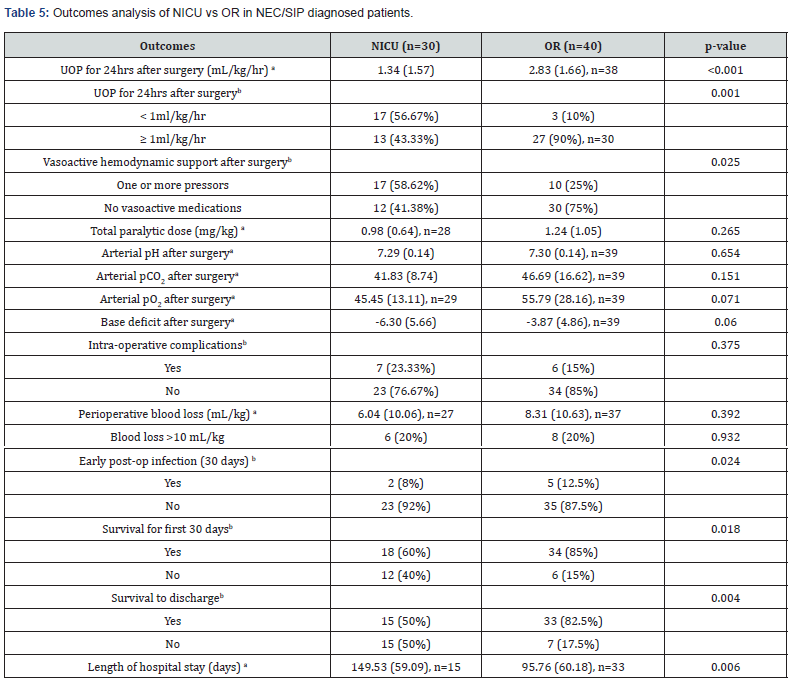
Values and p-values calculated for entire cohort, equal variances not assumed for NICU and OR groups
a Indicates data presented as mean (SD), b Indicates data presented as frequency (percentage).
Modifiable Factors for Overall Survival
Of all patients that survived to 30 days post-op, 97% had temperature monitoring during surgery compared to only 78% in the patient group that did not survive (p<0.001, Table 6). Of patients that survived to 30 days post-op, 76% had a heating device used during surgery, compared to 39% of patients in the group that did not survive (p<0.001). Of the NICU group, 6 (18.8%) patients had a documented heating device during surgery, compared to 104 (98.1%) patients in the OR group (p<0.001). There was no significant difference in minimum temperature recorded during the operation between survivors and non-survivors.
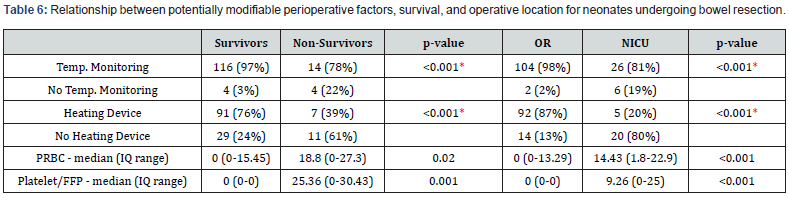
* Indicates Chi-squared analysis. Non-parametric analysis used for continuous variables.
Comparing the amount of packed red blood cells (PRBC) administered during surgery demonstrated a significant difference between patients that survived to 30 days and those who did not (p=0.019). Patients who survived to 30 days received a median amount of 0 mL/kg of PRBCs during surgery, whereas patients that did not survive to 30 days received a median of 18.8 mL/kg of PRBCs. Similarly, the median transfusion volume of platelets and FFP administered in the first 24 hours post-op was different between the survival group (0 mL/kg) and the non-survivors (25.4 mL/kg, p=0.001).
Modifiable factors that were studied but found to be nonsignificant in relation to survival included total paralytic dose, narcotic infusion vs PRN dosing, antibiotic use, type of ventilatory support, type of anesthesia, infant hat usage, and arterial line usage.
Discussion
Bowel resection operations often occur in the NICU at bedside for neonates instead of transferring the patient to the OR. This has been a widely accepted practice with other operations, such as congenital diaphragmatic hernia repair and PDA ligation, showing to be equally safe and effective when performed in the NICU [9,11]. However, little data has been published describing the outcomes or patient population of bowel resections that occur in the NICU versus the OR [12]. Our goal was to identify factors associated with having a bowel resection procedure in the NICU as opposed to the OR, describe outcomes, and analyze modifiable factors that might improve survival.
At our institution, we have anecdotally found that bowel resections in the NICU occur on more unstable patients with a higher risk of mortality. Hence, in contrast to some other studies on neonatal procedures in the NICU, we hypothesized that NICU operation patients would be more unstable and be associated with worse outcomes. This was held true in our data. Patients with a bowel resection in the NICU were less likely to survive to 30 days post-op than patients with the same operation in the OR. We found that patients with bowel resections in the NICU were generally a lower mean gestational age, lower weight, required more oxygen and hemodynamic support prior to surgery, had lower APGAR scores at 1 and 5 minutes, and were more acidotic prior to surgery. Not surprisingly, the NICU group appeared to be a more ill group of patients. These initial descriptive statistics were expected due to the current practice of limiting NICU operations to an as-needed basis for the patients too unstable to transport. We also determined that patients needing an operation in the NICU were more likely to have a diagnosis of spontaneous intestinal perforation or NEC, as opposed to malrotation, small bowel obstruction, and intestinal atresia. Again, the NICU operations were generally performed for more urgent diagnoses. It was likely the patients themselves were the main difference in survival in the NICU vs OR groups, with the fragility of the more premature and underweight neonates in the NICU group that led to their increased mortality.
We were interested in identifying an explanation for the increase in mortality associated with NICU surgery. Therefore, we analyzed post-op infections, intra-operative blood loss, and intra-operative complications. Intra-operative complications were more prevalent in the NICU group. While post-op infections and intra-operative blood loss showed no difference between the two groups, blood loss when defined as >10mL/kg was more evident in the NICU group. This points to blood loss playing an important role in survival. Blood loss in sick patients could be explained by a few different physiological paths including acidosis and hypothermia leading to coagulopathy [13]. Blood loss >10mL/ kg would also put the patient in the transfusion range, with blood product transfusion being known to worsen mortality in neonatal bowel disease [14]. These relationships should be investigated further.
Although the goal of this study was to describe all bowel resection operations, an isolated deep dive into severely ill patients proved to be interesting. When investigating only NEC/ SIP patients to control for illness severity, there was still an increase in mortality associated with NICU surgery, highlighting the fact that patients who remained in the NICU for surgery were more ill and physiologically unstable. In evidence of this, the bivariate analysis showed that these patients were smaller, developmentally younger, and on higher respiratory support. Interestingly, arterial pH and percentage of patients on vasoactive medication were similar preoperatively between NEC/SIP patients who had surgery in NICU vs OR. There was no difference in intraoperative complications or blood loss between the two groups, but in the early post-op period, we did see a higher percentage of NICU patients on vasoactive medications. These patients also had lower urine output for the first 24hrs post-op. This again suggests a higher physiologic toll on patients who stayed in the NICU for surgery. Interestingly, the OR group had higher incidence of early post-op infections, but this could be confounded by the fact that a higher percentage of NICU patients did not survive long enough to have the opportunity to develop an infection.
When further analyzing the causes of death for patients who did not survive to 30 days or discharge, it was evident that the majority of the deaths were related to the operative diagnosis rather than an external factor. An operative diagnosis of NEC was found to be a significant predictor of 30-day mortality on backward stepwise multivariable regression when combined with several other factors. This further supports the relationship between mortality and the indication for bowel resection. Interestingly, the proportion of NICU patients who survived 5 days postoperatively was about 80%, indicating that about half of the NICU deaths occurred after the early postoperative period and may have been unrelated to surgery itself. Still, the hazard ratio for mortality in the NICU group was a staggering 7.9 when compared to neonates who underwent bowel resection in the OR.
We also analyzed modifiable factors that correlated with survival rates. We hypothesized that temperature regulation and blood product administration are modifiable risk factors that would be associated with mortality. The NICU vs OR comparison of modifiable factors mirrored the survival vs non-survival comparison of modifiable factors. This relationship was expected, given that the majority of the patients that did not survive were in the NICU group. The OR and survival groups tended to overlap, while the NICU and non-survival groups overlapped. Temperature monitoring and usage of a heating device were both associated with increased survival. Multiple reports in literature show a relationship between hypothermia and worse outcomes [13,15,16].
Although thermoregulation has data-proven importance in neonates, our institution did not have a standardized hypothermia protocol during operations until 2018. As our data was collected retrospectively, the inconsistencies of the data recording process could confound these results. Although these seem like standard procedure, we would like to emphasize our institution shows room for improvement and believe other intuitions could as well.
Additional modifiable factors that showed correlation with survival were blood product and platelet/FFP administration. Naturally, in the era of data-driven transfusion thresholds, much of the decision making for perioperative blood transfusion is determined by institutional protocols. However, an admittedly unknown portion of the decision for blood transfusion is still based on acute changes in patient status and the subjective assessments of surgeons, anesthesiologists, and neonatologists. The NICU and OR groups had similar overall perioperative blood loss, but there was a higher percentage of patients in the NICU group who experienced >10mL/kg intraoperative blood loss. Blood transfusion is a known risk factor to cause and exacerbate NEC. The term transfusion-associated NEC was developed for this relationship and has been well described in the literature [14]. As NEC was the presenting etiology for bowel resection in many of our patients who had surgery in the NICU, this raises question of whether a higher volume of blood transfusion in the non-survivors a function of their baseline illness was or whether further postop transfusion could be an exacerbating factor of the ongoing illness, leading to increased inflammatory response and worse outcomes. Similarly, more platelet/FFP administration during the first 24 hours post-op was correlated with a decreased survival rate. The isolated cause of the decrease in survival rates with blood product transfusion in this population should be further analyzed in a study of its own.
The main weaknesses of this study revolve around the fact that it was a retrospective study at a single institution. The retrospective nature limited our ability to control the cohort and decision-making in the care of these very ill patients. We could not control the location of the operation, temperature maintenance, or blood product administration. Additionally, it was evident that the electronic charts in older patients were less complete, limiting data collection. Finally, our findings may not be generalizable to all institutions, since there are varying surgical, nursing, and anesthetic practices at different institutions. A prospectively managed multi-institutional cohort could provide a better opportunity to narrow down the root cause in differences in survival between operations performed in the two locations. Unfortunately, such a study would be nearly impossible, as the critically ill nature of these patients often dictates care decisions.
Conclusion
At our institution, the patient population undergoing bowel resections in the NICU had more severe markers of prematurity, were more critically ill, and had particular diagnosis patterns. Patients with operations in the NICU had significantly worse survival than neonates undergoing bowel resection in the operating room. The cause of this difference in survival is likely related to inherent differences in patients who were stable enough for transport to the operating room versus those who needed to remain in the NICU for surgery. These findings may be generalizable to institutions with similar practice patterns. We also identified several clinically relevant modifiable factors that were correlated with differences in survival in the overall cohort, including intraoperative temperature monitoring, temperature maintenance, and blood product administration, but warrant further studies to improve survival in both the NICU and OR.
Author Disclosure
The authors report no proprietary or commercial interest in any product mentioned or concept discussed in this article. The authors report no sources of support for this article.
Author Contributions
Lauren Howser was the project leader and the primary author for the manuscript. Carly Goehring and Eamaan Turk performed data collection. Cassandra Anderson and Sarah Fisher performed statistical analysis and application to the manuscript. Yan Han provided valuable statistical analysis. Dr. Brian Gray oversaw the entire project as the senior author and provided many original thoughts to the analysis. Authors declared to fulfill authorship criteria as devised by ICMJE and approved the final version. The authorship declaration form indicating individual contribution, submitted by the authors, is available with the editorial office.
Consent to Publication
Authors declared taking informed written consent for the publication of clinical photographs/material (not applicable to this project), from the legal guardian of the patient with an understanding that every effort will be made to conceal the identity of the patient, however it cannot be guaranteed.
References
- Wang YL, Jeng SF, Tsao PN, Chou HC, Chen CY, et al. (2015) Operating Room Within the Neonatal Intensive Care Unit-Experience of a Medical Center in Taiwan. Pediatrics & Neonatology 56(4): 220-225.
- Finer NN, Woo BC, Hayashi A, Hayes B (1993) Neonatal surgery: Intensive care unit versus operating room. Journal of Pediatric Surgery 28(5): 645-659.
- Kumar SS, Neogi S (2013) Bedside neonatal intensive care unit surgery- myth or reality! J Neonatal Surg 2(2): 20.
- He ZR, Lin TI, Ko PJ, Tey SL, Yeh ML, et al. (2018) The beneficial effect of air cleanliness with ISO 14644-1 class 7 for surgical intervention in a neonatal intensive care unit: A 10-year experience. Medicine 97(36): e12257.
- Al-Bassam A, Mallick M, Jado A (2008) Surgical procedures performed in the neonatal intensive care unit on critically ill neonates : Feasibility and safety. Annals of Saudi Medicine 28(2): 105-108.
- Geng Q, Wang Y, Li L, Guo C (2018) Early postoperative outcomes of surgery for intestinal perforation in NEC based on intestinal location of disease. Medicine 97(39): e12234.
- Goulet O, Baglin-Gobet S, Talbotec C, Fourcade L, Colomb V, et al. (2005) Outcome and Long-Term Growth After Extensive Small Bowel Resection in the Neonatal Period: A Survey of 87 Children. European Journal of Pediatric Surgery 15(2): 95-101.
- Sholadoye T, Mshelbwala P, Ameh E (2018) Presentation and outcome of treatment of jejunoileal atresia in Nigeria. African Journal of Paediatric Surgery 15(2): 84-87.
- Altokhais T, Gado A, Albassam A, Soomro M (2016) Bedside Neonatal Intensive Care Unit Correction of Congenital Diaphragmatic Hernia: Is Repair without Compromise? American Journal of Perinatology 33(9): 861-865.
- Lago P, Meneghini L, Chiandetti L, Tormena F, Metrangolo S, et al. (2005) Congenital Diaphragmatic Hernia: Intensive Care Unit or Operating Room? American Journal of Perinatology 22(4): 189-197.
- Lee LK, Woodfin MY, Vadi MG, Grogan TR, Ross PJ, et al. (2018) A comparison of postoperative outcomes with PDA ligation in the OR versus the NICU: a retrospective cohort study on the risks of transport. BMC Anesthesiology 18(1): 199.
- Frawley G, Bayley G, Chondros P (1999) Laparotomy for necrotizing enterocolitis: Intensive care nursery compared with operating theatre. Journal of Paediatrics and Child Health 35(3): 291-295.
- Trckova A, Stourac P (2018) Influence of perioperative hypothermia on blood clotting in children. Bratislava Medical Journal 119(5): 294-297.
- Nickel RS, Josephson CD (2015) Neonatal Transfusion Medicine. Clinics in Perinatology 42(3): 499-513.
- Billeter AT, Hohmann SF, Druen D, Cannon R, Polk HC (2014) Unintentional perioperative hypothermia is associated with severe complications and high mortality in elective operations. Surgery 156(5): 1245-1252.
- Qadan M, Gardner SA, Vitale DS, Lominadze D, Joshua IG, et al. (2009) Hypothermia and Surgery: immunologic mechanisms for current practice. Annals of Surgery 250(1): 134-140.






























