Does the Timing of Intubation and IMV Impact Clinical Outcomes in Adult COVID-19 Patients with ARDS: A Systematic Review and Meta-Analysis
Fu Li1, Xiaoqin Zhang2, Long Cheng Tao1, Lorraine Marsons3* and Phillip Dee3
1Department of Critical Care Medicine, The Third Affiliated Hospital of Shenzhen University, Shenzhen, China
2Guangdong Second Provincial General Hospital, China
3Faculty of Health, Education and Life Sciences, Birmingham City University, Birmingham, United Kingdom
Submission:February 03, 2023;Published:October 02, 2023
*Corresponding author: Lorraine Marsons, Faculty of Health, Education and Life Sciences, Birmingham City University, Birmingham, United Kingdom
How to cite this article:Fu L, Xiaoqin Z, Long Cheng T, Lorraine M, Phillip D. Does the Timing of Intubation and IMV Impact Clinical Outcomes in Adult COVID-19 Patients with ARDS: A Systematic Review and Meta-Analysis. Acad J Ped Neonatol 2023; 12(5): 555905. 10.19080/AJPN.2023.12.555905
Abstract
Background/Aim: COVID-19 can rapidly develop into acute lung injury, and even acute respiratory distress syndrome (ARDS), which has a high risk of death. Patients with ARDS often require intubation. However, the timing of intubation and its effect on clinical outcomes in COVID-19 ARDS (CARDS) patients remains unclear. Thus, the authors explored the impact of intubation time on clinical outcomes in COVID-19 patients with ARDS through a systematic review and meta-analysis.
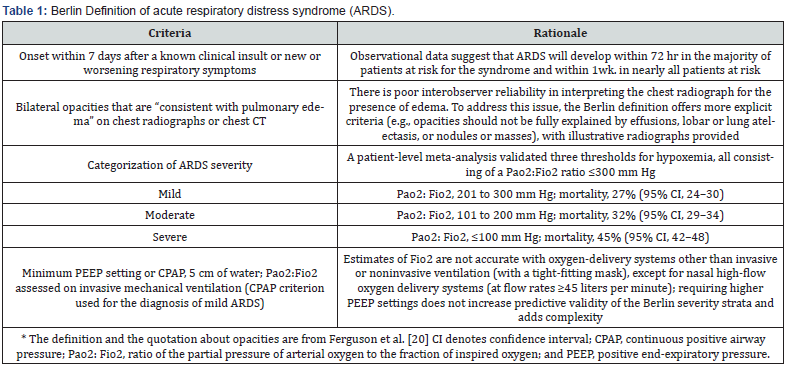
Materials and Methods: Research articles from PUBMED, CINAHL, MEDLINE, ProQuest Covid database, and Web of Science were searched through December 2021. All patients in the research met the Berlin criteria for ARDS. For the purposes of this review, “Early” intubation was defined as intubation within 24 hours of an ARDS diagnosis, while “Late” was defined as 24 or more hours after diagnosis. The primary outcome was ICU mortality, and secondary measures included length of ICU stay and duration of mechanical ventilation. The meta-analysis was performed using a random-effects model. The quality of cohort studies was assessed using the Newcastle-Ottawa Scale. The methodological quality of the overall evidence in this review was evaluated using the GRADE approach.
Results: After an extensive search, six cohort studies were ultimately included in the systematic review, altogether encompassing 2,739 patients with CARDS. A meta-analysis revealed statistically significant differences in mortality [risk ratio (RR)=0.78; 95% confidence interval (CI),0.69-0.88; Z=3.91, P < 0.0001)]. The mortality rate was 36.2% (817 deaths) in the early group and 48.2% (229 deaths) in the late group, respectively. Results of the narrative analysis showed that early intubation resulted in shorter ICU stays, which was statistically significant. However, no statistical difference was found in the duration of mechanical ventilation.
Conclusions: Early intubation can reduce mortality and length of ICU stay in adult COVID-19 patients with ARDS. However, the timing of intubation did not affect the duration of continuous mechanical ventilation.
Keywords: COVID-19; ARDS; Timing of Intubation; Invasive Mechanical Ventilator; Systematic Review; Mortality
Introduction
In December 2019, COVID-19 was identified as a new clinical syndrome caused by a novel coronavirus. The virus is transmittable through the respiratory tract and is highly contagious. Despite significant efforts to control the spread of COVID-19, it triggered a global pandemic [1-4], an epidemic of scale across international borders [5]. COVID-19 pneumonia may develop rapidly into acute respiratory distress syndrome (ARDS) with a high risk of death [6]. ARDS is an acute respiratory failure caused by increased pulmonary capillary permeability secondary to inflammatory oedema. It leads to alveolar flooding and subsequent deep hypoxemia, in which intrapulmonary shunt is the most important underlying mechanism [7]. However, ARDS caused by COVID-19 is different from ARDS with any other underlying cause. According to Huang et al. (2020) [8], the onset of ARDS associated with COVID-19 is between 8-12 days. There are two distinct phenotypes of COVID-19-associated ARDS (CARDS), L-type and H-type. Type L presents as pneumonia and is limited to mild inflammation of the subpleural interstation.
It is characterized by low elasticity, atelectasis, normal compliance, and low lung weight. On the other hand, patients with Type H meet typical ARDS criteria, including decreased lung compliance, hypoxemia, bilateral lung infiltration, and increased lung weight [9]. Li and Ma [10] have been reporting on respiratory support strategies for patients with CARDS during the past two years, but how exactly the timing of tracheal intubation and use of invasive mechanical ventilation impacts clinical outcomes is still unclear in patients with CARDS. Delayed intubation can cause autologous lung injury (SILI) due to high respiratory drive pressure [11]. However, intubating patients too early can also be associated with some complications, including ventilatorassociated pneumonia, airway injury, ventilator-induced lung injury, and hemodynamic disorders due to positive pressure ventilation [12]. Six primary studies [13-18] have indicated different results regarding the timing of intubation for patients with CARDS, and currently there is no systematic review relevant to this topic. Therefore, a systematic review is necessary to further explore how the timing of intubation impacts outcomes for these patients.
Materials and Methods
The PRISMA statement, which contains a 27-item checklist and four-phase flow chart [19], is used to help authors report systematic reviews and meta-analyses.
Eligibility Criteria
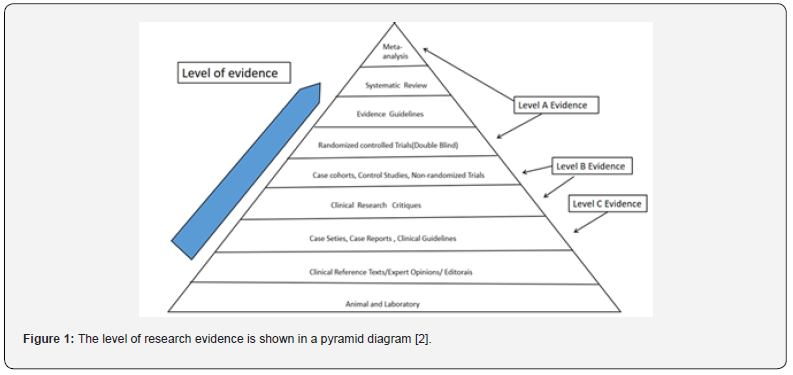
The population included in this systematic review was defined as adult patients (≥18 years old) with PCR-confirmed COVID-19 diagnoses who also had ARDS. ARDS was defined by the Berlin Criteria or American-European Consensus Conference (Table 1) [20]. Early intubation was defined as being intubated within 24 hours of being diagnosed with ARDS; Late intubation was defined as being intubated 24 or more hours after an ARDS diagnosis. The timing of intubation was also defined by authors of four original studies [21]. Systematic reviews and meta-analyses are in the upper echelon of the evidence-based medicine hierarchy of evidence, followed by randomized, controlled, double-blind studies, followed by cohort studies, case-control studies, case series, and case reports [2] (Figure 1). Randomized trial studies were not permitted due to potential ethical issues regarding the timing of intubation of COVID-19 ARDS patients [22]. Therefore, existing cohort studies and case-control studies were sought out to provide high-quality research evidence for this systematic review [23], (Figure 1).
Search Strategy
The authors only searched relevant scientific databases, which included PUBMED, CINAHL, MEDLINE, ProQuest Covid Databases, and Web of Science. Articles were retrieved from December 2019 to December 2021, and the language was restricted to English. In this systematic review, the search strategy developed by the authors consisted of a combination of keywords, medical subject headings (MeSH), free-text words, wildcards, acronyms, synonyms, and transatlantic terms. Boolean operators (“AND” “OR” and “NOT”) were used to combine the terms entered in each search field. Search strategy and keywords are described as follows (Table 2).
Study Selection
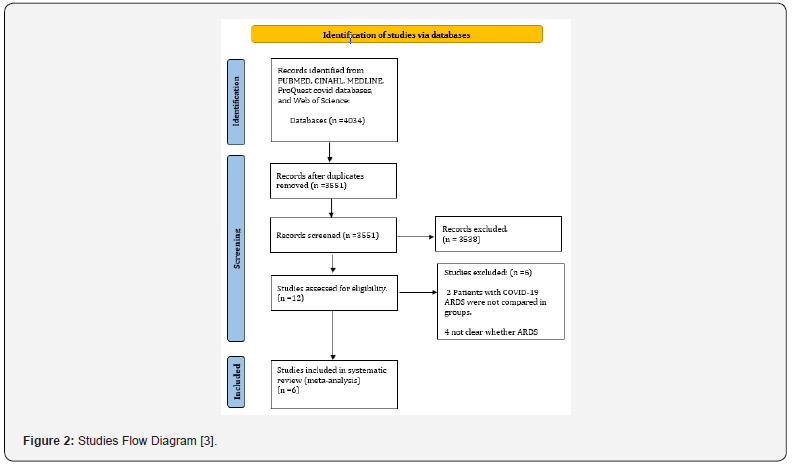
Two authors independently searched for relevant literature by executing the above search strategy and browsing abstracts or full texts to find potential articles. Detailed inclusion and exclusion criteria were used to screen the articles, and six primary research articles were finally selected as suitable for review.
Data Extraction and Risk of Bias Assessment
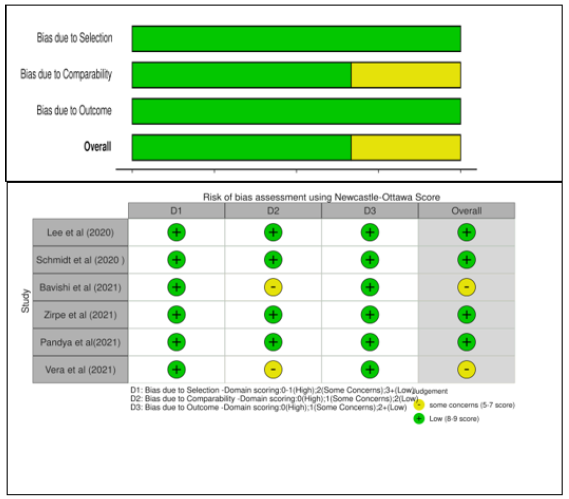
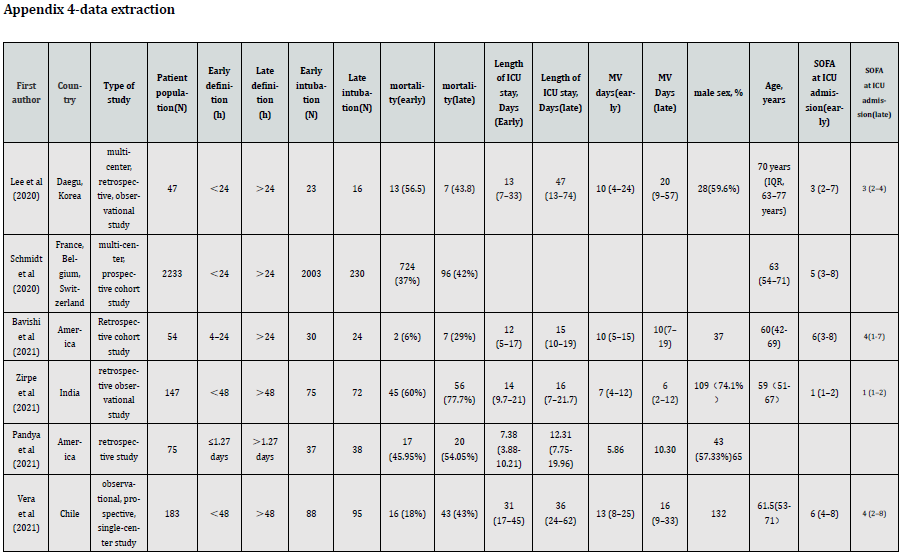
Two reviewers independently extracted and examined data from each included study. Extracted data included article title, author name(s), the date of publication, language, country, characteristics of participants, type of study, and data pertaining to the study’s outcome. Outcomes included mortality, length of ICU stay, and duration of ventilator use. The Newcastle-Ottawa Scale (NOS), developed by the University of Newcastle in Australia and the University of Ottawa in Canada, is a quality assessment tool for the systematic evaluation of non-randomized studies, especially for cohort and case-control studies [24]. The NOS cohort study version consists of eight multiple-choice questions involving topic selection and comparability, as well as outcome assessment or exposure. A star rating system is used to indicate the quality of the study, up to a maximum rating of nine stars. One star is awarded for each criterion if the reporting methodology is appropriate. Separate scales have been developed for cohort and case-control studies, which can help authors identify low-quality studies and inform sensitivity analyses or meta-regression [25]. NOS developers have examined NOS face and standard validity, reliability among evaluators, and evaluator burden. Surface validity has been assessed as strong by comparing each assessment item with its stem problem [26]. Therefore, NOS can be a helpful tool in assessing the quality of studies included in systematic reviews.

Data Synthesis
Stroup et al. published criteria for conducting and reporting meta-analyses of observational studies to improve the quality of reporting. Dichotomous data and risk ratio (RR) were chosen for data synthesis. The most common, a 95% confidence interval, is used to analysed mortality and favourable outcomes. Narrow confidence intervals are used to indicate that treatment estimates are relatively accurate [27]. In the second stage, the pooled (combined) intervention effect estimates are calculated as a weighted average of the estimated intervention effects in a single study. There are four methods for binary results meta-analysis, including three fixed-effect methods (Mantel-Haenszel, Peto, and inverse variance) and one random-effects method (Der Simonian and Laird inverse variance) [28]. For this systematic review, Rev Man software from the Cochrane Review was used for data analysis. The results were presented using forest maps.
Results
Study Selection.

Assessment Quality
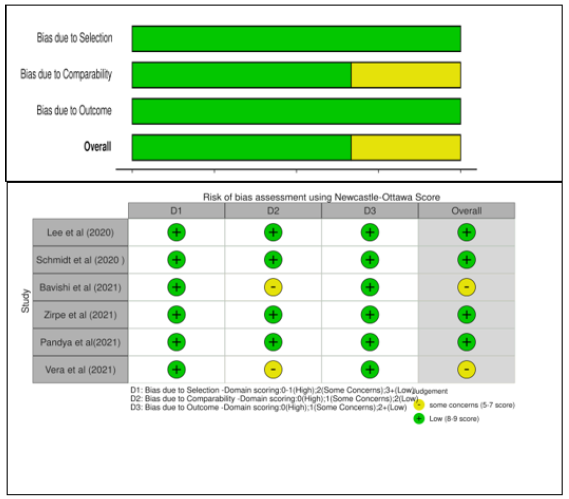
The Newcastle-Ottawa Scale was used to evaluate the quality of the six studies [32] (see Appendix 3). There are detailed evaluation records for each study, as well as summary tables for each of the six studies. The traffic-light plots and summary bar plots were created using the robvis tool (Figure 3), which is a web application for visualizing deviation risk assessment as part of a system assessment [33]. Selection criteria, comparability, and outcome (cohort) or exposure (case-control) were scored on a scale up to 9 (Figure 3).
Main Outcome
The forest plot showed that 2,731 participants across six studies were included in the meta-analysis with a combined RR = 0.78 (95% CI 0.69 to 0.88, Z=3.91, P < 0.0001) (Figure 4). Overall, the results showed that the mortality of the early intubation group was lower than the late intubation group, and the difference was statistically significant. Significant heterogeneity was observed (I2=63%), which indicated a large degree of variation between effect sizes in the included studies (Figure 4).
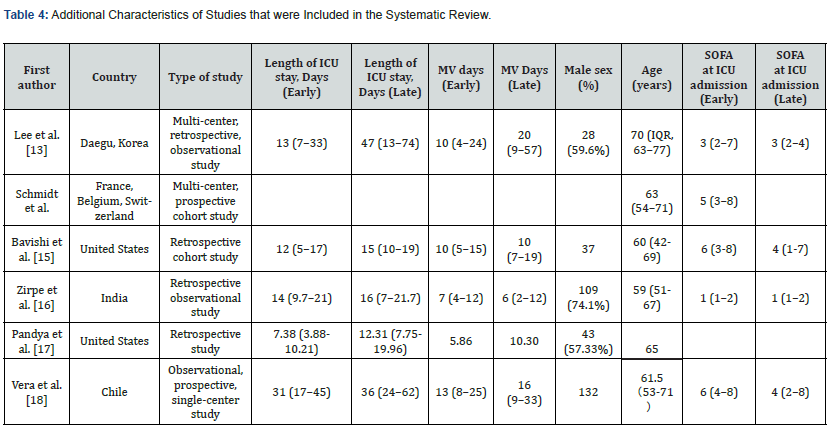
Length of ICU Stay
Schmidt et al. failed to extract the length of ICU stay of participants. No statistically significant differences were found between groups regarding the number of days spent in the ICU in three of the other studies (P > 0.05) [34,35]. Two studies indicated that there were significant differences between the groups (P< 0.05). Overall, since analysis found that early intubation results in shorter ICU stays, this was seen as statistically significant (Table 5).
Duration of Ventilation
Ventilation time was not extracted for the participants in one study Schmidt et al. The other five studies reported no significant difference in the duration of mechanical ventilation between the early intubation group and the late intubation group (P>0.05). Overall, the timing of intubation does not appear to impact the duration of mechanical ventilation of CARDS patients in the ICU (Table 5).
Complications
Patient complications were not reported separately in one study. Lee et al. observed that among patients treated with MV, the incidence of ventilator-associated pneumonia (VAP) in the early intubation group was often higher than that in the late intubation group, but no statistical significance was found (30.4%; N = 7 vs 6.2%; N = 1; P = 0.109). In research by the secondary infection rate was 13.3% in the early intubation group, while it was 22.2% in the late intubation group (P = 0.6). Additionally, AKI/RENAL failure was 21.3% in the early group and 18% in the late group (P = 0.1). A total of 16% of patients underwent tracheostomy in the early group, while the percentage rose to 25% in the late group (P = 0.1). Therefore, no statistically significant differences were found regarding secondary infection, Acute kidney injury (AKI), and interventional tracheotomy between intubation within 48 hours (early group) and intubation 48 hours after ICU admission (late group). Two studies did not describe patient complications.
Discussion
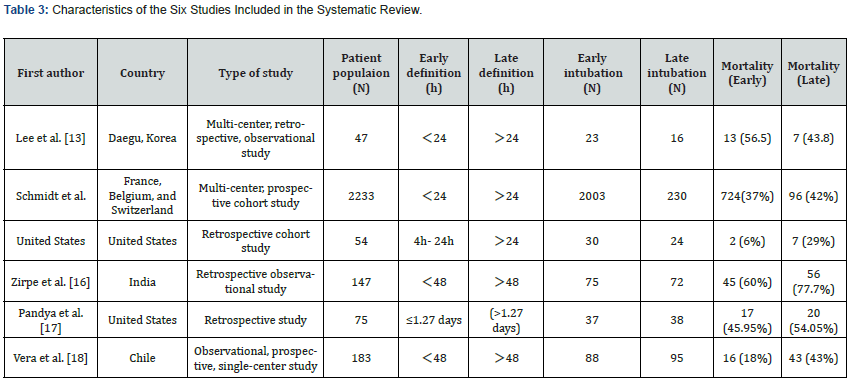
The purpose of this systematic review was to explore the effects of intubation time on clinical outcomes in COVID-19 patients with ARDS. Questions to investigate included whether late intubation increases ICU mortality, length of ICU stay, and duration of ventilator use. Results indicated that early intubation could reduce mortality and length of ICU stay in patients with CARDS. However, intubation time did not affect the duration of continuous mechanical ventilation in patients. There were obvious differences between the definitions of early intubation and late intubation in the six included studies. In three studies early intubation was defined as intubation within 24 hours after diagnosis of ARDS. Two studies defined early intubation as within 48 hours of diagnosis, while defined it as within 1.27 days. Most studies defined the early intubation time as within 24 hours after admission to an ICU [36,37]. However, according to the systematic review reported by the definition of early/late intubation time had no statistical difference in all-cause mortality between the two groups and did not influence the clinical outcomes of COVID-19 patients. Therefore, for the sake of homogeneity, early intubation group and late intubation group data was extracted for analysis according to the respective definitions included in the current study. However, the definition of early/late intubation time directly affected the number of participants between the two groups.
The Effects of Early and Late Intubation
The high mortality associated with late intubation may be related to lung injuries (P-SILI) unintentionally caused by the patients themselves. When COVID-19 patients’ respiratory support was insufficient, their lung function deteriorated in the first week [38]. ARDS is characterized by non-cardiogenic pulmonary oedema, decreased exchange volume of hypoxic blood, and normal gas related to V/Q imbalance, which leads to low respiratory compliance. Hypoxemia may cause patients to inhale spontaneously and violently, leading to lung injury caused by high trans-pulmonary pressure. Early intubation with pulmonary protective ventilation can prevent P-SILI [39]. In a study including 457 ARDS patients, the 60-day mortality rate of the late intubation subgroup (56%) was significantly higher than that of the early intubation group (36%) [40]. The mortality rate of the late intubation group continued to rise during the 2-year follow-up period, which was consistent with the results of the current study.
Chinese critical care experts also suggested that tracheal intubation should be done when critically ill patients are asymptomatic (persistent respiratory distress and/or hypoxemia) after standard oxygen therapy, which was also referred to in the COVID-19 guidelines for patient treatment [41]. Given the high risk of non-invasive respiratory support failure and the risk of virus particle atomization [42], Brown et al. also recommended that early tracheal intubation be performed for patients with respiratory failure who need ventilation support. Other factors influencing death included BMI, age, Sequential Organ Failure Assessment (SOFA) Score, and (Acute Physiology, Age, and Chronic Health Evaluation Ⅱ(APACHEⅡ)); however, no statistical difference was found between the two groups in the early/late stage. In a study by Pandya et al. The included population was characterized by a nasopharyngeal swab-confirmed COVID-19 patient with a mean age of 65 years. A median BMI of 31 was observed in the study, and all patients were more than 50% of the standard BMI and could be categorized as ‘obese’.
These were risk factors associated with mortality. In this study, the mortality rate of patients with mechanical ventilation was as high as 49%. The median age of non-survivors was higher than that of survivors (70 VS 59, p = 0.0006). The median ages of patients in the six included studies were 70, 63, 60, 59, 65, and 61.5, all of which were higher than 59. Therefore, elderly COVID ADRS patients were found to have a higher mortality rate. Compared with the United States, whose patients had a mortality rate of 16.6%, India’s mortality rate was much higher at 68.7%, which may be related to the level of economic development and medical care [43]. Overall, this systematic review found that patients with early intubation were prone to more severe illness, organ dysfunction, and higher SOFA and APACHE scores when diagnosed with ARDS compared to those with late intubation. These results may have great significance in clinical practice, by providing strong evidence for researchers and clinicians to consider when choosing when to intubate COVID-19 ARDS patients. This can assist in rationalizing the allocation of medical resources and reduce the mortality of patients.
Agreements and Disagreements with Other Studies and Reviews
No similar systematic reviews were found pertaining to the topic of this article. Navas-Blanco and Dudaryk (2020) agreed that early intubation can prevent adverse consequences due to delayed intubation in patients with CARDS. Two studies recommend early intubation for COVID-19 ARDS patients. However, a recent review by Papoutsi et al. found no statistically detectable difference in allcause mortality between patients undergoing either early or late intubation (3981 deaths; 45.4% versus 39.1%; RR 1.07,95% CI 0.99-1.15 p = 0.08). The same was true of MV duration (1892; MD- 0.58 days, 95% CI -3.06 to 1.89 days, p = 0.65). Intubation time may have had no effect on the mortality and morbidity of critically ill COVID-19 patients, which was inconsistent with the results summarized in this systematic review. In a study by Papoutsi et al. (2021), participants were critically ill patients with COVID-19. However, the population in this systematic review included ARDS patients with COVID-19.
Critique and Limitation
There are a few notable limitations to this study. For one, the reviewers only searched English-language articles, which can potentially lead to language bias. There may perhaps be articles related to this topic in other languages, but if so, these would have been excluded from the current review. In terms of secondary outcome data extraction, we contacted the authors of the six included studies by email, but failed to obtain specific data on length of ICU stays and MV duration. Therefore, the reliability of secondary measurement results may be reduced.
Conclusion
The findings of this systematic review conclude that early intubation for mechanical ventilation is beneficial to patients with COVID-19 ARDS. However, it appears that early intubation cannot reduce the overall duration of mechanical ventilation. The authors recommend immediate tracheal intubation for patients with moderate to severe COVID-19 ARDS. The treatment and management strategies of ARDS patients have been the continuing focus of researchers. In the face of COVID-19 pandemic, decreasing COVID-19 ARDS patients’ mortality remains an unsolved problem that needs further investigation. Further work is needed to improve research design and solve the problem of high heterogeneity and provide higher quality evidence
Appendix 1
Cinahl Search Strategy

Medline Search Strategy

ProQuest Search Strategy

Web of Science Search Strategy

Pubmed Searchhistory
Appendix 2
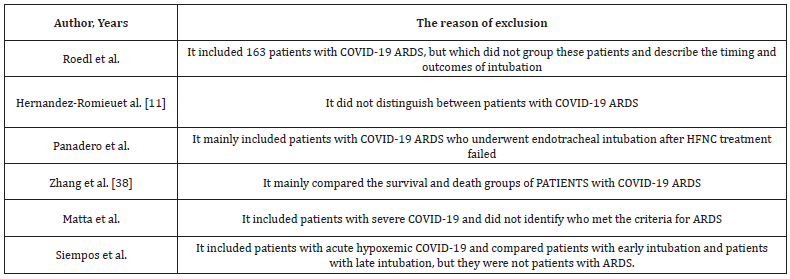
Appendix 3
Etailed Newcastle-Ottawa Scale of each included cohort study.
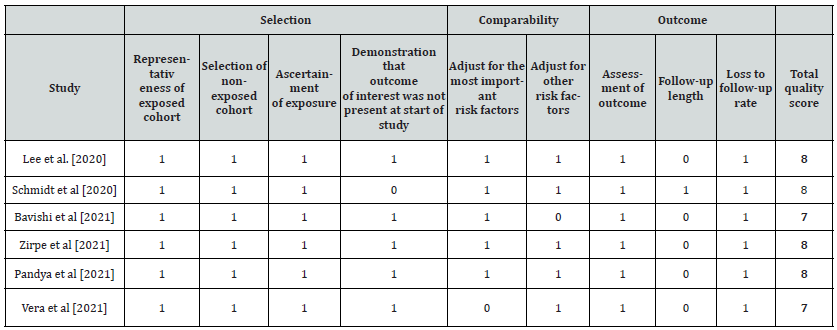
The quality of included studies was assessed by the Newcastle Ottawa scale. A study can be awarded a maximum of one star for each.numbered item within the Selection and Outcome categories and a maximum of two stars for Comparability.
Selection:
1) Representativeness of exposed cohort: 1, study population truly or somewhat representative of a community/ population-based study; 0, study population was sampled from a special population, that is, population from a company, hospital patients, data from the health insurance company or health examination organization, nurses.
2) Selection of non-exposed cohort: 1, drawn from the same community as the exposed cohort.
3) Ascertainment of exposure: 1, Clearly define early intubation time and late intubation time 0, The time of intubation was not clearly. defined.
4) Demonstration that outcome was not present at start of study: 1, SOFA score or APACHE II score have not significant difference
Comparability:
1) Whether a study adjusted for the most important factors deliberately.
2) Whether a study adjusted for other important risk factors.
Outcome:
1) Assessment of outcome: 1, The mortality events were confirmed by medical records or record linkage; 0, self-reported.
2) Was follow-up long enough for outcomes to occur: 1, duration of follow-up >=3 months; 0 if duration of follow-up <3 months.
3) Loss to follow-up rate: 1, complete follow-up or loss to follow up rate <=20 %; 0, follow-up rate < 80% or no description of those lost.
Modified Newcastle - Ottawa Quality Assessment Scale
Cohort Studies
Note: A study can be awarded a maximum of one star for each numbered item within the Selection and
Outcome categories. A maximum of two stars can be given for Comparability.

Schmidt et al (2020)

Bavishi et al (2021)

Zirpe et al (2021)

Pandya et al. (2021)

Study 6: Vera et al (2021)

Cohort Studies: Very Good Studies: 9-10 points;Good Studies: 7-8 points;Satisfactory Studies: 5-6 points.
Unsatisfactory Studies: 0 to 4 points.
References
- Wells GA SB, O’Connell D, Peterson J, Welch V, Losos M, et al. (2011) The Newcastle–Ottawa Scale (NOS) for assessing the quality of non-randomized studies in meta-analyses.
- Rosner AL (2008) EBM: thinking outside of the pyramid. Dynamic chiropractic 26(10): 19.
- Moher D, Liberati A, Tetzlaff J, Altman DG, Group P (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLOS medicine 6(7): e1000097-e1000097.
- Whittle JS, Pavlov I, Sacchetti AD, Atwood C, Rosenberg MS (2020) Respiratory support for adult patients with COVID-19. J Am Coll Emerg Physicians Open 1(2): 95-101.
- Porta MS, Greenland S, Hernán M, et al. (2014) A dictionary of epidemiology. Sixth ed. Oxford University Press.
- Wu C, Chen X, Cai Y, Xia J, Zhou X, et al. (2020) Risk Factors Associated with Acute Respiratory Distress Syndrome and Death in Patients with Coronavirus Disease 2019 Pneumonia in Wuhan, China. JAMA Intern Med 180(7): 934-943.
- Hanley C, Giacomini C, Brennan A, McNicholas B, Laffey JG (2022) Insights Regarding the Berlin Definition of ARDS from Prospective Observational Studies. Semin Respir Crit Care Med 43(3): 379-389.
- Huang C, Wang Y, Li X, Ren L, Zhao J, et al. (2020) Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 395(10223): 497-506.
- Montenegro F, Unigarro L, Paredes G, Moya T, Romero A, et al. (2021) Acute respiratory distress syndrome (ARDS) caused by the novel coronavirus disease (COVID-19): a practical comprehensive literature review. Expert Rev Respir Med 15(2): 183-195.
- Li X, Ma X (2020) Acute respiratory failure in COVID-19: is it "typical" ARDS? Critical care (London, England). 24(1): 198-198.
- Hernandez Romieu AC, Adelman MW, Hockstein MA, Robichaux CJ, Edwards JA, et al. (2020) Timing of Intubation and Mortality Among Critically Ill Coronavirus Disease 2019 Patients: A Single-Center Cohort Study. Critical care medicine 48(11): e1045-e1053.
- Rehman S, Shahiman MA, Khaleel MA, Holý O (2022) Does the intubation timeline affect the in-hospital mortality of COVID-19 patients? A retrospective cohort studies. Front Med (Lausanne) 9: 1023229.
- Lee YH, Choi KJ, Choi SH, Lee SY, Kim KC, et al. (2020) Clinical Significance of Timing of Intubation in Critically Ill Patients with COVID-19: A Multi-Center Retrospective Study. J Clin Med 9(9): 2847.
- COVID-ICU Group on behalf of the REVA Network and the COVID-ICU Investigators (2021) Clinical characteristics and day-90 outcomes of 4244 critically ill adults with COVID-19: a prospective cohort study. Intensive Care Med 47(1): 60-73.
- Bavishi A, Mylvaganam R, Agrawal R, Avery R, Cuttica M (2021) Early vs Late Intubation in COVID-19 acute respiratory distress syndrome: A Retrospective Study of Ventilator Mechanics, Computed Tomography Findings, and Outcomes. TP49 TP049 COVID: ARDS AND ICU MANAGEMENT. American Thoracic Society 2021: A2546-A2546.
- Zirpe KG, Tiwari AM, Gurav SK, Deshmukh AM, Suryawanshi PB, et al. (2021) Timing of Invasive Mechanical Ventilation and Mortality among Patients with Severe COVID-19-associated acute respiratory distress syndrome. Indian J Crit Care Med 25(5): 493-498.
- Pandya A, Kaur NA, Sacher D, O'Corragain O, Salerno D, et al. (2021) Ventilatory Mechanics in Early vs Late Intubation in a Cohort of Coronavirus Disease 2019 Patients With ARDS: A Single Center's Experience. Chest 159(2): 653-656.
- Vera M, Kattan E, Born P, Rivas E, Amthauer M, et al. (2021) Intubation timing as determinant of outcome in patients with acute respiratory distress syndrome by SARS-CoV-2 infection. Journal of Critical Care 65: 164-169.
- Moher D, Liberati A, Tetzlaff J, Altman DG; PRISMA Group (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med 6(7): e1000097.
- Ferguson ND, Fan E, Camporota L, Antonelli M, Anzueto A, et al. (2012) The Berlin definition of ARDS: an expanded rationale, justification, and supplementary material. Intensive Care Med 38(10): 1573-1582.
- Papoutsi E, Giannakoulis VG, Xourgia E, Routsi C, Kotanidou A, et al. (2021) Effect of timing of intubation on clinical outcomes of critically ill patients with COVID-19: a systematic review and meta-analysis of non-randomized cohort studies. Critical care (London, England) 25(1): 121.
- Joag K, Ambrosio G, Kestler E, Weijer C, Hemming K, et al. (2019) Ethical issues in the design and conduct of stepped-wedge cluster randomized trials in low-resource settings. Trials 20(Suppl 2): 703-703.
- Shah HM, Chung KC (2009) Archie Cochrane and his vision for evidence-based medicine. Plastic and reconstructive surgery 124(3): 982-988.
- Margulis AV, Pladevall M, Riera-Guardia N, Varas-Lorenzo C, Hazell L, et al. (2014) Quality assessment of observational studies in a drug-safety systematic review, comparison of two tools: the Newcastle-Ottawa Scale and the RTI item bank. Clinical epidemiology 6(default): 359-368.
- Luchini C, Veronese N, Nottegar A, Shin JI, Gentile G, et al. (2021) Assessing the quality of studies in meta-research: Review/guidelines on the most important quality assessment tools. Pharm Stat 20(1): 185-195.
- Hartling L, Milne A, Hamm MP, Vandermeer B, Ansari M, et al. (2013) Testing the Newcastle Ottawa Scale showed low reliability between individual reviewers. J Clin Epidemiol 66(9): 982-993.
- Higgins JPT, Thomas J, Chandler J, et al. (2019) Cochrane Handbook for Systematic Reviews of Interventions. 2 ed. Wiley Cochrane Series. John Wiley & Sons.
- O'Brien SF, Yi QL (2016) How do I interpret a confidence interval? Transfusion 56(7): 1680-1683.
- Lo CK-L, Mertz D, Loeb M (2014) Newcastle-Ottawa Scale: comparing reviewers to authors' assessments. BMC medical research methodology 14: 45-45.
- McGuinness LA, Higgins JPT (2021) Risk-of-bias VISualization (robvis): An R package and Shiny web app for visualizing risk-of-bias assessments. Res Synth Methods 12(1): 55-61.
- Lee SC, Son Kang J, Han CH, Park SC, Jung Ji Y (2021) Impact of COPD on COVID-19 prognosis: A nationwide population-based study in South Korea. Sci Rep 11(1): 3735.
- Bavishi AA, Mylvaganam RJ, Agarwal R, Avery RJ, Cuttica MJ (2021) Timing of Intubation in Coronavirus Disease 2019: A Study of Ventilator Mechanics, Imaging, Findings, and Outcomes. Crit Care Explor 3(5): e0415.
- Bauer PR, Gajic O, Nanchal R, Kashyap R, Martin-Loeches I, et al. (2017) Association between timing of intubation and outcome in critically ill patients: A secondary analysis of the ICON audit. Journal of critical care 42: 1-5.
- Mellado-Artigas R, Ferreyro BL, Angriman F, Hernández-Sanz M, Arruti E, et al. (2021) High-flow nasal oxygen in patients with COVID-19-associated acute respiratory failure. Crit Care 25(1): 58.
- Cruces P, Retamal J, Hurtado DE, Erranz B, Iturrieta P, et al. (2020) A physiological approach to understand the role of respiratory effort in the progression of lung injury in SARS-CoV-2 infection. Crit Care 24(1): 494.
- Gattinoni L, Chiumello D, Rossi S (2020) COVID-19 pneumonia: ARDS or not? Crit Care 24(1): 154.
- Kangelaris KN, Ware LB, Wang CY, Janz DR, Zhuo H, et al. (2016) Timing of Intubation and Clinical Outcomes in Adults With Acute Respiratory Distress Syndrome. Crit Care Med 44(1): 120-129.
- Zuo MZ, Huang YG, Ma WH, Xue ZG, Zhang JQ, et al. (2020) Expert Recommendations for Tracheal Intubation in Critically ill Patients with Noval Coronavirus Disease 2019. Chin Med Sci J 35(2): 105-109.
- Brown CA 3rd, Mosier JM, Carlson JN, Gibbs MA (2020) Pragmatic recommendations for intubating critically ill patients with suspected COVID-19. J Am Coll Emerg Physicians Open 1(2): 80-84.
- Palaiodimos L, Kokkinidis DG, Li W, Karamanis D, Ognibene J, et al. (2020) Severe obesity, increasing age and male sex are independently associated with worse in-hospital outcomes, and higher in-hospital mortality, in a cohort of patients with COVID-19 in the Bronx, New York. Metabolism 108: 154262-154262.
- Navas-Blanco JR, Dudaryk R (2020) Management of Respiratory Distress Syndrome due to COVID-19 infection. BMC Anesthesiology 20(1): 177.
- Marini JJ, Gattinoni L (2020) Management of COVID-19 Respiratory Distress. Jama 323(22): 2329-2330.
- Stern C, Kleijnen J (2020) Language bias in systematic reviews: you only get out what you put in. JBI Evid Synth 18(9): 1818-1819.






























