Superior Mesenteric Artery Syndrome in an Adolescent with No Significant Weight Loss: A Case Report
Christina Grabar1*, Madalin Berra2and Katherine Andreeff3
1From University of California Irvine School of Medicine, Orange, California, USA
2From University of California Irvine/CHOC Residency program, Orange, California, USA
3From CHOC Children’s, Orange, California, USA
Submission: October 10, 2021; Published: December 15 2021
*Corresponding author:Christina Grabar, From University of California Irvine School of Medicine, Orange, California, USA
How to cite this article:Christina G, Madalin B, Katherine A. Superior Mesenteric Artery Syndrome in an Adolescent with No Significant Weight Loss: A Case Report. Acad J Ped Neonatol 2021; 11(2): 555862. 10.19080/AJPN.2021.11.555862
Summary
This case report discusses a case of superior mesenteric artery (SMA) syndrome in a previously healthy 16-year-old male without significant weight loss or usual risk factors. This patient presented to the emergency department with 4 days of bilious vomiting and epigastric pain that started after ingestion of a large meal and intense exercise. During his illness he was seen at 3 different healthcare facilities before receiving a diagnosis at the fourth visit. SMA syndrome was initially suspected based on the location of obstruction on CT but was not confirmed until upper endoscopy was performed. In puberty, males have an increase in lean body mass with concurrent loss of adipose. With these changes, the angle between the aorta and the superior mesenteric artery can narrow with decrease in mesenteric fat.
The consumption of a large meal and intense exercise lead to an obstruction at the narrowed junction, which manifested in SMA syndrome. This patient’s SMA angle was measured at 16 degrees and the distance was 5 mm. This case demonstrates that pubertal growth spurt without increase in adipose tissue and acute ingestion of a large meal can trigger SMA syndrome, an angle less than 22 degrees and distance less than 8 mm strongly indicates SMA syndrome, and in cases of pediatric patients with recurrent abdominal symptoms the differential diagnosis should include SMA syndrome.
Background
Superior mesenteric artery syndrome is a relatively rare etiology of proximal gastrointestinal obstruction, with the incidence estimated at 0.1% to 0.3% [1]. It is characterized by compression of the third portion of the duodenum due to narrowing of the space between the superior mesenteric artery and abdominal aorta. The compression occurs primarily due to loss of the mesenteric fat pad surrounding the duodenum. Historically, the most common contributing factor to SMA syndrome is significant weight loss leading to loss of the mesenteric fat. Other factors include psychological disorders, anatomical and congenital anomalies, including spinal surgery for scoliosis, intestinal malrotation, and a more proximal insertion of the duodenum at the ligament of Treitz [2]. SMA syndrome typically presents with bilious emesis, postprandial epigastric pain, early satiety, anorexia, and nausea. Patients with SMA syndrome most commonly present after significant weight loss or following spinal surgery [3]. Therefore, it is abnormal to consider SMA syndrome in a patient that does not have the typical risk factors such as weight loss, spinal surgery, or psychological conditions.
Case Presentation
A previously healthy 16-year-old boy presented to the emergency room with acute bilious vomiting, chills, and epigastric pain. The patient had soccer tryouts the week prior to admission, he was able to perform as expected but reported feeling fatigued and lethargic during the physical activity. The patient denied having history of trauma to the abdomen during this time. Emesis and abdominal pain began abruptly after ingestion of a large meal following the conclusion of tryouts. Relieving factors included changes of positioning, such as lying in the fetal position. At baseline the patient was an avid eater and very active with participation in soccer. He reported a history of subjective difficulty gaining weight but denied any acute weight loss. He had never been diagnosed with malnutrition or failure to thrive an his BMI was in the 12th percentile on admission to the hospital. He reported one prior similar episode of bilious emesis a few months prior, which resolved within the day, he did not seek medical care at that time.
After two days of persistent bilious emesis occurring 20-30 times per day, the patient went to an emergency department. His symptoms were attributed to food poisoning, and he was given IV fluids and Zofran, he was able to briefly tolerate food by mouth after these interventions and was discharged home. However, the abdominal pain returned, and the vomitus became green, likely bilious, so patient went to an urgent care. He was given a similar diagnosis of food poisoning and was prescribed Reglan for symptomatic control. He trialed over-the-counter pro-biotics. His discomfort returned and he experienced a new symptom of shortness of breath. His family called emergency services and he was transferred to another ED by ambulance. He was treated with 100% O2, Haldol, Zofran, Benadryl, Reglan, which alleviated all of his symptoms including the dyspnea, and he was again sent home. His abdominal symptoms and emesis persisted at home, and the patient ultimately presented to our emergency department the following day
On presentation, his vital signs were stable. The patient had a slim, muscular build, with a height and weight of 177 cm and 56.1 kg, respectively. Abdominal exam revealed diffuse tenderness with guarding of the right lower quadrant with no rebound or distention. Complete blood count revealed mild leukocytosis with normal hemoglobin. Complete metabolic panel demonstrated hyperglycemic, hyperchloremic anion gap metabolic alkalosis with an acute kidney injury (AKI) due to dehydration. Liver function tests and inflammatory markers were within normal limits. Due to his right lower quadrant tenderness on initial exam, an ultrasound of the appendix was performed and was not able to visualize the appendix due to overlying fluid-filled bowel loops.
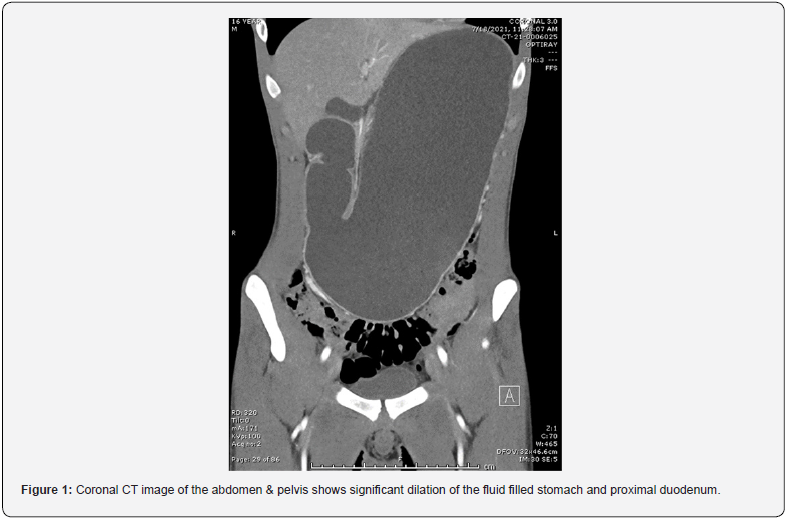
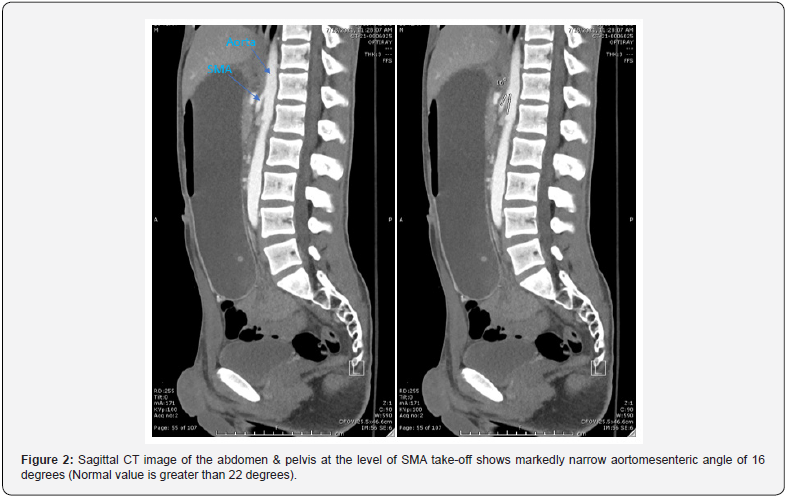
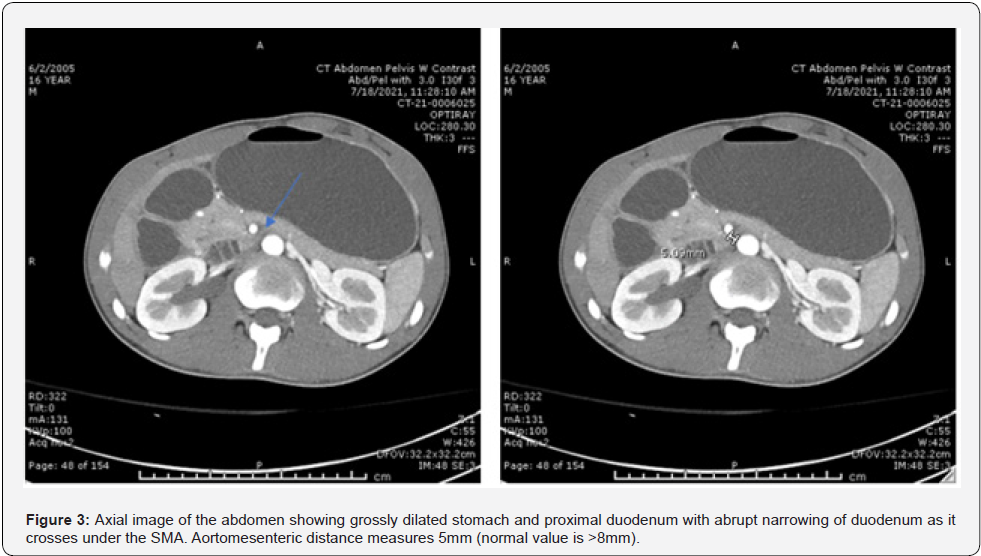
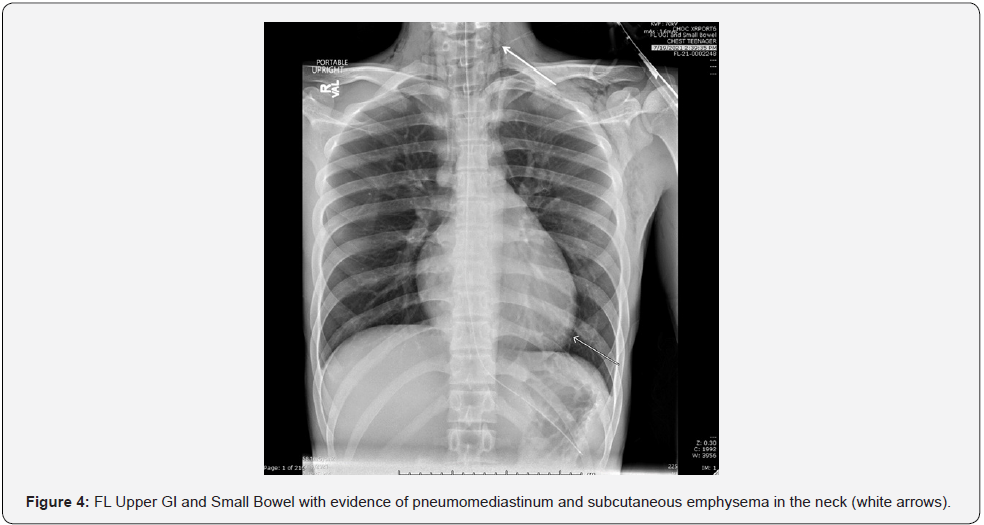
Following the inconclusive ultrasound, a CT scan with contrast of the abdomen and pelvis was performed. This revealed markedly distended stomach and duodenum (Figure 1) to the mid aspect of the transverse segment within normal remaining bowel loops and normal appendix. The location of the obstruction and narrowed aortomesenteric (AOM) angle and distance were anatomically consistent with SMA syndrome (Figures 2 & 3), but other etiologies of duodenal obstruction, such as duodenal web or other intraluminal process were possible. To relieve the patient’s abdominal distention, a Salem-sump was placed to suction with immediate output of gastric contents. The clinical history seemed inconsistent with SMA syndrome so further diagnostic steps were taken. An upper GI showed high grade partial obstruction at approximately the level of the D2-D3 junction with incidental pneumomediastinum with pneumopericardium and subcutaneous emphysema in the neck bilaterally, left axilla, and proximal left upper extremity (Figure 4).
Although there was low concern for esophageal perforation given the recent negative CT scan and the patient had no emesis or bloody output since admission, an esophagram was performed.
The esophagram demonstrated no evidence of esophageal leak or obstruction with grossly stable pneumopericardium and pneumomediastinum. These unexpected findings were determined to likely be due to transient Boeerhave tear from severe vomiting and retching. It is possible that his dyspnea and hyperventilation prior to admission could have been related to the development of pneumomediastinum from severe retching, as it can cause shortness of breath and chest pain when it first occurs.
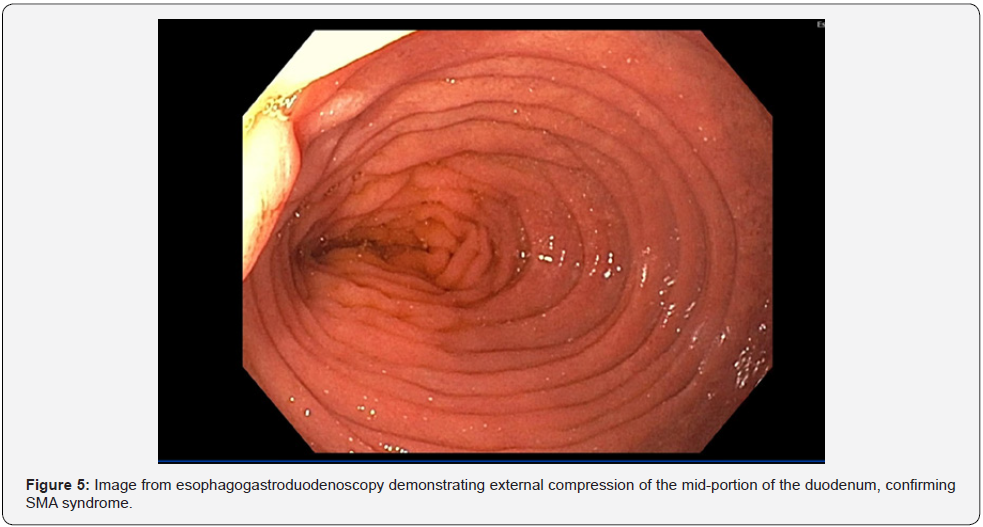
The following day an esophagogastroduodenoscopy showed external compression of the mid-portion of the duodenum (Figure 5), consistent with SMA syndrome, and nodular gastric antrum consistent with H. pylori.
To restore the mesenteric fat pad around the duodenum, aggressive caloric supplementation was pursued. A PICC line was placed and TPN was initiated with continuous monitoring of weight and labs. He was conservatively monitored for signs of refeeding syndrome, but magnesium, phosphate, and potassium all remained within normal limits. On hospital day 6 his Salem sump tube output had slowed significantly, and an attempt was made to clamp the tube. The patient tolerated this without issue. On hospital day 7, the Salem sump was removed, and he was allowed a clear liquid diet. His diet was advanced slowly and by hospital day 9 he was tolerating a soft surgical diet with adequate caloric intake. He experienced no nausea or vomiting during this time. His TPN was tapered and then discontinued. His weight was closely monitored, early on in his admission his weight dropped to a low value of 55.2 kg, at time of discharge on hospital day 11 he weighed 57.8 kg. At his first outpatient follow-up one week after discharge, the patient was cleared by GI to resume regular activity and to maintain a soft diet. Patient will be followed closely by both GI and his general pediatrician.
Discussion
During puberty, males typically undergo a significant increase in lean body mass with a concurrent loss of adipose [4]. Changes like this in body composition can lead to a narrowing of the aortomesenteric space and angle due to a decrease in the mesenteric fat density. Therefore, this study adds to prior literature indicating that a growth spurt is a risk factor for SMA syndrome [3,5,6] Possible triggers for compression include additional weight loss due to infection [6] and excessive ingestion of food [7]. In the current case, this patient had a history of difficulty gaining weight and had ingested an excessive amount of food after moderate physical activity. There have been other reports of excessive ingestion or food or water triggering SMA syndrome [5] and this case further supports this notion. Though the exact mechanism is unclear, the acute consumption of food lead to a transient obstruction that was mildly alleviated with position changes.
This case further supports the fact that significant weight loss is not a requirement for the development of SMA syndrome in pediatric patients [8]. Biank and Werlin found that of the 22 pediatric cases of SMA syndrome, 50% had no weight loss prior to becoming symptomatic [3]. The underlying etiology of SMA syndrome in the pediatric population is not fully understood, but this case supports prior hypotheses that inadequate weight gain relative to height can lead to a decrease in mesenteric fat and predisposes children and adolescents to develop SMA syndrome.
A child or adolescent presenting with bilious vomiting should be assumed to have an emergent intestinal obstruction until proven otherwise. Though it would be abnormal to present in adolescence, imaging such as an upper GI should be performed to rule out malrotation as a cause of bilious vomiting. In another case of SMA syndrome in a healthy adolescent, an upper GI was not performed prior to patient discharge, therefore malrotation was not ruled out and the underlying cause of intestinal obstruction was not identified [5]. It is vital to perform proper imaging when intestinal obstruction is suspected prior to patient discharge.
Symptoms of SMA syndrome are caused compression by the narrow angle of the superior mesenteric artery pressing against the duodenum. In conjunction with clinical suspicion, diagnosis of SMA syndrome can be facilitated by measurement of the angle between the aorta and SMA as measured on CT or ultrasound [8,9] The normal aortamesenteric angle is between 45 and 60 degrees and the normal AOM distance is between 10-20 mm [9,10]. AOM angles less than 22 to 28 degrees with an AOM distance between 2-8 mm is highly suggestive of SMA syndrome [11,12]. An angle less than 22 degrees in conjunction with one symptom of SMA syndrome (emesis, postprandial epigastric pain, anorexia, weight loss) has a sensitivity of 42.8% and specificity of 100%.13 A distance of 8 mm or less has a sensitivity and specificity of 100% [13]. This patient’s AOM angle was 16 degrees (Figure 2) and AOM distance of 5.09 mm (Figure 3), which strongly indicates SMA syndrome. AOM angle and distance have been used to confirm diagnosis of SMA [9] and were consistent with a diagnosis in this case. This patient’s atypical presentation led to further diagnostic studies such as EGD which further supported the diagnosis. Though the procedure was performed to ensure the patient did not have an esophageal web or other obstruction not evidenced by CT, with the sensitivity and specificity of the AOM angle and distance, an endoscopy might not have been needed to diagnose SMA syndrome.
In the pediatric population, recurrent abdominal symptoms with spontaneous resolution frequently occur in emergency care. This case is an example of this concept, as this patient went to three emergency care settings before imaging and admission. As previously stated, growth spurt without adequate weight gain and acute consumption of a large amount of food and water have been identified as triggers for SMA syndrome. With recurrent abdominal symptoms and growth spurts both being common in pediatrics, it is possible that SMA syndrome might be more prevalent in pediatric patients than previously thought [14,15]. Additionally, a prior case report demonstrated a patient with history of recurrent abdominal symptoms with spontaneous resolution and was discharged before CT results were reviewed and patient was diagnosed with SMA syndrome [5]. This prior study and the present case’s resolution of symptoms with positioning and fluids and medications at prior medical facilities imply that transient SMA syndrome may be responsible for some portion of unsolved abdominal symptoms in the pediatric population. Therefore, SMA syndrome should be included in the differential for children or adolescents with recurrent abdominal symptoms.
Learning points:
a) Weight loss is not a necessary risk factor for SMA syndrome in the pediatric population and should be included in the differential for adolescents with recurrent abdominal symptoms
b) Complications of SMA syndrome can potentially lead to esophageal rupture or pneumothorax due to forceful emesis and physicians should observe for pneumomediastinum and pneumopericardium on imaging
c) Excessive ingestion of food or water can trigger the compression of the SMA over the duodenum though the exact mechanism is unclear
d) Intermittent, but intractable bilious vomiting in a pediatric patient with several recent trips to healthcare providers should have a thorough workup, including abdominal CT to rule out SMA syndrome
e) AOM angle <22 degrees and AOM distance <8 mm is strongly suggestive of SMA syndrome and should be used when etiology of intestinal obstruction or abdominal symptoms is unclear.
References
- Van Horne N, Jackson JP (2021) Superior Mesenteric Artery Syndrome. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing.
- Welsch T, Büchler MW, Kienle P (2007) Recalling superior mesenteric artery syndrome. Dig Surg 24(3): 149-156.
- Biank V, Werlin S (2006) Superior mesenteric artery syndrome in children: a 20-year experience. J Pediatr Gastroenterol Nutr 42(5): 522-525.
- Holland Hall C, Burstein GR (2016) Adolescent physical and social development. In: Kliegman RM, Stanton BF, St Geme JW, Schor NF, . eds. Nelson textbook of pediatrics. 20th (edn). Philadelphia: Elsevier.
- Okamoto T, Sato T, Sasaki Y (2019) Superior mesenteric artery syndrome in a healthy active adolescent. BMJ Case Rep 12(8): e228758.
- Lee TH, Lee JS, Jo Y, Jae Hee Cheon, Yong Sung Kim, et al. (2012) Superior mesenteric artery syndrome: where do we stand today? J Gastrointest Surg 16(12): 2203-2211.
- Nishiguchi S, Shirobe T (2016) Acute abdominal pain caused by superior mesenteric artery syndrome in a healthy young boy. BMJ Case Rep 2016: bcr2016217512.
- Kothari TH, Machnicki S, Kurtz L (2011) Superior mesenteric artery syndrome. Can J Gastroenterol 25(11): 599-600.
- Kane KE, Koons AL (2017) The Aortomesenteric Angle as an Aid in Diagnosing Superior Mesenteric Artery Syndrome. Clin Pract Cases Emerg Med 1(2): 140-141.
- Keçeli M (2021) The Superior Mesenteric Artery Angle and Abdominal Main Vessel Diameters in Normovolemic Children: For Practical Sonographic Evaluation. Ultrasound Q 37(4): 343-348.
- Ranschaert, E Weerakkody Y (2021) Superior mesenteric artery syndrome. Radiopaedia.org.
- Agrawal GA, Johnson PT, Fishman EK (2007) Multidetector row CT of superior mesenteric artery syndrome. J Clin Gastroenterol 41(1): 62-65.
- Unal B, Aktaş A, Kemal G, Yasemin Bilgili, Sefa Güliter, et al. (2005) Superior mesenteric artery syndrome: CT and ultrasonography findings. Diagn Interv Radiol 11(2): 90-95.
- Waheed KB, Shah WJ, Jamal A, Hassan R Mohammed, Bilal Altaf, et al. (2021) Superior mesenteric artery syndrome: An often overlooked cause of abdominal pain! Saudi Med J 42(10): 1145-1148.
- Bakker ME, van Delft R, Vaessens NA, P R R Rosias (2014) Superior mesenteric artery syndrome in a 15-year-old boy during Ramadan. Eur J Pediatr 173(12): 1619-1621.






























