Romiplostim in Eltrombopag Refractory Thrombocytopenia Post Bone Marrow Transplant in a Child with MDS: A Case Report
Farah Malaeb, MD* and Miguel R. Abboud, MD
American University of Beirut, Beirut, Lebanon
Submission: October 21, 2021; Published: November 30 2021
*Corresponding author: Farah Malaeb, MD, American University of Beirut, Beirut, Lebanon, Tel: 0096170922613, 009611350000
How to cite this article:Farah M, Miguel A. Romiplostim in Eltrombopag Refractory Thrombocytopenia Post Bone Marrow Transplant in a Child with MDS: A Case Report. Acad J Ped Neonatol 2021; 11(1): 555859. 10.19080/AJPN.2021.11.555859
Abstract
Thrombocytopenia is a well-known complication post bone marrow transplant, whether primary or secondary. Refractory thrombocytopenia is not uncommon and over the past few years, thrombopoietin receptor agonists have emerged as new agents for the management of the above. Namely Eltrombopag and more recently Romiplostim. In children however, though Eltrombopag is being more frequently used, Romiplostim usage is still rarely reported for management of thrombocytopenia post bone marrow transplantation. Our experience with Romiplostim was successful in the management of refractory thrombocytopenia in a child with MDS post bone marrow transplantation.
Case Presentation
We report to you the case of our patient, who is currently a 5-year-old child with myelodysplastic syndrome, who was initially referred to us at 3 years of age for further workup of thrombocytopenia and anemia.
Initial bone marrow exam done showed a cellular marrow with dyserythropoiesis and 1% blasts, and a normal karyotype. One year after diagnosis, she underwent stem cell transplantation from her fully matched sibling donor. During this year, the child was transfusion dependent and required weekly packed RBC and monthly platelets transfusions. Bone marrow exam done prior to transplant showed a hypercellular marrow with markedly left shifted myelopoiesis, dyserythropoiesis and megakaryocytic hyperplasia and again, no increase in blasts.
She underwent stem cell transplantation from her fully matched sibling donor, conditioning chemotherapy Busulfan and Cyclophosphamide, GVHD prophylaxis with cyclosporin A and Methotrexate. Upon admission for transplant, the child was found to have refractory thrombocytopenia, requiring Rituximab prior to transplant with no significant improvement in her platelet count, in addition to IVIG post-transplant, with no major changes likewise. The child had a smooth transplant course otherwise, she engrafted on day +19. Eltrombopag was introduced, starting at 25 mg daily, then increased to 50 mg daily; the child continued to have severe thrombocytopenia requiring platelets transfusion every 5 days, when the platelets dropped below 10,000/mm3.
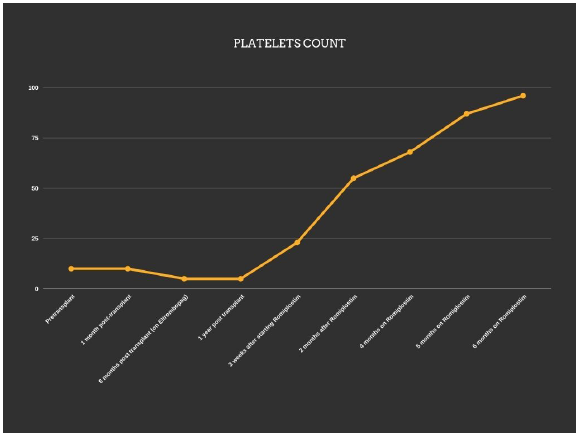
Bone marrow exam was done on day +35, showing a 70% marrow with maturing trilineage hematopoiesis and 99% chimerism. Repeat bone marrow exam on day 180 showed similar results. During that period, she continued to receive 50 mg of Eltrombopag daily, with no change in her platelet count. On day +200, she was started on Romiplostim at 2 mcg per kg subcutaneously once weekly. The patient’s platelets started to recover 3 weeks after starting Romiplostim and the last platelets transfusion she received was 2 weeks post Romiplostim initiation. Currently, over a year post transplant, the patient is still on Romiplostim, which was increased to 3 mcg per kg subcutaneously weekly and the platelets are consistently above 50,000/mm3 (refer to the graph below).
Of note, our patient has never had a life-threatening bleeding despite her persistent severe thrombocytopenia.
Discussion
Almost all patients who undergo HSCT develop pancytopenia after the conditioning regimen, and persistent thrombocytopenia frequently occurs during the posttransplant course because platelets are the last blood component to recover [1,2]. Thrombocytopenia could be either primary, where the platelet count never recovers, or secondary, where it recovers for a certain period and then thrombocytopenia recurs. Thrombocytopenia may be due to decreased platelet production, increased platelet Almost all patients who undergo HSCT develop pancytopenia after the conditioning regimen, and persistent thrombocytopenia frequently occurs during the posttransplant course because platelets are the last blood component to recover [1,2]. Thrombocytopenia could be either primary, where the platelet count never recovers, or secondary, where it recovers for a certain period and then thrombocytopenia recurs. Thrombocytopenia may be due to decreased platelet production, increased platelet of platelet production, from the stem cell through development of mature megakaryocytes and platelet release. Romiplostim is an important TPO-RA that has advanced treatment options for individuals with thrombocytopenia and acts by increasing platelet production and therefore increasing platelet counts [3].
Romiplostim binds to and activates the TPO-R on megakaryocyte precursors, activating multiple cell-signaling pathways, leading to enhanced cell growth and cell viability, which results in increased platelet production. Studies have also shown romiplostim to be effective in improving platelet counts in various preclinical and clinical settings, including chemotherapy induced thrombocytopenia and aplastic anemia [1,3]. The safety and efficacy of romiplostim has led to anecdotal use in both primary and secondary thrombocytopenia in the post-HSCT setting [1-3].
Lancman et al. [4] reported the use of Romiplostim in 5 patients with primary or secondary thrombocytopenia post HSCT, where response was achieved in 4 out of 5 patients with platelets count > 50 *10^9/L for 7 consecutive days, independent of platelets transfusion. However, none of those patients had MDS, and the median age was 39 years old [4].
In addition, Mahat et al reviewed 12 studies involving the use of romiplostim for prolonged post-HSCT thrombocytopenia; a platelet response of >50 × 10^9 /L free of platelet transfusion was observed in 40 out of the 49 patients (82%) overall [5].
Little has been published in pediatric age groups about the use of Romiplostim in the post-HSCT thrombocytopenia.
For instance, Maximova et al. [6] reported the safe and successful use of Romiplostim in secondary thrombocytopenia post-HSCT where 6 out of 7 children became transfusion independent during the second week of treatment with Romiplostim, knowing that none of those children had MDS [6].
The use of Romiplostim in children with thrombocytopenia post-HSCT whether primary or secondary is still under study and more prospective and RCT studies are required to better understand its safety and efficacy. However, in our experience with our patient, Romiplostim has helped us achieve a stable platelet count, where Eltrombopag failed, freeing our patient from her frequent platelet transfusions, with a relatively safe profile, as the child has no complaints so far from its use. The duration of treatment and the weaning schedule are yet to be determined based on the clinical status of the patient.
References
- Pulanic D, Lozier JN, Pavletic SZ (2009) Thrombocytopenia and hemostatic disorders in chronic graft versus host disease. Bone Marrow Transplant 44(7): 393-403.
- Kuter DJ (2014) Milestones in understanding platelet production: a historical overview. Br J Haematol 165(2): 248-258.
- Bussel JB, Soff G, Balduzzi A, Cooper N, Lawrence T, et al. (2021) A Review of Romiplostim Mechanism of Action and Clinical Applicability. Drug Des Devel Ther 15: 2243-2268.
- Lancman G, Coltoff A, Steinberg A (2020) Romiplostim for thrombocytopenia following allogeneic stem cell transplantation: A case series. Hematol Oncol Stem Cell Ther 13(3): 171-174.
- Mahat U, Rotz SJ, Hanna R (2020) Use of thrombopoietin receptor agonists in prolonged thrombocytopenia after hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 26(3): e65-e73.
- Maximova N, Zanon D, Rovere F, Maestro A, Schillani G, et al. (2015) Romiplostim for secondary thrombocytopenia following allogeneic stem cell transplantation in children. Int J Hematol 102(5): 626-632.






























