Echocardiography Guided Management of Pediatric Septic Shock Reduces the Incidence of Refractory Shock
Ahmed A El Nawawy, Asmaa H Abohassan and Hadir M Hassouna*
Department of Pediatrics, Alexandria University, Egypt
Submission: September 22, 2021; Published: November 29 2021
*Corresponding author: Hadir M Hassouna, lecturer of Pediatrics, Alexandria University, El Khartoom ssquare, Egypt, Tel: 002 01003568008
How to cite this article:Ahmed A El N, Asmaa H A, Hadir M H. Echocardiography Guided Management of Pediatric Septic Shock Reduces the Incidence of Refractory Shock. Two Case Reports. Acad J Ped Neonatol 2021; 11(1): 555858. 10.19080/AJPN.2021.11.555858
Abstract
Background: Established conventional interventions have failed to reverse refractory septic shock. For that reason, it is important to deliver a convenient approach that prevent progression of septic shock into refractory shock especially in resource-limited settings. There are few studies investigating refractory septic shock particularly in children.
Objective: To evaluate the role of echocardiography guided management of pediatric septic shock in reduction of the incidence of refractory shock.
Methods: A retrospective study including 581 children with septic shock admitted to a university hospital intensive care unit. Patients were divided into 2 groups. Conventional group were admitted from 1st of January 2009 to 31st December 2012 who were managed according to international guidelines of surviving sepsis campaign without echocardiography guidance. ECHO group were admitted from 1st of January 2013 to 31st December 2019 who were managed using echocardiography to optimize cardiac function and volume status. Refractory septic shock was defined as the association of high blood lactate with high vasoactive -inotropic support associated with myocardial dysfunction.
Results: Hundred eighty-five children (31.8%) were enrolled in the conventional group while 396 children (68.2%) were included in the ECHO group. The incidence of refractory septic shock was significantly higher among the conventional group (p=0.007). In the multivariate regression model, echocardiography guided management septic shock management was a protective independent factor against development of refractory septic shock [OR of 0.24 (95%CI of 0.06-0.98)].
Conclusion: Implementation of echocardiography in pediatric septic shock management can reduce incidence of refractory septic shock in children.
Introduction
Despite advances in understanding of the pathophysiology of septic shock in the last two decades, mortality due to septic shock in children is still high to reach 10-13 % in developed countries [1]. The corresponding figures are higher in units from resource-restricted countries reaching 18-24% in most units and even more in few other countries to be 34-58% [2,3].
Refractory septic shock (RSS) is known as the existence of hypotension, with end organ dysfunction that necessitate high-doses vasopressor support usually exceeds 0.5μg/kg/min norepinephrine or equivalent [4]. RSS is associated with mortality of up to 60%. Moreover, patients with vasopressor requirements more than 1μg/kg/min norepinephrine or equivalent with ongoing clinical worsening show higher fatality of 80–90% [5,6].
Early recognition and management are critical to reverse the shock. The Surviving Sepsis Campaign (SSC) guidelines in February 2020 included guidelines for antibiotic administration, fluid resuscitation, and the use of vasoactive agents [7,8]. Microcirculatory failure and accompanying ischemic sequels require alternative therapeutic strategies to improve outcomes in septic shock patients and avoid development of RSS.
Echocardiography is considered a key tool for the hemodynamic assessment in ICUs through identification of the pathophysiology and categorization of shock as distributive, hypovolemic, obstructive, or cardiogenic which all may be present in septic shock [9]. Therein lies its importance in identification of causes hemodynamic instability and thus prompt guidance of therapy. Incorporation of serial echocardiography in management of pediatric septic shock provided early recognition of septic myocardial dysfunction and hypovolemia that was not apparent on clinical assessment and resulted in faster shock reversal, less fluid overload, shorter hospital stay, and lower mortality compared with the group without serial echocardiography [10].
The rational of the current study was to evaluate the role of echocardiography guided management of pediatric septic shock in reduction of the incidence of RSS.
Methods
After approval of the university ethical committee (IRB number 00007555, April 2019), a retrospective cohort study was carried out in a 9-beded pediatric intensive care unit of a university teaching hospital. Consents from patients’ parents or legal guardians were waived as the study was retrospective and as the results were innominate.
Consecutive children aged 1 month to 12 years were included if they had septic shock on PICU admission. Pediatric septic shock was defined as severe infection leading to cardiovascular dysfunction (including hypotension, need for treatment with a vasoactive medication, or impaired perfusion) [4,5]. Patient who had myocarditis, cardiomyopathy, congenital heart disease, trauma, as well as incomplete data were excluded. Data were collected from the computerized filling system of the PICU.
Patients were divided into 2 groups.
Conventional group: patients admitted from 1st of January 2009 to 31st of December 2012 who were managed according to international guidelines of surviving sepsis campaign without echocardiography guidance [11].
ECHO group: patients admitted from 1st of January 2013 to 31st of December 2019 who were managed using echocardiography to optimize cardiac function and volume status.
Refractory septic shock (RSS) was defined as: the association of high blood lactate with high vaso-inotrope doses associated with myocardial dysfunction [12] high blood lactae was considered when blood lactate >8 mmol/L or 1 mmol/L lactate increase after 6h of resuscitation. High vaso-inotrope doses were counted when vasoactive inotropic score (VIS) was greater than 200 [12]. Vasoinotrope score was calculated as (epinephrine + norepinephrinein mcg/kg min) × 100 + (dobutamine + dopamine in mcg/kg min) + (vasopressin in mcg/kg min) × 10,000 + (milrinone in mcg/kg min) × 10] [13]. Myocardial dysfunction was defined as the occurrence of a resuscitation-responsive cardiac arrest in pediatric intensive care unit or cardiac ultrasound findings with left ventricle ejection fraction (LVEF) <25% or a cardiac index <2.2 L/min/m2 [12].
Clinical variables including Pediatric Index of Mortality 2 (PIM2) score at intensive care admission [14], daily Pediatric Logistic Organ Dysfunction 2 (PELOD2) score [15], shock reversal time (defined as maintenance of SBP <5th centile for age or >70 mmHg from 1 mo-1yr, [age ×2+70] from 1-10 years, and a SBP of at least 90 mmHg in children >10 years; without vasopressor support for at least 24 hours) [16] were extracted from the patient data. Disseminated intravascular coagulopathy (DIC) score was calculated using the the International Society on Thrombosis and Haemostasis (ISTH) scoring system which include prothrombin time, fibrinogen, platelet count, and D-dimer. A score of 5 or greater indicated overt DIC [17]. The patients’ outcome, duration of invasive mechanical ventilation and PICU stay were recorded.
For the ECHO group, transthoracic echocardiography was performed, using Echocardiography Doppler Philips HD 11XE system (model: Philips 989605325131, USA). Detailed echocardiographic findings using 2 dimensional (B mode), M mode, pulsed Doppler were obtained. Therapy was adjusted in the ECHO group according to the echocardiographic findings. Firstly, to guide fluid therapy; when IVC was collapsed or >50% respirophasic variation, fluid boluses were continued and when IVC was normal or full of minimal respirophasic variation, fluid boluses were discontinued or slowed. Secondly, when septic myocardial dysfunction (SMD) was present, inotropes were started. Ascending and descending titration of inotropes was guided by the echocardiography findings. Echocardiography was done at time of admission and serially before and after changes in treatments then daily till patient is discharged or deceased.
Statistical methods
Data were collected, revised, coded and fed to statistical software SPSS IBM version 20. For comparison between two independent groups (conventional and echo groups); Chi square test and Monte Carlo exact probability were used to test for differences at percentages while Mann-Whitney test and independent samples t-test were used for comparing median and means, respectively. Expected variables affecting RSS, namely, age, sex, source of infection, PIM2 score, PELOD2 score, DIC, serum lactate and VIS, were candidate factors for univariate analysis for predicting the most significant factors associated with RSS. Those significant on univariate analysis were subjected to a multivariate logistic regression model to identify independent variables affecting development of RSS.
Results
Total PICU admissions were 2512 during the study period in which 768 (30.5%) children had septic shock. Hundred eightyseven (24.3%) children were excluded as pre-specified exclusion criteria. Five hundred eighty-one children (75.6%) were included in which 185 children (31.8%) were enrolled in the conventional group while 396 children (68.2%) were included in the ECHO group (Figure 1). Comparison between the two groups as regard the base line characteristics and assessment was shown in Table 1. Laboratory findings were shown in Table 2. There was no significant difference between the two groups as regards initial serum lactate. However, rising lactate ≥1 mmol/L after 6 hours significantly predominate among children in the conventional group.
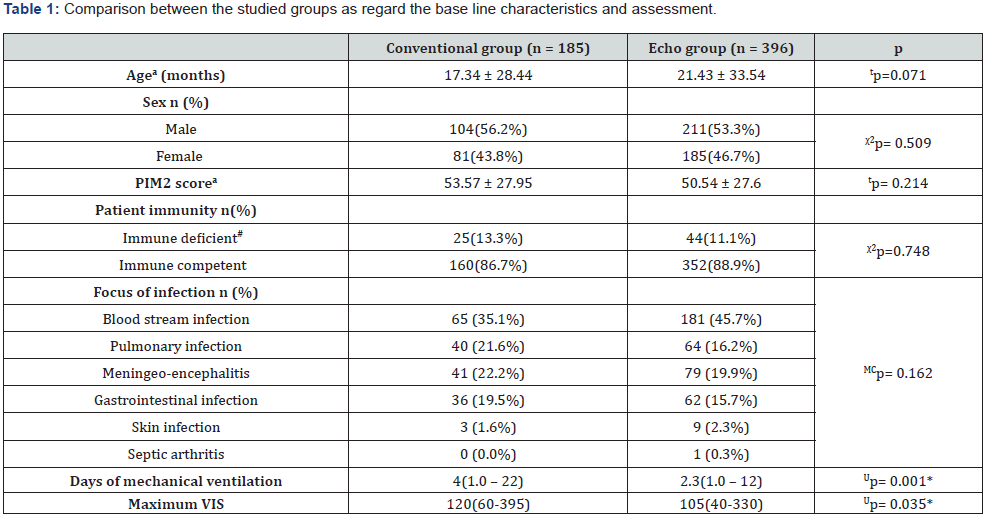
a: mean ± SD, b: Median (range), #: Recent/current long-term steroid use, severe malnutrition or leukemia, PIM2: Pediatric Index of Mortality 2, VIS: vasoactive inotropic score, Up: p value of Mann Whitney test , tp: p value of independent samples t-test, χ2p: p value of Chi square test , MCp: value of Monte carlo exact probability *: Statistically significant at p ≤ 0.05
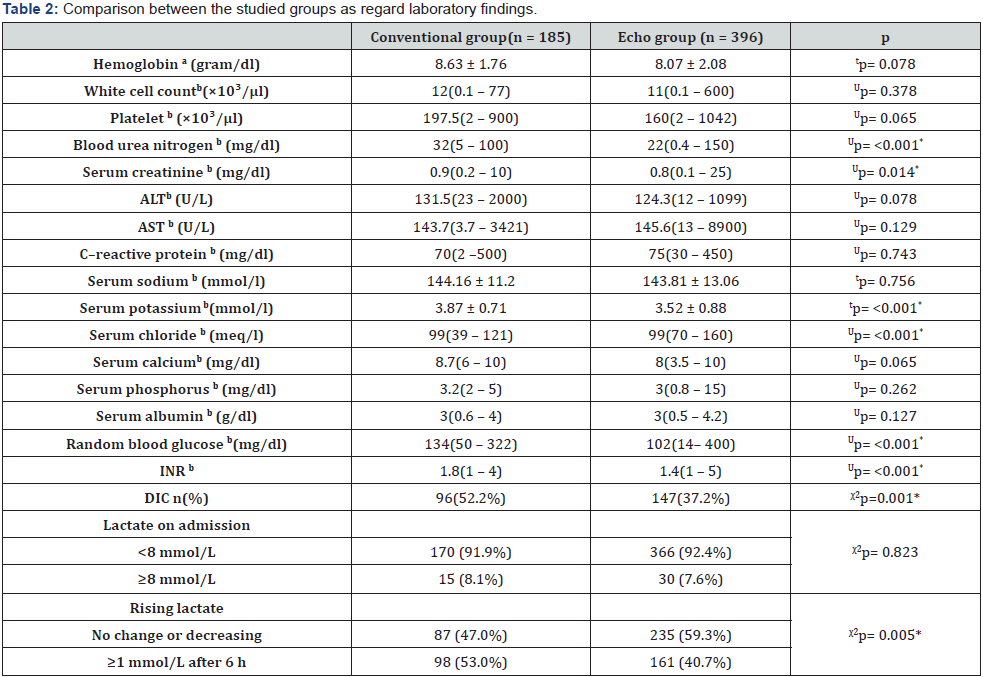
a: mean ± SD, b: Median (range), INR: international normalizing ratio, DIC: disseminated intravascular coagulopathy, Up:: p value of Mann Whitney test , tp: p value of independent samples t-test, χ2p: p value of Chi square test, *: Statistically significant at p ≤ 0.05
As the primary aim of the study, the incidence of RSS was significantly higher among the conventional group (p=0.007). Correspondingly, the incidence of shock reversal was significantly higher among the ECHO group(p=0.043) with appreciably shorter shock reversal time(p=0.047). Although the PELOD2 score did not differ significantly on the first two days of admission, PELOD2 score was markedly improving on the third day of admission in the ECHO group(p=0.006). Length of PICU stay was significantly shorter in the ECHO group (p=0.002). Mortality was higher in the conventional group. Yet, it was not statistically significant (p=0.093). However, mortality due to RSS was significantly higher in the conventional group (p=0.007) (Table 3).
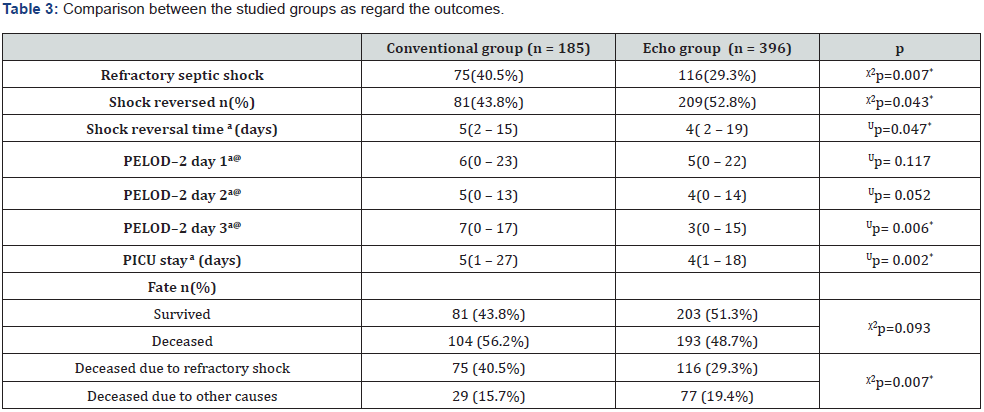
a: Median (range), PELOD2: Pediatric logistic organ dysfunction 2, Up:: value of Mann Whitney test, χ2p: value for Chi square test, @: PELOD-2 was implemented 2016 but all retrospective data before 2016 were recalculated as PELOD-2 score to unify the result, *: Statistically significant at p ≤ 0.05.
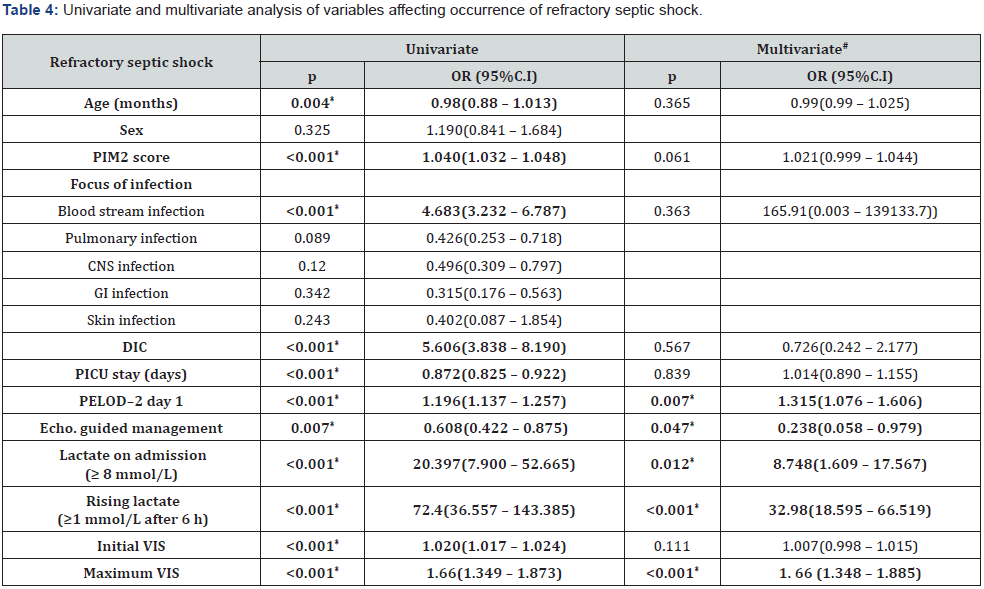
PIM2: Pediatric Index of Mortality 2, DIC: disseminated intravascular, PICU: pediatric intensive care unite, PELOD2: Pediatric logistic organ dysfunction2, VIS: vasoactive inotropic score, OR: Odd`s ratio, CI: Confidence interval, #: Multiple logistic regression model was used
*: Statistically significant at p ≤ 0.05
Table 4 summarized the univariate and multivariate analysis for the variables affecting RSS for the total sample. Univariate analysis revealed that younger age, high PIM2 s,core, high PELOD2 day 1, blood steam infection , DIC, initial lactate (≥ 8 mmol/L), rising lactate (≥1 mmol/L after 6 hours), high initial and maximum VIS as well as not using echocardiography guided management were risk factors of RSS. While multidimensional data provided by multivariate analysis revealed that only high PELOD2 day 1, initial lactate ≥8 mmol/L, rising lactate and high maximum VIS were risk factors of RSS, whereas echocardiography guided management of pediatric septic shock was a protective factor that safeguard against development of RSS.
Discussion
“Prevention is better than cure”. Management of patients with refractory septic shock remains a great challenge. Helpless established conventional interventions have failed to reverse RSS. For that reason, it is important to deliver a convenient approach that prevent progression of pediatric septic shock into RSS especially in resource-limited settings where extracorporeal membrane oxygenation (ECMO) treatment is impossible.
Vital signs and clinical evaluation poorly differentiate between cold and warm shock and inaccurately identify children with cardiac dysfunction.Therefore, the new pediatric SSC guidelines (2020) recommended against the use of clinical parameters to categorize children as being in warm versus cold shock. Advanced hemodynamic monitoring (eg, arterial blood pressures, central venous oxygen saturations) in addition to clinical assessment may provide more reliable guidance. Using echocardiography to assess the hemodynamic state and guide changes in therapy became fundamental in the management of pediatric septic shock. It provides necessary data that cannot be acquired by clinical examination solely. Serial echocardiography allowed optimal adjustment of therapy, significantly improved all hemodynamic parameters, and reduced shock reversal time [10]. Shock reversal is the primary aim that should be accomplished whenever a child having septic shock is recognized. Han YY et al. [18] found that each additional hour of persistent shock was associated with >2 folds increased odds of mortality [18]. Septic shock if not reversed often progress to multiple organ dysfunction syndrome (MODS). Several clinical trials revealed mortality from 40% to 75% in patients with MODS resulting from from sepsis [19]. In the current study, the percentage of children succeeded in shock reversal was significantly higher in the echo group with a median of 1 day earlier than that in the conventional group. Early shock reversal in the echo group was associated with a significant reduction in PICU stay among survivors of the echo group compared with those of conventional group with a median of 1 day earlier than that in the conventional group. Although the echo group showed 7.5% lower mortality than the conventional group, it was statistically insignificant because mortality in critically ill patients is multifactorial. Yet deaths due to unresolved shock was significantly lower in the echo group.
In the current study, echo group showed significant improvement of lactate level after 6 hours of care. Failure of lactate to normalize during critical illness had been associated with increased risk of major adverse events [20]. In the current study, DIC was significantly higher in the conventional group compared to the echo group as echocardiography guided therapy improves patient hemodynamics and normalize lactate as early as possible compared with conventional method. Acidosis accelerates fibrinogen degradation, leading to a potential deficit in fibrinogen availability which participate in the development of DIC [21].
In the conventional group, fluid resuscitation was administered according to the previous international guidelines in which the target was ‘‘adequate blood pressure and clinical monitors of cardiac output including heart rate, quality of peripheral pulses, capillary refill, level of consciousness, peripheral skin temperature, and urine output.’’ Pediatric fluid resuscitation guidelines used in the conventional group were based on the concept of empiric fluid loading using vital signs as a marker of response to treatment. The host response to sepsis in children is highly variable and dynamic during the disease course, and vital signs are a poor predictors of disease severity or response to treatment. As such, using a “one size fits all” approach in fluid resuscitation seems hazardous. Although intravenous fluid boluses remain a cornerstone of the resuscitation of children with septic shock, an increasing number of publications have highlighted the increased morbidity and mortal ity associated with aggressive fluid administration. The recent SSC panel (2020) therefore made a recommendation that takes the health care setting into account. Fluid administration needs to be titrated to signs of perfusion and organ dysfunction and should be discontinued if signs of fluid overload develop. In contrast, in settings where intensive care is not available, fluid resuscitation should be restricted to children with hypotension using more judicious fluid amounts of 10–20 mL/kg during the first hour.Certainty of intravascular volume is important because over-resuscitation may lead to longer mechanical ventilation days, prolonged ICU stays and worse outcomes [22]. Optimized fluid therapy guided by echocardiography which has been very useful in the resuscitation of shocked patients, monitoring their fluid demands, and predicting recurrence of shock with avoidance of fluid overload. In ECHO group, there was significant reduction in days of mechanical ventilation and PICU stay as well as better outcome.
Maximum VIS was markedly lower in the echo group compared with the conventional group. Unnecessary doses of vasopressors/ inotropes would be avoided because echocardiography allowed tailoring therapy according to the condition of each patient. Gaies et al. concluded that high maximum VIS was strongly associated with a poor outcome compared with patients with a low maximum VIS [13].
The primary aim of the study was to evaluate the role of echocardiography in reducing the incidence of refractory septic shock among pediatric septic shock patients. Although definition of RSS was broader in the echo group that included cardiac ultrasound findings in defining myocardial dysfunction, the incidence of RSS was significantly lower owing to early and accurate management of septic myocardial dysfunction as well as appropriate volume optimization. Factors affecting development of RSS were studied in the current study. In the multivariate regression model, the independent factors that increased the risk of the patient to develop refractory septic shock were high PELOD-2 on the day of admission, high maximum VIS, high initial lactate as well as rising lactate level after 6 hours of resuscitation. While using echocardiography in management of septic shock was a protective independent factor against development of refractory septic shock.
Although, there is limited study worldwide to identify risk factors for occurrence of refractory septic shock, there are other studies identify risk factors of early and late deaths in septic shock in adult patient. They found that increasing age, diabetes mellitus, malignancy, lack of pathogen identification, and higher admission SOFA score remained independent risk factors of early death. Among patients who survived the first 3 days, independent determinants of late death included increasing age, cirrhosis, lack of pathogen identification, and previous corticosteroid treatment [23]. To our knowledge, the risk factors of developing RSS in children were not yet studied. However, mortality risk factors among children and newborns with refractory septic shock who required ECMO treatment were studies. Sole’ et al. found that vasoplegic pattern shock or S. pneumoniae sepsis were mortality risk factors in children and newborns with RSS who required venoarterial ECMO treatment [24]. While Workerman et al. [25] included cardiac failure or extracorporeal cardiopulmonary resuscitation as indication for extracorporeal life support (ECLS) cannulation ,having a new neurologic event following cannulation, acquiring a new infection following cannulation, inability to normalize pH in the 48 hours following ECLS cannulation and requiring higher daily volume of platelet transfusions as significant risk factors for mortality in children with refractory pediatric septic shock who are supported with ECLS [25].
Limitations
The limitation of the study is being a retrospective single centre study. However, it is one of the scarce studies that investigate RSS in children. Also, large sample size was included with minimal data loss. There were no significant differences in the baseline characteristics of the two studied groups minimizing bias.
Conclusion
Implementation of echocardiography in pediatric septic shock management reduced incidence of refractory septic shock in PICU.
References
- Watson RS, Carcillo JA, Linde-Zwirble WT, Clermont G, Lidicker J, et al. (2003) The epidemiology of severe sepsis in children in the United States. Am J Respir Crit Care Med 167(5): 695-701.
- Hartman ME, Linde-Zwirble WT, Angus DC, Watson RS (2013) Trends in the epidemiology of pediatric severe sepsis. Pediatr Crit Care Med 14(7): 686-693.
- Weiss SL, Fitzgerald JC, Pappachan J, Wheeler D, Jaramillo-Bustamante JC, et al. (2015) Global epidemiology of pediatric severe sepsis: the sepsis prevalence, outcomes, and therapies study. Am J Respir Crit Care Med 191(10): 1147-1157.
- Bassi E, Park M, Azevedo LCP (2013) Therapeutic strategies for high-dose vasopressor-dependent shock. Crit Care Res Prac. 2013: 654708.
- Brown SM, Lanspa MJ, Jones JP, Kuttler KG, Li Y, et al. (2013) Survival after shock requiring high-dose vasopressor therapy. Chest. 143(3): 664–671.
- Brand DA, Patrick PA, Berger JT, Ibrahim M, Matela A, et al. (2017) Intensity of vasopressor therapy for septic shock and the risk of in-hospital death. J Pain Symptom Manag 53(5): 938–943.
- Weiss SL, Peters MJ, Alhazzani W, Agus MSD, Flori HR, et al. (2020) Surviving Sepsis Campaign international guidelines for the management of septic shock and sepsis‐associated organ dysfunction in children. Pediatr Crit Care Med 21(2): e52‐ e106.
- Cruz A, Lane R, Balamuth F, Aronson PL, Ashby DW, et al. (2020) Updates on pediatric sepsis 1(5): 981-993.
- Gaspar HA, Morhy SS (2015) The role of focused echocardiography in pediatric intensive care: a critical appraisal. Biomed Res Int 2015: 596451.
- El-Nawawy AA, Abdelmohsen AM, Hassouna HM (2018) Role of echocardiography in reducing shock reversal time in pediatric septic shock: a randomized controlled trial. J Pediatr (Rio J) 94(1): 31–39.
- Dellinger RP, Levy MM, Rhodes A, Annane D, Gerlach H, et al. (2013) Surviving Sepsis Campaign: international guidelines for management of severe sepsis and septic shock, 2012. Intensive Care Med 39(2): 165-228.
- Morin L, Ray S, Wilson C, Remy S, Benissa MR, et al. (2016) Refractory septic shock in children: a European Society of Paediatric and Neonatal Intensive Care definition. Intensive Care Med 42(12): 1948-1957.
- Gaies MG, Gurney JG, Yen AH, Napoli ML, Gajarski RJ, et al. (2010) Vasoactive–inotropic score as a predictor of morbidity and mortality in infants after cardiopulmonary bypass. Pediatr Crit Care Med 11(2): 234-238.
- Slater A, Shann F, Pearson G, for the PIMSG (2003) PIM2: a revised version of the Paediatric Index of Mortality. Intensive Care Med 29(2): 278-285.
- Leteurtre S, Duhamel A, Salleron J, Grandbastien B, Lacroix J, et al. (2013) PELOD-2: an update of the PEdiatric logistic organ dysfunction score. Crit Care Med 41(7): 1761-1773.
- Sligl WI, Milner DA, Sundar S, Mphatswe W, Majumdar SR (2009) Safety and efficacy of corticosteroids for the treatment of septic shock: a systematic review and meta-analysis. Clin Infect Dis 49(1): 93-101.
- Taylor FB, Toh CH, Hoots WK, Wada H, Levi M (2001) Towards definition, clinical and laboratory criteria, and a scoring system for disseminated intravascular coagulation. Thromb Haemost 86(5): 1327-1330.
- Han YY, Carcillo JA, Dragotta MA, M Levi, Scientific Subcommittee on Disseminated Intravascular Coagulation (DIC) of the International Society on Thrombosis and Haemostasis (ISTH) (2003) Early reversal of pediatric-neonatal septic shock by community physicians is associated with improved outcome. Pediatrics 112: 793–799.
- https://www.sciencedirect.com/science/article/pii/B9780323449427002028
- Domico M, Allen M (2016) Biomarkers in pediatric cardiac critical care. Pediatr Crit Care Med 17(8): S215-S221.
- Martini WZ (2009) Coagulopathy by hypothermia and acidosis: mechanisms of thrombin generation and fibrinogen availability. J Trauma 67(1): 202-208; discussion 208-9. doi: 10.1097/TA.0b013e3181a602a7.
- Kelm D, Perrin J, Cartin-Ceba R, Gajic O, Schenck L, et al. (2015) Fluid overload in patients with severe sepsis and septic shock treated with early-goal directed therapy is associated with increased acute need for fluid-related medical interventions and hospital death. Shock 43(1): 68-73.
- Daviaud F, Grimaldi D, Dechartres A, Charpentier J, Geri G, et al. (2015) Timing and causes of death in septic shock. Ann Intensive Care 5(1): 16.
- Solé A, Jordan I, Bobillo S, Moreno J, Balaguer M, et al. (2018) Venoarterial extracorporeal membrane oxygenation support for neonatal and pediatric refractory septic shock: more than 15 years of learning. Eur J Pediatr 177(8): 1191-1200.
- Workman JK, Bailly DK, Reeder RW, Dalton HJ, Berg RA, et al. (2020) Risk Factors for Mortality in Refractory Pediatric Septic Shock Supported with Extracorporeal Life Support. ASAIO J 66(10): 1152-11 60.






























