Comparison of Brainstem Auditory Function at Term Between Premature Babies with Neonatal Chronic Sublethal Hypoxia and Those Without the Hypoxia
James Ken Jiang1 Cui Wong2, Ze Dong Jiang2* and Jin Wang2
1School of Physiology, Pharmacology and Neuroscience, University of Bristol, Bristol, UK
2Division of Neonatology, Children’s Hospital of Fudan University, Shanghai, China
Submission: October 20, 2021; Published: November 11, 2021
*Corresponding author: Ze Dong Jiang, Division of Neonatology, Children’s Hospital of Fudan University, Shanghai, China
How to cite this article: Jiang K J, Wang C, Jiang Z D, Wang J. Comparison of Brainstem Auditory Function at Term Between Premature Babies with Neonatal Chronic Sublethal Hypoxia and Those Without the Hypoxia. Acad J Ped Neonatol 2021; 10(4): 555855. DOI: 10.19080/AJPN.2021.10.555855
Abstract
Background: Neonatal chronic sublethal hypoxia is a major perinatal condition to affect the immature brain. We compared brainstem auditory function between premature babies with neonatal chronic sublethal hypoxia and those without the hypoxia to assess the effect of chronic sublethal hypoxia on the immature auditory function.
Methods: At 39-40 weeks of postconceptional age, 88 premature babies were recruited and studied with brainstem auditory evoked response at various click rates. Forty-two babies who experienced chronic sublethal hypoxia due to neonatal chronic lung disease served as the study group, and 46 babies who did not experience the hypoxia as the control group.
Results: The response threshold did not differ significantly between the study and control groups, although it was slightly lower in the study group. The latencies of the response waves I and III in the study group were slightly shorter than in the control group, without any significance at any click rates. Wave V latency was slightly longer than in the control group. However, the I-V interpeak interval in the study group was significantly longer than in the control group at all click rates (all p <0.01). There were small differences between the two groups in the amplitudes of waves I, III and V, with no statistical significance at any click rates. No systematically significant differences were found between the two groups in the V/I and V/III amplitude ratios.
Conclusion: There was no major difference in peripheral auditory function between the premature babies with chronic sublethal hypoxia and those without the hypoxia. However, premature babies with chronic sublethal hypoxia had prolonged central conduction time, suggesting poorer central or brainstem auditory function.
Keywords: Auditory abnormality, Chronic sublethal hypoxia, Neonatal hypoxia, Premature baby
Introduction
The auditory system is very sensitive to hypoxia and hypoxemia which disturb the metabolism of neurons, depresses the electrophysiological function of synapses, and interferes with nerve conduction [1-4]. Experiments in animal models have shown that chronic sublethal hypoxia (CSH) adversely affects the immature cerebral cortex [4-9]. It is possible that the immature central auditory system is also affected by CSH. In newborn babies, hypoxia is particularly prevalent among those who are born very prematurely [10,11]. With the increase in survival rate for critically ill premature babies, there is an increased concern of brain damage and neurodevelopmental disorders, including auditory problems, in the survivors.
Considerable evidence suggests that neurodevelopmental disorders in babies born very prematurely often link to hypoxic events during the perinatal period. These babies often undergo chronic or prolonged periods of sublethal hypoxia [12]. In babies born very prematurely, a typical clinical problem that is associated with CSH is chronic lung disease (CLD). It is a major lung disease that causes hypoxaemia of pulmonary origin in babies who are born very prematurely [13-17]. Babies who suffer CLD often experience frequent episodes of hypoxaemia or prolonged hypoxaemia, which plays an important role in the development of brain damage and neurological impairment in premature babies with CLD.
A previous study using the brainstem auditory evoked responses (BAERs) compared brainstem auditory function between premature babies with neonatal CLD and normal term babies [18]. These babies were found to have major abnormalities in the BAER components that mainly reflect functional status of the central auditory system, suggesting poor myelination and synaptic dysfunction in the central, specifically brainstem, auditory pathway. The abnormality may well be related to or due to CSH that occurs during the process of CLD. However, since these babies were born very prematurely, one cannot exclude a possible effect of premature birth on the auditory system which contributes the abnormality found in these babies. To minimize such a possible confounding effect, we compared the BAER in premature babies who had neonatal CLD with those who did not have CLD. Any differences between them are likely to reflect the adverse effect of CSH on the immature auditory system in babies born prematurely.
Subjects and Methods
Subjects
The study (CLD) group was comprised of 42 CLD premature babies who required supplementary oxygen or ventilatory support beyond 36 weeks of postconceptional age to maintain PaO2 > 50 mmHg. All babies had clinical signs of chronic lung respiratory disease and radiographic evidence of CLD (persistent strands of density in both lungs), which still existed when they were recruited for this study. These inclusive criteria were the same as previously reported [18]. The gestational age ranged between 25 and 32 weeks (28.8 ± 1.9 weeks) and birthweight ranged between 585 and 1980 g (1,093 ± 297 g). The control (non-CLD) group is comprised of 42 premature babies who did not have any evidence of neonatal CLD. Their gestational age ranged between 25 and 32 weeks (29.2 ± 1.9 weeks) and birthweight ranged between 598 and 2025 g (1,256 ± 368 g).
These babies were recruited from the Children’s Hospital of Fudan University. Any babies who had other major perinatal complications were excluded to minimize any confounding effects on the BAER [3,19]. Parental consent was obtained for each baby before study entry. All babies were tested with BAER at 39-40 weeks of postconceptional age; 39.6 ± 0.4 weeks in the study group, and 39.7 ± 0.4 weeks in the control group, which was almost the same.
Protocols of recording BAER
The protocols are the same as previously described [3,18-20]. The recording was conducted using a Spirit 2000 Evoked Potential System (Nicolet Biomedical Inc. Madison, WI, USA) was used to record and analyse the BAER. The babies lay supine in a cot in a quiet room. Before BAER recording the auditory meatus was inspected and cleaned of any vernix or wax. The recording commence shortly after the baby fell asleep naturally, often after a feed, without using any sedatives.
Three gold-plated disk electrodes were placed at the middle forehead (positive), the ipsilateral earlobe (negative) and the contralateral earlobe (ground), respectively. Inter-electrode impedances were maintained at <5 kW. The acoustic stimuli were rarefaction clicks, which were generated by rectangular pulses 100 µsec in duration and delivered monaurally to the left ear through a TDH 39 earphone. Duplicate recordings were made in response to each stimulus condition to examine reproducibility. The clicks were presented at the order of 21, 51 and 91/s in the first run and in reverse order in the second run.The intensity level of the clicks was 60 dB nHL for all babies. These study procedures were approved by the Central Oxford Research Ethics Committee.
Sweep duration was 12 ms. The evoked brain responses to 2,048 clicks were amplified, bandpass filtered between 100 and 3000 Hz, and inputted to the averager. If the data exceeded 91% of the sensitivity parameter setting (51 µV), that sweep (artefact) was automatically rejected by the system. During the averaging, both the ongoing filtered EEG and the running averaged BAER were monitored. Sampling was discontinued whenever there were excessive muscle artefacts on the monitoring oscilloscope.
Analysis of data
As previous described [3], measurement of thee latency of each BAER wave (I, III and V) was made the onset of click stimuli to the peak of each wave. Interpeak interval (I-V, I-III and III-V) was calculated as the time between any two wave peaks. Wave I amplitude was measured from the peak of wave I to the lowest trough between waves I and III, and wave III amplitude was from the trough to the peak of wave III. Measurement of wave V amplitude was made from the peak of wave V to the following trough. The amplitude ratio of waves V and I and III (i.e. V/I and V/III amplitude ratios) were also calculated.
The measurements of the above BAER variables from two replicated BAER recordings to each stimulus condition were averaged for further analyses. Mean and standard deviation of each BAER variable at each stimulus condition were compared between groups using the Student t test were used. A 2-tailed value of p <0.05 was considered statistically significant. The statistical analysis was performed using SPSS package version 22.
Results
The threshold of BAER was determined by establishing the lowest intensity of the clicks which produced visible and reproducible wave V with an amplitude between 0.04 and 0.10mV. No significant difference was found in BAER threshold between the study and control groups, although the threshold was lower in the study group (12.4±9 dB nHL) than in the control group (14.1±8 dB nHL).
Figures 1-3 show boxplot of the latencies for waves I, III and V, respectively, at 21-91/s clicks in CLD and non-CLD groups. Figure 4 show boxplot of the I-V interpeak interval in CLD and non-CLD groups and the results of statistical comparison between the two groups. Figures 5-7 show boxplot of the amplitudes for waves I, III and V, respectively, in CLD and non-CLD groups. Figures 8 and 9 show boxplot of the V/I and V/III amplitude ratios, respectively, in CLD and non-CLD groups and the results of statistical comparison in the V/I ratio between the two groups.
There were small differences in the latencies of BAER wave V components at various click rates between the study and control group. At 21/sclick rate, the latencies of BAER waves I and III in the study group tended to be shorter than in the control group (Figures 1 and 2). These differences between the two groups did not reach statistical significance. Wave V latency was slightly longer in the study group (Figure 3). However, the I-V interpeak interval in the study group is significantly longer than in the control group (p <0.01) (Figure 4). There were small differences in the amplitudes of waves I, III and V between the study and control groups (Figures 5-7). None of the wave amplitudes differed significantly. The V/I amplitude ratio in the study group was significantly smaller than in the control group (Figure 8). No significant difference was found in the V/III amplitude ratio between the two groups, although it was slightly smaller in the study group (Figure 9).
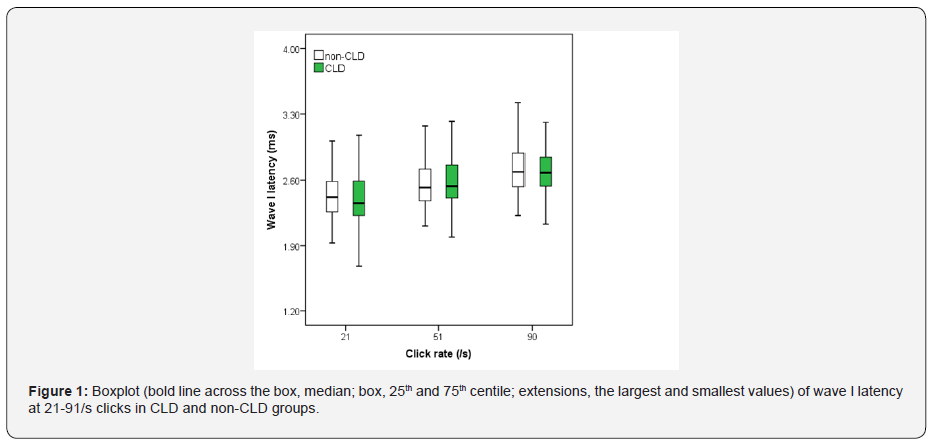
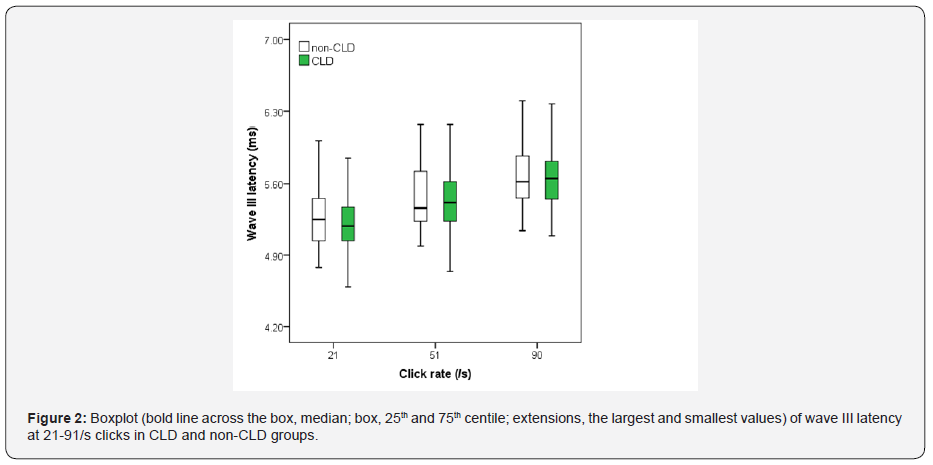
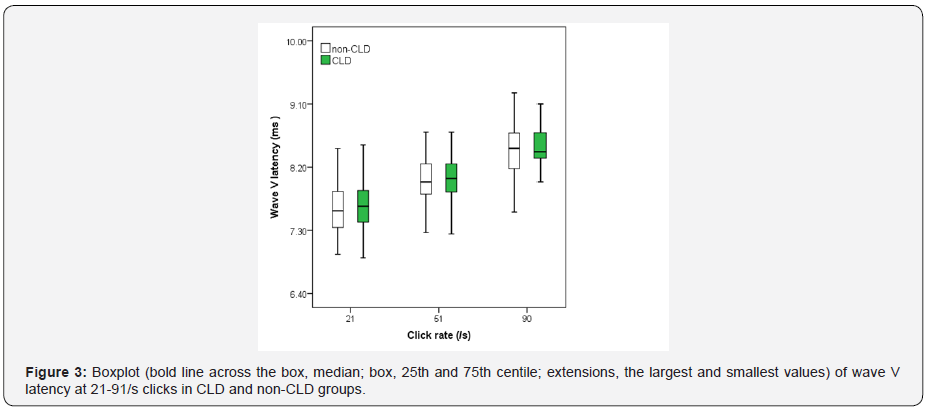
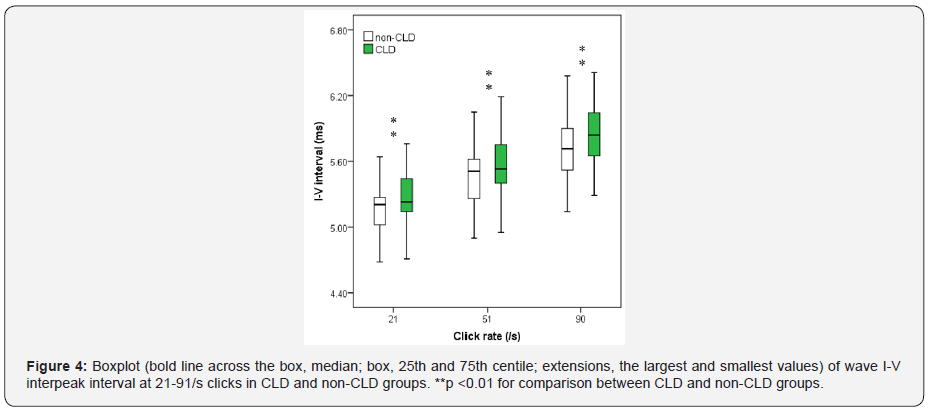
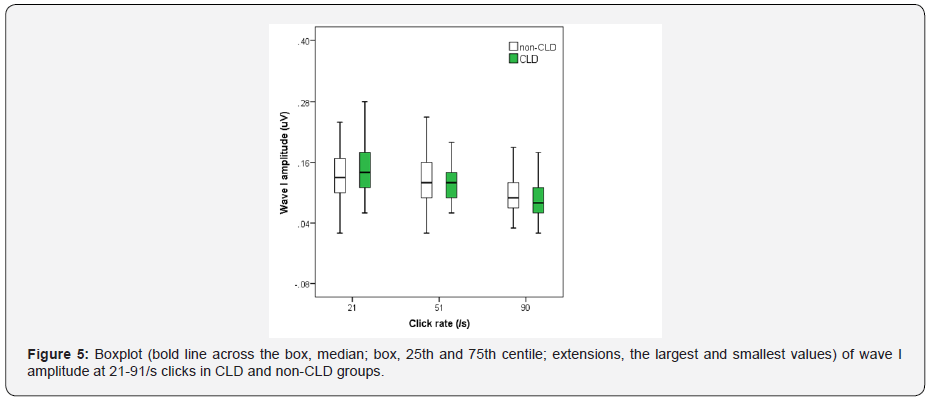
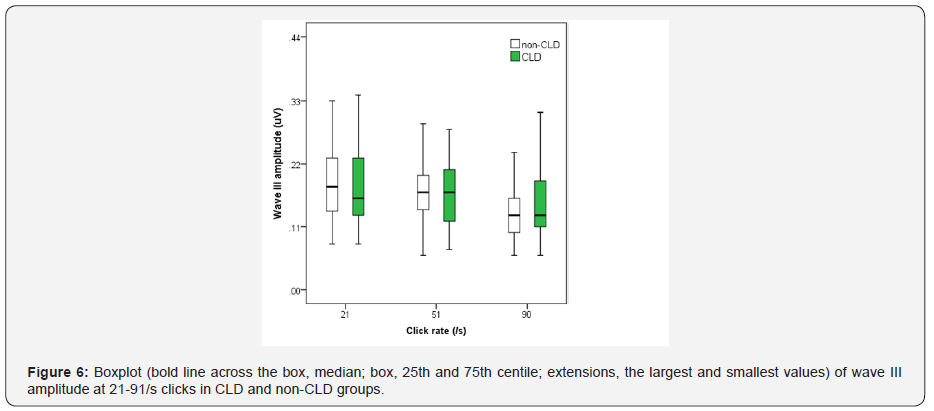
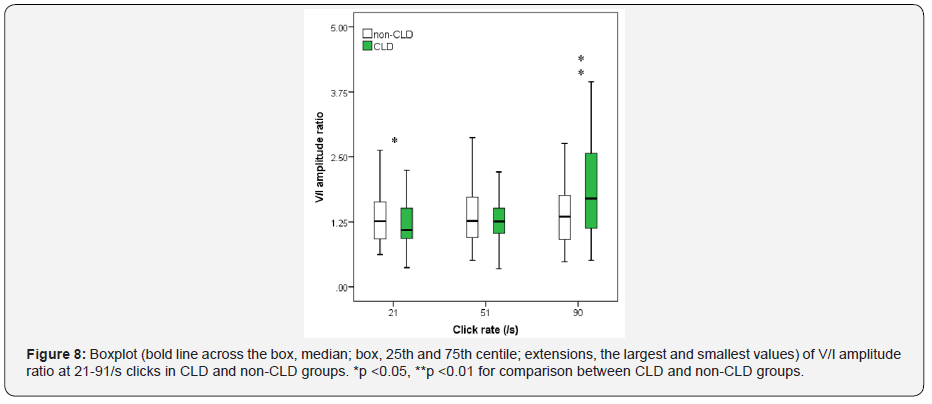
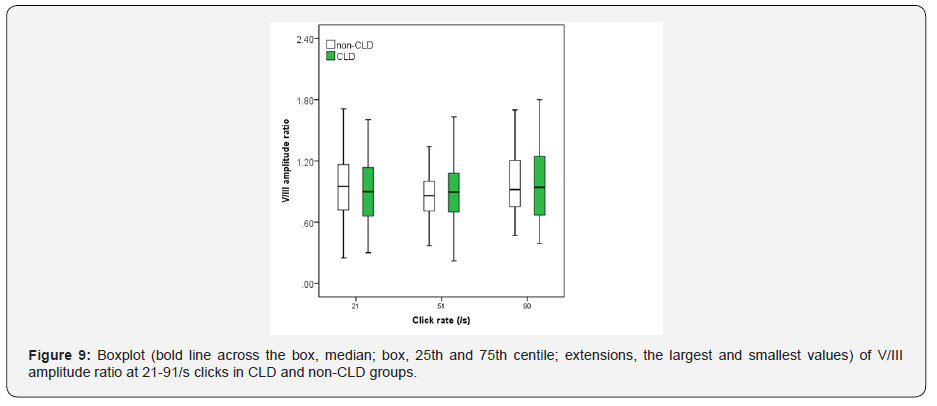
As the click rate was increased, all BAER wave latencies and the I-V interpeak interval were increased whereas wave amplitudes were decreased (Figures 1-9). At higher click rate of 51/s and 91/s, the latencies of BAER waves I, III and V were all generally similar in the study and control group, with only small differences (Figures 1 and 2). The I-V interpeak interval in the study group is significantly longer than in the control group at both 51/s and 91/s (p <0.01 and 0.01) (Figure 4). The amplitudes of waves I, III and V showed small differences between the two groups, without any statistical significance at either 51/s or 91/s (Figures 5-7). The V/I amplitude ratio in the study group was similar to that in the control group at 51/s clicks but was significantly greater than in the control group (p <0.01) (Figure 8). The V/III amplitude ratio was similar in the two groups at both 51/sand 91/s (Figure 9).
Discussion
In human subjects, the BAER has been used as an important tool to examine functional integrity and development of the brainstem auditory pathway in babies after perinatal hypoxia or hypoxia-ischaemia [2,4,19-26]. In experimental animal models, the BAER has been shown that to be is very sensitive to arterial blood oxygen levels and acute hypoxia or hypoxia-ischaemia [4,27-30]. Therefore, in both human and animal experiments the BAER has been demonstrated as an important tool to research into the influence of hypoxia or hypoxia-ischaemia on the developing auditory system, including the neural pathway and the cochlea. More recently, the BAER has also been shown to be a valuable method to investigate the effect of prolonged or chronic hypoxaemia on the auditory system to shed light on the influence of CSH on the auditory system [3,4,18].
Hypoxaemia has a direct effect on the cochlea and an indirect effect by way of cardiovascular collapse and cerebral ischaemia [30]. Persistent, particularly permanent, hearing impairment is primarily caused by prolonged periods of hypoxic-ischaemic insult secondary to the hypoxia, instead of primary or direct hypoxic injury, and the complicated factors associated with hypoxia. Animal experiments revealed that BAER abnormalities following hypoxia are mainly due to ischaemia even when the initial insult is hypoxic alone [30]. In the present BAER study, we found some differences between the premature babies with neonatal CLD and those without CLD. The BAER threshold in the CLD babies was relatively lower than in the non-CLD babies. The latencies of BAER waves I and III, which is significantly affected by hearing threshold, in the CLD babies were slightly shorter than in the non-CLD babies. This is apparently related to the relatively lower BAER threshold in the CLD babies.
The BAER threshold and the latencies of waves I and III predominately reflect peripheral auditory function. Our findings in these BAER variables suggest no major difference exists in peripheral auditory function between the CLD and non-CLD babies at 40 weeks of postconceptional age. Thus, CSH associated with neonatal CLD is unlikely to exert any significantly additional adverse effect on peripheral auditory function at term age. This is different from acute severe hypoxia that often affects peripheral auditory function, including cochlea and auditory nerve [3,19,31-34]. During the neonatal period, one-third of babies who suffer perinatal hypoxia-ischemia have peripheral auditory abnormality [35]. Peripheral auditory abnormality is not uncommon in babies born vary prematurely [34,36]. The BAER threshold in our premature CLD and non-CLD babies tended to be higher than in normal term babies whose BAER threshold is usually at 10 dB nHL or lower. This higher threshold suggests minor to moderate peripheral auditory abnormality in our premature babies.
A major finding in the present study is that the I-V interval in the CLD babies was significantly longer than in the non-CLD infants at all click rates. This interval, the most widely used BAER variable, reflect neural conduction time along the auditory pathway in the brainstem. The relative increase in the I-V interval in our CLD babies, when compared with the non-CLD babies, suggests that and CSH associated with neonatal CLD significantly affects the central auditory pathway in premature babies. This increase is less significant than what was found when comparing the I-V interval between premature CLD babies and normal term babies [18,37]. Clearly, premature birth and associated perinatal conditions result in longer I-V interval, whereas the CSH associated with CLD exerts additional effect that leads to even longer I-V interval. The longer I-V interval is suggestive of an impairment or delay in myelination of the central auditory pathway, which is well related to the frequent episodes of hypoxaemia or prolonged hypoxaemia occurring during the course of neonatal CLD. This finding is comparable with what was found in animal experiments that prolonged or chronic sublethal hypoxia can result in severe impairments in corticogenesis in the developing brain and a significant decrease in subcortical white matter [6].
Hypoxia often occurs during early life and damages the central nervous system such as the auditory system, resulting in neurological impairment and neurodevelopmental disorders [3,19,38-45]. During the course of CLD, the frequent episodes of hypoxaemia or prolonged hypoxaemia in neonatal CLD inevitably leads to CSH [14,37]. The auditory impairment in neonatal CLD could be related to various perinatal risk factors, but CSH must play a significantly adverse role in the impairment. Since none of our CLD babies had any other concomitant major brain pathology that may confound the results, the major BAER abnormalities found in our CLD babies are most likely to be mainly attributed to CSH due to the frequent episodes of hypoxaemia or prolonged sublethal hypoxaemia during the course of CLD.
In addition to the latencies of BAER wave components, the amplitudes of waves I, III and V and the V/I amplitude ratios in our CLD babies did not show any significant differences from those in the non-CLD babies. Although the V/I amplitude ratio in the CLD babies was significantly different from that in the non-CLD babies at 21/s and 91/s clicks, the differences were not systematic. Therefore, there were no major and systematic differences in BAER amplitude variables between the two groups of premature babies. The CSH associated with neonatal CLD does not exert any additional adverse effect on the amplitudes of BAER wave components. It appears that the neural origins of the amplitudes in premature babies are not significantly affected by the CSH associated with CLD.
Conclusion
Our premature CLD babies did not show any major differences in BAER variables that mainly reflect peripheral auditory function from the premature non-CLD babies. There seems to be no major difference in peripheral auditory function between premature babies with CSH and those without CHS. However, the I-V interval in the CLD babies was significantly longer than in the non-CLD babies. Clearly, premature babies with CSH are associated with poorer central, more specifically brainstem, auditory function. CSH occurring during the premature period exert adverse effect on myelination and functional status of the central auditory system.
Acknowledgements
We thank the doctors and nurses at the Division of Neonatology, Children’s Hospital of Fudan University for their enthusiastic assistance in recruiting subjects and collecting data.
References
- Johnston M, Trescher WH, Ishida A, Nakajima W (2001) Neurobiology of hypoxic-ischemic injury in the developing brain. Pediatr Res 49(6): 735-741.
- Jiang ZD (2008) Brainstem electrophysiological changes after perinatal hypoxia ischemia. In: Hämäläinen E (Ed) New Trends in Brain Hypoxia Ischemia Research. Nova Science Publishers, New York, USA, Pp. 203-220.
- Jiang ZD (2015) Evoked potentials in pediatric brainstem lesions. In: G Galloway (Ed) Clinical Neurophysiology in Pediatrics: A Practical Approach to Neurodiagnostic Testing and Management. Demos Medical Publishing, LLC, New York, USA, Pp. 187-213.
- Jiang ZD (2010) Damage of chronic sublethal hypoxia to the immature auditory brainstem. In: Fiedler D, Krause R, editors, Deafness, Hearing Loss, and the Auditory System. Nova Science Publishers, New York, USA, Pp. 159-180.
- Curristin SM, Cao A, Stewart WB, Zhang H, Madri JA, et al. (2002) Disrupted synaptic development in the hypoxic newborn brain. Proc Natl Acad Sci USA 99(24): 15729-15734.
- Ment LR, Schwartz M, Makuch RW, Stewart WB (1998) Association of chronic sublethal hypoxia with ventriculomegaly in the developing rat brain. Dev Brain Res 111(2): 197-203.
- Schwartz MLS, Vaccarino F, Chacon M, Yan WL, Ment LR, et al. (2004) Chronic neonatal hypoxia leads to long term decreases in the volume and cell number of the rat cerebral cortex. Semin Perinatol 28(6): 379-388.
- Stewart WB, Ment LR, Schwartz M (1997) Chronic postnatal hypoxia increases the numbers of cortical neurons. Brain Res 760(1-2): 17-21.
- Weiss J, Takizawa B, McGee A, Stewart WB, Zhang H, et al. (2004) Neonatal hypoxia suppresses oligodendrocyte Nogo-A and increases axonal sprouting in a rodent model for human prematurity. Exp Neurol 189(1): 141-149.
- Hack M, Wright LL, Shankaran S, Tyson JE, Horbar JD, et al (1995) Very-low-birth-weight outcomes of the National Institute of Child Health and Human Development Neonatal Network, November 1989 to October 1990. Am J Obstet Gynecol 172(2 Pt 1): 457-464.
- Vohr BR, Msall ME (1997) Neuropsychological and functional outcomes of very low birth weight infants. Semin Perinatol 21(3): 202–220.
- Poets CF, Stebbens VA, Richard D, Southall DP (1995) Prolonged episodes of hypoxia in preterm infants indetectable by cardiorespiratory monitors. Pediatrics 95(6): 860-863.
- Cutz E, Chiasson D (2008) Chronic lung disease after premature birth. N Engl J Med 358(7): 743-745.
- Greenough A, Milner AD (2005) Pulmonary disease of the newborn: Chronic lung disease. In: Rennie JM, editor. Roberton’s Textbook of Neonatology. (4th edn), Elsevier Churchill Livingstone, Edinburgh, Schotland Pp. 554-572.
- Mazloum D, Moschino L, Bozzetto S, Baraldi E (2014) Chronic lung disease of prematurity: long-term respiratory outcome. Neonatology 105(4): 352-356.
- Tracy MK, Berkelhamer SK (2019) Bronchopulmonary Dysplasia and Pulmonary Outcomes of Prematurity. Pediatr Ann 48(4): e148-e153.
- Sahni M, Bhandari V (2020) Recent advances in understanding and management of bronchopulmonary dysplasia. F1000Res 9: F1000 Faculty Rev-703.
- Jiang ZD, Brosi DM, Wilkinson AR (2006) Brainstem auditory function in very preterm infants with chronic lung disease: Delayed neural conduction. Clin Neurophysiol 117(7): 1551-1559.
- Wilkinson AR, Jiang ZD (2006) Brainstem auditory evoked response in neonatal neurology. Semin Fet Neonatol Med 11(6): 444-451.
- Jiang ZD, Brosi DM, Wilkinson AR (2021) Postnatal functional status of the brainstem auditory pathway in term infants after perinatal hypoxia-ischemia. J Matern Fetal Neonatal Med doi: 10.1080/14767058.2020.1836618.
- Jiang ZD, Brosi DM, Wilkinson AR (2010) Differences in impaired brainstem conduction between neonatal chronic lung disease and perinatal asphyxia. Clin Neurophysiol 121(5): 725-733.
- Jiang ZD, Brosi DM, Wilkinson AR (2010) Relationship between neonatal brainstem auditory function during the neonatal period and depressed Apgar score. J Matern-Fet Neonat Med 23(9): 973-979.
- Jiang ZD, Brosi DM, Chen C, Wilkinson AR (2009) Brainstem response amplitudes in neonatal chronic lung disease and differences from perinatal asphyxia. Clin Neurophysiol 120(5): 967-973.
- Friss HE, Wavrek D, Martin WH, Wolfson MR (1994) Brainstem auditory evoked responses to hypercarbia in preterm infants. Electroencephalog Clin Neurophysiol 90(5): 331-336.
- Jiang ZD, Brosi DM, C Chen, Wilkinson AR (2009) Impairment of perinatal hypoxia-ischaemia to the preterm brainstem. J Neurolog Sci 287(1-2): 172-177.
- Jiang ZD, Brosi DM, Wilkinson AR (2009) Depressed brainstem auditory electrophysiology in preterm infants after perinatal hypoxia-ischemia. J Neurol Sci 281(1-2): 28-33.
- Freeman S, Sohmer H, Silver S (1991) The effect of stimulus repetition rate on the diagnostic efficacy of the auditory nerve-brain-stem evoked response. Electroencephalog Clin Neurophysiol 78(4): 284-290.
- Inagaki M, Kaga M, Isumi H, Hirano S, Takashima S, et al. (1997) Hypoxia-induced ABR changes and heat shock protein expression in the pontine auditory pathway of young rabbits. Brain Res 757(1): 111-118.
- Jiang ZD, Woung GM, Shao XM, Wilkinson AR (2005) Dynamic changes in brainstem auditory electrophysiology in newborn piglets after ischaemia. Pediatric Res 58: 426.
- Sohmer H, Freeman S, Gafni M, Goitein K (1986) The depression of the auditory nerve-brainstem evoked response in hypoxemia - mechanism and site of effect. Electroenceph Clin Neurophysiol 64(4): 334-338.
- Mencher LS, Mencher GT (1999) Neonatal asphyxia, definitive markers and hearing loss. Audiology 38(6): 291-295.
- Jiang ZD (2015) Neural conduction impairment in the auditory brainstem and the prevalence in term babies in intensive care unit. Clin Neurophysiol 126: 1446-1452.
- Kountakis SE, Skoulas I, Phillips D, Chang CYJ (2002) Risk factors for hearing loss in neonates: a prospective study. Am J Otolaryngol 23(3): 133-137.
- Newton V (2001) Adverse perinatal conditions and the inner ear. Semin Neonatol 6(6): 543-551.
- Jiang ZD, Brosi DM, Wang J, Shao XM, Wilkinson AR (2004) One-third of term babies after perinatal hypoxia-ischaemia have transient hearing impairment: dynamic change in hearing threshold during the neonatal period. Acta Paediatr 93(1): 82-87.
- Wang C, Jiang ZD (2015) Brainstem auditory response findings in very preterm babies in the intensive care unit. Neonatology 107(2): 157-160.
- Jiang JK, Wang C, Jiang ZD (2021) Brainstem auditory impairment in very preterm infants who experience chronic sublethal hypoxia. Arch Neurol Neurosci 11(3): ANN.MS.ID.000761.
- Dixon G, Badawi N, Kurinczuk JJ, Keogh JM, Silburn SR, et al. (2002) Early developmental outcomes after newborn encephalopathy. Pediatrics 109(1): 26–33.
- Gonzalez FF, Miller SP (2006) Does perinatal asphyxia impair cognitive function without cerebral palsy? Arch Dis Child Fetal Neonatal Ed 91: F454-F459.
- Levene MI (2001) The newborn infant. In: Levene MI, Chervenak FA, Whittle M, editors. Fetal and neonatal neurology and neurosurgery (4th edn), Churchill-Livingstone, Edinburgh, Scotland, Pp 471-504.
- Levene MI, Evans DJ (2005) Neurological problems in the newborn: Hypoxic-ischaemic brain injury. In: Rennie JM, (Ed) Roberton’s Textbook of Neonatology (4th ed), Elsevier Churchill Livingstone, Edinburgh, Scotland, Pp. 1128-1148.
- Marlow N, Rose AS, Rands CE, Draper ES (2005) Neuropsychological and educational problems at school age associated with neonatal encephalopathy. Arch Dis Child 90(5): 380–387.
- Rennie JM, Hagmann CF, Robertson NJ (2007) Outcome after intrapartum hypoxic ischaemia at term. Semin Fetal Neonatal Med 12(5): 398-407.
- Volpe JJ (2001) Hypoxic-ischemic encephalopathy: Clinical aspects. In: Volpe JJ (Ed) Neurology of the Newborn (4th edn) WB Saunders, Philadelphia, USA Pp. 331-394.
- Shima Y, Kumasaka S, Migita M (2013) Perinatal risk factors for adverse long-term pulmonary outcome in premature infants: comparison of different definitions of bronchopulmonary dysplasia/chronic lung disease. Pediatr Int 55(5): 578-581.






























