Clitoromegaly in Premature Infants: Is It Truly Pathologic?
Meenal Gupta, MD1*, Charis Chambers, MD2, Katherine O’Flynn O’Brien MD2, Laurel Hyle JD, MPH3, Michael E Scheurer, PhD4, Lefkothea Karaviti, MD1 and Oluyemisi Adeyemi-Fowode, MD2
1Department of Pediatrics, Section of Diabetes and Endocrinology, Baylor College of Medicine/Texas Children’s Hospital, USA
2Department of Obstetrics and Gynecology, Division of Pediatric and Adolescent Gynecology, Baylor College of Medicine/Texas Children’s Hospital, USA
3Chief, Ethics, Texas Children’s Hospital, Department of Pediatrics, Baylor College of Medicine, USA
4Center for Epidemiology and Population Health, Department of Pediatrics, Baylor College of Medicine, USA
Submission: March 24, 2021; Published: August 12, 2021
*Corresponding author: Meenal Gupta, Department of Pediatrics, Division of Endocrinology, University of Washington, Seattle Children’s Hospital, 4800 Sand Point Way NE, Seattle, WA, 98105, USA, Tel: 206-987-5627; Fax: 206-985-3373
How to cite this article: Gupta M, Chambers C, O’Flynn O’Brien K, Hyle L., Scheurer M.E., et al. Clitoromegaly in Premature Infants: Is It Truly Pathologic?. Acad J Ped Neonatol. 2021; 10(3): 555843. DOI: 10.19080/AJPN.2021.10.555843
Abstract
Background: In premature infants, normative data for clitoral size are limited and transient clitoromegaly has been reported. Consequently, the potential for over-diagnosis is high, leading to increased healthcare burden and parental stress. We aimed to determine if clitoromegaly in premature infants is associated with hormonal dysfunction and whether lower birth weight and prematurity are associated with formal consults for perceived clitoromegaly.
Methods: This retrospective, institutional chart review included premature female infants with perceived clitoromegaly born between January 2012 and December 2018. Based on receiving a consult for clitoromegaly, patients were divided into ‘formal consult’ and ‘no formal consult’ groups. True clitoromegaly was defined as clitoral length >9 mm or clitoral width >6 mm. In the ‘no formal consult’ group, we assessed persistence of clitoromegaly based on the discharge examination.
Results: 29 patients met inclusion criteria: 15 in the ‘formal consult’ group and 14 in the ‘no formal consult’ group. Only 3 patients in the ‘formal consult’ group had true clitoromegaly: one had a completely normal evaluation, and two had results consistent with immaturity of the adrenal glands. In the ‘no formal consult’ group, only 3 patients had clitoromegaly noted on discharge and outcome was unknown for 1. Multi-variable logistic regression showed that extreme prematurity (p=0.04) and history of IUGR (p=0.04) independently increased the likelihood of a formal consult.
Conclusion: The majority of clitoromegaly in premature infants is not associated with hormonal dysfunction. Extreme prematurity and a history of IUGR increase the likelihood of a formal consult for perceived clitoromegaly.
Keywords: Clitoromegaly, Prematurity, Ambiguous Genitalia, Transient Clitoromegaly, Iugr, Extreme Prematurity, Clitoromegaly Consult
Abbreviations: 17-OHP: 17-hydroxyprogesterone; 17-OHPreg: 17-hydroxypregnenolone; 3b-HSD: 3-b-hydroxysteroid dehydrogenase; A: Androstenedione; ACTH: Adrenocorticotropic hormone; AGA: Appropriate for gestational age; AMH: Anti-müllerian hormone; CA: Chronological age; CAH: Congenital adrenal hyperplasia; DHEA: Dehydroepiandrostenedione; DHEA-S: Dehydroepiandrostenedione sulfate; DOC: Deoxycorticosterone; DSD: Disorders of sexual differentiation; EMR: Electronic medical record; FSH: Follicle stimulating hormone; GA: Gestational age; GU: Genito-urinary; IUGR: Intra-uterine growth restriction; LGA: Large for gestational age ; LH: Luteinizing hormone; LMP: Last menstrual period; NBS: Newborn screen; NICU: Neonatal intensive care unit; PAIS: Partial androgen insensitivity syndrome; PMA: Post menstrual age; POR: P450-oxidoreductase; SGA: Small for gestational age; T: Testosterone; TCH: Texas Children’s Hospital; US: Ultrasound
Background
Clitoromegaly, or abnormal enlargement of the clitoris, can be congenital or acquired and is usually associated with excess androgen exposure. Neonatal clitoromegaly can result from hormonal causes such as in-utero androgen exposure of fetal, maternal, or placental origin. Occasionally, non-hormonal conditions like certain syndromes (e.g. Beckwith-Wiedemann Syndrome, etc.), field defects, cysts or tumors of the clitoris may result in clitoromegaly [1]. The most common pathologic cause of neonatal clitoromegaly is congenital adrenal hyperplasia (CAH) with an incidence of about 1:10,000 to 1:20,000 [2-4]. However, the exact incidence of neonatal clitoromegaly is not known due to lack of a standard definition, subjectivity in visual assessment and inconsistencies in measurement technique.
Although a standard definition of clitoromegaly is lacking, generally, based on normative data in various populations, a clitoral length >9 mm and a clitoral width >6 mm in full-term infants are considered abnormal [5-14]. Wide racial and ethnic differences exist with some studies suggesting a clitoral length cut-off of 10 mm[10] while others suggesting 6 mm [11]. Several of these studies have also shown an inverse correlation between birth weight and clitoral size [6,7,9,11,13,15]. Limited normative data for clitoral size in the premature infant population, particularly the extreme prematurity group (<28 weeks gestation at birth), lead to extrapolation of aforementioned criteria for determination of clitoromegaly [8-13]. This extrapolation does not account for the important distinction that clitoral size in premature infants is typically larger than that that in term infants [8,10-13] and can result in over-diagnosis of clitoromegaly in these infants.
In 2019, 1 in every 10 infants born in the United States was preterm [16]. The increasing incidence and higher survival rates of premature infants [17-19] have resulted in increasing consults for perceived clitoromegaly. The birth of a neonate with ambiguous genitalia or suspected disorder of sexual differentiation (DSD) is considered a medical and social emergency [2,20]. Genital ambiguity along with risk of potentially fatal complications from CAH-related adrenal crisis make neonatal clitoromegaly a cause of major concern for primary physicians as well as patients’ families. In addition, uncertainty related to the diagnosis and fear of stigmatization cause substantial psychological stress for parents [21-23]. Most of these infants undergo an extensive evaluation to rule out hormonal pathology [24,25].
Several studies have reported transient clitoromegaly in premature infants, but the exact mechanisms are not well understood. High circulating androgen levels have been associated with transient clitoromegaly in infants born prematurely [4,24,26,27]. One of the proposed mechanisms for these transiently elevated androgen levels is the persistence of fetal adrenal zone activity until 42 weeks PMA (post-menstrual age) regardless of gestational age (GA) at birth [28,29]. Adrenocorticotropic hormone (ACTH) is not the sole regulator of fetal adrenal zone activity [29] and more recently, in vivo and in vitro studies have shown that kisspeptin-Kiss1R signaling may play a role in fetal adrenal gland development and steroidogenesis [30]. Another mechanism, proposed by Greaves et al. [31] in a case series of four premature infants with transient clitoromegaly, is a transient activation of the pituitary-gonadal axis leading to luteinizing hormone (LH) surge or an increased LH: follicle stimulating hormone (FSH) ratio. The authors further postulated that higher or prolonged kisspeptin surge in some extremely premature infants [32], possibly generated from brain or placenta [33], led to this gonadotropin surge and ovarian programming [31]. In the immediate postnatal period (6-10 weeks), premature female infants have been shown to have considerably higher LH and FSH levels compared to term infants indicating an interruption in maturation of hypothalamic-pituitary-gonadal axis caused by premature birth [34]. Zimmer et al. [35] described 5 cases of fetal transient clitoromegaly noted at 15-16 weeks gestation on obstetric ultrasound (US) with disappearance of clitoromegaly by 22-26 weeks gestation and normal female external genitalia at birth. Similarly, prenatal sonographic diagnosis of clitoromegaly in 16 patients at mean GA of 26.5 weeks was postnatally confirmed in only 6 patients indicating possibility of transient fetal clitoromegaly [36]. Other proposed mechanisms include: altered binding proteins in some very low birth weight infants, variation in end organ sensitivity and localized conversion of fetal steroids to more potent androgens at the site of action in the genitalia [26,31]. The apparent prominence of clitoris in some premature infants may be due to the fact that the clitoris is fully developed by 27 weeks of gestation while surrounding labial tissues are not [8]. Moreover, lack of vulvar subcutaneous fat or clitoral edema in premature infants may be falsely perceived as clitoromegaly [4].
Historically, the management of atypical genitalia was often based on a paternalistic model focused on physician, parental and societal comfort rather than a patient-centric approach. This raises serious ethical concern at baseline and the concern is heightened by feedback provided by adult patients who have experienced surgical interventions that they believe have negatively impacted their health [37,38]. Ethical concerns centered on patient autonomy, justice, benefit, and burden persist regarding irreversible procedures in the absence of known patient benefit.
In premature infants, the phenomenon of transient clitoromegaly and potential for over-diagnosis highlight the importance of streamlining the evaluation based on probable etiology and overall clinical condition. Understanding the etiology is crucial to avoid unnecessary patient discomfort, parent psychological stress, healthcare costs, and resource outlay for follow-up. We hypothesized that the majority of clitoromegaly in premature infants is not associated with an underlying hormonal dysfunction. We also hypothesized that in premature infants with perceived clitoromegaly, lower birth weight and lower gestational age are associated with a formal consult for clitoromegaly.
Methods
The aims of this retrospective chart review were to evaluate the percentage of hormonal dysfunction in premature infants with perceived clitoromegaly and identify factors associated with a formal consult for clitoromegaly in these patients. The study protocol was approved by the Baylor College of Medicine institutional review board.
Inclusion criteria
Infants with perceived clitoromegaly, sex female/unknown, born at Texas Children’s Hospital (TCH), main campus location and admitted to the Neonatal Intensive Care Unit (NICU) from January 2012 to December 2018 were included in this study. Perceived clitoromegaly was defined as documentation of clitoromegaly in the electronic medical record (EMR) by the NICU team at any time during the admission. Preterm (<37 weeks gestation at birth) and term (³37 weeks gestation at birth) infants were included in the initial review. The subsequent analyses included only the preterm infants.
Exclusion criteria
Male infants and infants born at outside hospitals were excluded from the study. The latter were excluded due to the lack verifiable birth history and anthropometric data as well as to avoid referral bias. Term infants were excluded after the initial review.
Data collection and classification of patients
A comprehensive search of the EMR was conducted using the following: relevant ICD-10 codes; stand-alone search terms “clitoris”, “clitoromegaly” and “cliteromegaly”; aforementioned search terms coupled with consult orders for pediatric endocrinology, pediatric gynecology, pediatric urology, or genetics; and procedure codes for laboratory tests including 17-hydroxyprogesterone (17-OHP) and a lab panel for CAH diagnoses used at our institute (CAH profile 6). A secure database was created via the TCH-REDcap® (Research Electronic Data Capture) web application [39,40] and patients meeting any of the above search criteria were included in chart review.
Patients that met inclusion criteria were divided into the ‘formal consult’ and the ‘no formal consult’ groups (Figure 1). The ‘formal consult’ group consisted of patients for whom the NICU team had requested a formal consult for clitoromegaly from any of the DSD experts (subspecialists in pediatric endocrinology, pediatric gynecology, pediatric urology, or genetics). Patients with consults for ambiguous genitalia or abnormal newborn screen (NBS) for CAH without documented findings or concern for clitoromegaly were excluded. The ‘no formal consult’ group consisted of patients with documented clitoromegaly divided into the following sub-groups– patients that did not receive a formal consult from a DSD expert were in the ‘no consult’ sub-group; those that received a consult, but not for clitoromegaly were in the ‘consult reason: other’ sub-group.
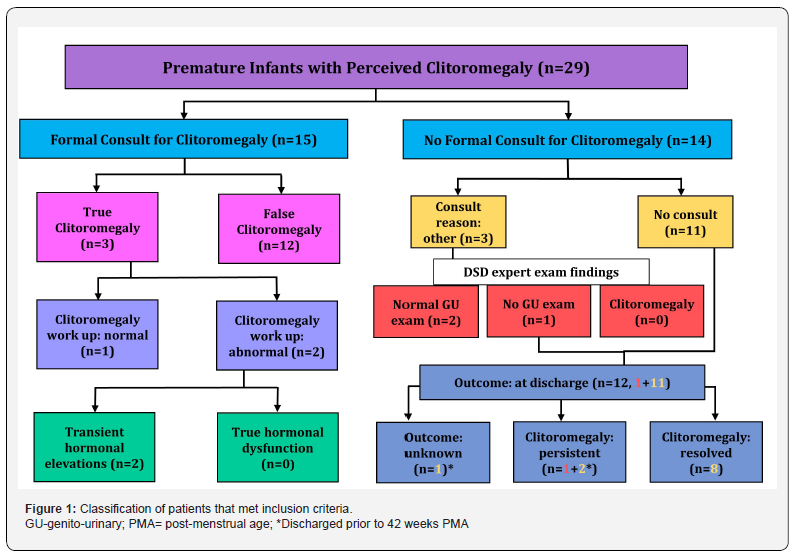
Data pertaining to prenatal and maternal history, anthropometry, demographics, clinical course, and physical examinations were collected. These data included birth weight, birth length, GA at birth, race, ethnicity, NBS results for CAH, maternal age, maternal signs of virilization, maternal use of progestins, singleton/multiple birth, history of intrauterine growth retardation (IUGR), size for GA, genito-urinary (GU) exam findings and length of NICU stay. Clitoromegaly-related work up including laboratory and radiological investigations was also reviewed. Laboratory data included levels of 17-OHP, 17-hydroxypregnenolone (17-OHPreg), dehydroepiandrostenedione (DHEA), dehydroepiandrostenedione sulfate (DHEA-S), androstenedione (A), 11-deoxycortisol, deoxycorticosterone (DOC), LH, FSH, estradiol, testosterone (T), anti-Müllerian hormone (AMH), renin, aldosterone, cortisol, and genetic tests. Radiological data included any pelvic and abdominal imaging results. Additional data were collected for patients in either group who were examined by a DSD experts. These data included weight, length on day of exam (±5 days from exam), post-menstrual age (PMA) on day of exam, GU exam findings, clitoral length, clitoral width, follow-up exam findings if any. In the ‘consult reason: other’ sub-group, GU exam findings in the DSD expert consult note were used to determine if patient had true clitoromegaly. For patients in this sub-group without a documented GU exam in the expert consult note and for patients in the ‘no consult’ sub-group, the NICU documented discharge examination was used to assess persistence of clitoromegaly.
Patients in the ‘formal consult’ group were sub-divided into two groups based on the GU exam in the consult note (Figure 1). The ‘true clitoromegaly’ group consisted of patients with clitoral length >9 mm or clitoral width >6 mm. Patients not meeting these measurement criteria and those with clitoral edema or prominent clitoral hood were classified under ‘false clitoromegaly’. The ‘true clitoromegaly’ group patients were further divided into ‘work up- normal’ and ‘work up-abnormal’ sub-groups based on the abnormal flags in the EMR for the tests in theclitoromegaly-related work up. Patients in the ‘work up-abnormal’ sub-group were further divided into two groups based on whether the test results were indicative of a true hormonal dysfunction (e.g., CAH) or a transient hormonal elevation related to immaturity of the adrenal gland.
Statistical analyses
The study population was described and differences between the ‘formal consult’ and ‘no formal consult’ groups were determined using the chi-squared test or Fisher’s exact test for categorical variables and Student’s t-test for continuous variables. Univariable and multivariable logistic regression were used to determine factors that increased the likelihood of a formal consult. All variables with a significance level below 0.20 in univariate analysis were considered for inclusion in the multivariable model. The likelihood ratio test was used to determine which variables were most predictive of a formal consult in the multivariable regression model [41].
Results
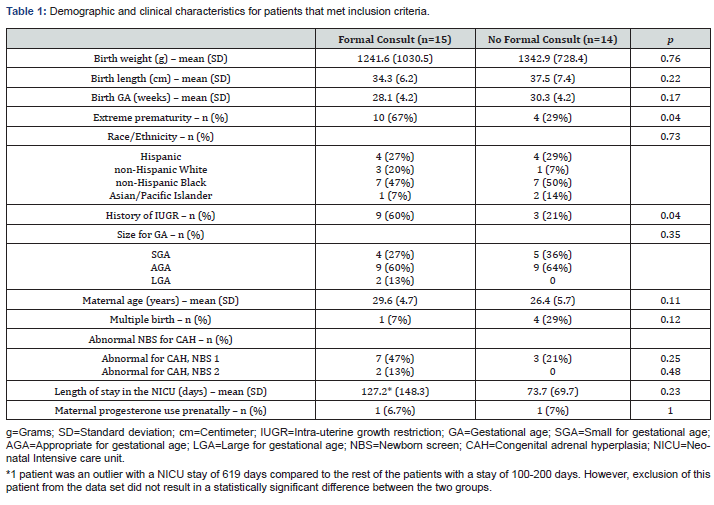
We identified 29 patients that met inclusion criteria: 15 in the ‘formal consult’ group and 14 in the ‘no formal consult’ group (Table 1). No statistically significant differences were found between the two groups in terms of anthropometric data, demographics, maternal factors, NBS for CAH or length of NICU stay. An IUGR history was more common in the ‘formal consult’ group (60%) vs. ‘no formal consult’ group (21%) (p=0.04). Extreme prematurity was also more common in the ‘formal consult’ group (67%) vs. ‘no formal consult’ group (29%) (p=0.04).
Factors associated with the likelihood of a formal consult
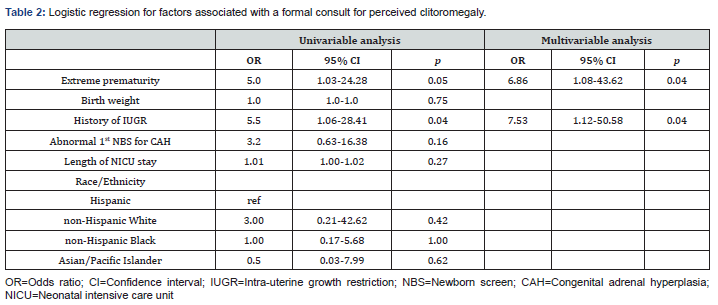
Univariable logistic regression showed that a history of IUGR (OR 5.5, CI 1.06-28.41, p=0.04) increased the likelihood of a formal consult in a premature infant with perceived clitoromegaly (Table 2). Multi-variable logistic regression showed that extreme prematurity (OR 6.86, CI 1.08-43.62, p=0.04) and a history of IUGR (OR 7.53, CI 1.12-50.58, p=0.04) increased the likelihood of a formal consult in these patients.
True vs. false clitoromegaly in the ‘formal consult’ group
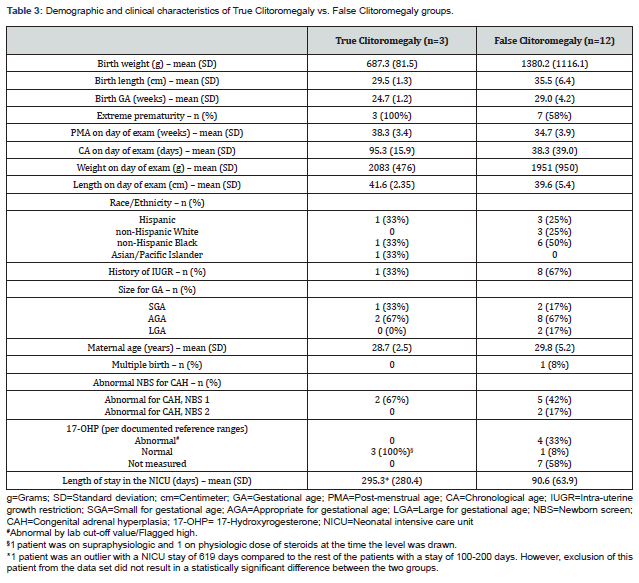
Only 3 patients in the ‘formal consult’ group had true clitoromegaly. The remaining 12 patients had false clitoromegaly (Table 3). Patients in the ‘true clitoromegaly’ group had lower birth weight, birth length and birth GA compared to those in the ‘false clitoromegaly’ group. Of note, however, patients in the ‘true clitoromegaly’ group were older (mean age 95.3±15.9 days) and more mature (mean PMA of 38.3±3.4 weeks) on the day of consult compared to those in the ‘false clitoromegaly’ group (mean age 38.3±39 days; mean PMA 34.7±3.9 weeks). Racial and ethnic diversity existed in both groups. However, the ‘false clitoromegaly’ group had a higher percentage of non-Hispanic Blacks (50%). Extreme prematurity was more common in the ‘true clitoromegaly’ group (100%) versus the ‘false clitoromegaly’ group (58%). Conversely, a history of IUGR was more common in the ‘false clitoromegaly’ group (67%) compared to the ‘true clitoromegaly’ group (33%). Most infants in both groups were appropriate for gestational age (AGA). On the first NBS for CAH, abnormalities were reported in both groups: 67% in the ‘true clitoromegaly’ versus 42% in the ‘false clitoromegaly’ group. However, on the second NBS for CAH, abnormalities were reported in no infants in the ‘true clitoromegaly’ group versus 17% in the ‘false clitoromegaly’ group.
True clitoromegaly sub-group
A review of the work-up in the ‘true clitoromegaly’ group revealed a normal 17-OHP level in all 3 patients and none of them had electrolytes derangements concerning for CAH. The clinical course and laboratory evaluation for each is outlined below:
Patient 1
Patient 1 (birth GA: 25 6/7 weeks) was 3-6-months chronological age (CA), equivalent to 42-49 weeks PMA, when the adrenal hormones were tested. Longitudinal reference ranges for this time frame are not available in premature infants. Therefore, available reference ranges in term infants of similar CA or preterm infants at a much younger CA were used for comparison [42-46]. The first 17-OHP level (69 ng/dL) was drawn at PCA 41 6/7 weeks, 5 days after a 6-day course of dexamethasone (~27 mg/m2/day hydrocortisone equivalent) (HC-E). The second 17-OHP level (81 ng/dL) was drawn a week later while the patient was on prednisone (~15 mg/m2/day HC-E). While the 17-OHP levels were normal, androstenedione and 11-deoxycortisol were mildly elevated at 274 ng/dL and 224 ng/dL respectively, based on term infant data [43,46]. Transient borderline elevations were noted in testosterone levels (33-36 ng/dL) [19,46] that decreased to 2 ng/dL by 7.5 months CA. Both NBS for CAH showed normal results, and a normal uterus and ovaries were noted on pelvic US. Follow up GU examinations reported prominent clitoral hood but no clitoromegaly.
Patient 2 and 3
The remaining patients (Patient 2 and Patient 3) in this group had a normal work up in terms of the 17-OHP level, karyotype (46, XX), normal uterus on US, and NBS for CAH (first abnormal; second normal). Patient 2 had normal levels of 17-OHP (145.5 ng/dL), androstenedione (83.1 ng/dL) and testosterone (5.1 ng/dL) at PCA 28 2/7 weeks while receiving physiologic hydrocortisone (~10 mg/m2/day). Results of a 125 µg ACTH stimulation test at 34 5/7 weeks PMA (CA=2.6 months) showed baseline and stimulated ratios of 17-OHPreg/17-OHP at 6 and 12, respectively and those of DHEA/A at 15 and 27, respectively. One month later, morning levels of these adrenal hormones were significantly lower and the baseline ratios of 17-OHPreg/17-OHP and DHEA/A were 16 and 11, respectively (Supplemental data, Table 4). These results were consistent with reduced 3-b-hydroxysteroid dehydrogenase (3b-HSD) activity related to the immaturity of the adrenal gland. Patient 2 did not have a follow up GU exam documented. Patient 3 showed significant reduction in clitoral size (from 15x10 mm to 10x1.5 mm) on the follow up exam.
False clitoromegaly sub-group
Four of the 12 patients in the ‘false clitoromegaly’ group had a 17-OHP level that flagged as abnormal; all 4 had a history of IUGR.
Patient A and B
Two patients- Patient A and Patient B- had multiple congenital anomalies and transitioned to palliative care with subsequent death at the CA of 11 and 25 days, respectively. Patient A had prenatal evidence of skeletal dysplasia and phenotypic features of Antler-Bixley syndrome. GU exam showed posterior labial fusion without clitoromegaly. A 125 µg ACTH stimulation test was performed at 36 6/7weeks PMA (CA=7 days) and critical trio whole-exome sequencing (WES) test was also ordered. However, the test results were not obtained until after the patient’s death and therefore, not formally interpreted. With undetectable baseline and stimulated levels of DHEA, A and T, the ACTH stimulation test results were concerning for possible 17, 20 lyase deficiency (Supplemental data, Table 5). The critical trio WES test results were notable for compound heterozygous pathogenic variant in the P450 oxidoreductase gene (POR) gene. Thus, the patient had ambiguous genitalia likely related to DSD from a POR deficiency but not clitoromegaly per se. Patient B had a prominent clitoral hood and cloacal anomaly but no clitoromegaly on GU exam. A 125 µg ACTH stimulation test performed at 31 4/7 weeks PMA (CA=2 days) showed baseline and stimulated ratios of 17-OHPreg/17-OHP at 5 and 21, respectively and those of DHEA/A at 2 and 15, respectively which were consistent with reduced 3b-HSD activity (Supplemental data, Table 5). A repeat stimulation test was recommended in 4-6 weeks, however patient died prior to that time period.
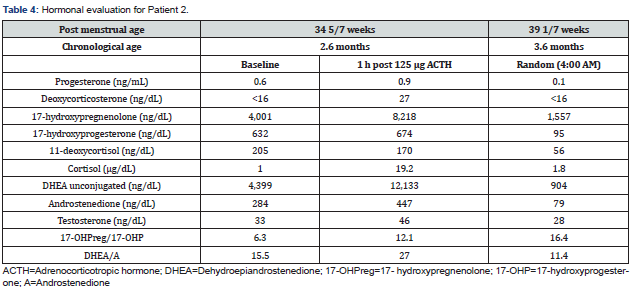
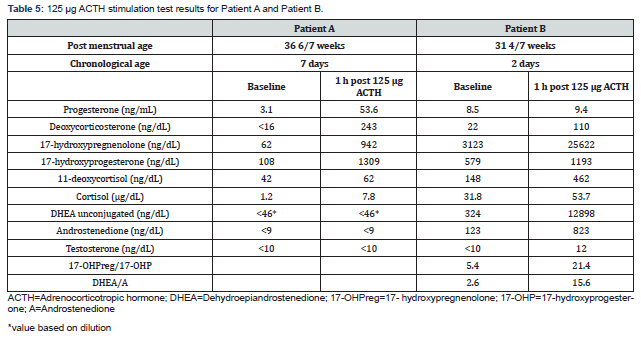
Patient C and D
Patients C and D did not meet measurement criteria for true clitoromegaly. Patient C had a 17-OHP level of 1021 ng/dL at 32 weeks PMA (CA=1.5 months) that decreased to 109 ng/dL by 41 weeks PMA. A high dose ACTH stimulation test was also performed albeit incorrectly so could not be interpreted and genetic test for common CAH mutations was negative. Lastly, Patient D had elevated levels of 17-OHP (2127 ng/dL) and 11-deoxycortisol (1439 ng/dL) at 31 6/7 weeks PMA (CA=34 days) with subsequent normalization of 17-OHP level to 221 ng/dL at 38 2/7weeks PMA.
Among the remaining 8 patients in the ‘false clitoromegaly’ group, 5 had no clitoromegaly related work-up. 1 patient had normal 17-OHP (43 ng/dL) and 11-deoxycortisol (98 ng/dL) levels. Another patient had a left-sided ovarian cyst with prominent myometrium on pelvic US with resolution of the cyst noted a month later. And the remaining patient had a 46, XX karyotype, negative results from a fluorescence in situ hybridization analysis of the SRY gene, and normal adrenal glands and cloacal malformation noted on pelvic MRI.
Outcomes in ‘no formal consult’ group
Of the 14 patients in the ‘no formal consult’ group, 11 were in the ‘no consult’ sub-group and 3 were in the ‘consult reason: other’ sub-group. In the ‘consult reason: other’ sub-group, 2 patients were classified as having ‘false clitoromegaly’ based on expert consult note documentation of a normal GU exam. The remaining patient had no GU exam documented in the expert consult note so clitoromegaly noted in the NICU discharge summary was used as evidence of persistence. In assessing the persistence of clitoromegaly in the ‘no consult’ sub-group, 8/11 patients had normal discharge GU exams indicating that clitoromegaly documented earlier in their clinical courses was either transient or false. One patient had no documented discharge GU exam, thus, the outcome was unknown. However, this patient was discharged prior to 42 weeks PMA. Overall, only 3/14 patients in the ‘no formal consult’ group had clitoromegaly noted on discharge, and 2 of these 3 patients were also discharged prior to 42 weeks PMA.
Discussion
Our study showed that the majority of clitoromegaly in premature infants is not associated with a true hormonal dysfunction. Of the 29 patients that met criteria, only 3 patients had true clitoromegaly and a pathological cause of androgen exposure was identified in none of them. While concurrent steroid administration could have falsely suppressed 17-OHP levels in patient 1 and 2, suppression from pathologic levels to completely normal levels would be highly unlikely. Additional findings on the evaluation also pointed toward a transient hormonal elevation related to immaturity of the adrenal gland as did improvement in clitoromegaly noted in 2 of the 3 patients that had a follow up exam.
We also found that a history of IUGR and extreme prematurity increased the likelihood of a formal consult in premature infants with perceived clitoromegaly. It is possible that the generally poor health in infants in these categories [47,48], compared to the more mature or appropriately growing infants, prompted the primary physician to call in a consult. Relative clitoromegaly due to lack of subcutaneous vulvar fat could also be a contributing factor [4,8]. Management of an infant with atypical genitalia is an unfamiliar and challenging scenario for most clinicians [49]. Therefore, provider experience, or lack thereof, could be a major determinant in whether a consult is called for an infant with perceived clitoromegaly. Interestingly, a history of IUGR was more common in the ‘false clitoromegaly’ group versus the ‘true clitoromegaly’ group. This finding suggested that IUGR might have affected the perception of clitoromegaly rather than existence of true pathology. Moreover, studies have shown that growth restricted infants actually produced lower 17-OHP levels and had a disproportionately small fetal adrenal zone [50,51].
False clitoromegaly was confirmed in approximately half the patients (48%): 12 in the ‘formal consult’ group and 2 in the ‘consult reason: other’ sub-group. A total of 8 (27%) patients, from the ‘no formal consult’ group, had either transient or false clitoromegaly. Three of the remaining 4 patients in this group with either persistent clitoromegaly (n=2) or outcome unknown (n=1) were discharged prior to completion of 42 weeks PMA (Figure 1). Clitoromegaly in these 3 patients could have also been transient or false, given the lack of consults for perceived clitoromegaly and persistence of fetal adrenal zone activity until 42 weeks PMA noted in previous studies [4,28]. The high percentage of false clitoromegaly noted in our study is indicative of inconsistencies in examination technique and need for provider education.
Our study findings also highlight the variability amongst specialists with regards to evaluation of clitoromegaly. Two-thirds of the consults, including those for patients in the ‘true clitoromegaly’ group, were received when patients were >4 weeks CA thus eliminating possibility of salt-wasting forms of CAH. However, a variety of biochemical and imaging studies, including 17-OHP, were performed in both true and false clitoromegaly groups. The need for a work-up, particularly in the false clitoromegaly group, could have been influenced by consulting physician’s experience, lack of familiarity with the phenomenon of transient clitoromegaly in premature infants and concern for missing rare causes of DSD or the concerns voiced by the primary team or parents.
Transient hormonal elevations noted in the clitoromegaly related work-up across both true and false clitoromegaly groups are reflective of the immaturity of the adrenal gland. Patient 2 (true clitoromegaly) and Patient B (false clitoromegaly) both had evidence of reduced 3b-HSD activity likely due to the predominance of fetal adrenal zones at birth [4,52,53]. Elevations in 11-deoxycortisol were also noted in both groups- Patient 1 (true clitoromegaly) and Patient D (false clitoromegaly). These findings could be related to reduced 11b-hydroxylase activity observed in premature infants [45,54,55]. It is well known that 17-OHP levels are higher in premature infants [54-56] and in stressed/sick infants [44,57] often leading to false-positive results on the NBS for CAH. Therefore, use of GA cut-off criteria for CAH newborn screening has been recommended [58]. Falsely elevated 17-OHP could also result from fetal adrenal zone steroids’ interference with immunoassays [59]. However, in present study, 17-OHP values were obtained by the liquid chromatography-tandem mass spectrometry (LC-MS/MS) technique, so interference is unlikely. Most importantly, the lack of long-term longitudinal reference ranges makes it difficult to accurately interpret the androgen levels in the context of prematurity. Although the 17-OHP levels depend on PMA, the more premature the infant, the longer it takes for 17-OHP values to reach those observed in healthy term infants. Therefore, term infant reference ranges are not strictly applicable to premature infants who have reached term PMA [60]. Patients C and D in the false clitoromegaly group had transiently elevated 17-OHP levels flagged as abnormal in the EMR; however, Patient C’s 17-OHP level at 1.5 months CA (1021 ng/dL) was actually normal [60]. Further, reduced 11b-hydroxylase activity could have contributed to transiently elevated 17-OHP in Patient D [55]. Moreover, transient 17-hydroxyprogesteronemia– a condition with transient elevations in 17-OHP levels for a few months without any clinical symptoms or genetic mutations related to CAH– has been reported. This occurs more frequently in premature infants, although it was thought to be independent of gestational age [61].
Why only some of these premature infants developed transient clitoromegaly whereas others did not, remains an important question. In most reported cases, transient clitoromegaly in premature infants was noted in the first few months of life [4,24,26,27,31] and not at birth. This timing was consistent with the timing of consults (CA=1-3 months) for our study patients. If true clitoromegaly is present at birth, an evaluation may be warranted as noted by Castets et al. [62]. However, the authors’ suggestion that clitoral size remains stable after 24 weeks GA is debatable. Firstly, unlike ours, their study was cross-sectional and did not follow individual patients longitudinally. Secondly, term infants constituted 75% of their study population with extremely premature infants comprising only 3%. As mentioned previously, development of transient clitoromegaly in premature infants could be related to patient-specific variables like altered binding proteins, excessive kisspeptin surge affecting adrenal or ovarian androgen production, variation in end-organ sensitivity, or localized conversion of fetal steroid to potent androgens, etc. [26,30-32].
Another question that parents and caregivers might have is regarding the duration of clitoromegaly and any long-term effects. A case series by Williams et al. [25] described 8 girls (mean age: 6 years) referred for clitoromegaly not caused by a DSD. Clitoromegaly was present at birth in 7 of the 8 patients, but clitoral size at birth was not reported for any of the patients. All 8 patients had a history of prematurity with 7 being extremely premature. Of note, however, 5 of the 8 patients only had excess clitoral skin which would not classify as true clitoromegaly. 6 of these patients were managed conservatively and 2 were referred to DSD clinic due to parental concerns. Contrastingly, in our study, 2 of the 3 patients with true clitoromegaly, who had a documented follow up exam, showed a reduction in clitoral size by 45 weeks PMA (~CA=4.5 months). Similarly, most other cases of transient clitoromegaly in premature infants reported a resolution or improvement in clitoral size by CA of 6 months-1 year [4,24,26,27,31]. Regarding long-term effects, it is important to note that reduced fetal growth in girls has been associated with pronounced adrenarche, hyperinsulinism and functional ovarian hyperandrogenism later in life [63]. It remains unclear whether transient clitoromegaly, particularly in infants with a history of IUGR, might have any long-term effects due to fetal re-programming of the hypothalamic-pituitary-adrenal axis. Addressing this issue will require longitudinal follow up studies [64,65].
Of note, no patients in the premature infant population in our study had CAH. However, our initial data-set that included a review of term infants identified three patients with classical CAH. The overall incidence of classical CAH in Texas is 1:16008 [56] and the average number of female births per year at our institute is approximately 3200. Consequently, the identification of three female infants with classical CAH born at our institute over a 7-year period is consistent with national data plus the tertiary referral status of our institute.
Limitations of our study include a small sample size of patients with true clitoromegaly. While this precluded statistical comparisons between the true versus false clitoromegaly groups, it does highlight the fact that perceived clitoromegaly in this age group is mostly false clitoromegaly. Multi-institutional studies conducted over a longer time-period would be needed to capture more patients with true clitoromegaly and assess patient factors associated with true clitoromegaly. Exclusion of male infants from our study could have potentially led to missing infants with 46, XY DSD like 5-alpha-reductase deficiency, partial androgen insensitivity syndrome (PAIS), etc. who were assigned a male sex at birth. However, these conditions rarely present as isolated clitoromegaly.
Ethical considerations
In patients with neonatal clitoromegaly, the uncertainty related to diagnosis and concern for stigmatization could potentially have lifelong ramifications for patients and caregivers [21-23]. Therefore, it is important to base the management approach on an ethical framework. The importance of mindful, open communication cannot be overstated. Streuli et al. [66] conducted a study with third-year medical students as prospective parents and demonstrated the impact of contrasting professional counseling on timing of surgery in infants with DSD. In this study, all participants believed their decision-making was based on their personal attitude despite the fact that 66% of those who were randomized to watch the ‘medicalized’ video chose early surgery versus 23% of the group who watched the ‘demedicalized’ video. Parental concerns about ‘fixing’ the atypical genital appearance in infants with isolated clitoromegaly may be centered around social constructs, fear of stigmatization, or religious or cultural beliefs. Moreover, they may be negatively impacted by lack of evidence-based knowledge. The timing of genitoplasty has been a topic of intense debate, even in confirmed cases of CAH, because data are insufficient for determining whether the benefits of early genitoplasty outweigh the risks [20,67]. Therefore, based on the principles of beneficence, non-maleficence, autonomy, and justice, early or irreversible surgical intervention is not advisable in premature infants where the likelihood of clitoromegaly being a transient phenomenon is higher. Similarly, in cases of isolated clitoromegaly without other signs of virilization or concern for classical CAH, extensive evaluation may not be needed; instead, a work-up can be pursued if clitoromegaly does not improve by the time the infant approaches term GA. Since infants cannot be autonomous decision makers, respect for parental decision-making needs to be understood within a larger context of a patient-centered approach. The focus should be on the long-term ‘best interests’ of the patient and the concept of an ‘open future’ which seeks to preserve as many autonomous decision-making opportunities as reasonably practicable to pediatric patients [37,67-69]. In communicating with parents/caregivers, it is important for physicians to adopt a sensitive approach and provide culturally appropriate education grounded in evidence-based medicine, and avoid medicalization of a potentially transient condition [23].
Conclusion
This single institution retrospective study addressed the knowledge gap regarding clitoromegaly in premature infants highlighting that the majority of these cases are not truly pathologic. Less than a tenth of patients in this study had true clitoromegaly indicating the need for objectivity in examination techniques. Moreover, heterogeneity in the approach to evaluation requires attention, as do associated costs. Standardizing the definition and evaluation of clitoromegaly in premature infants will improve patient care, help mitigate parental stress and reduce the healthcare burden related to over-diagnosis. To this end, larger studies are needed on normative data for clitoral size in premature infants, particularly the extreme prematurity group. Clinical decision-making could be optimized with the development of a risk prediction model for pathologic clitoromegaly based on patient factors and a larger sample size. Until such time, careful conservative management based on overall clinical picture and potential risk of DSD is important. At the same time, open lines of communication should be maintained between the healthcare team and parents/guardians regarding transient clitoromegaly versus possibility of DSD to help alleviate the stress related to uncertainty.
References
- Iezzi ML, Lasorella S, Varriale G, Zagaroli L, Ambrosi M, et al. (2018) Clitoromegaly in Childhood and Adolescence: Behind One Clinical Sign, a Clinical Sea. Sex Dev 12(4): 163-174.
- Kaye C, Cunniff C, Firas J, Moeschler J, Panny S, et al. (2000) Evaluation of the newborn with developmental anomalies of the external genitalia. American Academy of Pediatrics. Committee on Genetics. Pediatrics 106(1): 138-142.
- Speiser PW, Arlt W, Auchus RJ, Baskin LS, Conway GS, et al. (2018) Congenital adrenal hyperplasia due to steroid 21-hydroxylase deficiency: An endocrine society clinical practice guideline. J Clin Endocrinol Metab 103(11): 4043-4088.
- Greaves R, Kanumakala S, Read A, Zacharin M (2004) Genital abnormalities mimicking congenital adrenal hyperplasia in premature infants. J Paediatr Child Health 40(4): 233-236.
- S E Oberfield, A Mondok, F Shahrivar, J F Klein, LS Levine (1989) Oberfield Clitoral size in full term infants. Am J Perinatol 6(4): 453-454.
- Kutlu HA, Akbiyik F (2011) Clitoral length in female newborns: A new approach to the assessment of clitoromegaly. Turkish J Med Sci 41(3): 495-499.
- Asafo-Agyei SB, Ameyaw E, Chanoine J-P, Zacharin M, Nguah SB (2017) Clitoral size in term newborns in Kumasi, Ghana. Int J Pediatr Endocrinol 2017: 6, doi: 10.1186/s13633-017-0045-y.
- Riley WJ, Rosenbloom AL (1980) Clitoral size in infancy. J Pediatr 96(5): 918-919.
- Litwin A, Aitkin I, Merlob P (1991) Clitoral length assessment in newborn infants of 30 to 41 weeks gestational age. Eur J Obstet Gynecol Reprod Biol 38(3): 209-212.
- Jarrett OO, Ayoola OO, Jonsson B, Albertsson-Wikland K, Ritzen M (2015) Country-based reference values and international comparisons of clitoral size in healthy Nigerian newborn infants. Acta Paediatr 104(12): 1286-1290.
- Mondal R, Chatterjee K, Samanta M, Hazra A, Ray S, et al. (2016) Clitoral length and anogenital ratio in Indian newborn girls. Indian Pediatr 53(4): 299-303.
- Yokoya S, Kato K, Suwa S (1983) Penile and clitoral size in premature and normal newborns, infants and children. Horumon To Rinsho 31(12): 1215-1220.
- Won SY, Koh MW, Lee Tae Hyung, Eun Mi Jung, Kim Jung Sook, et al. (2001) The Clitoral Size of the Female Korean Newborn. Yeungnam Univ J Med 18(2): 287-292.
- Sane K, Pescovitz OH (1992) The clitoral index: A determination of clitoral size in normal girls and in girls with abnormal sexual development. J Pediatr 120(2 Pt 1): 264-266.
- Adekoya AO, Fetuga MB, Jarrett OO, Ogunlesi TA, Chanoine J-P, et al. (2019) Clitoral sizes and anogenital distances in term newborns in Nigeria. Int J Pediatr Endocrinol 2019: 5. doi: 10.1186/s13633-019-0069-6.
- Martin JA, Hamilton BE, Osterman MJK, Driscoll AK (2019) Births: Final data for 2018. Natl Vital Stat Rep 68(13): 1-47.
- Myrhaug HT, Brurberg KG, Hov L, Markestad T (2019) Survival and impairment of extremely premature infants: A Meta-analysis. Pediatrics 143(2).
- Saigal S, Doyle LW (2008) An overview of mortality and sequelae of preterm birth from infancy to adulthood. Lancet 371(9608): 261-269.
- Greaves RF, Pitkin J, Ho CS, Baglin J, Hunt RW, et al. (2015) Hormone Modeling in Preterm Neonates: Establishment of Pituitary and Steroid Hormone Reference Intervals. J Clin Endocrinol Metab 100(3): 1097-1103.
- Lee PA, Houk CP, Ahmed SF, Hughes IA, Achermann J, et al. (2006) Consensus statement on management of intersex disorders. Pediatrics 118(2): e488-e500.
- Crissman HP, Warner L, Gardner M, Carr M, Schast A, et al. (2011) Children with disorders of sex development: A qualitative study of early parental experience. Int J Pediatr Endocrinol 2011(1): 10.
- Madeo AC, O’Brien KE, Bernhardt BA, Biesecker BB (2012) Factors associated with perceived uncertainty among parents of children with undiagnosed medical conditions. Am J Med Genet A 158A(8): 1877-1884.
- Anthony E, Baratz AB, Boney C, Brown DR, Byne W, et al. (2006) Clinical guidelines for the management of disorders of sex development in childhood. Consortium of te Management of Disorders of Sex Development. 2006. 2 p.
- Dumont T, Black AY, Ahmet A, Fleming NA (2009) Isolated Transient Neonatal Clitoromegaly with Hyperandrogenism of Unknown Etiology. J Pediatr Adolesc Gynecol 22(5): e142-e145.
- Williams CE, Nakhal RS, Achermann JC, Creighton SM (2013) Persistent unexplained congenital clitoromegaly in females born extremely prematurely. J Pediatr Urol 9(6 PART A): 962-965.
- Midgley PC, Azzopardi D, Oates N, Shaw JC, Honour JW (1990) Virilisation of Female Preterm Infants. Arch Dis Child 65(7 Spec No): 701-703.
- Couch R, Girgis R (2012) Postnatal Virilization Mimicking 21-Hydroxylase Deficiency in 3 Very Premature Infants. Pediatrics129(5): e1364-e1367.
- Midgley PC, Russell K, Oates N, Shaw JCL, Honour JW (1996) Activity of the adrenal fetal zone in preterm infants continues to term. Endocr Res 22(4): 729-733.
- Midgley PC, Russell K, Oates N, Holownia P, Shaw JCL, et al. (1998) Adrenal function in preterm infants: ACTH may not be the sole regulator of the fetal zone. Pediatr Res 44(6): 887-893.
- Katugampola H, King PJ, Chatterjee S, Meso M, Duncan AJ, et al. (2017) Kisspeptin is a novel regulator of human fetal adrenocortical development and function: A finding with important implications for the human fetoplacental unit. J Clin Endocrinol Metab 102(9): 3349-3359.
- Greaves R, Hunt RW, Zacharin M (2008) Transient anomalies in genital appearance in some extremely preterm female infants may be the result of foetal programming causing a surge in LH and the over activation of the pituitary-gonadal axis. Clin Endocrinol (Oxf) 69(5): 763-768.
- Han SK, Gottsch ML, Lee KJ, Popa SM, Smith JT, et al. (2005) Activation of gonadotropin-releasing hormone neurons by kisspeptin as a neuroendocrine switch for the onset of puberty. J Neurosci 25(49): 11349-11356.
- Herbison AE, Clarkson J, Campbell R (2014) Kiss and tell in the brain-kisspeptins and the neural control of fertility.
- Tapanainen J, Koivisto M, Vihko R, Huhtaniemi I (1981) Enhanced Activity of the Pituitary-Gonadal Axis in Premature Human Infants. J Clin Endocrinol Metab 52(2): 235-238.
- Zimmer EZ, Blazer S, Blumenfeld Z, Bronshtein M (2012) Fetal Transient Clitoromegaly and Transient Hypertrophy of the Labia Minora in Early and Mid Pregnancy. J Ultrasound Med 31(3): 409-415.
- Cheikhelard A, Luton D, Philippe-Chomette P, Leger J, Vuillard E, et al. (2000) How accurate is the prenatal diagnosis of abnormal genitalia? J Urol 164(3 Pt 2): 984-987.
- Kon A (2015) Ethical Issues in Decision-Making for Infants with Disorders of Sex Development. Horm Metab Res 47(5): 340-343.
- Crouch NS, Creighton SM (2004) Minimal surgical intervention in the management of intersex conditions. J Pediatr Endocrinol Metab 17(12): 1591-1596.
- Harris PA, Taylor R, Thielke R, Payne J, Gonzalez N, et al. (2009) Research electronic data capture (REDCap)-A metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform 42(2): 377-381.
- Harris PA, Taylor R, Minor BL, Elliott V, Fernandez M, et al. (2019) The REDCap consortium: Building an international community of software platform partners. J Biomed Inform 95:103208.
- Hosmer DW, Lemeshow S (2000) Applied Logistic Regression. (2nd edn), John Wiley & Sons, Inc, Hoboken, NJ, USA.
- Nykänen P, Heinonen K, Riepe FG, Sippell WG, Voutilainen R (2010) Serum concentrations of adrenal steroids and their precursors as a measure of maturity of adrenocortical function in very premature newborns. Horm Res Paediatr 74(5): 358-364.
- Wiener D, Smith J, Dahlem S, Berg G, Moshang T (1987) Serum adrenal steroid levels in healthy full-term 3-day-old infants. J Pediatr 110(1): 122-124.
- Lee MM, Rajagopalan L, Berg GJ, Moshang T (1989) Serum Adrenal Steroid Concentrations in Premature Infants. J Clin Endocrinol Metab 69(6): 1133-1136.
- Hingre RV, Gross SJ, Hingre KS, Mayes DM, Richman RA (1994) Adrenal steroidogenesis in very low birth weight preterm infants. J Clin Endocrinol Metab 78(2): 266-270.
- Garagorri JM, Rodríguez G, Lario-Elboj ÁJ, Olivares JL, Lario-Muñoz Á, et al. (2008) Reference levels for 17-hydroxyprogesterone, 11-desoxycortisol, cortisol, testosterone, dehydroepiandrosterone sulfate and androstenedione in infants from birth to six months of age. Eur J Pediatr 167(6): 647-653.
- Glass HC, Costarino AT, Stayer SA, Brett CM, Cladis F, et al. (2015) Outcomes for extremely premature infants. Anesth Analg 120(6): 1337-1351.
- Garite TJ, Clark R, Thorp JA (2004) Intrauterine growth restriction increases morbidity and mortality among premature neonates. Am J Obstet Gynecol 191(2): 481-487.
- Davies JH, Cheetham T (2017) Recognition and assessment of atypical and ambiguous genitalia in the newborn. Arch Dis Child 102(10): 968-974.
- Bolt RJ, van Weissenbruch MM, Popp-Snijders C, Sweep CGJ, Lafeber HN, et al. (2002) Fetal Growth and the Function of the Adrenal Cortex in Preterm Infants. J Clin Endocrinol Metab 87(3): 1194-1199.
- Farzad Mohajeri Z, Aalipour S, Sheikh M, Shafaat M, Hantoushzadeh S, et al. (2019) Ultrasound measurement of fetal adrenal gland in fetuses with intrauterine growth restriction, an early predictive method for adverse outcomes. J Matern Neonatal Med 32(9): 1485-1491.
- Riepe FG, Mahler P, Sippell WG, Partsch CJ (2002) Longitudinal study of plasma pregnenolone and 17-hydroxypregnenolone in full-term and preterm neonates at birth and during the early neonatal period. J Clin Endocrinol Metab 87(9): 4301-4306.
- Lashansky G, Saenger P, Fishmant K, Gautier T, Mayes D, et al. (1991) Normative Data for Adrenal Steroidogenesis in a Healthy Pediatric Population: Age-and Sex-Related Changes after Adrenocorticotropin Stimulation. J Clin Endocrinol Metab 73(3): 674-686.
- Nomura S (1997) Immature adrenal steroidogenesis in preterm infants. Early Hum Dev 49(3): 225-233.
- Kamrath C, Hartmann MF, Boettcher C, Wudy SA (2014) Reduced activity of 11β-hydroxylase accounts for elevated 17α-hydroxyprogesterone in preterms. J Pediatr 165(2): 280-284.
- Therrell BL, Berenbaum SA, Manter-Kapanke V, Simmank J, Korman K, et al. (1998) Results of Screening 1.9 Million Texas Newborns for 21-Hydroxylase-Deficient Congenital Adrenal Hyperplasia. Pediatrics 101(4): 583-590.
- Huysman MWA, Hokken-Koelega ACS, De Ridder MAJ, Sauer PJJ (2000) Adrenal Function in Sick Very Preterm Infants. Pediatr Res 48(5): 629-633.
- Van Der Kamp HJ, Oudshoorn CGM, Elvers BH, Van Baarle M, Otten BJ, et al. (2005) Cutoff levels of 17-α-hydroxyprogesterone in neonatal screening for congenital adrenal hyperplasia should be based on gestational age rather than on birth weight. J Clin Endocrinol Metab 90(7): 3904-3907.
- Jean F, Yves Le B, Jérôme Guéchot, Nicolas H, Marie-Anne M (2017) A Liquid Chromatography/Tandem Mass Spectometry Profile of 16 Serum Steroids, Including 21-Deoxycortisol and 21-Deoxycorticosterone, for Management of Congenital Adrenal Hyperplasia. J Endocr Soc 1(3): 186-201.
- Linder N, Davidovitch N, Kogan A, Barzilai A, Kuint J, et al. (1999) Longitudinal measurements of 17alpha -hydroxyprogesterone in premature infants during the first three months of life. Arch Dis Child - Fetal Neonatal Ed. 81(3): F175-8.
- Cavarzere P, Samara-Boustani D, Flechtner I, Dechaux M, Elie C, et al. (2009) Transient hyper-17-hydroxyprogesteronemia: A clinical subgroup of patients diagnosed at neonatal screening for congenital adrenal hyperplasia. Eur J Endocrinol 161(2): 285-292.
- Castets S, Nguyen K-A, Plaisant F, Prudon MB, Plotton I, et al. (2021) Reference values for the external genitalia of full-term and pre-term female neonates. Arch Dis Child - Fetal Neonatal Ed. 106(1): 39-44.
- Ibáñez L, Potau N, Francois I, de Zegher F (1998) Precocious Pubarche, Hyperinsulinism, and Ovarian Hyperandrogenism in Girls: Relation to Reduced Fetal Growth. J Clin Endocrinol Metab 83(10): 3558-3562.
- Paul A, Deans R, Viner R, Creighton S (2011) Pubertal Development and Sexuality in Female Adolescents Born Preterm: A Review of the Literature. Int J Adolesc Med Heal 23(3): 175-179.
- Barker DJP (1995) The fetal and infant origins of disease. Eur J Clin Invest 25(7): 457-463.
- Streuli JC, Vayena E, Cavicchia-Balmer Y, Huber J (2013) Shaping parents: Impact of contrasting professional counseling on parents’ decision making for children with disorders of sex development. J Sex Med 10(8): 1953-1960.
- Harris RM, Chan YM (2019) Ethical issues with early genitoplasty in children with disorders of sex development. Curr Opin Endocrinol Diabetes Obes 26(1): 49-53.
- Gillam LH, Hewitt JK, Warne GL (2010) Ethical principles for the management of infants with disorders of sex development. Horm Res Paediatr 74(6): 412-418.
- Wiesemann C, Ude-Koeller S, Sinnecker GHG, Thyen U (2010) Ethical principles and recommendations for the medical management of differences of sex development (DSD)/intersex in children and adolescents. Eur J Pediatr 169(6): 671-679.






























