Diagnosis of Pierre Robin’s Sequences: How to Judiciously Apply Additional Investigations
Maman Mathilde1, Loron Gauthier1, Bednarek Nathalie1, Dubernard Xavier2, Doco Fenzy Martine3,4, Delecroix Quentin5,Poli Merol Marie Laurence6 and Francois Caroline2,5,6*
1Pédiatrie, American Memorial Hospital, 47 rue Cognacg Jay, 51100 Reims, France
2ORL, Hôpital Robert Debré, Rue du Général Koenig, 51100 Reims, France
3Génétique, Hôpital Robert Debré, Rue du Général Koenig, 51100 Reims, France
4EA 3801, Hôpital Robert Debré, Rue du Général Koenig, 51100 Reims, France
5Chirurgie plastique reconstructrice et esthétique, Hôpital Maison Blanche, 45 rue Cognacq Jay 51100 Reims
6Chirurgie pédiatrique, American Memorial Hospital, 47 rue Cognacq Jay, 51100 Reims
Submission:April 17, 2020; Published:March 18, 2021
*Corresponding author: Pr Caroline FRANCOIS, Plastic Reconstructive and Aesthetic Surgery, University of Champagne Ardennes, Hôpital Maison Blanche, 45 rue Cognacq Jay, 51100 Reims, France, Tel: +33 (0)6 82873893, +33 (0)3 26783194
How to cite this article: Maman M, Loron G, Bednarek N, Dubernard X, Doco F M, et al. Diagnosis of Pierre Robin’s Sequences: How to Judiciously Apply Additional Investigations. Acad J Ped Neonatol. 2021; 10(2): 555836. DOI: 10.19080/AJPN.2021.10.555836
Abstract
There are currently no published recommendations for “standard” assessment to be performed in neonatal period in a patient with Pierre Robin sequence (PRS). This situation leads to an increasing number of investigations and a lack of consistency in the timeliness of these investigations. Primary objective of our study was to synthesize national and local practices regarding neonatal diagnostic management in children with PRS. Secondary objective was to propose a prioritization of paraclinical examinations for etiological diagnosis and severity according to clinical presentation. A multicentric survey across french PRS network was conducted to assess stated practices of initial investigation in infants presenting with PRS. A retrospective study was conducted in our center over seven years to assess observed practices as well as results get from thoses additional investigations. Both the single-centre and multicentre studies showed a wide discrepancy in the management of patients with PRS. In our single-center study, 30 children were looked over. Echocardiography was the most common diagnostic test (10 abnormalities/27 tests). Other examinations are of interest according to the clinical picture, the severity and require for some a prioritization in time. We propose a standardized screening of infants presenting with PRS, to assess both aetiology and severity of the disease: nocturnal oximetry, echocardiography, congenital deafness screening (OAS/PEA), ENT, ophthalmological and genetic consultation. In most severe cases of PRS, or abnormal clinical feature, or abnormal antenatal ultrasound, cerebral MRI, genetic testing, and renal ultrasound might be performed.
Keywords:Sequence of Pierre Robin, Cleft palate, Diagnosis
Introduction
The Pierre Robin sequence (PRS) is a congenital malformation combining retrognathism, glossoptosis and more or less a velopalatal cleft. Its prevalence varies from 1/8500 to 1/14000 births [1].
This PRS can be isolated (40% of cases), syndromic (40% of cases) or associated (association with other malformations not related to a known and described syndrome) (20% of cases) [1-4].
The absence of published recommendations and the fear of a syndromic or associated form frequently may lead to an excess of additional investigations for most of these patients. It seems necessary to perform only necessary and/or urgent investigations in order to reduce patient and/or parental stress, use of invasive, hazardous, or expensive investigations.
The primary objective of this study was to synthesize national and local practices regarding neonatal diagnostic management in children with PRS.
The secondary objective was to propose recommendations for prioritizing diagnostic severity and etiological examinations according to the clinical presentation and severity of the disease (grade 1, 2 or 3 according to Cole’s classification) [5].
These 3 grades are defining such as: children with no obvious clinical signs of respiratory distress falls into grade 1, those with intermittent signs of respiratory distress fall into grade 2 and the children concern by grade 3 are subject to moderate to severe respiratory distress when nursed supine.
Patients and Methods
Our work is divided into two parts
A multicenter survey of stated practices was conducted using a Google Drive® questionnaire. It was emailed to the 22 French PRS network (including competence and references centers for this specific rare diseases) and one Swiss Team
PRS Care in France belongs to network of rare diseases, organized as reference and competence centers. Reference centers are composed of highly specialized teams with expertise on a specific pathology that promote advanced care, research, and teaching. They are also in charge for reallocate financing to competence centers. Competence centers are locally disseminated structures which provide care for rare disease).
This questionnaire collected information about
- Respondent’s specialty (medical or surgical)
- Additional investigation performed: echocardiography, cranial ultrasound, magnetic resonance imaging (MRI), renal ultrasound and whole skeleton radiography night oximetry, polysomnography, linguistic or pharyngeal electromyography.
- Timeframe for consultations (genetic, ophthalmological and otorhinolaryngological (ENT).
- Frequency of investigation: was it performed always, never, or based on clinical findings only.
- Expected results of each investigation.
A retrospective study was conducted, to assess observed practices and results get from those additional investigations. We included patients who were hospitalized between 2009 and 2016 in our neonatal unit were PRS requiring hospitalization and investigation are systematically admitted.
For each patient, gender, antenatal clinical and ultrasound data, postnatal clinical features were extracted from medical charts. Each investigation performed was reported as well as their detailed results.
Severity of Pierre Robin’s sequences was graded as described by Cole, following three grades of increasing severity [5].
Results
multicentric survey of stated practices
We obtained 27 answers to our questionnaire from 22 teams in 19 different French centres and 1 Suisse team (1 to 3 answers per centre).
Two rare disease reference centres, 19 rare disease competence centres and a Swiss team answered our questionnaire.
Of these responses, 16 questionnaires were completed by surgeons and 11 by pediatricians (Figure 1).
The results obtained are summarized in Figure 2.
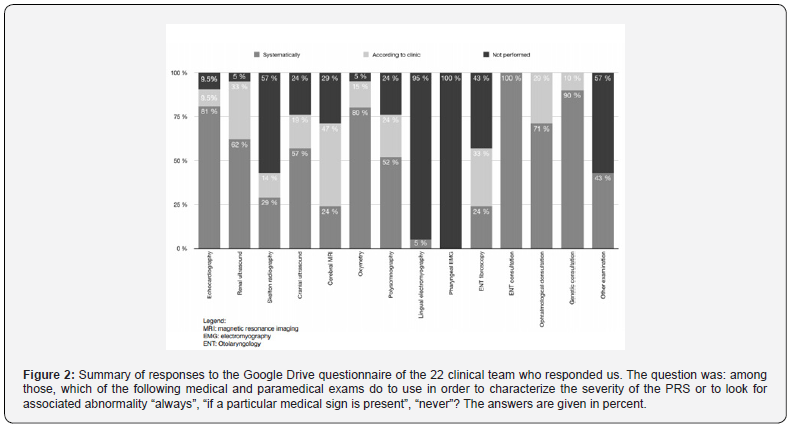
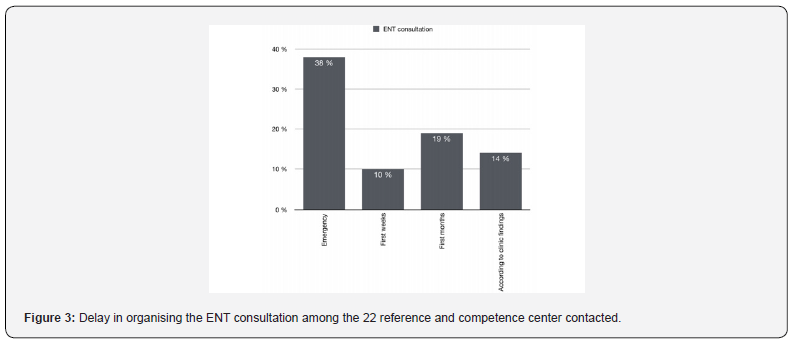
Discrepancy among centers is remarkable, as well as among physician’s specialty (Figure 2)/ The ENT consultation was systematically organized in all teams, but the time frame within which it was organized varied greatly from one team to another (Figure 3).
Echocardiography, oximetry, ophthalmological consultation, genetic consultation is frequent investigation, as they are systematically required for 81%, 80%, 71%, 90% of responders, respectively.
To the opposite the Lingual or Pharyngeal EMG is not used by any center (lack of access is frequently reported).
Thirty-eight percent of the responding teams organize an ENT consultation in emergency when diagnosing a PRS (the 100% are not reached on this graphic due to some incomplete responses on our questionnaire).
Retrospective monocentric study
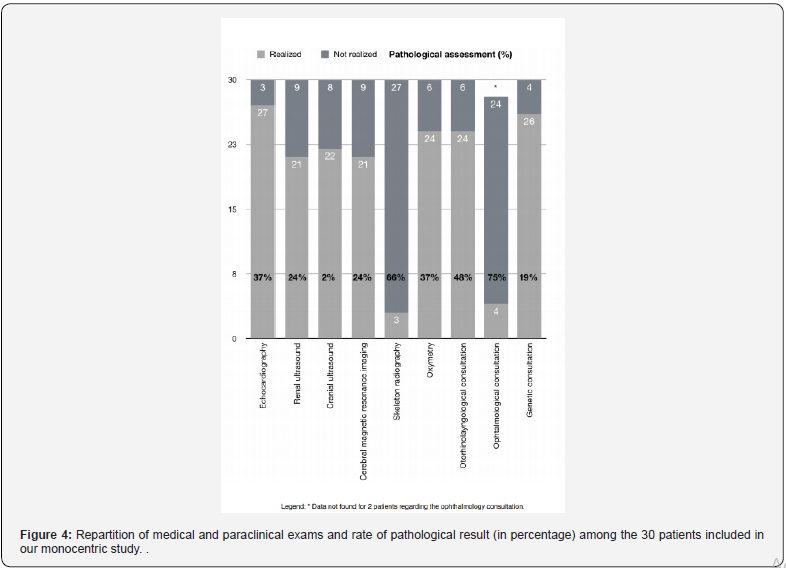
The results of the study can be observed in Table 1 and Figure 4.
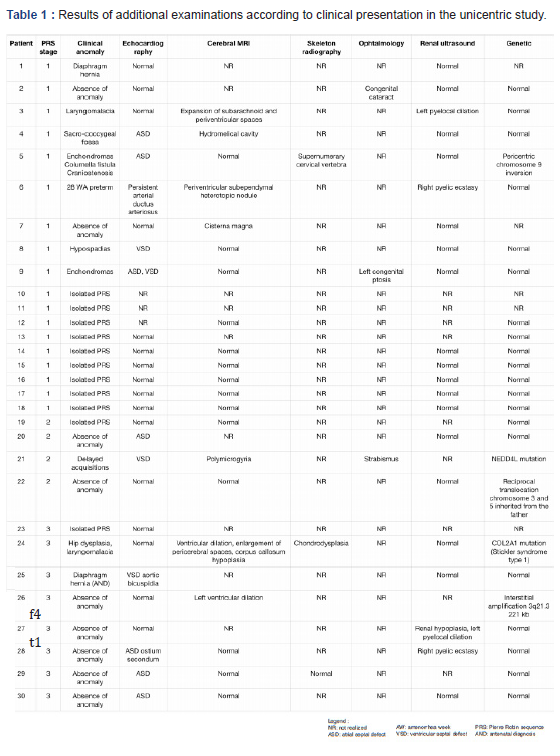
From 2009 to 2016, 30 patients were hospitalized in neonatal intensive care unit with PRS (Table 1).
Sex ratio was 12 males to 18 females. Velopalatal cleft was systematically reported in those patients (Table 1).
We reported 18 Grade 1 PRS (60%), 4 Grade 2 (13,3%) and 8 Grade 3 PRS patients (26,6%). Among our studied patients only 2 had an antenatal suspicion of PRS (6.67%).
Oxymetry was performed in 80 % of cases, to assess severity of the disease. Echocardiography, renal ultrasound, cranial ultrasound, cerebral magnetic resonance imaging, ENT and genetic consultations were respectively provided in 90%, 70%, 73%, 70%, 80% and 86% of infants presenting with PRS.
Skeleton radiography and ophthalmological consultation were rarely performed during initial hospitalization (6,6% and 13,3%).
In the monocentric, retrospective of observed practices, we report that 19 over 30 patients presents abnormal result on one or more additional investigation. The anomalies found were mostly associated, as in the case of the patient with Stickler’s syndrome, who had several associated anomalies suspected on clinical examination (chondrodysplasia, delayed acquisitions, or ophthalmic anomaly).
Discussion
The management of patients diagnosed with PRS at birth is currently not standardized. The teams involved carry out a variable number of investigations, either systematically or triggered by abnormal clinical features and/or abnormal findings on systematic ultrasound scans during pregnancy.
This study revealed a significant discrepancy in the diagnostic management of patients within t secondary objective of this study was to propose a prioritization of these examinations.
It seemed useful to us to divide our assessment into two objectives: severity (to guide the therapeutic management of patients) and etiology (to look for abnormalities in a syndromic or associated form).
Severity assessment
Oximetry and polysomnography
The systematic nocturnal oximetry for each patient with PRS is for us, as in the literature [6-8], a key-element of the assessment. This will guide respiratory management (head-elevated supine position, head-elevated prone position, need for appropriate management with ventilatory assistance such as Continuous Positive Airway Pressure (CPAP) during sleep or waking phases). Monitoring with oximetry control or even polysomnography has shown sufficient efficacy (in terms of weight gain and apnea/ hypopnea index) of conservative versus surgical treatment (glossopexy, osteo-mandibular distraction). It therefore seems essential that all these patients benefit from oximetry in order to assess their respiratory impairment and adapt their management.
Polysomnography makes it possible to guide the therapeutic management of patients on the respiratory level but also on the nutritional level: [9]
- Oximetry alone: significant respiratory symptomatology in the supine position but no desaturations in the head-elevated prone position and no nutritional difficulties.
- Oximetry and polysomnography: no respiratory distress in the ventral proclive but persistent dietary difficulties after the 2nd week of life. If obstructive and severe apneas: nasopharyngeal tube and management of feeding difficulties.
- Polysomnography, ENT investigations (flexible fibroscopy and bronchoscopy): persistence of symptoms despite optimal management (nasopharyngeal tube) [9].
Laryngoscopy and bronchoscopy should be proposed in a second step if respiratory symptoms persist despite optimal management (nasopharyngeal tube). These investigations therefore do not have their place in the first-line diagnostic assessment of PRS with respiratory disorders successfully managed by positioning [9].
Polysomnography therefore seems indicated in children with persistent disorders despite optimal management, i.e., patients with grade 2 or higher according to the classification of Cole et al. [9].
Lingual and pharyngeal electromyography
Lingual and pharyngeal electromyography (EMG) is of interest in the clinical evaluation of patients to characterize the severity of the disease and the risks of normal feeding in these children [10-11]. However, only a few centers have routine access to these examinations. Dynamic electromyography makes it possible to evaluate the neuromuscular elements involved in the suction-deglutition reflex. It allows a qualitative evaluation of the swallowing sucking disorder and highlights an incoordination between the oral and pharyngeal phases of swallowing. A motor dysfunction of the esophagus may also be partly responsible by affecting the esophageal time of swallowing [10-12].
Significant nerve damage to the lingual EMG would predict syndromic or associated PRS [11].
Isolated PRS have moderate to minimal impairment of the motor function of swallowing [12,13].
It seems interesting to carry out a lingual and pharyngeal EMG in case of prolonged eating disorders. The completion of this examination will depend on the accessibility of the child, there is no recommended time limit, it will be adapted to the overall care of the child.
Etiological assessment
Genetic consultation and monitoring
Consultation and follow-up in genetics should be systematic in such patients. The time of first contact with a genetic team depends on the condition and the associated manifestations (associated dysmorphia, significant failure...). The conduct of genetic investigations should be discussed on a case-by-case basis after reviewing the family history, the complete clinical examination and the antenatal ultrasound record [13,14]. Unless a medical sign is detected at birth, an emergency systematic genetic study does not seem necessary. In the literature, many genetic abnormalities have been found in combination in patients with PRS during routine genetic tests. In addition, during follow-up in genetic counselling, according to some studies, up to 18% of patients initially diagnosed as isolated forms are in fact syndromic forms. The morphotype is refined with the child’s development and most of the time allows to orientate the research [13,14]. All these elements point to the importance of genetic counselling for all patients. Nor should we hesitate to repeat these consultations during the child’s growth.
The most common syndromes encountered in association with PRS are: Stickler syndrome (14.1% of PRS [2,13-15], velocardiofacial syndrome and Treacher-Collins syndrome [1,2,13].
These three syndromes would represent 65% of the syndromic forms of PRS [12].
More than 50 syndromes have been described to date. All these syndromes have the particularity of being accompanied by a particular morphotype that makes it possible to orient paraclinical research and explorations after a careful clinical examination in search of particular signs [15].
Even if the PRS is frequently isolated, the request for a genetic consultation must be systematic, without urgency in the absence of an associated clinical call point and from birth if not, in order to refine etiological diagnoses and provide genetic counselling to the family for the risk of recurrence.
Cranial ultrasound (CrUS)
CrUS is a good test for neurological follow-up of premature infants, but does not appear to be a gold standard in screening for morphological abnormalities that may be associated with PRS. Indeed, our study showed that abnormalities detected by brain MRI were not seen on CrUS in most cases.
The CrUS allows a fairly good median line analysis for associated anomalies if performed by an experienced physician. However, it allows only a limited analysis of the rest of the cerebral parenchyma.
The CrUS has not proven its effectiveness in diagnosing PRS and remains an unscramble examination, however, it should not be carried out urgently. The reference examination is the cerebral MRI which is the only one that guarantees a fine analysis of the median line, the pituitary gland, the optical chiasma and in the search for gyration anomalies.
Cerebral MRI
MRI is an examination that requires premedication for children over three months old. In our study, the MRI abnormalities found were correlated to a clinical call point. This leads us to the proposal of this examination only in the case of abnormal neurological examination or antenatal diagnosis of cerebral malformations.
However, it is important for all patients, whether or not an MRI has been performed, to continue the follow-up of these children in specialized consultation until the age of acquisition and, as the case may be, to offer follow-up to the early medical and social action centre.
Echocardiography
Cardiac damage associated with PRS is a factor that can aggravate symptomatology. Respiratory distress may be aggravated by underlying heart disease. Heart disease can lead to food intake difficulties and poor weight gain. The presence of associated heart disease also guides genetic diagnosis.
Some syndromes associated with PRS may be associated with heart disease. In this study, nearly 40% of underlying anomalies were found on systematic reviews. Echocardiography is noninvasive and can be performed at the patient’s bedside, so it seems interesting to perform post-natal echocardiography for all patients with PRS [13,14].
Renal ultrasound
In our series, the systematic use of kidney ultrasound in all patients, except in cases with antenatal anomalies, revealed minor anomalies (moderate pyelectasis...). However, these anomalies did not lead to any specific management. In this context, we recommend that a renal ultrasound be performed in the event of suspected renal antenatal abnormalities, or other associated clinical signs that may be related to renal malformation (pretragian cysts and fistulas...).
ENT counseling
In case of abnormalities of the outer ear or abnormalities in the screening for hearing disorders at birth (auditory evoked potentials): ENT consultation should be organised as a matter of urgency.
In case of PRS with difficulties in ventilatory management, it is advisable to request an ENT consultation with naso-fibroscopy to look for glottic involvement aggravating respiratory phenomena (tracheomalacia, laryngomalacia) [16-18]
ENT follow-up is systematically recommended for children with cleft palate. Children with a cleft (PRS or other cause) have a higher frequency of seromucosal ear infections that can cause hearing damage [7].
Systematic follow-up should be organised from the age of 6 months for all patients with cleft palate. The follow-up is adapted by the ENT according to the clinical findings with an annual examination at least up to 7 years, to be continued if needed in the event of a particular pathology.
Ophthalmology consultation
The association with a Stickler syndrome in the context of Pierre Robin sequences (estimated at 10 to 14% of Pierre Robin sequences (Figure 5)) encourages the greatest caution regarding ocular anomalies [13].

A consultation with a paediatric ophthalmologist must be organised in the first year of life to look for abnormalities [13,14]. If during the complete clinical examination, an anomaly is found (ptosis, permanent strabismus, absence of eye pursuit...), then an earlier consultation should be organized. Thereafter, it is advisable for these children to provide regular ophthalmological follow-up [13,19].
Other reviews
Skeletal radiography is often performed systematically (in our study, and according to the questionnaire, in many centres). This examination is radiant, long and painful for patients. In our study, the anomalies found were suspected during the patient’s clinical examination. We propose that skeletal radiography should only be performed in the event of abnormalities in the clinical examination.
In our population, few patients have been treated with a hormone test. This assessment will only be done on a case-by-case basis if the patient shows suggestive signs of an anomaly in the hypothalamic-pituitary axis.
Management algorithm and synthesis
This work leads us to suggest a systematic algorithm to choose which medical exams ask when taking in charge a newborn PRS. This algorithm aims to limit the number of exams and their cost. There are chosen according to the PRS’s grade and associated medical signs (antenatal or post-natal anomaly).
Conclusion
There is a wide disparity in practice at the national level but also within each centers depending on the discipline (paediatrician or surgeon) with regard to the additional examinations requested. In practice, we offer a “standard” etiological assessment for all patients: echocardiography, congenital deafness screening, ENT consultation, ophthalmological consultation and genetic consultation. This assessment will be completed by a cerebral MRI, a genetic test (karyotype, CGH array, search for a particular mutation), a renal ultrasound, in case of grade 3 PRS, or in case of antenatal or clinical ultrasound anomalies at the post-natal examination. The radiological assessment of the skeleton and the hormonal assessment are only to be carried out on clinical orientation. MRI is also an examination to be proposed for the possibility of reaching the median line (pituitary gland) and associated gyration anomalies that may assist in etiological diagnosis.
It is always possible, during the multidisciplinary monitoring of rare disease competence centres, to complete the assessment with new examinations if necessary. The clinical examination always remains the basis of our reflection to guide and prioritize requests for additional examinations.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
References
- Gangopadhyay N, Mendonca D, Woo A (2012) Pierre Robin Sequence. Semin Plast Surg 26(02): 76–82.
- Basart H, Paes EC, Maas SM, van den Boogaard M-JH, van Hagen JM, et al. (2015) Etiology and pathogenesis of robin sequence in a large Dutch cohort. Am J Med Genet A 167(9): 1983-1992.
- Doray B, Badila-Timbolschi D, Schaefer E, Fattori D, Monga B, et al. (2012) Epidemiology of orofacial clefts (1995-2006) in France (Congenital Malformations of Alsace Registry). Arch Pédiatrie 19(10): 1021-1029. [Article in French].
- Melkoniemi M, Koillinen H, Männikkö M, Warman ML, Pihlajamaa T, et al. (2003) Collagen XI sequence variations in nonsyndromic cleft palate, Robin sequence and micrognathia. Eur J Hum Genet 11(3): 265-270.
- Cole A, Lynch P, Slator R (2008) A New Grading of Pierre Robin Sequence. Cleft Palate Craniofac J 45(6): 603-606.
- Albino FP, Wood BC, Han KD, Yi S, Seruya M, et al. (2016) Clinical Factors Associated with the Non-Operative Airway Management of Patients with Robin Sequence. Arch Plast Surg 43(6): 506-511.
- Glynn F, Fitzgerald D, Earley MJ, Rowley H (2011) Pierre Robin sequence: An institutional experience in the multidisciplinary management of airway, feeding and serous otitis media challenges. Int J Pediatr Otorhinolaryngol 75(9):1152-1155.
- Linz A, Bacher M, Kagan K-O, Buchenau W, Arand J, et al. (2011) Pierre Robin Sequenz: Pränatale Diagnostik und interdisziplinäre Therapie. Z Für Geburtshilfe Neonatol 215(03): 105-108.
- Reddy VS (2016) Evaluation of upper airway obstruction in infants with Pierre Robin sequence and the role of polysomnography--Review of current evidence. Paediatr Respir Rev 17: 80-87.
- Renault F, Baudon J-J, Galliani E, Flores-Guevara R, Marlin S, et al. (2011) Facial, lingual, and pharyngeal electromyography in infants with Pierre Robin sequence. Muscle Nerve 43(6): 866-871.
- Baudon J-J, Renault F, Goutet J-M, Flores-Guevara R, Soupre V, et al. (2002) Motor dysfunction of the upper digestive tract in Pierre Robin sequence as assessed by sucking-swallowing electromyography and esophageal manometry. J Pediatr 140(6): 719-723.
- Côté A, Fanous A, Almajed A, Lacroix Y (2015) Pierre Robin sequence: Review of diagnostic and treatment challenges. Int J Pediatr Otorhinolaryngol 79(4): 451-464.
- Rathé M, Rayyan M, Schoenaers J, Dormaar JT, Breuls M, et al. (2015) Pierre Robin sequence: Management of respiratory and feeding complications during the first year of life in a tertiary referral centre. Int J Pediatr Otorhinolaryngol 79(8): 1206-1212.
- Izumi K, Konczal LL, Mitchell AL, Jones MC (2012) Underlying Genetic Diagnosis of Pierre Robin Sequence: Retrospective Chart Review at Two Children’s Hospitals and a Systematic Literature Review. J Pediatr 160(4): 645-650.e2.
- Evans AK, Rahbar R, Rogers GF, Mulliken JB, Volk MS (2006) Robin sequence: A retrospective review of 115 patients. Int J Pediatr Otorhinolaryngol 70(6): 973-980.
- Marques IL, de Sousa TV, Carneiro AF, Barbieri MA, Bettiol H, et al. (2001) Clinical experience with infants with Robin sequence: a prospective study. Cleft Palate-Craniofacial J 38(2): 171-178.
- Marques IL, Prado-Oliveira R, Leirião VHV, Jorge JC, de Souza L (2010) Clinical and Fiberoptic Endoscopic Evaluation of Swallowing in Robin Sequence Treated With Nasopharyngeal Intubation: The Importance of Feeding Facilitating Techniques. Cleft Palate Craniofac J 47(5): 523-529.
- Luis Seguias (2019) The Role of the Otolaryngologist in a Craniofacial Interdisciplinary Team. Glob J Oto 19(3): 556013.
- Witmer MT, Vasan R, Levy R, Davis J, Chan RVP (2012) Bilateral maculopathy associated with Pierre Robin sequence. J Am Assoc Pediatr Ophthalmol Strabismus 16(4): 409-410.






























