High Levels of Serum Soluble Selectin-E Is Associated with Frequency of Vaso-Occlusive Crises and Haematological Parameters in Children with Sickle Cell Anaemia
Olufemi Samuel Smith1*, Lateef Salawu2, Samuel Ademola Adegoke3, Olabamiji Abiodun Ajose4, Rahman Ayodele Bolarinwa2, Tewogbade Adedeji4, Adewuyi Temidayo Adeniyi5 and Adebayo Kazeem Oderinu1
1Department of Chemical Pathology, Obafemi Awolowo University (OAU) Teaching Hospitals Complex, Ile-Ife, Nigeria
2Department of Haematology and Immunology, Obafemi Awolowo University Ile-Ife, Nigeria
3Department of Paediatrics and Child Health, Obafemi Awolowo University Ile-Ife, Nigeria
4Department of Chemical Pathology, Obafemi Awolowo University Ile-Ife, Nigeria
5Department of Paediatrics, Ekiti state University Teaching Hospital, Ado- Ekiti
Submission:July 09, 2020; Published:March 17, 2021
*Corresponding author: Olufemi Samuel Smith (MBBS, FWACP, MSc Immunology), Department of Chemical Pathology, Obafemi Awolowo University Teaching Hospitals Complex, Ile-Ife, P.M.B. 5538, Osun State, Nigeria, Tel: +234806-167-5606
How to cite this article: Smith OS, Salawu L, Adegoke SA, Ajose OA, Bolarinwa RA, et al. High Levels of Serum Soluble Selectin-E Is Associated with Frequency of Vaso-Occlusive Crises and Haematological Parameters in Children with Sickle Cell Anaemia. Acad J Ped Neonatol. 2020; 10(1): 555835. DOI: 10.19080/AJPN.2021.10.555835
Abstract
Background: Pathophysiology of vaso-occlusive crises (VOC) includes activation and elaboration of adhesion molecules such as selectins. We therefore determined the relationship between soluble selectin-E in children with sickle cell anaemia (SCA) and frequency of VOC and hematological parameters.
Methods: It is a cross sectional, case control study. Forty-five patients each with SCA in VOC, in steady state and HbAA controls were studied. Soluble selectin-E was analysed using ELISA. Student’s t test, pearsons correlation and linear regression analysis were used in data analysis as appropriate.
Results: Children with SCA in VOC had higher (837.9±336.9ng/L) soluble selectin-E than those in steady state (605.5±257.6ng/L) and HbAA controls (406.2±142.7ng/L). Soluble selectin-E was significantly higher in children with SCA in VOC than in steady state. (t=3.678, p<0.001). Soluble selectin-E level correlated positively with frequency of VOC in SCA patients in VOC (r=0.483, p=0.001) and in steady state (r=0.396, p=0.007). It also had positive correlation with leucocyte and platelet counts in children with SCA in VOC (r=0.425, p=0.004) and (r=0.455, p=0.002) respectively; and in steady state (r=0.366, p=0.014) and (r=0.557, p=0.001) respectively. Linear logistic regression showed selectin-E as an independent predictor of frequency of VOC in SCA children who presented with VOC (OR=0.450, 95% CI=0.001- 0.004, p=0.013).
Conclusion: The study observed that soluble selectin- E is higher in patients with SCA and related to frequency of VOC and heamatological parameters.
Keywords:Selectin-E, Sickle Cell Anaemia, Vaso-occlusive crisis
Introduction
Sickle cell disease (SCD) is the general term for a group of disorders characterized by the presence of sickle haemoglobin (HbS). It is an autosomal recessive genetic disorder involving point mutation in beta globin gene chain on chromosome 11. This leads to substitution of hydrophilic glutamic acid by hydrophobic valine at the sixth position of the beta-globin chain [1]. Sickle cell anaemia (SCA), which is the occurrence of homozygous β-globin S gene mutation (HbSS), is the most common severe inherited disorder of humans and is predominant among people from Africa, Asia and Mediterranean countries [2]. Nigeria has one of the highest burdens of SCA with a prevalence of two to three percent [2].
One of the major clinical manifestations of SCA is vasoocclusive phenomena [3]. A classic view of SCA focused on the outcome of primary genetic defect with polymerisation of haemoglobin within the red cell as a major cause of vasoocclusive crises [4]. A more holistic view now sees the disease as an inflammatory disorder with increase circulation of cytokines such as IL-1 and TNF- α and subsequent activation of cells such as endothelial cells, platelets, and leucocytes, not affected by the genetic mutation [5]. The physiologic role of endothelial cells includes production of pro- and anti- coagulant, regulation of vascular pressure and blood flow. However, when bound by sickled red cell, they become abnormally activated and express adhesion molecules such as selectins and integrins which bind to sialyl- Lewis X that is expressed on neutrophils, platelets, monocytes and T lymphocyte [4]. This enhances the process of rolling and attachment of polymorphonuclear cells, particularly neutrophils, in the initial phase of inflammation that predispose to VOC [6].
Previous studies have also reported that activation of endothelial cells lead to tissue factor release, activation of coagulation cascade, formation of platelet-monocyte and plateletneutrophil aggregates during vaso-occlusive process [7]. Central to the contribution of these non-erythrocyte cells in the development of vaso-occlusive crises is the involvement of endothelial cell [6]. Adhesion molecules (selectin-E, ICAM-1, VCAM-1) are normally expressed on endothelial cells at low concentration. However, increased expression occurs during endothelial cell activation as a result of inflammation. This is accompanied by shedding of soluble adhesion molecules such as selectins from endothelial cells into the blood [8,9]. Like inflammatory cytokines, soluble adhesion molecules play important role in the recruitment of white blood cells and platelet at sites of inflammation and binding of these cells to endothelium of venules [10].
Soluble adhesion molecules now serve as markers of endothelial cell activation and dysfunction during inflammatory processes and their serum levels have been previously utilized in evaluating relationship between endothelial cell dysfunction and haemolytic rate, pulmonary hypertention and end organ dysfunction in patients with SCA [8], however to the best of authors’ knowledge, there is no study that has evaluated the relationship between soluble adhesion molecules (selectin) and frequency of vaso-occlusive crises which is the commonest clinical presentation of children with SCA in Nigeria. The main goal of this study therefore is to investigate the relationship between soluble selectin-E, and severity of disease using frequency (number) of vaso-occlusive crises, a major clinical manifestation in Nigerian children with SCA and haematological parameters.
Subjects and Methods
Study Design and population
This study was a cross-sectional comparative study. Study patients (cases) were children with SCA aged one to fifteen years and confirmed by haemoglobin electrophoresis. Forty-five children with SCA in steady state were consecutively recruited from the Paediatric SCD Clinic; while 45 SCA patients with vasoocclusive crises were recruited from the children emergency unit as they present consecutively for management. None of the participants were studied twice both in steady state and during VOC event.
For the purpose of this study, steady state was defined as period without any acute illness, pain and infection for at least 4 weeks before recruitment and no history of blood transfusion in the preceding 3 months [11].
Vaso-occlusive crises was defined as onset of pain of unknown origin that lasts at least four hours, and which requires hospital visit and therapy [12]. The frequency of vaso-occlusive crises within the period of twelve months before recruitment into the study was gotten through clinical history and confirmed from the patient’s medical records.
SCA patients on hydroxyurea, on chronic blood transfusion programme, those with recent blood transfusion in the preceding 3 months and those with chronic disease and chronic infection were excluded from the study.
Also, forty-five age- and sex- matched HbAA children confirmed by Hb electrophoresis were recruited consecutively as controls. They were age- and gender- matched apparently healthy haemoglobin AA children who attend the Children Welfare Clinic (CWC) for pre-school entry medical tests, accompanied SCA patients to the SCD clinic and children of consenting staff of the hospital.
SCA patients with PCV ≥ 30% were classified as no anaemia, those with PCV of 26-29.9% as mild anaemia, those with PCV of 20-25.9% as moderate anaemia, and those with PCV <20% as severe anaemia [13]. SCA patients with WBC count of <4 x109/L have leucopenia, those with WBC of 4-11 x109/L have normal WBC count and those with WBC >11 x109/L have leucocytosis [14]. SCA patients with platelet count < 100 x109/L are classified as having thrombocytopenia, those with platelet count of 100 - 400 x109/L as normal platelet count and those with platelet count > 400 x109/L as having thrombocytosis [14].
SCA patients were grouped into two depending on the number of pain episode in the twelve months prior to recruitment into the study; those with less than 3 episode(s) and those with 3 or more pain episodes. Children with SCA who had 3 or more pain episodes in a 12-month period were regarded as having severe disease [15].
Ethical consideration
The ethics clearance for this study was obtained from the Ethics and Research Committee of the institution in which the study was conducted (Ethics Reference Number- IRB/IEC/0004553). The participants were fully informed about the study both verbally and with the use of patient’s information sheet. Written informed consents and ascent were obtained from the parents/ caregivers and patients above age seven respectively
Data collection
Relevant demographic information was obtained from the subjects who satisfied the inclusion criteria using a pre-designed proforma and medical charts. Data on socio-demographic characteristics (age, gender and socioeconomic class), painful episodes in the previous twelve months before recruitment were obtained and verified from medical records.
Six milliliters of venous blood was collected from each participant following standard aseptic procedure of venipuncture. Out of this, 3.5 milliliters was dispensed into a plain bottle and was centrifuged at 1,500 revolutions per min for 15minutes after allowing for clot retraction. The resultant supernatant serum was separated into another plain bottle. This was stored at -70 °C for quantitative analyses of serum (soluble) selectin-E in batches using enzyme linked immunosorbent assay (ELISA) method; kit manufactured by SPAN Biotech Ltd, Unit 5, Building 3, Sunshine Aloha, Bantian Industrial Park, Banxuegang Road, Loggang Shenzhen 518129, China. The remaining 2.5 milliliters was dispensed into dipotassium Ethylenediamine tetra acetic acid (K2EDTA) anticoagulant bottle for analysis of full blood count and differentials using automated analyser (SYSMEX KX 21 Haematology Analyser) at the Institute of Human Virology Nigeria Laboratory within the study centre.
Statistical analysis of data
Data collected was analyzed using descriptive and inferential statistics contained in the Statistical Package for the Social Sciences (SPSS), version 20.0 (SPSS Chicago Inc., IL, U.S.A). Means and standard deviations were compared using the independent student’s t-test and analysis of variance (ANOVA). The degree of correlation of continuous data was determined by Pearson’s correlation analysis. Linear regression analysis was carried out to examine the independent effect of soluble selectin-E, age, gender, and hematological variables on frequency of VOC. Statistical significance was established when “p” value is less than 0.05.
Results
General and Demographic characteristics of study population
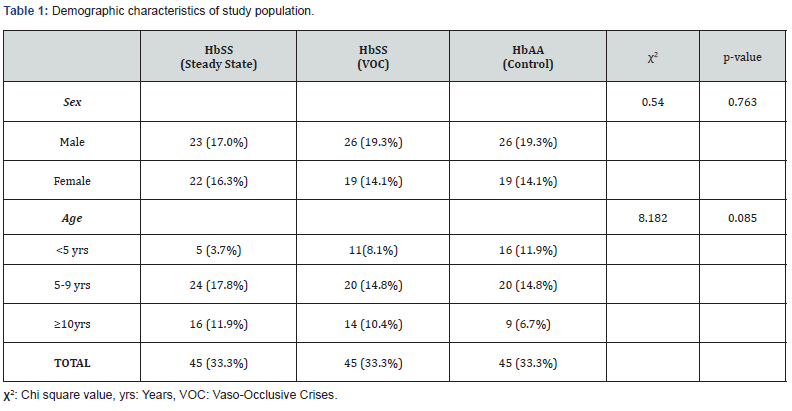
Table1 shows the sex and age distribution of the study population. A total of 135 SCA children between the age of 1 and 15 years were recruited into the study. They included 45 in vasoocclusive crises, 45 in steady state and 45 age- and sex-matched apparently healthy haemoglobin AA controls. Overall, there were 75 (55.6%) males and 60 (44.4%) females. No significant difference exists in the sex distribution within the three groups (χ2=0.54, p=0.763). The mean age of the children with SCA in vasoocclusive crises was 7.69±3.92 years, while that of children with SCA in steady state was 8.18±3.32 years and that controls was 6.60±3.14 years. There was no significant difference in the means of the ages in the three groups (F=2.426, p=0.092). The patients were grouped into three age groups: 1-4 years, 5-9 years, and 10-15 years. Majority of the study participants were between the ages of 5 years and 9 years (47.7%) while 23.7% and 28.9% were under-fives and adolescent, respectively. There was no significant difference in the age group distributions (χ2=8.182, p=0.085).
The mean serum selectin-E and haematological parameters in the study participants
The serum soluble Selectin-E ranged from 398.9 ng/L to 1664.4 ng/L in children with SCA in vaso-occlusive crises, 230.2 ng/L to 1337.0 ng/L in children with SCA in steady state and 102.5ng/L to 836.0 ng/L in controls. Using independent student t- test, the mean serum Selectin-E concentration was significantly higher in children with SCA in VOC when compared to that of children with SCA in steady state alone with p value also <0.001 (Table 2).
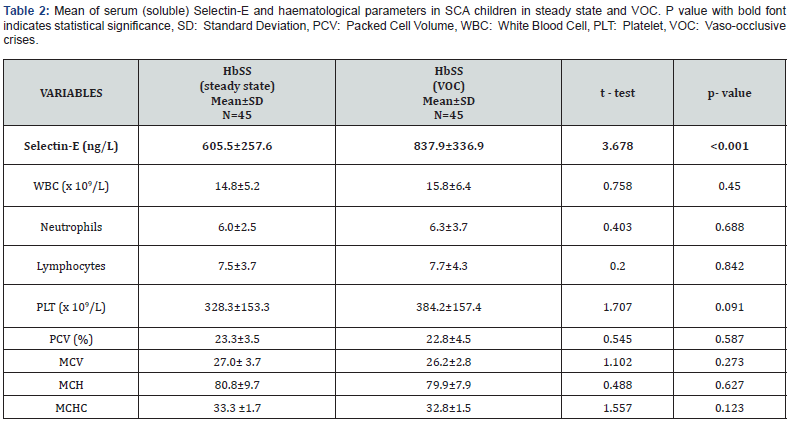
The haematocrit level is lowest in children with SCA in VOC (22.8±4.5%) and highest in healthy controls (36.5±2.7%); There was no significant difference in the haematocrit level in children with SCA in VOC (22.8±4.5%) when compared to those in steady state (23.3±3.5%). (t=0.545, p=0.587) (Table 2). On the contrary, the platelet count was highest in children with SCA in VOC 384.2±157.4 x 109/L and lowest in controls (245.9±73.6 x 109/L); Also, the total WBC was highest in children with SCA in VOC 15.8±6.4 x 109/L and lowest in controls (6.0±1.8 x 109/L). The platelet count and total white cell count were statistically similar between children with SCA in VOC and those in steady state; (t=1.707, p=0.091), (t=0.758, p=0.450) respectively (Table 2). The other haematological parameters are shown in Table 2.
Relationship between serum soluble Selectin-E and frequency of vaso-occlusive crises
In children with SCA in VOC, 35 (77.8%) had <3 annual pain episode(s) while 10 (22.2%) had 3 or more pain episodes. Also, in children with SCA in steady state, 31(68.9%) %) had <3 annual pain episode(s) and 14 (31.1%) had 3 or more pain episodes.
In patients with SCA in VOC, the mean soluble selectin-E was significantly higher in patients who had 3 or more pain episodes in 12 months preceding recruitment into the study (1050.0±447.9 ng/L) when compared to those with <3 pain episodes (775.9±309.3ng/L) (t=2.374, p=0.022) (Table 3). This is similar in patients with SCA in steady state, with the mean soluble selectin-E being significantly higher in patients who had 3 or more pain episodes in 12 months preceding recruitment into the study (777.3±277.0 ng/L) when compared to those with <3 pain episodes (544.6±204.8ng/L); (t=2.977, p=0.005) (Table 3).
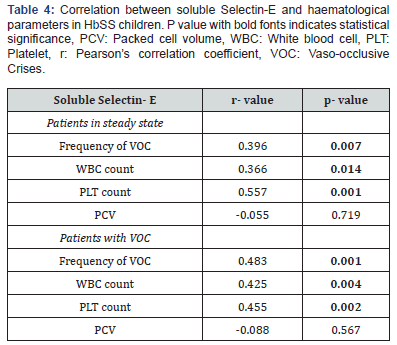
In addition, there was significant positive correlation between serum soluble Selectin-E concentration and the number of VOC the patients had 12 months preceding recruitment for SCA patients in vaso-occlusive crises (r=0.483, p=0.001) and those in steady state (r=0.396, p=0.007) (Table 4).
Relationship between serum soluble Selectin-E and haematological parameters
Twenty-four (26.7%) children with SCA had WBC counts within normal values while 66 (73.3%) had leukocytosis. None of them had leukocytopenia. In children with SCA in VOC, the serum concentration of soluble Selectin-E in patients with leucocytosis (892.1±355.8 ng/L) was significantly higher compared to patients with normal WBC count (648.4±158.5 ng/L) (t=2.093, p=0.042). Also, in children with SCA in steady state, the serum concentration of soluble Selectin-E in those with leukocytosis (683.7±263.2 ng/L) was significantly higher compared to those with normal WBC count (467.9±194.1 ng/L) (t=2.742, p=0.009) Table 3.
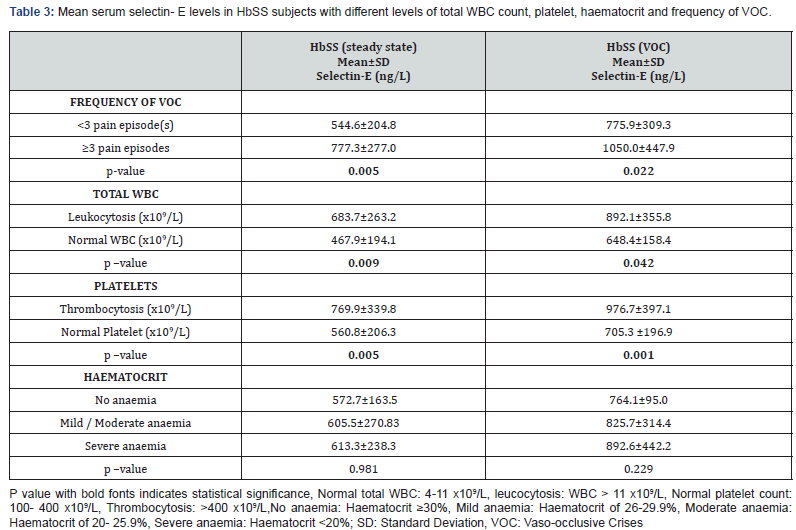
In all SCA participants, 56 (62.2%) had platelet counts within normal values, while 34 (37.8%) had thrombocytosis. None of the patients with SCA had thrombocytopenia. In SCA patients in vasoocclusive crises, the serum soluble Selectin-E concentration in patient with thrombocytosis (976.7±397.1 ng/L) was significantly higher when compared to those with normal platelet count (705.3±196.9 ng/L) (t=2.885, p=0.001). Also, in SCA patients in steady state, the serum soluble Selectin-E concentration in patient with thrombocytosis (769.9±339.8 ng/L) was significantly higher when compared to those with normal platelet count (560.8±206.3 ng/L) (t=2.002, p=0.005) (Table 3).
In all SCA participants, 5 (5.6%) had no anaemia, 66 (73.33%) had mild/ moderate anaemia and 19 (21.11%) had severe anaemia. The serum levels of soluble selectin E in patients with different groups of haematocrit (No anaemia, mild/ moderate anaemia and severe anaemia) was similar in children with SCA in steady state (F=0.019, p=0.981) and in those in VOC (F=0.769, p=0.229) (Table 3).
The serum soluble Selectin-E concentration had significant positive correlation with total WBC count in children with SCA in VOC (r=0.425, p=0.004) and in children with SCA in steady state
(r=0.366, p=0.014). Also, there was significant positive correlation between serum soluble Selectin-E concentration and platelet counts in children with SCA in VOC (r=0.455, p=0.002) and in children with SCA in steady state (r=0.557, p=0.001). However, there was no correlation between serum soluble Selectin-E concentration and haematocrit in SCA children in VOC (r=-0.088, p=0.567) and in SCA children in steady state (r=-0.055, p=0.719) (Table 4) (Figure 1-2).
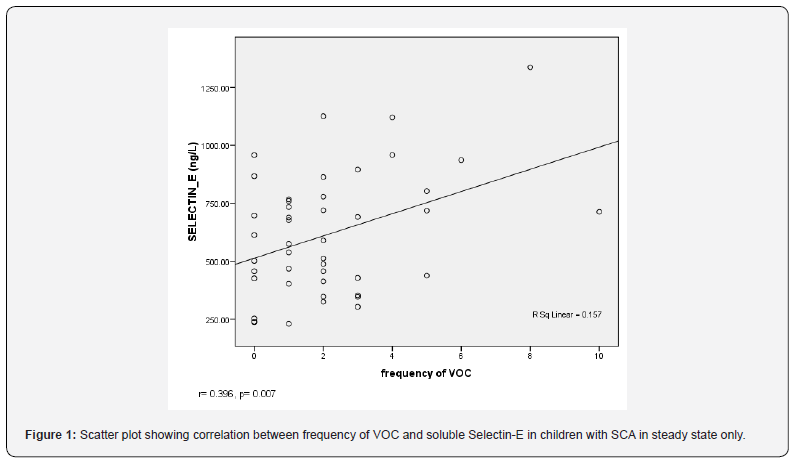
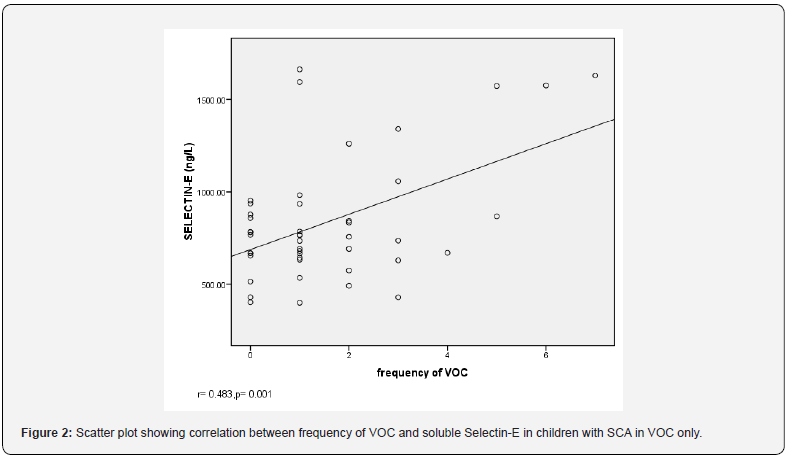
Linear regression analysis
Linear regression analysis was done to examine the independent effect of soluble selectin, age, gender, hematological variables on frequency of VOC. In this regression model, frequency of VOC was taken as the outcome variable while age, gender, soluble selectin, PCV, total WBC and platelet count were taken as independent or predictive variables. In children with SCA in VOC, only selectin-E was found to be an independent predictor of frequency of VOC (OR=0.450, 95% CI=0.001-0.004, p=0.013). Other variables tested in the model were not found to predict frequency of VOC (Age, OR -0.055, 95% CI -0.144- 0.096, p=0.689; gender, OR=0.212, 95% CI=-0.271- 1.716, P=0.149; platelet count, OR=-0.024, 95% CI=-0.004- 0.003, p=0.880; total WBC count, OR=0.114, 95% CI=-0.063- 0.124, p=0.512; PCV, OR=0.107, 95% CI=-0.077- 0.164, p=0.471).
Discussion
The pathophysiologic mechanism involved in the clinical manifestations of sickle cell disease (SCD) is complex and multi-factorial. Endothelial cell activation is reported to have a central role to play during vaso-occlusive crises [4,16]. Although studies have been conducted on the relationship of biomarkers of endothelial cell activation with pulmonary hypertension renal insufficiency, and end organ dysfunction [8], to the best of the authors’ knowledge, there are very few studies on the association between soluble selectin-E (which is a proven marker of endothelial cell activation) and VOC as well as hematological parameters (viz-a-viz haematocrit level, leucocyte and platelet count). This study was, therefore, conducted to demonstrate if any relationship exists between one of the products of endothelial cell activation (Selectin-E) and disease severity in children with sickle cell anaemia (SCA) using frequency of vaso-occlusive crises and haematologic profile.
In this study, serum soluble Selectin-E was observed to be significantly higher in SCA patients with VOC compared to patients in steady state. This may suggest that though both group of patients have ongoing inflammatory processes, expectedly, it is more severe in patients with VOC than in those in steady state. The concentration of serum soluble Selectin-E in patients with SCA in steady state was also observed to be significantly higher than in their apparently healthy age- and sex-matched controls. Kato et al. [8] in 2015 reported that patients with SCA had higher serum selectin-E compared to HbAA controls [8]. Al Najjar et al in 2017 who studied markers of endothelial dysfunction and leucocyte activation in Saudi and non-Saudi haplotypes of SCD in 2017 reported that patients with SCA in acute vaso-occlusive crises had higher serum level of selectin-E compared to those in steady state [17]. Sickle cell disease is a well-recognized state of chronic inflammation with increased production of cytokines such as IL- 1β, IL-6, IL-17 and TNF-α [18]. These pro-inflammatory cytokines enhance endothelial cell activation and release of Selectin-E [19].
As observed from this study, other workers have also found that children with SCA have higher total white cell count even in the absence of bacterial infection [20]. This has been associated with more severe disease manifestations such as cerebrovascular accident, acute chest syndrome and increase mortality [21-23]. Aside from increase in white cell count, there is associated slow rolling, intra-luminal crawling, paracellular and transcellular migration and increase adhesion of these leucocytes, most especially granulocytes, monocytes and memory CD4+ subpopulation of T-cells, to endothelial cells. These processes are enhanced by both circulating and endothelial selectin-E and they play a direct role in the pathogenesis of sickle cell vaso-occlusion [24-26]. Previous studies have reported that this is due to increase release of circulating Selectin-E which sends activating signals that cause up-regulation of β-2 integrin and MAC-1 in the inflamed vessel [27]. This therefore suggests that selectin-E may contribute to blockage of blood vessels by leucocytes thereby predisposing these patients to VOC.
In this present study, we observed that the mean serum (soluble) selectin-E in sickle cell anaemia patients with thrombocytosis was significantly higher compared to those with normal platelet count. Also, there was a positive significant correlation between serum soluble selectin-E and platelet count. Increase platelet count, platelet activation and hypercoagulability contribute to pathophysiology of SCA and activated platelets are involved in inducing the expression of major endothelial cell adhesion molecules such as ICAM-1 and selectin-E [28]. Studies by Proenca-Ferreira et al have demonstrated adherence of activated platelet to endothelial cells and stimulation of leukocyteendothelial cell interaction during inflammation [29]. This cooperation between endothelial cells, leucocytes, and platelets under the influence of selectin-E may contribute to severity of disease in patients with SCA.
Interestingly in this present study, there was no significant difference in the mean serum soluble Selectin-E concentration in the four classes of haematocrit level and there was no significant correlation between serum soluble Selectin-E and haematocrit level. Hidalgo et al. [27] suggested that selectin-E participates in generating a secondary wave of activating signals leading to polarised expression of activated CD11a/ CD18, also called MAC-1, that mediates the capture and adhesion of sickled erythrocytes on vessel wall [27]. Previous studies also showed unique endothelial expression of Fc receptor for the Fc portion of immunoglobulin coated sickled red cell and increased interaction between subendothelial matrix laminin and red blood cell CD44+ [30]. Furthermore, there exist a strong correlation between red cell adhesion to endothelial cell and clinical severity [31]. However, this current study did not find any association between serum soluble Selectin-E and haematocrit. Further molecular studies in Nigerian children are needed to elucidate these contradictory findings.
Acute painful episodes are one of the major clinical event in patients with SCA. They are predictor of early death and measure of disease severity [32]. The rate of painful episodes varies widely among patients and those with 3 or more painful episodes within 12 months period have been classified to have severe disease [15]. This study founds its uniqueness in determining the association/ relationship between soluble selectin-E and one of the markers of disease severity, frequency of VOC. The study observed that patients with 3 or more pain episodes in the last 12 months preceding recruitment into the study had significantly higher levels of soluble sectin-E compared to those with <3 pain episodes. Also, it was observed that the number of vaso-occlusive crises episodes in children with SCA over the 12 months preceding recruitment into the study correlated positively with serum soluble Selectin-E level. Furthermore, in this present study, soluble selectin-E was seen to be an independent predictor of frequency of pain episodes in children with SCA. This suggests that soluble selectin-E is related to increased frequency of VOC in these patients. Previous studies have shown that selectin-E is central to activation of factors such as recruitment and adhesion of activated leucocyte, vascular clogging by heterotypic cell- cell aggregate composed of sickled red cell and platelet proliferation and activation that initiate the development of vaso-occlusive crises [33]. Kato et al. [8] in 2005 reported that there is increased expression of selectin-E on endothelial cell and its release into the circulation were associated with pulmonary hypertension, multiorgan dysfunction and increase risk of mortality in patients with SCA [8]. Charles et al. [10] in 2018 also reported an increase in soluble endothelial molecules in patients presenting with VOC. Kuryliszyn et al. [34] 2005 observed that soluble selectin-E was increased in patients with systemic sclerosis. They also reported that organ systemic involvement in patients with systemic sclerosis is associated with an altered function of endothelial cells [34]. One of the biochemical factors that facilitate blood flow and maintains homeostasis is suppression of circulating endothelial molecules by nitric oxide (NO) [35]. The increase in soluble selectin-E may therefore be attributed to decrease in NO precursors and impaired bioavailability of NO [10].
The association of elevated soluble selectin E with increased frequency of VOC, leucocytosis and thrombocytosis seen in this study shows the part soluble selectin E plays in disease severity in patients with SCA, since both leucocytosis and thrombocytosis exist in most patients in vaso-occlusive crises and are also risk factors for developing VOC [7].
This study was limited by the fact that it is a cross sectional study. A longitudinal study that compares the level of selectin-E in the same patient when present in vaso-occlusive crises and when they are in steady state might be more revealing. Also, this study is subject to recall bias, especially on information on frequency of significant pain episodes.
In conclusion, this study has shown that soluble Selectin-E is elevated in patients with SCA compared to non-SCA individuals. It has also showed that children with SCA in VOC had higher soluble Selectin-E compared to children with SCA in steady state. It also showed the relationship between Selectin-E, haematological parameter (WBC count and platelet count) and VOC with elevated plasma selectin-E levels being associated with worsening clinical manifestations such as increased frequency of vaso-occlusive crises, leukocytosis and thrombocytosis in patients with SCA. These findings suggest that the level of soluble selectin-E, a biomarker of endothelial cell dysfunction, may be used as a predictor of frequency of vaso-oclusive crises in children with SCA and its routine assay may be advised.
Also, in developing countries like Nigeria, several treatment options such as the use of analgesics, antioxidant supplements, anti-sickling agents and hydroxyurea exist to reduce frequency of vaso-occlusive crises, more research on the effect of selectin inhibitors on frequency of VOC in patients with SCA with the hope of instituting them in the routine management of these patients is therefore desirable to further improve the clinical profile and well-being of these patients.
References
- Hoffbrand AV (2007) Genetic disorders of haemoglobin. In: Hoffbrand AV, Moss PAH (Eds) Essential hematology. (6th edn), wiley-blackwell, Oxford, Pp. 89-107.
- Sickle Cell Anaemia (2006) World Health Organisation.
- Quinn CT (2013) Sickle Cell Disease in Childhood: From Newborn Screening Through Transition to Adult Medical Care. Pediatr Clin North Am 60(6): 1363-1381.
- Frenette PS, Atweh GF (2007) Sickle cell disease: old discoveries,new concepts, and future promise. J Clin Invest 117(4): 850-858.
- Mohan JS, Lip GYH, Wright J, Bareford D, Blann AD (2005) Plasma levels of tissue factor and soluble E-selectin in sickle cell disease: relationship to genotype and to inflammation. Blood Coagulation & Fibrinolysis 16(3): 209-214.
- Hebbel RP, Osarogiagbon R, Kaul D (2004) The endothelial biology of sickle cell disease: Inflammation and a chronic vasculopathy. Microcirculation 11(2): 129-151.
- Manwani D, Frenette PS (2013) Vaso-occlusion in sickle cell disease: pathophysiology and novel targeted therapies. Blood 122(24): 3892-3899.
- Kato GJ, Martyr S, Blackwelder WC, Nichols JS, Coles WA, et al. (2005) Levels of soluble endothelium-derived adhesion molecules in patients with sickle cell disease are associated with pulmonary hypertension, organ dysfunction, and mortality. Br J Haematol 130(6): 943-953.
- Anna S, Yi L, Paul B, Stephana C, Elizabeth W, et al. (1997) Circulating activated endothelial cells in sickle cell anaemia. N Engl J Med 337(22): 1584-1590.
- Charles A, Eric SD, Fredericka S, Bartholomew D, Gifty BD, et al. (2018) Levels of Soluble Endothelium Adhesion Molecules and Complications among Sickle Cell Disease Patients in Ghana. Diseases 6(29): 1-7.
- Juwah AI, Nlemadim EU, Kaine W (2004) Types of anaemic crises in paediatric patients with sickle cell anaemia seen in Enugu, Nigeria. Arch Dis Child 89(6): 572-576.
- Ballas SK, Lieff S, Benjamin LJ, Dampier CD, Heeney MM, et al. (2010) Definitions of the phenotypic manifestations of sickle cell disease. Am J Hematol 85(1): 6-13.
- De Baun MR, Vichinsky E (2007) Nelson textbook of paediatrics. (18th edn), Saunders Elsevier, Philadelphia, USA.
- Ezeilo G (1981) White blood cell count in healthy Africa. Nig Med Pract 2: 73-78.
- Adegoke SA, Kuti BP (2012) Evaluation of clinical severity of sickle cell anaemia in Nigeria children. J Appl Hematol 4(2): 58-64.
- Kaul DK, Finnegan E, Barabino GA (2009) Sickle red cell-endothelium interactions. Microcirculation 16(1): 97-111.
- Al Najjar S, Adam S, Ahmed N, Qari M (2017) Markers of endothelial dysfunction and leucocyte activation in Saudi and non-Saudi haplotypes of sickle cell disease Ann Hematol 96(1): 141-146.
- Adegoke SA, Smith OS, Adekile AD, Figueiredo MS (2017) Relationship between serum 25-hydroxyvitamin D and inflammatory cytokines in paediatric sickle cell disease. Cytokine 96: 87-93.
- Zhang J, Defelice A, Hang JP, Colatsky T (2010) Surrogate Markers for Drug-induced Vascular Injury. Toxicol Pathol 38(6): 856-871.
- Akinola NO, Stevens SME, Franklin IM, Mash GB, Stuart J (1992) Subclinical ischaemic episodes during the steady state of sickle cell disease. J Clin Patho 45(10): 902-906.
- Platt OS, Brambilla DJ, Rosse WF, Milner PF, Castro O (1994) Mortality in sickle cell disease. Life expectancy and risk factors for early death. N Engl J Med 330(23): 1639-1644.
- Vichinsky EP, Styles LA, Colangelo LH, Wright EC, Castro O, et al. (1997) Acute chest syndrome in sickle cell disease: clinical presentation and course. Cooperative Study of Sickle Cell Disease. Blood 89(5): 1787-1792.
- Wun T (2001) The Role of Inflammation and Leukocytes in the Pathogenesis of Sickle Cell Disease; Haemoglobinopathy. Haematology 5(5): 403-412.
- Ley K, Laudanna C, Cybulsky MI, Nourshargh S (2007) Getting to the site of inflammation: the leukocyte adhesion cascade updated. Nat Rev Immunol 7(9): 678-689.
- Ates A, Kinikli G, Turgay M, Duman M (2004) Serum-Soluble Selectin Levels in Patients with Rheumatoid Arthritis and Systemic Sclerosis. Scandenavian J of Immun 59(3): 315-320.
- Turhan A, Weiss LA, Mohandas N, Coller BS, Frenette PS (2002) Primary role for adherent leukocytes in sickle cell vascular occlusion: a new paradigm. Proc Natl Acad Sci U S A 99(5): 3047-3051.
- Hidalgo A, Chang J, Jang JE, Peired AJ, Chiang EY, Frenette PS (2009) Heterotypic interactions enabled by polarized neutrophil microdomains mediate thromboinflammatory injury. Nat Med 15(4): 384-391.
- Conran N, Franco-Penteado CF, Costa FF (2009) Newer aspects of the pathophysiology of sickle cell disease vaso-occlusion. Hemoglobin 33(1): 1-16.
- Proenca Ferreira A, Brugnerotto A, Garrido VT, Dominical VM, Vital DM, et al. (2014) Endothelial Activation by Platelets from Sickle Cell Anemia Patients. PLoS One 9(2): e89012.
- Joneckis CC, Ackley RL, Orringer EP, Wayner EA, Parise LV (1993) Integrins and glycoprotein IV (CD36) are expressed on circulating reticulocytes in sickle cell anemia. Blood 82(12): 3548-3555.
- Hebbel RP, Eaton JW, Steinberg MH (1980) Erythrocyte adherence to endothelium in sickle-cell anemia: a possible determinant of disease severity. N Engl J Med 302(18): 992-995.
- Steinberg MH (2005) Preicting clincal severity in sickle cell anaemia. Br J Heamatology 129(4): 465-486.
- Frenette PS, Manwami D (2013) Vaso-occlusion in sickle cell disease: pathophysiology and novel targeted therapies. Hematology Am Soc Hematol Educ Program 2013: 362-369.
- Kuryliszyn A, Moskal P, Klimiuk A, Sierakowski S (2005) Soluble adhesion molecules (sVCAM-1, sE-Selectin), vascular endothelial growth factor (VEGF) and endothelin-1 in patients with systemic sclerosis: Relationship to organ systemic involvement. Clin Rheumatol 24(2): 111-116.
- De Caterina R, Libby P, Peng HB, Thannickal VJ, Rajavashisth TB, et al. (1995) Nitric oxide decreases cytokine-induced endothelial activation. Nitric oxide selectively reduces endothelial expression of adhesion molecules and proinflammatory cytokines. J Clin Invest 96(1): 60-68.






























