The Effect of Antiviral Therapy for Congenital Cytomegalovirus (CMV) on Children Hearing Loss
Jong woo chung1, Yehree KTM1, Lojain Alsiwat2*, Latefa Alareek2, Suzan Alshehri2
1Department of Otorhinolaryngology, College of Medicine, University of Ulsan, Asan Medical City Seoul, Korea
2 College of Medicine, King Saud University, King Saud University Medical City, Saudi Arabia.
Submission:April 02, 2020; Published:May 04, 2020
*Corresponding author: Lojain Alsiwat, College of Medicine, King Saud University, King Saud University Medical City, PO Box 242069, Riyadh 11322, Saudi Arabia
How to cite this article: Jong Woo Chung*, Yehree KTM, Lojain Alsiwat, Latefa Alareek, Suzan Alshehri. The Effect of Antiviral Therapy for Congenital Cytomegalovirus (CMV) on Children Hearing Loss. Acad J Ped Neonatol. 2020; 9(4): 555825. DOI: 10.19080/AJPN.2020.09.555825
Abstract
Introduction: Congenital Cytomegalovirus (CMV) is the most common viral pathogen encountered in newborns and the most prevalent congenital infection in human beings.
Objective: To evaluate existing studies on the effect of antiviral treatment on long- term hearing loss in infants with congenital Cytomegalovirus infection and how it can aid in the prevention of such devastating outcomes.
Methods: Systematic review was performed by obtaining papers using a four-step procedure to ensure that all included studies meet the desired criterion. Additionally, PI was used to test the effect of antiviral treatment on hearing loss.
Results: The average PI among studies used was 0.4 indicating that the use of antiviral treatment improved hearing loss with an average of 40%. Also, Cochran’s Q test was insignificant which indicates homogeneity between studies.
Conclusion: CMV infections are the most important cause of nongenetic hearing loss, neurodevelopmental delay, intellectual impairment, and multisystem organ failure in children. The reviewed studies showed that the antiviral treatment (ganciclovir) could be used as a treatment for patients including children and infants who are diagnosed with hearing loss due to CMV infection.
Keywords: Congenital Cytomegalovirus; CMV; Hearing loss; Antiviral therapy; Systematic review
Introduction
Congenital Cytomegalovirus (CMV) is the most common viral pathogen encountered in newborns and the most prevalent congenital infection in human beings. It is a Herpes virus that has been documented to be linked to sensorineural hearing loss (SNHL) as it appears in 30-65% of children infected [1,2]. In fact, it has been documented as the leading cause of non-genetic neurosensory hearing loss in developed countries [3]. The pathophysiology of the congenital CMV-related SNHL is not fully understood. There suggested direct cytopathic effects and a localized inflammatory response of the stria vascularis [4]. Also, local reactivation of the virus in this site may explain the late nature of hearing deterioration. CMV infection manifests no significant clinical findings at birth, as 90% of infants will be asymptomatic at that time [5-7]. Approximately 33% to 50% of SNHL due to congenital CMV infection is late-onset hearing loss. What makes SNHL in these patients quite challenging to diagnose is the fluctuating and progressive nature, for it can present as unilateral high-frequency losses or profound bilateral losses. Not only can the clinical features vary, but also the time of presentation. It has been documented that the majority of children develop late-onset loss during the preschool and early school years. Although the disease often tends to leave an irreversible injury, antiviral therapy of newborns with CMV infection has been an option available to clinicians to reduce the impact of the disease’s sequels or even help to prevent the deterioration when started early on. The suggestion that antiviral therapy might be useful in newborns with congenital CMV infection first was raised in numerous reports in the late 1960s and early 1970s [8-11]. Additionally, in 1980, ganciclovir was first introduced for the treatment of congenital CMV infection and it has proved to be safe, effective, and well-tolerated [12- 20]. There are Many antiviral medications such as ganciclovir and its prodrug, valganciclovir, foscarnet, and cidofovir. However, controlled clinical studies suggested that ganciclovir therapy was able to limit the neurodevelopmental manifestations, including SNHL. Also, studies showed that antiviral agents could help limit the deterioration in hearing loss in children with congenital CMV infection; in fact, it has been reported in the literature to be linked to improvement in audiometric studies. The effect of treatment with antiviral therapy such as ganciclovir/valganciclovir may be related to a decrease in the CMV viral load [21-23]. Treatment of the reactivated virus may halt viral replication in the inner ear which would otherwise lead to irreversible damage. Based on such encouraging findings, ganciclovir/valganciclovir has been established as the treatment of infants with symptomatic congenital CMV infection [24,25]. Furthermore, several studies document the possibility of prevention of hearing deterioration with ganciclovir [26] and improvement of other neurological symptoms [27,28]. It was also showed that the ganciclovir/ valganciclovir treatment beyond the neonatal period mighty be beneficial in preventing further deterioration of inner ear damage in children with late-onset hearing loss of congenital CMV infection. Congenital infection caused by human Cytomegalovirus (CMV) is a common occurrence, but its significance is underappreciated. It has conferred a substantial medical and economic burden on society. In recent years, an awareness of its impact in newborns, which includes sensorineural hearing loss (SNHL), has increased. Therefore, we believe it is essential to inform possible approaches to prevent complications. The SNHL remains a catastrophic complication of the disease. Nonetheless, data on the use of antiviral agents for the prevention of SNHL in children with congenital CMV remain relatively insufficient, because of the lack of prospective data. It is still vague to some clinicians whether it will provide long-term benefit for congenital or perinatal acquired CMV infection and its squeal such as SNHL which we intend to focus on in our paper. So far, many studies have found that patients with congenital CMV benefit from antiviral treatment. They reported a delay and limitation go the worsening of their hearing loss and the data available served as observational evidence of the benefits of antiviral treatment in these children [29]. We collected and evaluated existing data on the effect of antiviral therapy on long term hearing loss on infants with congenital Cytomegalovirus infection and how it can aid in the prevention of such devastating outcomes [30].
Methods
Literature search
We obtained papers for this review using a four-step procedure. First, we performed a search of the peer-reviewed literature using PubMed and identified potential studies for inclusion using the following keywords: “antiviral therapy” and “CMV” and “hearing loss”. Second, we analyzed abstracts for all studies identified and excluded papers that did not satisfy selection criteria. Third, we analyzed the full-text version of all remaining studies and excluded those that did not meet selection criteria. Finally, we started reviewing the results. The PRISMA flow diagram showed in Figure 1 below shows the procedure followed to identify studies used in this research.
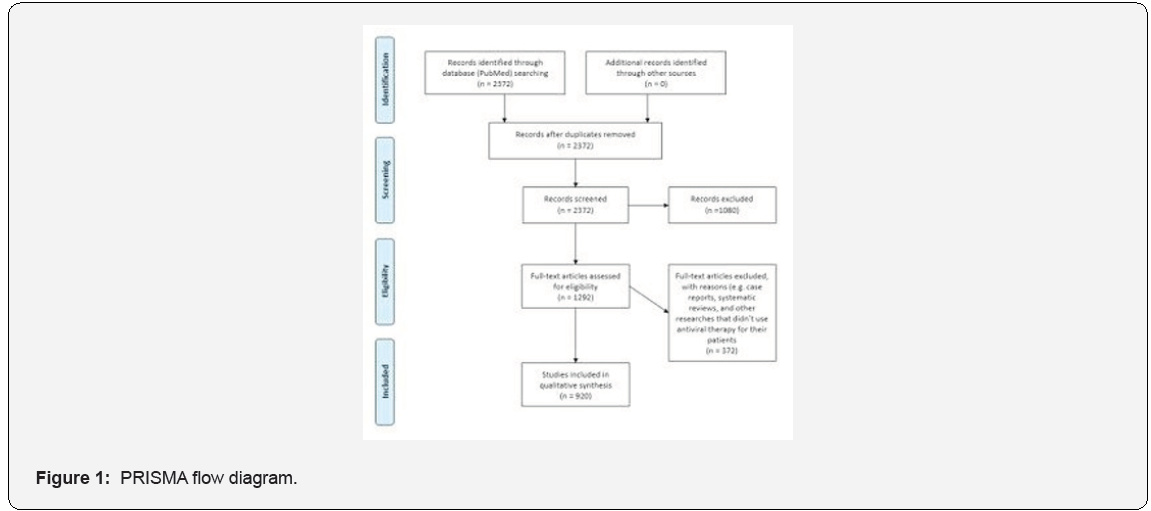
Selection criteria
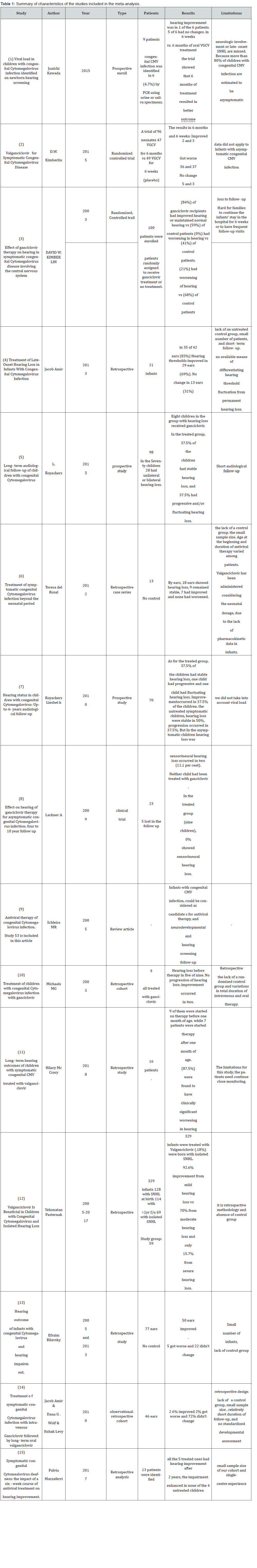
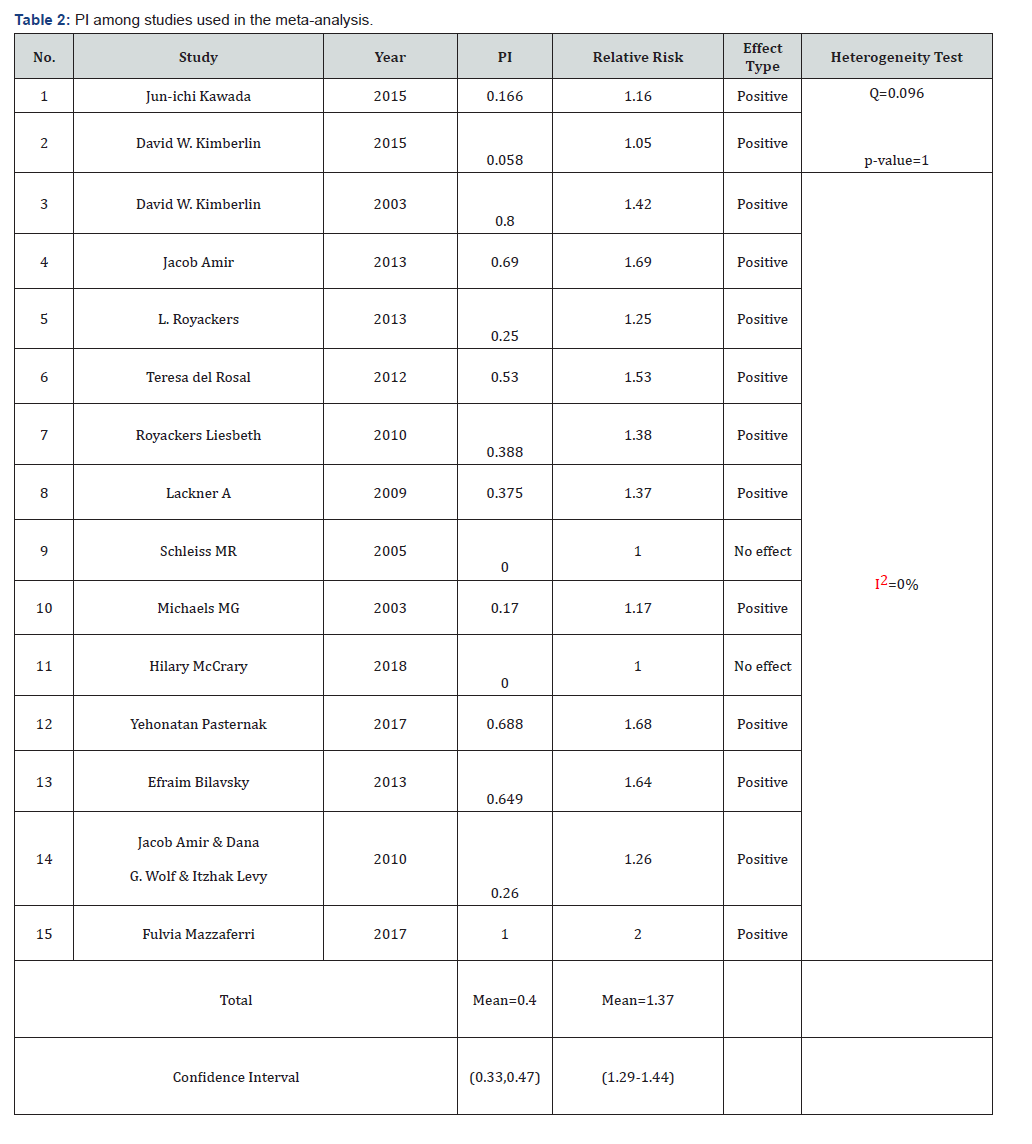
We collected 52 published papers. We excluded case reports, systematic reviews, and other researches that didn’t use antiviral therapy for their patients. We reviewed fifteen original research papers from 2003-2018. Table 1 shows the analyzed data of the papers.
Data extraction
The data were extracted from eligible studies by the investigators found through the PubMed database.
Statistical methods
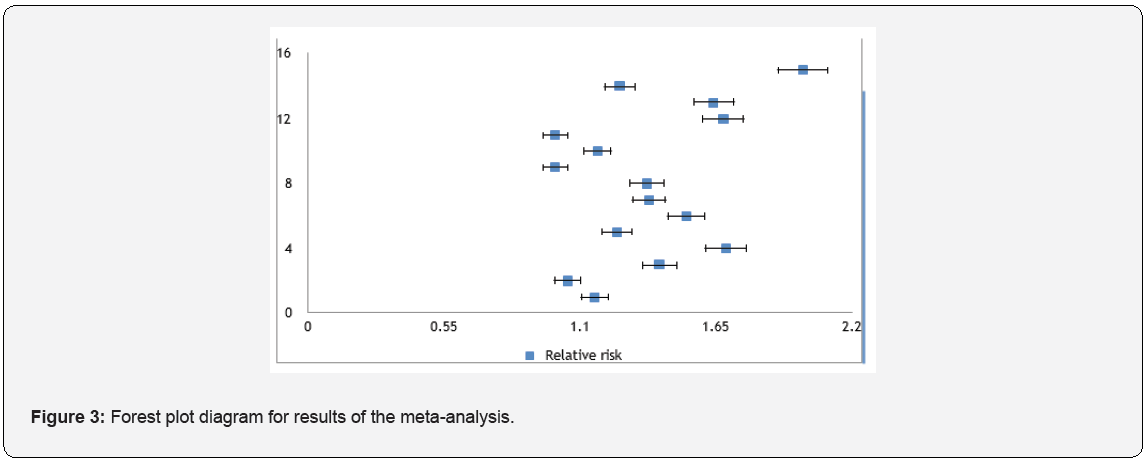
The percent of improvement (PI) expresses the effect-size in each study. PI represents the number of improved cases relative to the sample size. PI was used to test the effect of antiviral treatment on hearing loss. If PI=0, then there is no effect which means there is no improvement. If PI=1, then all cases improved. In general, the larger the PI value, the greater the effect.
Heterogeneity
In order to test the homogeneity between studies included in the meta-analysis, Cochran’s Q is used. The Cochran’s Q is computed by summing the squared deviations of each study’s estimate from the overall meta-analytic estimate using the following formula:
I 2 = 100% × (Q − d f )/Q,
where d f = k −1 and k is the number of studies included in the analysis. I2 lies between 0% and 100%, where a value of 0% indicates no heterogeneity and larger values show increasing heterogeneity. (Table 1)
Results
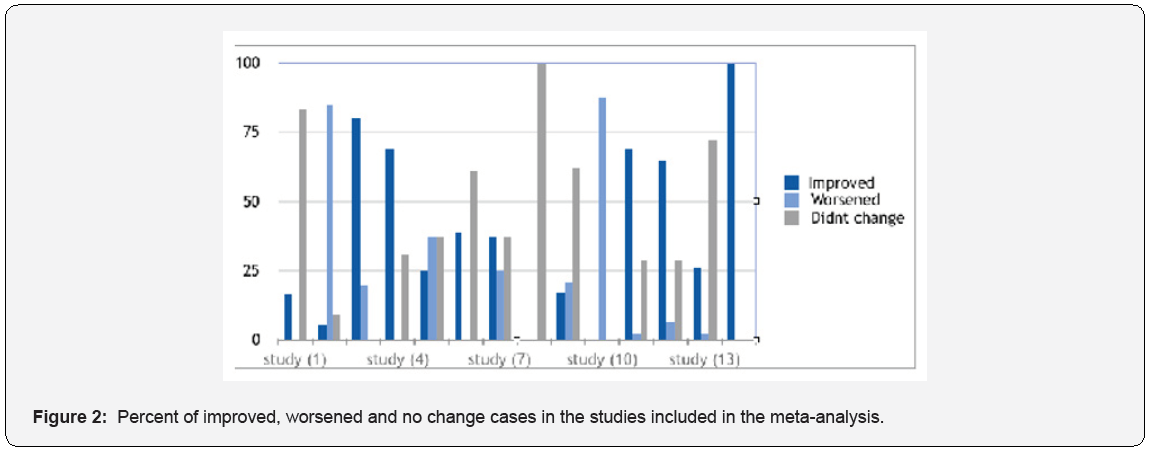
The 15 research papers from 2003-2018 summarized in Table 1 include 3 prospective studies, 3 randomized control trials, 8 retrospective studies, and 1 review article. We categorized the results after the antiviral therapy to improved, didn’t improve, no change as shown in Figure 2. In article number (1) only 6 patients were observed, 1 had an improvement after the therapy and 5 had no change. Whereas in article (2), a total of 96 neonates 47 VGCV for 6 months 2 had an improvement, 36 had worsening hearing loss, and 5 had no change versus 49 VGCV for 6 weeks (placebo) where 3 had an improvement, 37 had worsening hearing loss, and 3 had no change. In article (3) 25 received the treatment 20 had an improvement, 5 had no change after one year follow up. In another study 29 out of 42 ears had an improvement, 13 ears had no change. In study number (5), 2 had an improvement, 3 had worsening, and 3 had no change. In the other study that was done in 2012, 7 out of 26 ears had an improvement, 18 had worsening hearing loss and 9 ears had no change. In article number (7), 8 patients received the treatment 3 had and improvement, 2 had worsening hearing loss and 3 had no change in the hearing. Whereas in study number (8), all of the 10 patients who received the treatments had no change in the hearing loss. In study number (9), 4 out of 24 had an improvement in the hearing loss, 5 had deteriorated and 17 had no change. 9 patients in study (10) received the treatment 2 had an improvement and the rest had no change in the hearing loss. Surprisingly in study number (11), 87.5% of the patients who received the treatments had worsening in the hearing. In another study were 80 affected ears at baseline, (68.8%) improved and (2.5%) deteriorated, both from moderate-to-severe hearing loss. In another, out of the 77 ears which received treatment, 50 ears improved, 5 worsened, and 22 did not change. In another study, out of 46 ears 26 % improved, 2% worsened and 72% did not change. In the last study, all the 5 treated ones achieved significant hearing improvement after 2 years, whereas the impairment enhanced in none of the 4 untreated children. A summary of the results of the meta- analysis for the included studies are shown in Table 2 shown below. The average PI among studies used in the meta-analysis was 0.4 or 40%, which means that that use of antiviral treatment improved hearing loss with an average percent of 40%. Two studies resulted in no effect while 13 studies resulted in positive effect. The Cochran’s Q test was insignificant which indicates homogeneity between studies. Also, I 2 index was % indicating that there is no observed heterogeneity between studies. (Figure 2 & 3). Shows that in most studies included, the percent of improvement was moderate or less than average and five studies only showed high percent of improvement.
Discussion
Ganciclovir is known to be one of the most common treatments of hearing loss due to CMV. The results showed high percentages of positive effect of ganciclovir in improving hearing loss in children and infants, especially in the long term. 85% of treated children showed improvement or maintained original hearing loss when using ganciclovir for 6 months while only 17% of the treated children showed improvement or maintained original hearing loss when using ganciclovir for 6 weeks. Also, the results from the 6-month treatment trial showed a significant improvement in audiologic and neurodevelopmental outcomes. Therefore, patients with severe SNHL require a long-term ganciclovir treatment. The long term, 6-month treatment trial, decreased the worsening of hearing loss from 68% to 21% [3]. Ganciclovir can also be used as a treatment of late- onset hearing loss in infants with congenital Cytomegalovirus infection. 69% of infants using oral valganciclovir showed improvement in hearing loss due to CMV while 31% of these infants showed no change in hearing [22]. After 1 year of using ganciclovir, children with congenital hearing loss showed 25% to 30% improvement in the hearing threshold. That improvement took place during or shortly after treatment. Thus, there is enough evidence to prove that ganciclovir has the propensity to increases the likelihood of improvement in hearing and reduces the likelihood of deterioration in hearing. In a 4 to 10 years follow-up, none of the children treated with ganciclovir has sensorineural hearing loss whereas 11% of the children in the untreated control group showed sensorineural hearing loss. Studies also showed that there is no significant difference hearing outcome between oral and intravenous treatment. Sometimes the use of ganciclovir in treating hearing loss has no significant effect on patients. In a study conducted by Amir, Wolf, and Levy, 72% of the infants treated by a long-term oral ganciclovir for congenital Cytomegalovirus infection showed no change in the symptoms. Worsening of hearing usually has a low occurrence percentage (between 2%-16%). However, recently one study showed significant negative effect of using ganciclovir in treating hearing loss, where 87.5% of the treated children were found to have clinically significant worsening in hearing after the treatment. Ganciclovir can also have other side effects in the long run, such as central venous catheter/site infection, catheter malfunction and moderate neutropenia. These possible side effects are doserelated and can be easily managed by dose reduction. Despite all the negative side effects of the use of ganciclovir, this systematic review determined that the use of antiviral treatment improved hearing loss by 40% on average. Synthesis of the studies on results for children with congenital CMV show no observed heterogeneity between studies. As is the case in most research, all the reviewed studies had limitations. One of the common limitations is the loss of follow-up due to the number of required follow-ups that make it hard for family to continue in studies. Also, the lack of untreated control group and small sample size in multiple studies caused difficulties comparing groups to obtain an accurate effect of ganciclovir. Sometimes the retrospective nature of the design can add to the limitations of the study. Missing data and information related to the patients, such as other neurologic involvement, lateonset SNHL, pharmacokinetic data, is another common limitation in similar studies. Other times, the studies do not consider important aspects, such as accounting for the viral load.
Another limitation of the review process includes enclosing to few search motors, searching only in English language. There is still a need for better screening tool for congenital CMV infection. Thus, other studies are needed to obtain screening tools that are cost effective and simple to use by the general population.
Conclusion
CMV infections are the most important cause of nongenetic hearing loss, neurodevelopmental delay, intellectual impairment, and multisystem organ failure in children. It is a global health threat affecting approximately 0.3–1.7% of all newborns. The reviewed studies and experiments showed more positive effects than negative effects for using antiviral treatments for hearing loss in children and infants with CMV infections. Hence, after reviewing 15 well- performed studies and obtaining an overage of 40% improvement in hearing loss using antiviral treatment (ganciclovir), ganciclovir could be used as a treatment for patients including children and infants who are diagnosed with hearing loss due to CMV infection.
References
- Williamson WD, Desmond MM, LaFevers N, Taber LH, Catlin FI, et al. (1982) Symptomatic congenital Cytomegalovirus: disorders of language, learning and hearing. Am J Dis Child136(10):902-905.
- Pass RF, Fowler KB, Boppana S (1991) Clinical importance of cytomegalo- virus infection: an In: LandiniMP, editor. Progress in Cytomeg- alovirus Research New York: Elsevier Science Publishers 3-10.
- Kimberlin DW, Lin CY, Sánchez PJ, Demmler GJ, DanknerW,et al. (2003) Effect of ganciclovir therapy on hearing in symptomatic congenital Cytomegalovirus disease involving the central nervous system: a randomized, controlled trial. The Journal of pediatrics 143(1): 16-25.
- Dahle AJ, Fowler KB, Wright JD, Boppana SB, Britt WJ, et al. (2000) Longitudinal investigation of hearing disorders in children with congenital Cytomegalovirus. J Am AcadAudiol 11(5): 283-290.
- McCracken GH, Shinefield HMR, Cobb K, Rausen AR, Dische R, et al. (1969)Congenital cytomegalic inclusion disease: a longitudinal study of 20 patients. Am J Dis Child117(5):522-539.
- Pass RF, Stagno S, Myers GJ, Alford CA (1980) Outcome of symptomatic congenital Cytomegalovirus infection: results of long-term longitudinal follow-up. Pediatrics66(5):758-762.
- Noyola DE, Demmler GJ, Nelson CT, Griesser C, Williamson WD,et al. (2001) and the Houston Congenital CMV Longitudinal Study Group. Early predictors of neurodevelopmental outcome in symptomatic congenital Cytomegalovirus infection. J Pediatr138(3):325-331.
- Plotkin SA, Stetler H (1969) Treatment of congenital cytomegalic inclusion 1993 disease with antiviral agents. Antimicrobial Agents Chemother 9: 372-379.
- Feigin RD, Shackelford PG, Haymond MW, DeVivo DC (1971) Floxuridine treatment of congenital cytomegalic inclusion disease. Pediatrics 48(2): 318-322.
- Kraybill EN, Sever JL, Avery GB, Movassaghi N (1972) Experimental use of cytosine arabinoside in congenital Cytomegalovirus infection. J Pediatr80(3): 485-487.
- Mc Cracken GH, LubyJP (1972) Cytosine arabinoside in the treatment of congenital cytomegalic inclusion disease. J Pediatr 80(3): 488-493.
- Attard-Montalto SP, English MC, Stimmler L, Snodgrass GJ (1993) Ganciclovir treatment of congenital Cytomegalovirus infection: a report of two cases.Scand J Infect Dis 25(3): 385-388.
- Stronati M, Revello MG, Cerbo RM, et al. (1995) Ganciclovir therapy of congenital human Cytomegalovirus hepatitis. Acta Paediatr84(3): 340-341.
- Vallejo JG, Englund JA, Garcia-Prats JA, Demmler GJ (1994) Ganciclovirtreat- ment of steroid-associated Cytomegalovirus disease in a congenitally infected neonate. PediatrInfect Dis J 13(3): 239-241.
- Junker A, Matheson D, TingleA, Thomas E (1991) Immune responses after ganciclovir and immunoglobulin therapy of infants. Pediatr Infect Dis J10(3): 256-258.
- Reigstad H, Bjerknes R, MarkestadT, Myrmel H (1992) Ganciclovir therapy of congenital Cytomegalovirus disease. Acta Paediatr81(9):707-708.
- MichaelsMG, Greenberg DP, Sabo DL, Wald ER (2003) Treatment of children with congenital Cytomegalovirus infection with Pediatr Infect Dis J 22(6): 504-509.
- Nigro G, Scholz H, Bartmann U (1994) Ganciclovir therapy for symptomatic congenital Cytomegalovirus infection in infants: a two-regimen experi- ence. J Pediatr 124(2): 318-322.
- Crowley B (2002) Ganciclovir treatment of symptomatic congenital cytomeg- alovirus infection. J Antimicrob Chemother 50: 435-436.
- Nigro G, Krzysztofiak A, Bartmann U (1997) Ganciclovir therapy for Cytomegalovirus-associated liver disease in immunocompetent or im- munocompromised children. Arch Virol 142(3):573-580.
- Fischler B, Casswall TH, Malmborg P, Nemeth A (2002) Ganciclovir treatment in infants with Cytomegalovirus infection and cholestasis. J Pediatr Gastroenterol Nutr 34(2): 154-157.
- Amir J, Wolf DG, Levy I (2010) Treatment of symptomatic con- genital Cytomegalovirus infection with intravenous gan- ciclovir followed by long-term oral valganciclovir. Eur J Pediatr169(9):1061-1067.
- Kimberlin DW, Acosta EP, Sánchez PJ (2008) National Institute of Allergy and Infectious Diseases Collaborative Antiviral Study Group. Pharmacokinetic and pharmaco- dynamic assessment of oral valganciclovir in the treatment of symptomatic congenital Cytomegalovirus disease. J Infect Dis 197(6):836-845.
- Kimberlin DW, Lin C-Y, Sanchez PJ (2003) Effect of ganciclovir therapy on hearing in symptomatic congenital Cytomegalovirus dis- ease involving the central nervous system: A randomized, controlled trial. J Pediatr143:16-25.
- Nassetta L, Kimberlin D, Whitley R (2009)Treatment of con- genitalCytomegalovirus infection: implications for future therapeutic strategies. J AntimicrobChemother 63(5):862-867.
- Oliver SE, Cloud GA, Sanchez PJ (2009) Neurodevelopmental outcomes following ganciclovir therapy in symptomatic congenital cytomega- lovirus infections involving the central nervous system. J Clin Virol46(4):22-26.
- Kimberlin DW, Chin-Yu Lin, Sanchez PJ, Demmler GJ, DanknerW, et (2003)Effect of ganciclovir therapy on hearing in symptomatic congenital cytomegalo- virus disease involving the central nervous system: a ran- domized, controlled trial. J Pediatr143(1):16-25.
- Whitely RJ, Cloud G, Gruber W, Storch GA, Demmler GJ, et al. (1997) Ganciclovir treatment of symptomatic congenital Cytomegalovirus infection:results of a phase II J Infect Dis175(5):1080-1086.
- Nigro G, Scholz H, Bartmann U (1994) Ganciclovir therapy for symptomatic congenital Cytomegalovirus infection in infants: a two regimen experience. J Pediatr 124(2): 318-322.
- Pasternak Y, ZivL, Attias J, Amir J, Bilavsky E (2018) Valganciclovir is beneficial in children with congenital Cytomegalovirus and isolated hearing loss. The Journal of pediatrics 199:166-170.






























