Cell Ratios in Traumatic Cerebrospinal Fluid. Do They Have Predictive Value for Meningitis?
Ali Ahmed Raba1,2*, Jean Donnelly1
1 Department of General Paediatrics, Child Health Ireland at Crumlin, Ireland
2School of Medicine and Medical Sciences, University College Dublin, Ireland
Submission: February 05, 2020; Published: February 19, 2020
*Corresponding author: Ali Ahmed Raba, School of Medicine and Medical Sciences, University College Dublin, Dublin, Ireland
How to cite this article: Ali Ahmed Raba, Jean Donnelly. Cell Ratios in Traumatic Cerebrospinal Fluid. Do They Have Predictive Value for Meningitis?. Acad J Ped Neonatol. 2020; 9(2): 555813. DOI: 10.19080/AJPN.2020.09.555813
Abstract
Background: A cerebrospinal fluid (CSF) red to white cell correction ratio of 500:1 is often quoted as a preliminary result for determining potential positive/negative CSF cultures. The aim of this study is to examine the diagnostic utility of correcting the white blood cell (WBC) counts in traumatic Cerebrospinal fluid based on 500:1 ratio and to evaluate this adjustment in identifying bacterial meningitis.
Method: We performed a cohort study on children ≤3 months who were admitted to Our Lady’s Children’s Hospital, Crumlin, with suspected sepsis from the 1st October 2017 to the 5th October 2018. Cerebrospinal fluid WBC counts were adjusted downward for traumatic lumbar punctures (LP) (RBCs > 500/mm3) using WBC to RBC (500:1) ratio.
Result: A total of 117 infants (age: 39.4±21.7 days, male: 85(72.6%), traumatic LP: 35(29.9%)) were enrolled in this study. Among 117 infants, 3 (2.6%) had positive CSF cultures for bacterial meningitis, 28 (23.9%) had viral meningitis. Compared with the unadjusted cerebrospinal fluid WBC count, the adjusted one had substantially better specificity (by 8.4%) and positive predictive value (by 30%) with a similar sensitivity and negative predictive value for bacterial meningitis
Conclusion: Adjustment of WBC counts in traumatic LPs is associated with a significant reduction in the false positive cases without an increase in false negatives.
Keywords:Bloody Cerebrospinal fluid; Infant; Meningitis; Traumatic Lumbar Puncture
Abbreviations: CSF: Cerebrospinal Fluid; LP: Lumbar Puncture; RBCs: Red Blood Cells; PPV: Positive Productive Value; NPP: Negative Productive Value; WBC: White Blood Cell; ED: Emergency Department; IQR: Interquartile Range; HHV-6: Human Herpesvirus-6
Introduction
Acute meningitis is a common infection of the meningeal membrane lining the brain. It is predominantly viral in etiology and is responsible for approximately 82–94% of cases of meningitis in children. It is also considered a mild form of meningitis that often resolves spontaneously with supportive care only [1,2]. Bacterial meningitis however, despite advances in medical care including empirical antibiotics and vaccines, is still associated with serious sequalae and mortality rates of 10–50% of patients, depending on the causative organism [3]. Therefore, a rapid and accurate diagnosis of acute bacterial meningitis is vital to prevent the serious sequelae, and also to avoid unnecessary exposure to antibiotics or prolonged hospitalisation [4,5]. Examination of the cerebrospinal fluid (CSF) through lumbar puncture (LP) remains the gold standard method to confirm bacterial meningitis in any patient with suspected meningitis, as the clinical symptoms and signs are often nonspecific and unreliable [6,7]. Traumatic LPs however, are common in children (10-40%) and complicate the clinician’s ability to interpret CSF white blood cell (WBC) counts [6,8]. Although there were several methods for correction of WBC count in a traumatic LP, a CSF red to white cell correction ratio of 500:1 is the most used method for adjustment of the white cell count in traumatic CSF. The reliability of this approach to rule out bacterial meningitis remains controversial. A large retrospective study showed that adjustment of CSF WBC using 500:1 ratio does not aid in the diagnosis of bacterial meningitis in neonates [9]. Another multicentre study found that correction of the CSF WBC count reduces the number of infants with CSF pleocytosis while it can underestimate the number of white cells in the CSF and therefore under diagnose meningitis [8]. The aim of the present study to examine the ability of the ability of corrected CSF WBC to identify infants with bacterial meningitis.
Methods
This was a single centre cohort study of children who were admitted to Our Lady’s Children’s Hospital, Crumlin, with suspected sepsis between the 1st October 2017 to the 05th October 2018. We included infants ≤60 days of age who presented to the emergency department (ED) and who had an LP performed with CSF culture and CSF cell counts. Infants with incomplete CSF data were excluded from the study. In infants with more than one LP only the first LP was included in the study. Our primary outcome was bacterial meningitis, as defined by an infant with a positive CSF culture for a bacterial pathogen. We defined traumatic LP as a CSF RBC count ≥500cells/mm3 [9,10]. CSF WBC counts were adjusted downward for traumatic lumbar punctures using white to red cells (500:1) ratio. For example, if the CSF RBC count was 10,000 and the CSF WBC count was 70, then the adjusted CSF WBC count of 50 would be used (70-10,000/500). Demographics (age, gender), CSF parameters including RBC, WBC, protein, glucose and CSF culture, and blood inflammatory markers (WBC, CRP), and blood culture result were collected for each baby. Statistical analysis was performed using SPSS statistical software version 22.0. Descriptive statistics were used for all demographic variables of interest using frequency distribution and percentage. Mean and standard deviation (SD) were used for parametric numeric data, while non-parametric numeric data were summarised using median and interquartile range (IQR). Receiver operating characteristic (ROC) curves were constructed for unadjusted and adjusted WBC count. We calculated sensitivity, specificity, positive productive value (PPV) and negative productive value (NPV) of unadjusted and adjusted WBC count in traumatic and non-traumatic CSF for predicting bacterial meningitis. We used a CSF WBC count cutoff value of 10cells/mm3 and this was considered as significant CSF pleocytosis. We examined the ability of the adjusted CSF WCC count to detect infants with bacterial meningitis. Our results were summarized using p-value and 95% confidence intervals. P-values < 0.05 was be considered statistically significant.
Results
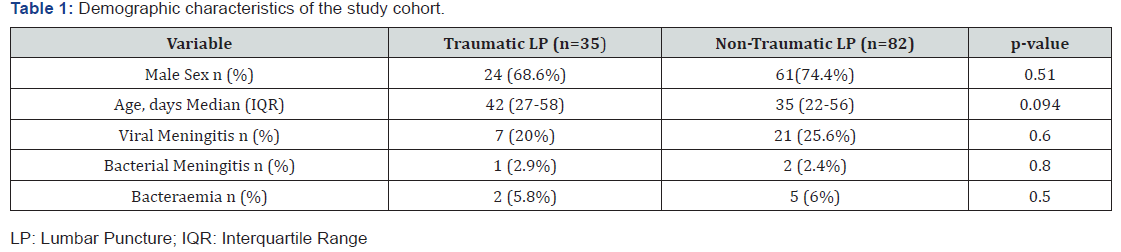
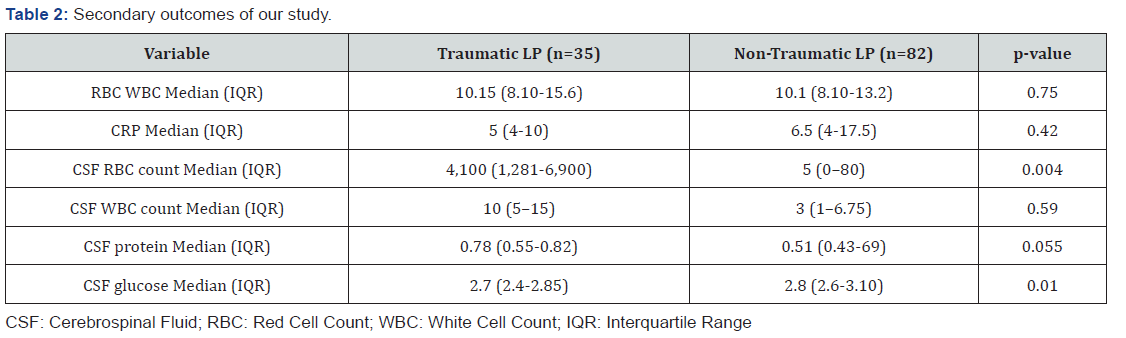
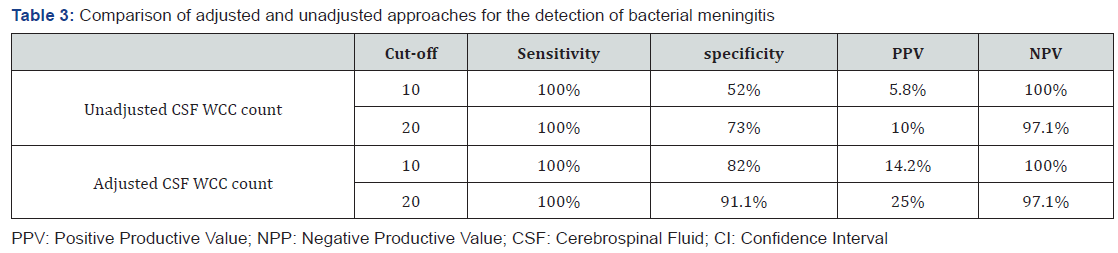
During the study period, a total of 117 infants who had a LP, were included in this study. Median patient age was 39 days (Interquartile Range (IQR) 24–35 days) and there were 85 males (72.6%) and with a male to female ratio 2.6:1. Among 117 infants, 3 (2.6%) had positive CSF cultures for bacterial meningitis (group B streptococcus was detected from a CSF of 2 (1.7%) infants and 1 (0.9%) had pneumococcal bacterial meningitis) and 28 (23.9%) had viral meningitis (11 (9.4%) had Enterovirus RNA meningitis, 10 (8.5%) had parechovirus and 7 (6%) had Human Herpesvirus-6 (HHV-6). Table 1 shows the demographic characteristics of our cohort. Of the 117, 35 (29.9%) infants had a traumatic LP, and infants with a traumatic LP were older than those with nontraumatic LP (42 (27-58) vs 35 (22-56), P: 0.094). The median CSF RBC count for infants with traumatic LPs was 4,100 (1,281– 6,900), while for those without traumatic LPs was 5 (0–80). The median CSF WBC count for infants with traumatic LPs was 10 (5–15) and for infants with non-traumatic LPs was 3 (1–6.75) (p:0.5) (Table 2). Before the adjustment of CSF WBC count, infants with bacterial meningitis had significant higher CSF WBC count of 3,990 (1,997–4,825), than those without meningitis, 4 (2–10), (p<.045). However, after adjustment, the difference of WBC count in CSF between two groups were not statistically significant (p:0.057) (Table3). The sensitivity, specificity, and positive and negative predictive values for the non-traumatic and traumatic LPs for prediction of bacterial meningitis at different cut-offs are presented in Table 3. In comparison with the unadjusted CSF WBC count, the adjusted LPs had similar sensitivity and NPV for bacterial meningitis, while specificity and PPV slightly improved after correction. NPV and PPV of adjusted CSF WBC count at cut off value of 10 were 100% and 14.2%, respectively (sensitivity 100%, specificity 82%), while NPV and PPV of unadjusted one were 100% and 5.8%, respectively (sensitivity 100%, specificity 52%). Similarly, at cut off value of 20, NPV and PPV of adjusted CSF WCC count were 97.1% and 25%, respectively (sensitivity 100%, specificity 91.1%), while NPV and PPV of unadjusted one were 97.1% and 10%, respectively (sensitivity 100%, specificity 73%) (Table 3).
Discussion
The presence of blood in CSF is associated with increased protein levels and cell count which leads to a reduction in the diagnostic value of LP. The prevalence of traumatic lumbar puncture in our study was (29.9%), which is similar to that which has previously been reported [9-12]. The RBC cut-off values for of traumatic LP varies in previous studies. For example, some studies defined the traumatic LP as a CSF RBC ≥10,000 cells/mm3, [8,13] while other defined as CSF specimens with ≥500 RBCs/mm3 [9,10,14]. In our study, CSF with RBC count ≥500 cells/mm3 was considered a traumatic LP as this is the cut off value that is used in our institution. Several studies assessed different approaches to correct the WCC count in traumatic CSF. Some authors used ratio 500 or 1000 RBCs to 1 WBC in the CSF. While other adjusted CSF WCC by using the ratio of RBCs to WBCs in the peripheral blood [8,9,15]. We found that application of the 500 CSF RBCs per 1 CSF WBC correction ratio for infants with a traumatic LP does not reduce the sensitivity of the CSF WCC for bacterial meningitis. In addition to this, our study revealed that using a 500:1 correction substantially reduced the number of infants who were classified as having CSF pleocytosis without misclassifying any case of bacterial meningitis. In other words, CSF WCC correction reduced the false positives (patients without bacterial meningitis who unnecessarily receive antibiotics) without an increase in false negatives. Greenberg et al. [9] conducted a large study including 2519 neonates (<28 days) with a traumatic lumbar puncture. On contrary to our findings, they found that correction for a traumatic LP using a 500:1 correction substantially reduced the sensitivity of CSF WBC for bacterial meningitis by 4%. Moreover, another study included examined the 500:1 ratio method for prediction of meningitis in children from 1-month to 18-year-old with traumatic LP (RBCs ≥500 cells/mm3). They found that the adjusted CSF WCC was not significantly better than the unadjusted one [14]. Our study revealed that increasing the cut-off from 10 to 20 WCC associated with an increase in the specificity by 9.1% and the PPV by 10.8%, while the NNP decreased by 2.9% and the sensitivity remained the same. These finding are like that showed by Greenberg et al, [9] who found that increasing the cut-off to 20, increasing the specificity but the sensitivity was decreased. There are a couple of limitations in this study. Firstly, the sample size was small, as the sample was drawn from one hospital and only over a limited time period. Secondly, we assessed only a 1:500 ratio method to adjust CFS WCC and no other methods were examined in our study. Thirdly, although a clinician usually does LP prior to administering antibiotics, it was not always clear which infants received antibiotics prior to LP. Lastly, we included infants with traumatic LP with no upper limit of RBC count in CSF. However, the strength of our study relates to the fact that it utilised a recent data set obtained from a setting with strict guidelines for the management of infants with suspected meningitis.
Conclusion
The results of this study indicate that application of a 500 CSF RBCs per 1 CSF WBC adjustment for the traumatic LP significantly increases specificity without reducing the sensitivity. Therefore, utilisation of the corrected CSF WBC count could lead to a reduced rate and duration of unnecessary antibiotic exposure and associated with a reduced hospitalizations rate for many infants without bacterial meningitis. However, all available data should be used when deciding whether to initiate treatment in an infant with suspected meningitis and bloody CSF. We believe that further larger studies are required in this area to examine the best method of WBC correction in traumatic LPs.
References
- Nigrovic LE, Kuppermann N, Malley R (2002) Development and validation of a multivariable predictive model to distinguish bacterial from aseptic meningitis in children in the post-Haemophilus influenzae era. Pediatrics110(4):712-71
- McGill F, Griffiths MJ, Solomon T (2017) Viral meningitis: current issues in diagnosis and treatment. Curr Opin Infect Dis30(2):248-2
- Aronin SI, Peduzzi P, Quagliarello VJ (1998) Community-acquired bacterial meningitis: risk stratification for adverse clinical outcome and effect of antibiotic timing. Ann Intern Med129(11):862-86
- Pingree EW, Kimia AA, Nigrovic LE (2015) The effect of traumatic lumbar puncture on hospitalization rate for febrile infants 28 to 60 days of age. Acad Emerg Med22(2):240-24
- Chauhan D, Mokta K, Kanga A, Grover N (2018) Epidemiology, clinical profile and role of rapid tests in the diagnosis of acute bacterial meningitis in children (aged 1-59 months). Neurol India66(4):1045-104
- Nazir M, Wani WA, Kawoosa K, Dar SA, Malik M, et al. (2018) The Diagnostic Dilemma of Traumatic Lumbar Puncture: Current Standing of Cerebrospinal Fluid Leukocyte Corrections and Our Experience with Cerebrospinal Fluid Biomarkers. J Child Neurol33(7):441-44
- Bonadio W (2014) Pediatric lumbar puncture and cerebrospinal fluid analysis. J Emerg Med46(1):141-1
- Lyons TW, Cruz AT, Freedman SB, Neuman MI, Balamuth F, et al. (2017) Interpretation of Cerebrospinal Fluid White Blood Cell Counts in Young Infants with a Traumatic Lumbar Puncture. Ann Emerg Med69(5):622-6
- Greenberg RG, Smith PB, Cotten CM, Moody MA, Clark RH, et al. (2008) Traumatic lumbar punctures in neonates: test performance of the cerebrospinal fluid white blood cell count. Pediatr Infect Dis J27(12):1047-10
- Mazor SS, McNulty JE, Roosevelt GE (2003) Interpretation of traumatic lumbar punctures: who can go home? Pediatrics111(3):525-52
- Fielkow S, Reuter S, Gotoff SP (1991) Cerebrospinal fluid examination in symptom-free infants with risk factors for infection. J Pediatr119(6):971-97
- Schreiner RL, Kleiman MB (1979) Incidence and effect of traumatic lumbar puncture in the neonate. Dev Med Child Neurol21(4):483-48
- Lyons TW, Cruz AT, Freedman SB, Arms JL, Aronson PL, et al. (2017) Correction of Cerebrospinal Fluid Protein in Infants with Traumatic Lumbar Punctures. Pediatr Infect Dis J36(10):1006-100
- Bonsu BK, Harper MB (2006) Corrections for leukocytes and percent of neutrophils do not match observations in blood-contaminated cerebrospinal fluid and have no value over uncorrected cells for diagnosis. Pediatr Infect Dis J25(1):8-11.
- Osborne JP, Pizer B (1981) Effect on the white cell count of contaminating cerebrospinal fluid with blood. Arch Dis Child56(5):400-40






























