The Growth History of a Girl carrying ABCC8 Gene Mutation resulting in Congenital Hyperinsulinism
Jianfang Zhu, Hong Chen, Yanlan Fang, Chunlin Wang* and Li Liang
Department of Pediatrics, The first Affiliated Hospital of Zhejiang University School of Medicine, China
Submission: November 06, 2018; Published: January 04, 2019
*Corresponding author: Chunlin Wang, The first Affiliated Hospital of Zhejiang University School of Medicine79 Qingchun Road, Hangzhou 310006, China
How to cite this article: Jianfang Z, Hong C, Yanlan F, Chunlin W, Li Liang. The Growth History of a Girl carrying ABCC8 Gene Mutation resulting in Congenital Hyperinsulinism. Acad J Ped Neonatol. 2019; 7(3): 555771. DOI: 10.19080/AJPN.2019.07.555771
Abstract
Objective: Congenital hyperinsulinism (CHI) is the most common cause of severe, persistent hypoglycemia in newborns and infancy. Increasingly more mutations of genes were found to be associated with CHI.
Methods: The following were performed on a female infant diagnosed to suffer from CHI. PCR and directly sequencing for mutation detection of all the 39 exons and the flanking intron–exon boundaries of ABCC8 gene, a whole-body PET CT-scan with [18F]-L-Dihydroxyphenylalanine (DOPA) to detect lesions of organs, clinicopathologic and immunohistochemical analysis for pathological diagnosis. Blood glucose was then continuously monitored, and she was followed up every six months.
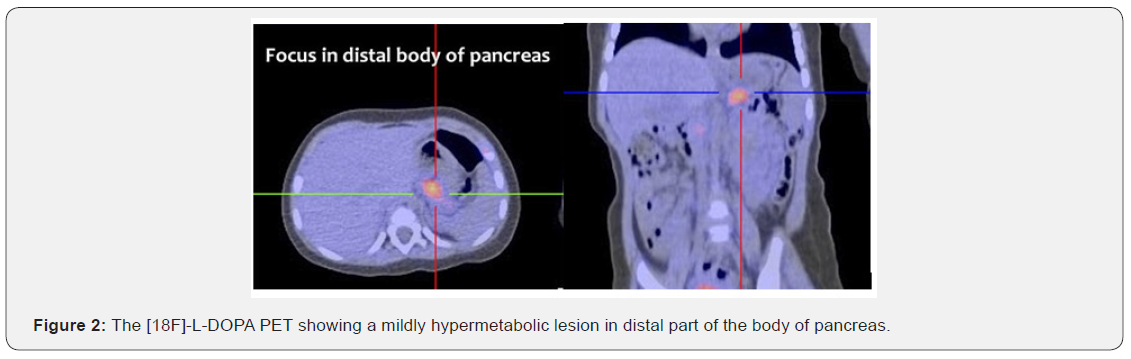
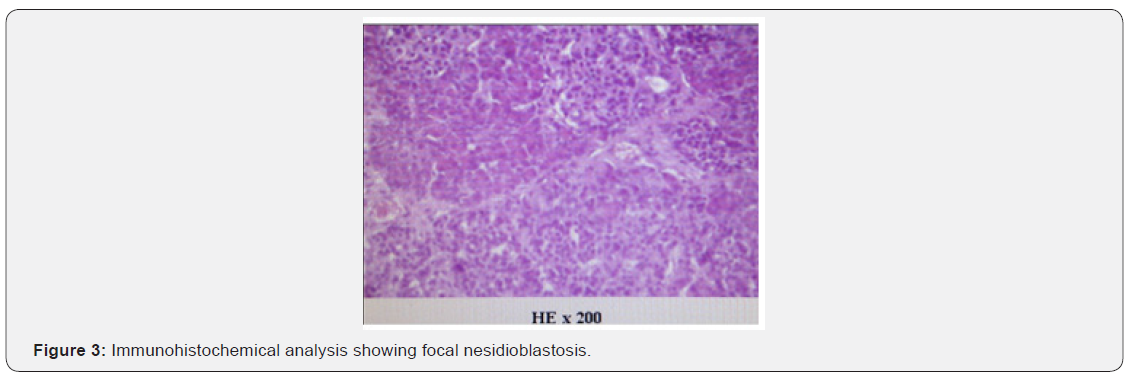
Results: Genetic analysis of the girl revealed a heterozygous ABCC8 R598X mutation. The whole-body PET CT-scan with [18F]-L-DOPA revealed a mildly hypermetabolic lesion in distal part of the body of pancreas.After the partial lesion resection of pancreas, clinicopathologic and immunohistochemical analysis prompted focal nesidioblastosis. Then the girl remained euglycemic with normal feeding, now is six years old with normal growth rate.
Declaration of Interest Section: We reported a girl with R598X mutation of ABCC8 gene resulting in CHI. [18F]-L-DOPA PET can help to identify focal and diffuse forms of CHI effectively. Then surgery can help to control hypoglycemia.
Keywords: CHI; Persistent hypoglycemia; [18F]-L-DOPA PET; ABCC8
Introduction
Congenital hyperinsulinism (CHI) or persistent hyperinsulinemic hypoglycemia (HH) of infancy is the most critical cause of persistent hypoglycemia in infants beyond the first few days after birth [1]. Currently, mutations in 12 genes have been reported to cause CHI and more recently there have been isolated case reports of potential novel genetic mechanisms in patients with CHI and other syndromic features [2]. The most common and severe forms of CHI are due to mutations in the ABCC8 and KCNJ11 genes, encoding the sulfonylurea receptor 1 (SUR1) and inward rectifier potassium channel Kir6.2 (Kir6.2) subunits of the ATP sensitive potassium (KATP) channel, respectively [3,4]. It is well known that [18F]-L-Dihydroxyphenylalanine (DOPA) positron emission tomography (PET) has an established role in the identification of focal and diffuse forms of CHI, and the localization of the hyperinsulinemic focus [5]. Here, we report a baby carrying a heterozygous mutation, R598X, in the ABCC8 gene associated with persistent hypoglycemia during the infant period. The focal lesion of pancreas was diagnosed using the [18F]-L-DOPA PET. Then she was restored to health successfully with partial lesion resection of pancreas by laparoscopic surgery.
Case Report
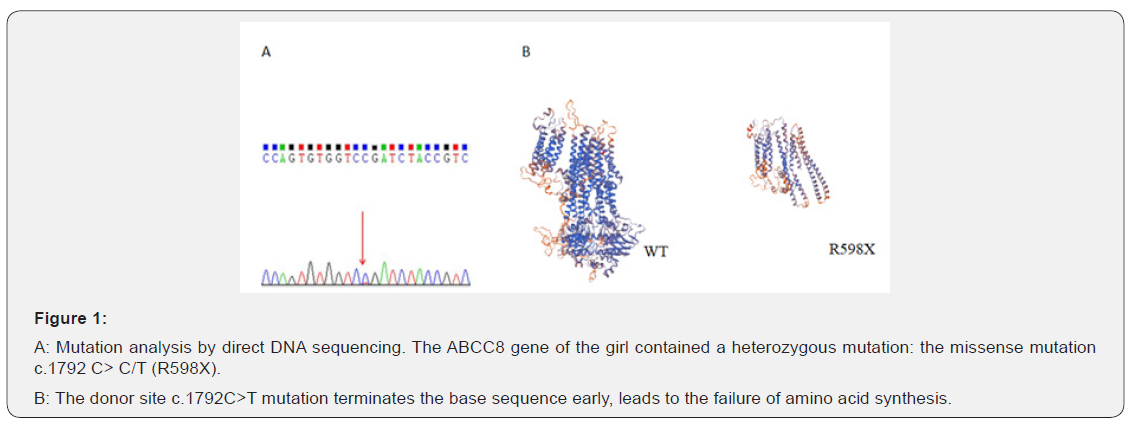
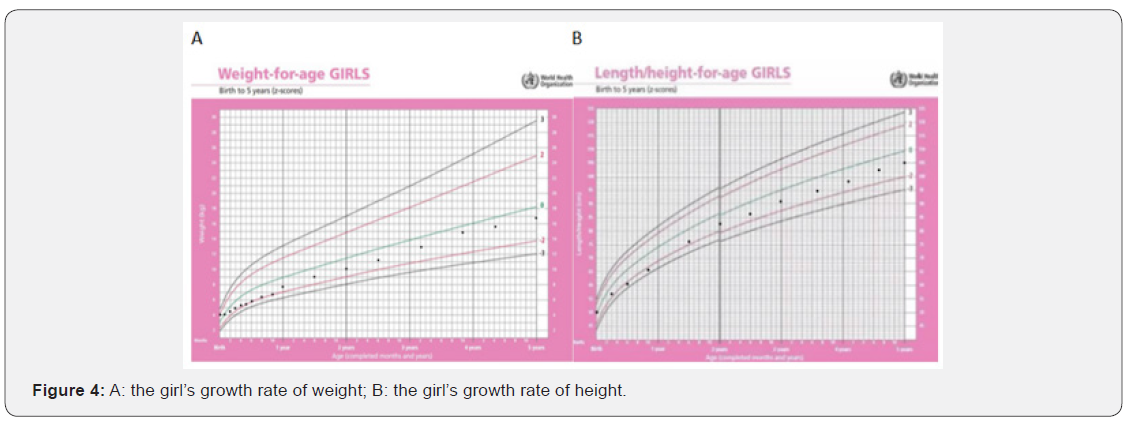
An 11-day-old female infant was admitted with persistent hypoglycemia since 11 hours after born. The infant was born at term through normal vaginal delivery. She was the first-born of healthy non-consanguineous parents, with a birth weight of 4.05kg. The mother’s serum glucose was always normal even when gestation. Eleven hours after birth, the neonate developed symptomatic episodes of hypoglycemia, presenting as lethargy, pallor and sweat (capillary glucose from 1.8 to 2.1mmol/L). She started on intravenous glucose infusion immediately with the increasing dose until the total glucose dose 11.5mg/kg/min. The sepsis screen was negative and blood culture was sterile. As the infant increased her oral intake over the following days, the glucose infusion could be tapered and discontinued, and the baby was discharged from the hospital. She did well until 6-month-old when she presented three episodes of tonic clonic seizures, and biochemical and hormonal tests revealed the following, serum glucose 1.4mmol/L, serum insulin 14uIU/ml, negative urine ketone and glucose, normal serum electrolytes, C-peptide and hormone levels (GH, TSH, FT4, and cortisol). Serum aminoacidogram and free fattyacid levels were also normal. Meanwhile, abdominal ultrasound examination and magnetic resonance cholangiopancreatography (MRCP) failed to reveal any evidence of pancreatic hyperplasia. The insulin/ glucose ratio (0.56) increased and the diagnosis of CHI was confirmed. In view of persistent hypoglycemia, the patient was started on hydrocortisone injection at an initial dose of 4mg/kg/ day and oral diazoxide (5mg/kg/day). The serum glucose could not be controlled until the hydrocortisone injection and oral diazoxide both increased to 15mg/kg/day, but it decreased again on tapering of hydrocortisone. In the meantime, corn starch was fed to the patient with milk. At the beginning of the ninth month, the baby was started on subcutaneous octreotide injection (5μg/ kg/day) because of recurrent hypoglycemia, but she could not maintain euglycemia despite high dose of 15μg/kg/day. Genetic analysis of the infant revealed a heterozygous ABCC8 missense mutation, c.1792 C> C/T (R598X) (Figure 1), affecting premature transcription termination of exon 12. A whole-body PET CTscan with [18F]-L-DOPA revealed a mildly hypermetabolic lesion in distal part of the body of pancreas when she was 10 months old (Figure 2). Then partial lesion resection of pancreas was performed by laparoscopic surgery when she was 10.5 months old. Clinicopathologic and immunohistochemical analysis prompted it to be focal nesidioblastosis (Figure 3). After surgery, no drugs were used, and the girl maintained euglycemia with feeding as normal children for 6 years. Blood glucose was continuously monitored, and she was followed up every six months. The girl’s growth lagged significantly before the operation, but after surgery it was normal, as shown in (Figure 4).
Discussion
Hypoglycemia is the main clinical feature of CHI and leads to a high risk of seizures and brain damage. Symptoms revealing hypoglycemia are various and depend on the severity of hypoglycemia and age of the patient ranging from asymptomatic hypoglycemia revealed by routine blood glucose monitoring to life-threatening hypoglycemic coma or status epilepticus [6]. Consequently, timely diagnosis and adequate treatments are crucial. This includes a glucose load by mouth or intravenous injection, at the time of hypoglycemia, to correct it. Then a treatment to prevent future episodes of hypoglycemia must be conducted, which may contain frequent and glucose-enriched feeding, hydrocortisone, diazoxide and octreotide.
In clinical practice, when medical or dietary therapies are ineffective, insulinoma should be vigilant. Imageology and genetics can help to identify diffuse or focal forms of CHI. In view of high frequency of missed diagnosis by normal imageology, such as abdominal ultrasound examination and MRCP, [18F]-L-DOPA PET can help to diagnose effectively. It has been estimated that [18F]- L-DOPA PET is 96 % accurate in differentiating between focal and diffuse disease and 100 % accurate in localizing the focal lesion. Indeed, the high sensitivity of this method could aid surgeons to perform a curative partial resection of pancreas without the high risk of long-term diabetes mellitus [5,7].
As many recent reports have shown, the phenotype associated with ABCC8 and KCNJ11 mutations ranges from asymptomatic macrosomia, persistent HH in infants to neonatal diabetes. In adults, these mutations may also be critical causes of dominantly inherited early-onset diabetes mellitus [8-12]. The mutations of ABCC8 could be dominant, recessive. All 39 exons and the flanking intron–exon boundaries of ABCC8 gene were amplified by PCR and directly sequenced in our infant and her parents. A heterozygous R598X ABCC8 mutation was identified in the baby, but not in the parents. The parents had no hypoglycemia in childhood or diabetes mellitus at present. Mutations of this gene could cause hyperinsulinism that is responsive or unresponsive to treatment with diazoxide [13,14]. The baby in our case was unresponsive to treatment with diazoxide.
In the present report, the heterozygous R598X mutation of ABCC8 gene is indicated to be one cause of CHI. Although the girl now has normal blood sugar and growth rate after surgery for 6 years, there is still a risk for developing diabetes mellitus later in life and needs to be kept in long-term observation.
Acknowledgments
The authors sincerely thank and acknowledge Prof. Pik-to Cheung, Department of Paediatrics and Adolescent Medicine, University of Hong Kong, for providing suggestion to diagnose by [18F]-L-DOPA PET in their hospital. And we also thank David Nkoma, whose native language is English, for correcting English grammar, style, and usage.
Declaration of Interest Section
This study was supported by National Science Foundation of China (No. 11571309), Zhejiang provincial TCM science and technology program (No. 2018ZQ031) and Zhejiang ProvincialNatural Science Foundation (No. LQ19H310002).
References
- Yorifuji T (2014) Congenital hyperinsulinism: current status and future perspectives. Ann Pediatr Endocrinol Metab 19(2): 57-68.
- Demirbilek H, Hussain K (2017) Congenital Hyperinsulinism: Diagnosis and Treatment Update. J Clin Res Pediatr Endocrinol 9(Suppl 2): 69-87.
- Thomas PM, Cote GJ, Wohllk N, Haddad B, Mathew PM, et al. (1995) Mutations in the sulfonylurea receptor gene in familial persistent hyperinsulinemic hypoglycemia of infancy. Science 268(5209): 426-429.
- Thomas P, Ye Y, Lightner E (1996) Mutation of the pancreatic islet inward rectifier Kir6.2 also leads to familial persistent hyperinsulinemic hypoglycemia of infancy. Hum Mol Genet 5(11): 1809-1812.
- Hardy OT, Hernandez-Pampaloni M, Saffer JR (2007) Diagnosis and localization of focal congenital hyperinsulinism by 18F-fl uorodopa PET scan. J Pediatr 150(2): 140-145.
- Arnoux JB, Verkarre V, Saint-Martin C, Montravers F, Brassier A, et al. (2011) Congenital hyperinsulinism: current trends in diagnosis and therapy. Orphanet J Rare Dis 6: 63.
- Hardy O, Hernandez-Pampaloni M, Saffer JR, Scheuermann JS, Ernst LM, et al. (2008) Accuracy of [18F] Fluorodopa Positron Emission Tomography for Diagnosis and Localizing Focal Congenital Hyperinsulinism. J Clin Endo Metab 92(12): 4706-4711.
- Kapoor RR, Flanagan SE, James CT, McKiernan J, Thomas AM, et al. (2011) Hyperinsulinaemic hypoglycaemia and diabetes mellitus due to dominant ABCC8/KCNJ11 mutations. Diabetologia 54(10): 2575-2583.
- Klee P, Bellanné-Chantelot C, Depret G, Llano JP, Paget C, et al. (2012) A novel ABCC8 mutation illustrates the variability of the diabetes phenotypes associated with a single mutation. Diabetes Metab 38(2): 179-182.
- Riveline JP, Rousseau E, Reznik Y, Fetita S, Philippe J, et al. (2012) Clinical and metabolic features of adult-onset diabetes caused by ABCC8 mutations. Diabetes Care 35(2): 248-251.
- Oçal G, Flanagan SE, Hacihamdioğlu B, Berberoğlu M, Siklar Z, et al. (2011) Clinical characteristics of recessive and dominant congenital hyperinsulinism due to mutation(s) in the ABCC8/KCNJ11 genes encoding the ATP-sensitive potasium channel in the pancreatic beta cell. J Pediatr Endocrinol Metab 24(11-12): 1019-1023.
- Al Mutair AN, Brusgaard K, Bin-Abbas B, Hussain K, Felimban N, et al. (2013) Heterogeneity in Phenotype of Usher-Congenital Hyperinsulinism Syndrome: Hearing loss, retinitis pigmentosa, and hyperinsulinemic hypoglycemia ranging from severe to mild with conversion to diabetes. Diabetes Care 2013; 36(3): 557-561.
- Macmullen CM, Zhou Q, Snider KE, Tewson PH, Becker SA, et al. (2011) Diazoxide-unresponsive congenital hyperinsulinism in children with dominant mutations of the β-cell sulfonylurea receptor SUR1. Diabetes 60(6): 1797-1804.
- Snider KE, Becker S, Boyajian L, Shyng SL, MacMullen C, et al. (2013) Genotype and phenotype correlations in 417 children with congenital hyperinsulinism. J Clin Endocrinol Metab 98(2): E355-E363.






























