Noma Neonatorum Caused by Acinetobacter Sepsis in a Premature Baby
Şükriye Yılmaz* and Erdal Peker
Tekden Privacy Hospital, Turkey
Submission: March 15, 2018; Published: August 14, 2018
*Corresponding author: Şükriye Yılmaz, Tekden Privacy Hospital, Velioğlu street, No:77 34000, Bağcılar, İstanbul.
How to cite this article: Şükriye Y, Erdal P. Noma Neonatorum Caused by Acinetobacter Sepsis in a Premature Baby. Acad J Ped Neonatol. 2018; 7(1): 555762. DOI: 10.19080/AJPN.2018.07.555762
Abstract
Noma neonatorum is characterized by a gangrenous phenomen affecting mucocutaneous junctions of mouth, nasal and anal area and occasionally, the eyelids and scrotum. Especially it is observed during the first few weeks of neonatal life [1-2] and is mainly linked to Pseudomonas aeruginosa (P. aeruginosa), prematurity, and low birth weight. No established treatment in the few reported cases, and mortality is nearly inevitable [3]. In this report we have presented in a premature with noma neonatorum caused by Acinetobacter baumannii (A. baumannii).
Keywords: Noma neonatorum; Acinetobacter baumannii, Prematurity
Abbrevations: CBC: Compled Blood Cell; CPAP: Continuous Positive Airway Pressure; CRP: C-Reactive Protein; INR: Internalization Normalization Ratio; NICU: Neonatal Intensive Care Unit; PEEP: Positive End Expiratory Pressure
Case Report
We admitted a female twin delivered at 31weeks’ gestation in another hospital referred to our hospital. Apgar scores were 6-7-7 at 1-5-10mins, and the weight was 1260g. On examination, baby have mild respiratory distress, cardiac auscultation was normal, neonatal reflexes mildly depressed. Other systemic examinations were remarkable. The baby was stabilized with continuous positive airway pressure (CPAP) with PEEP 6 and was transferred to the neonatal intensive care unit (NICU). Compled blood cell (CBC), biochemistry analysis, C-reactive protein (CRP), procalsitonin, blood culture was taken, and the baby was treated empirically with ampicillin and gentamicin. Cranial ultrasound revealed grade 2 intraventricular hemorrhage. On day 6 she developed increasing respiratory distress and hypotension. A sepsis evaluation was performed including blood cultures, and a CBC had a hemoglobin of 16.1g/dl, platelets of 43,000mm3 and a white blood cell count of 33.860/l with 74.3% segmented neutrophils, 15.5% lymphocytes, 5.9% monocytes, 3.9% eosinophils. CRP elevated 205mg/L (normal range <5mg/L), and procalcitonin levels was 36.8ng/ml (normal range <0.05ng/ml). Treatment changed with vancomycin, meropenem, colisitine and liposomal amphotericin B. Pentoxifyline added her treatment. On day 8 a pustular rash appeared in the anal regions and the general condition of the patient has deteriorated. In addition, she developed severe hypotension necessitating pressor support with dopamine, dobutamine, epinephrine and norepinephrine. Blood culture result of multi drug resistant A. baumannii and antibiotic sensitivity test was performed which showed A. baumannii sensitive to only ciprofloxacin, tetracycline and colisitine. Based on the culture and sensitivity test ciprofloxacin added the treatment. Supportive treatments were added fresh frozen plasma, thrombocytes and erythrocytes product packets. Swabs were taken from the pharynx, the rectum, and both eyes and were found as a negative. On day 9 the perianal and genital region was also erythematous and edematous around the necrotic center (Figure 1).
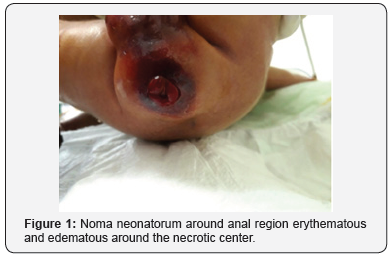
Baby develops acute renal failure with creatinine 4.6mg/dL, anuria. Other laboratory analysis was CBC had a hemoglobin of 9.6g/dl, platelets of 79 000mm3 and a white blood cell count of 65.079/l with 86.8% segmented neutrophils, prothrombin time 33.9sec with Internalization normalization ratio (INR) of 2.83. Hepatic markers were abnormal. During the ensuing two days anal and genital necrotic lesion was grew up. On day 11 patient with severe acidosis and refractory hypotension, whose general condition was disrupted during follow-up, she was died although all supportive treatment.
Discussion
In the original series of 35 patients, the lesions were seen in the majority of cases in the orofacial region (27 of 35 patients), as well as in the anogenital region in 7 of 35 patients [4]. In 2014, Raimondi et all were reported a case of a 28day-old female infant was born with extremely low birth weight. She has diffuse gingival noma, initially misdiagnosed as thrush. P.aeruginosa was grown from all swabs but cultures were sterile. The strain showed resistance to penicillins, carbapenems, aminoglycosides, sulfamethoxazole-trimethoprim, and nitrofurantoin while being sensitive to colistin. A regimen of intravenous and topical colistin was followed for 3 weeks [3].
Nayak et all recently described a 25day-old Indian female infant that referred with facial tissue destruction and fever. Swab test showed Gram negative bacilli. P. aeruginosawas isolated. They had treated her with netilmicin, amikacin, clindamycin and topical clindamycin cream [6].
Freeman at all delivered a case 26 weeks’ gestation twin and and the weight was 808g. On day 15 she received a noma neonatorum diagnosis in perianal regions. She was treated with intravenous meropenem and tobramycin based on antibiotic susceptibility test results, and topical silver sulfadiazine was applied to the skin lesions. She was discharged on day of Life 90. They suggest that noma neonatorum represents a neonatal form of ecthyma gangrenosum. The etiologic agent in almost all of the cases is the same (P. aeruginosa) and the histopathology similar [7].
Our case was different
A level III European NICU, extremely low birth weight, and multi drug rezistance A. baumannii was grown from blood culture. Yet, it is an environment where P.aeruginosacolonization is a feared and not uncommon event, and neonatologists need to have an appropriate index of suspicion for noma to prevent its mortality or morbidity.
Conclusion
pediatricians and neonatologist’s necessity to be consider that multidrug-resistant nosocomial bacteria such as P. aeruginosa might observe with a destroyer mucosal lesions without any systemic symptoms. Appropriate antibiotic, such as colisitine, may be useful in that cases unfortunately we vanish our case causes multiorgan failure and A. baumannii sepsis.
References
- Parikh TB, Nanavati RN, Udani RH (2006) Noma neonatorum. IndianJ Pediatr 73: 439-440.
- Lin JY, Wang DW, Peng CT, Tsai FJ, Chiou YM et al. (1992) Noma neonatorum: an unusualcase of noma involving a full-term neonate. Acta Paediatr 81(9): 720-722.
- Raimondi F, Veropalumbo C, Coppola C, Maddaluno S, Ferrara T, et al. (2015) Noma Neonatorum From Multidrug-Resistant Pseudomonas aeruginosa: An Underestimated Threat? J Pediatric Infect Dis Soc 4(3): e25-e27
- Ghosal SP, Sen Gupta PC, Mukherjee AK, Choudhury M, Dutta N, et al. (1978) Noma neonatorum: its aetiopathogenesis. Lancet 2(8084): 289-291.
- Nayak PA, Nayak UA, Khandelwal V, Gupta A (2013) Noma neonatorum. BMJ Case Rep
- Freeman AF, Mancini AJ, Yogev R (2002) Is noma neonatorum a presentation of ecthyma gangrenosum in the newborn? Pediatr Infect Dis J 21(1): 83-85.
- Atiyeh BS, Hashim HA, Rubeiz MT, Hamdam AM, Bitar FF, et al, (1998) Necrotising infection of the orofacial tissues in neonates (noma neonatorum) Case report. Scand J Plast Reconstr Surg Hand Surg 32(3): 343-345.






























