Procedure Location Affects the Cost of Balloon Atrial Septostomy Procedures without Compromising Care
Fabio Savorgnan1, Nicholas B Zaban1, Michael M Ross2 and John P Breinholt3
1 Department of Pediatric, Baylor College of Medicine, Indiana
2Department of Pediatric, University of Colorado, Colorado
3Department of Pediatric, University of Texas Health Science Center at Houston, Texas
Submission: September 10, 2016; Published: August 30, 2017
*Corresponding author: Fabio Savorgnan, Department of Pediatric, Baylor College of Medicine, Texas Children Hospital, Indianapolis, Indiana.
How to cite this article: Fabio S, Nicholas B Z, Michael M R, John P B. Procedure Location Affects the Cost of Balloon Atrial Septostomy Procedures without Compromising Care. Acad J Ped Neonatol. 2017; 5(4): 555725. DOI: 10.19080/AJPN.2017.05.555725
Abstract
Background:Balloon Atrial Septostomy (BAS) is a common procedure performed in infants with dextro-transposition of the great arteries (d-TGA). BAS improves oxygenation by improving mixing of oxygenated and deoxygenated blood at the atrial level prior to definitive repair via the arterial switch operation. The focus on healthcare costs is increasingly in the news and is broadly discussed in medicine and medical training.
Purpose:This study compares safety and cost-effectiveness of balloon Atrial Septostomy (BAS) for patients with d-Transposition of the Great Arteries (dTGA) performed at the patient bedside vsthe catheterization laboratory (cathlab).
Methods:Neonates with dTGA who underwent BAS from 10/2000 to 1/2014 met inclusion criteria. Medical and procedural records, echocardiograms, and catheterization data were reviewed. Comparisons between the 2 procedural locations included patient demographics, pre and post procedure oxygen saturations, and safety outcomes. The two locations were compared using t-tests for most continuous variables and Fisher’s exact tests for all categorical variables. Wilcoxon rank sums tests and analysis of covariance (ANCOVA) model for time variable and oxygen saturation respectively.
Results:No safety or outcome benefit was identified for BAS performed at the bedside or cathlab for patients with dTGA.
Conclusion:BAS performed at the bedside and in the cathlab have similar safety outcomes and efficacy. Bedside BAS can be performed at substantially decreased cost relative to BAS performed in the cathlab. Bedside BAS has the advantages of not expose the patient to radiation and not introducing risks of patient manipulation and transfer.
Condensed abstract:This is the largest study evaluating the safety and cost-effectiveness of bedside BAS. In our study we found that BAS performed at the bedside and in the catheterization lab had similar safety outcomes and efficacy. Bedside BAS in the neonatal intensive care unit has the advantages of not introducing risks of patient manipulation and transfer, and does not expose the patient to radiation. Bedside BAS also has the advantage of being significantly less expensive than BAS performed in the cathlab. The next step would be to perform a prospective randomized study comparing cost-efficacy of BAS performed at bedside versus in the catheterization lab.
Keywords:Balloon atrial septostomy; D-Transposition of the great arteries; Catheterization laboratory; bedside procedure; Cost effectiveness
Introduction
Balloon Atrial Septostomy (BAS) is a common procedure performed in infants with dextro-Transposition of the Great Arteries (d-TGA). BAS improves oxygenation by improving mixing of oxygenated and deoxygenated blood at the atrial level prior to definitive repair via the arterial switch operation. BAS improves cerebral oxygenation and reduces oxygen requirements [1,2], and lowers in-hospital mortality relative to patients who do not receive BAS [3]. BAS can be performed in the cardiac catheterization laboratory (cathlab) using fluoroscopy, or can be performed at the patient bedside in the Neonatal Intensive Care Unit (NICU) under echocardiography guidance [4-7]. The decision where to perform BAS is often based on physician or institutional preference, with safety concerns or desire to visualize coronary artery anatomy often cited when the cathlab is preferred.
Procedure billing for bedside septostomy involves cardiologist professional fees, echocardiography guidance and interpretation, and catheter equipment resulting in charges of approximately $9,200 at the study institution. Sedation for bedside BAS is incorporated into daily hospital charges from the NICU. Billing for BAS performed in the cathlab includes the same charges from the bedside procedure, plus separate billing from anesthesiology (approximately $2,000), and cathlab facility charges (approximately $18,000) resulting in a total charge of approximately $29,000.
This study compares the outcomes of BAS performed in the catheterization laboratory to those performed at the bedside. Moreover, it compares the healthcare costs of the procedure in the different locations to help determine if healthcare cost savings should be a determinant in location choice based on patient outcomes.
Methods
We reviewed all patients at a single institution who underwent BAS from October 2000 to January 2014. Medical records including echocardiograms, cardiac catheterization and operative reports were reviewed. Patients were excluded if they did not have a diagnosis of d-TGA, if they had a diagnosis of coarctation of the aorta in addition to d-TGA, or if they had a diagnosis of double outlet right ventricle. We recorded the BAS location of each patient, cathlab versus bedside. We collected the following data: gestational age, age at BAS, age at arterial switch operation, presence of a prenatal diagnosis of d-TGA, and procedural use of ventilator support or inotropes. Pre-procedure and postprocedure oxygen saturations were recorded. Complications related to BAS were documented, as well as the occurrence of catheterization in the cathlab specifically to evaluate coronary anatomy. Discrepancies from the echocardiography defined coronary anatomy and operative report were identified.
Statistical analysis was performed to compare patient characteristics for each procedural location. The two locations were compared using t-tests for most continuous variables and Fisher’s exact tests for all categorical variables. Since age at procedure, age at surgery, and days between procedure and surgery were nonparametric, Wilcoxon rank sums tests were used to compare the two locations on these characteristics. An analysis of Covariance (ANCOVA) model was used to compare the two locations on the change in oxygen saturation levels after adjusting for the pre-procedure level.
Results
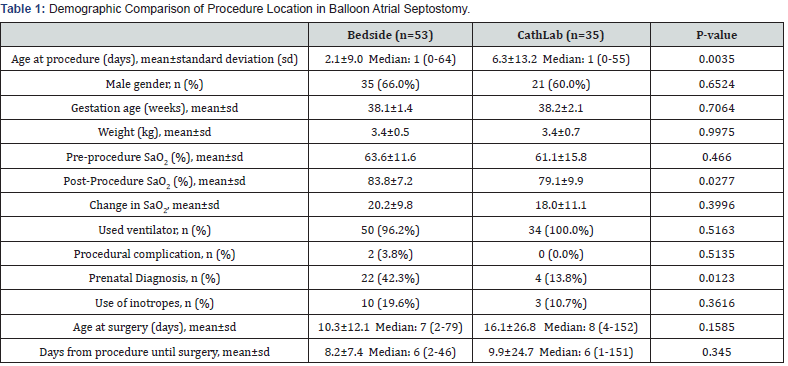
We identified 88 patients who had a BAS performed for d-TGA. 53 patients (60.2%) had a BAS performed at the bedside, and 35 patients (39.8%) had a BAS performed in the cathlab. Table 1 compares patient characteristics between the two groups. There was no difference between gender, gestational age, weight, or use of inotropes between the two locations. Cathlab patients were 4 days older than those at bedside (cathlab: 6.3±13.2 days; bedside: 2.1±9.0 days; p=0.004). More bedside BAS patients had a prenatal diagnosis of d-TGA than those performed in the cathlab (42% vs. 14%; p=0.012). There was no difference in pre-procedure oxygen saturation between the groups. Patients who had a BAS performed at the bedside had a higher post procedure oxygen saturation than those performed in the cathlab (83.8% vs. 79.1%; p=0.028). However, there was no difference in the pre- and post- procedure oxygen saturation difference between the patient groups (20.2% vs. 18.0%; p=0.40). There was no difference between age at arterial switch surgery (10.3 vs. 16.1; p=0.16) or the number of days from BAS until surgery (8.2 vs. 9.9; p=0.35).
There were two complications that occurred during bedside BAS. One patient developed supraventricular tachycardia during the procedure and was cardioverted with catheter manipulation, with no clinical sequelae. Another patient had a large coronary sinus resulting in difficulty with procedural imaging. Thus, the procedure was aborted and the patient was referred to the cathlab for BAS. There were no complications for BAS performed in the cathlab. There was no difference in complication rates at the cathlab versus bedside.
Twenty-four patients were identified with a coronary variant. The most common coronary anomaly was a circumflex branch arising from the right coronary artery, which was found in 16 of 24 (67%). Fourteen of the 24 coronary anomaly patients (58%) had the anomaly correctly identified by echo prior to procedure. One patient was diagnosed with a coronary variant by echo but was found to have normal coronary arteries in the operating room. Five patients who had bedside BAS had a cardiac catheterization afterwards to define coronary anatomy. Of these five, one had a coronary variant. Twenty-two of 53(41%) bedside BAS patients did not have coronary anatomy clearly defined by echocardiography; six of these patients had coronary artery variants identified at surgery. These six patients did not have a cardiac catheterization prior to surgery. There were no operative complications related to coronary anatomy.
Discussion
BAS is an important, life saving procedure that promotes hemodynamic stability in anticipation of corrective surgery. BAS can be successfully performed in the catheterization laboratory, or under echo cardiographic guidance at the bedside. The choice of modality is usually determined by institutional, or practitioner, preference. Clinical discussions regarding the ideal choice largely focus on safety. This study compares BAS outcomes based on the procedural locations of the cathlab versus the bedside.
The two groups had similar characteristics including gestational age, weight, sex, use of inotropes, and pre-procedure oxygen saturation. Bedside BAS was performed earlier than cathlab BAS, which may be due the ease of organizing the procedure at bedside relative to coordinating the cathlab procedure that is dependent on a schedule. Why more patients with prenatal diagnoses underwent bedside procedures compared to unknown diagnoses at birth is unknown. Whether earlier BAS held clinical benefit was not evaluated in this study.
There were two complications in our patient cohort, both of which were in the bedside BAS group. One was supraventricular tachycardia, converted with catheter manipulation; the other was the inability to complete the procedure with solely ultrasound guidance. Supraventricular tachycardia or other atrial arrhythmias are well-described complications caused by the presence of catheters introduced into the right atrium, whether they are procedural equipment or central venous lines. Moving the offending object, or less commonly, performing chemical or electrical cardio version often achieves successful treatment. Nevertheless, these events did not create a significant difference with the cathlab group [8-10]. Neither patient had long-term sequelae attributed to the bedside procedure. Our study did not demonstrate a difference in safety outcomes between procedure location, consistent with prior studies [11]. Prior studies have suggested that BAS was associated with a higher risk of stroke, but more recent studies and meta-analyses do not show a correlation with BAS and brain injury [12,13]. These studies did not distinguish procedural location as a variable.
Coronary anatomy remains a concern for patients with d-TGA due to the reimplantation of the coronary arteries during the arterial switch operation. There were coronary variants in 27% of patients in our study, most common being a circumflex originating from the right coronary artery. In this study, 22 patients who had a pre-procedure echocardiogram that did not clearly demonstrate coronary anatomy, then underwent a bedside BAS, did not undergo catheterization to identify coronary anatomy prior to arterial switch operation. Six of these patients were found to have coronary anomalies at the time of the arterial switch, but there were no complications related to coronary anatomy. Found that patients with d-TGA and single coronary or intramural coronary anatomy had a higher mortality compared with other types of coronary anatomy. More recent studies did not show a correlation between coronary anatomy and mortality in patients with d-TGA who underwent arterial switch operation [14,15,16]. In (Brown 2001) [17] suggested that newer coronary reimplementation techniques have minimized coronary anomalies as a significant risk factor for mortality in patients undergoing the arterial switch operation [17]. This suggests that having a cardiac catheterization to identify coronary anatomy prior to arterial switch operation is not always necessary. As such, the key advantage of the cathlab may be superfluous to the operative management of these patients.
The focus on healthcare costs is increasingly in the news and is broadly discussed in medicine and medical training. Nevertheless, specific discussion of how to reduce costs is not often realized. Concerns are raised that patient safety and outcomes should outweigh any cost advantage of one specific therapy over another. Discussions with hospital and practice billing departments revealed a large difference between what is billed in a cathlab procedure versus a bedside procedure in the NICU. Resource utilization during bedside BAS is more efficient because sedation and ventilator management is provided by the attending ICU physician. Their fees are related to their daily service commitment, and the need for additional services, resulting in manpower and billing, are absent. The cathlab utilizes an additional team of nurses and technicians, and includes standard fees associated with the service. This team is not needed, except for perhaps one individual to assist the cardiologist, during bedside BAS. Importantly, the technical fees associated with cathlab use are not assessed. In the cathlab, echo assistance may or may not be used. If echo is not used, the technical and professional fees would not be assessed,compared to those fees always associated with bedside BAS. This would reduce the overall cost of the cathlab procedure, however, cathlab would maintain higher charges than bedside BAS due to the cathlab facility fee. Importantly, the argument of care over cost is lost in light of our findings that the outcomes at both locations were equivalent.
This is the largest study evaluating the safety and costeffectiveness of bedside BAS. In our study we found that BAS performed at the bedside and in the catheterization lab had similar safety outcomes and efficacy. Bedside BAS in the neonatal intensive care unit has the advantages of not introducing risks of patient manipulation and transfer, and does not expose the patient to radiation. Bedside BAS also has the advantage of being significantly less expensive than BAS performed in the cathlab. Bedside BAS can also be performed at referral hospitals to stabilize patients with d-TGA and allow them to remain at the referral hospital for several days prior to transfer to tertiary care centers [18]. Although BAS in the cardiac catheterization lab has the advantage of characterizing coronary anatomy, some studies suggest [19] that coronary anomalies are not a significant risk factor for mortality in the arterial switch operation. In the current era of quality and cost containment, we advocate that bedside BAS offers equivalent safety with the benefit of considerable cost savings compared to the cathlab [20].
References
- Van der Laan ME, Verhagen EA, Bos AF, Berger RM, Kooi EM (2013) Effect of balloon atrial septostomy on cerebral oxygenation in neonates with transposition of the great arteries. Pediatr Res 73(1): 62-67.
- Hiremath G, Natarajan G, Math D, Aggarwal S (2011) Impact of balloon atrial septostomy in neonates with transposition of great arteries. J Perinatol 31(7): 494-499
- Mukherjee D, Lindsay M, Zhang Y, Lardaro T, Osen H, et al. (2010) Analysis of 8681 neonates with transposition of the great arteries: outcomes with and without Rashkind balloon atrial septostomy. Cardiol Young 20(4): 373-380.
- Bullaboy CA, Jennings RB Jr, Johnson DH (1984) Bedside balloon atrial septostomy using echocardiographic monitoring. Am J Cardiol 53(7): 971.
- Baker EJ, Allan LD, Tynan MJ, Jones OD, Joseph MC, et al. (1984) Balloon atrial septostomy in the neonatal intensive care unit. Br Heart J 51(4): 377-378.
- Steeg CN, Bierman FZ, Hordof AJ, Hayes CJ, Krongrad E, et al. (1985) “Bedside” balloon septostomy in infants with transposition of the great arteries: new concepts using two-dimensional echocardiographic techniques. J Pediatr 107(6): 944-946.
- Zellers TM, Dixon K, Moake L, Wright J, Ramaciotti C (2002) Bedside balloon atrial septostomy is safe, efficacious, and cost-effective compared with septostomy performed in the cardiac catheterization laboratory. Am J Cardiol 89(5): 613-615.
- Verheij G, Smits-Wintjens V, Rozendaal L, Blom N, Walther F, et al. (2009) Cardiac arrhythmias associated with umbilical venous catheterisation in neonates. BMJ Case Rep pii: bcr04, 1778, doi: 10.1136/bcr.04.2009.1778.
- Green C, Yohannan MD (1998) Umbilical arterial and venous catheters: placement, use, and complications. Neonatal Netw 17(6): 23-28.
- Hodzic S, Golic D, Smajic J, Sijercic S, Umihanic S, et al. (2014) Complications Related to Insertion and Use of Central Venous Catheters (CVC). Med Arch 68(5): 300-303.
- D’Orsogna L, Lam J, Sandor GG, Patterson MW (1989) Assessment of bedside umbilical vein balloon septostomy using two-dimensional echocardiographic guidance in transposition of great arteries. Int J Cardiol 25(3): 271-277.
- Beca J, Gunn J, Coleman L, Hope A, Whelan LC, et al. (2009) Preoperative brain injury in newborn infants with transposition of the great arteries occurs at rates similar to other complex congenital heart disease and is not related to balloon atrial septostomy. J Am Coll Cardiol 53(19): 1807-1811.
- Pasquali SK, Hasselblad V, Li JS, Kong DF, Sanders SP (2002) Coronary artery pattern and outcome of arterial switch operation for transposition of the great arteries: a meta-analysis. Circulation 106(20): 2575-2580.
- Qamar ZA, Goldberg CS, Devaney EJ, Bove EL, Ohye RG (2007) Current risk factors and outcomes for the arterial switch operation. Ann Thorac Surg 84(3): 871-878.
- Sarris GE, Chatzis AC, Giannopoulos NM, Kirvassilis G, Berggren H, et al. (2006) The arterial switch operation in Europe for transposition of the great arteries: a multi-institutional study from the European Congenital Heart Surgeons Association. J Thorac Cardiovasc Surg 132(3): 633-639.
- Hong SJ, Choi HJ, Kim YH, Hyun MC, Lee SB, et al. (2012) Clinical features and surgical outcomes of complete transposition of the great arteries. Korean J Pediatr 55(10): 377-382.
- Brown JW, Park HJ, Turrentine MW (2001) Arterial switch operation: factors impacting survival in the current era. Ann Thorac Surg 71(6): 1978-1984.
- Martin AC, Rigby ML, Penny DJ, Redington AN (2003) Bedside balloon atrial septostomy on neonatal units. Arch Dis Child Fetal Neonatal Ed 88(4): 339-340.
- Lin AE, Di Sessa TG, Williams RG (1986) Balloon and blade atrial septostomy facilitated by two-dimensional echocardiography. Am J Cardiol 57(4): 273-277.
- Polito A, Ricci Z, Fragasso T, Cogo PE (2012) Balloon atrial septostomy and pre-operative brain injury in neonates with transposition of the great arteries: a systematic review and a meta-analysis. Cardiol Young 22(1): 1-7.






























