Pulmonary Hypertension in Children with Esophageal Varices: Frequency and Relation to N-Terminal Pro B-Type Naturetic Peptide
Haitham El Bashir1*, Mariam M El Hawli1, Hanaa Masoud1, Afnan Albahri2, Mostafa Refaat2 and VishwanathaKini3
1Developmental & Rehabilitation Section, Hamad Medical Corporation, U.A.E
2Weill Cornell Medical College, Qatar Foundation, U.A.E
3Radiology Department, Hamad Medical Corporation, U.A.E
Submission: May 25, 2016 ; Published: August 12, 2016
*Corresponding author: Haitham El Bashir, Rehabilitation Department, Al Jalila Children’s Specialty Hospital, PO Box 7662, Dubai, United Arab Emirates,
How to cite this article: Haitham El B, Mariam M El H, Hanaa M, Afnan A, Mostafa R, et al . Brain White Matter Changes in Four Patients with Classical 005 Homocystinuria: Two of Them were Reversible. Acad J Ped Neonatol. 2016; 1(5): 555571. DOI: 10.19080/AJPN.2016.01.555571
Abstract
Classical homocystinuria (CHU) due to Cystathionine Beta-Synthase (CBS) deficiency is rare autosomal recessive inborn error of amino acids metabolism causing accumulation of methionine and homocysteine. The clinical manifestations of CHU may include neurological deficits mostly due to thromboembolic event so the most common brain radiological findings are secondary to stroke. The management is based on pharmacological treatment and dietary restriction of methionine. We present four patients with pyridoxine non-responsive CHU, who were found to have white matter changes on brain MRI, with a review of their clinical characteristics and neurological outcome. All patients had an underlying homozygous mutation of c.1006C>T (p.R336C) in the CBS gene, hypermethioninemia due to poor adherence to diet restriction, and were diagnosed before the implementation of the National Metabolic Newborn Screening Program in Qatar. Dramatic reductions in methionine levels because of better compliance were associated with reversibility of the brain white matter lesions in two patients.
Introduction
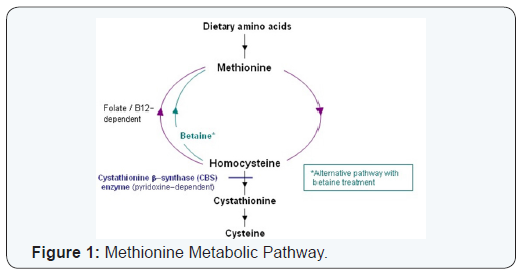
Classical homocystinuria (CHU) due to Cystathionine Beta-Synthase (CBS) deficiency (OMIM 236200) is a rare autosomal recessive in born error of amino acids metabolism [1]. Degradation of dietary methionine produces an intermediate compound known as homo cytosine that is mostly re-methylated to methionine. Homocysteine is also converted to cystathionine by the action of CBS which requires pyridoxine (Vitamin B6). Cystathionine Beta-Synthase deficiency due to CBS gene mutation leads to accumulation of both methionine and homocysteine, (Figure 1) [2]. The estimated prevalence of Classical homocystinuria is between 1/20,000 and 344,000. However, in Qatar it is approximately 1/1,800 births, the highest in the world [3-5]. Untreated patients may present with multisystem manifestations, such as vision problems due to myopia and ectopia lentis, osteoporosis, skeletal deformities mimicking Marfan syndrome, intellectual disability, and thromboembolic events. Neurological presentations may include seizures, extra pyramidal signs, psychiatric disturbance and focal neurological deficit due to infarction [5-7].
Neuro-imaging is not routinely done for patients with classical homocystinuria unless clinically indicated. The most common brain radiological findings are secondary to stroke: infarction, atrophy, and venous occlusion [7]. Classical homocystinuria is classified into pyridoxine responsive and none responsive types, homozygous mutation of c.1006C>T (p.R336C) in the CBS gene was attributed for most of pyridoxine non-responsive homocystinuria among Qatari patients [4].
Protein (Methionine) restricted diet and pharmacological treatment with pyridoxine (Vitamin B6), Betaine, hydroxy-cobalamine (Vitamin B12), and folic acid, are the corner stone in managing patients with classical homocystinuria. The aim is to control biochemical abnormalities and prevent complications.
Newborn screening is essential to improve clinical outcomes and patient’s quality of life by allowing early diagnosis and management. Poor compliance to diet restriction and medical treatment leads to hypermethioninemia and serious complications [8,9].
Case Series
Case 1
An 18 year old, male was diagnosed with classical homocystinuria by the age of 7 years. He was born at term after uncomplicated pregnancy and a smooth antenatal history. He was hospitalized at age of 5 months for acute bronchiolitis and at 10 months with intussusceptions that was reduced by barium enema.
At 7 years of age, he presented with history of limited vision and poor school performance. Physical examination showed, tall stature, slender body constitution with height falling at the 90th percentile and weight falling just below 50th centile, long face, long tibial bones, but no scoliosis or kyphosis. Ophthalmological assessment revealed bilateral lens subluxation so homocystinuria was suspected and later on confirmed after serum amino acids tests: homocysteine blood level 153.8umol/L, (normal 4-12mol/L) and methionine serum level 495umol/L (normal 43-223umol/L). The patient was started on treatment immediately: Betaine (Tri-methyl glycine) 5 gm/day orally, Folic acid 5mg/day orally, vitamin B6 (pyridoxine) 300 mg/day orally, Vitamin B12 (Hydroxy-cobalamine) 1000ug/week IM injection and restricted protein diet, but he had poor compliance mainly to diet. Later the diagnosis of CHU pyridoxine non-responsive type was confirmed by genetic study that confirmed homozygous mutation of c.1006C>T (p.R336C) in CBS gene.
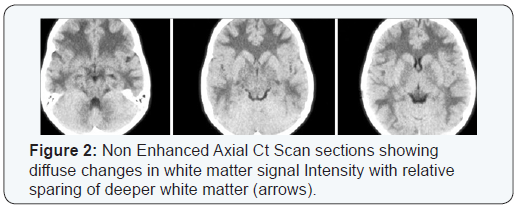
At the age of 9, he was found to have increased muscle tone with exaggerated deep tendon reflexes in both lower limbs. A brain CT scan showed diffuse reduced attenuation of the white matter noted all over the brain parenchyma including the cerebellar white matter sparing of the overlying cortex and basal ganglia (Figure 2). A brain MRI was requested to confirm the findings but patient traveled abroad for bilateral lens implantation. The patient was evaluated again at the age of 13 years. At that time he was a full time student, with learning problems, speech and language difficulties.
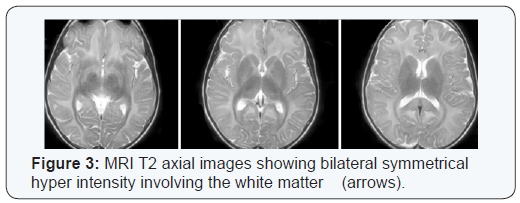
He was able to walk, run, and go up & down stairs independently, and was independent in activities of daily living. IQ test revealed the following: verbal reasoning 37 (Average), visual abstract reasoning 36 (average), quantitative reasoning 31 (average), short term memory 25 (average) and overall IQ 59 (mental retardation-mild). Brain MRI at that period showed diffuse increased signal intensity on T2 and FLAIR of the cerebral per ventricular white matter bilaterally in a symmetrical pattern, there was also increased signal intensity of the cerebral white matter bilaterally on diffusion weighted images. No evidence of intracranial hemorrhage identified. The ventricles and extra axial CSF spaces appeared unremarkable. There was a focal area of cystic changes noted in the right cerebral per ventricular white matter keeping with an old insult (Figure 3).The MRA of the cerebral vessels showed no radiological evidence of vasculitis. The tortuosity of the optic nerves noted bilaterally.
Case 2
A 17 year old girl was diagnosed with classical homocystinuria at the age of 6 years. The diagnosis was suspected for the first time due to incidental finding of bilateral lens dislocation. Homocysteine level at diagnosis was 173.1umol/L and methionine level was 622umol/L. Later CHU pyridoxine non-responsive type was confirmed by isolation of c.1006C>T (p.R336C) CBS gene mutation. Family history reported no parental consanguinity. The patient had normal neonatal, infantile, and early childhood stages. On examination (age 6 years), she had arachenodactyly, limited cognitive function, speech was normal, and no abnormal neurological findings were detected .I.Q test was refused by parents. Soon after the diagnosis, she was started on medication: Betaine, Vitamin B6 (pyridoxine), folate, vitamin B12sub> (hydroxycobalamin) IM injections, and restricted diet but the patient was not compliant. Hearing assessment was reported as normal and she was followed up by ophthalmology for abnormal vision.
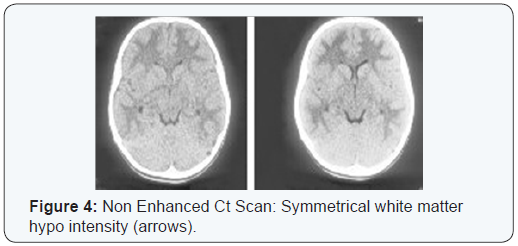
At the age of 7 years, the patient was admitted to the pediatric ward with one month history of intermittent, generalized, compressing type headache that was not associated with vomiting or nausea. A brain CT reported as exaggerated white matter hypo density of the supra tutorial brain symmetrically involved both hemispheres with no focal areas of infarction or hemorrhage (Figure 4). A Brain MRI done in the same period also revealed bilateral symmetrical diffuse white matter abnormality characterized by poor myelination. Electromyography and Nerve Conduction Study were normal. Echocardiogram was requested showed mild mitral valve prolapse with no hemodynamic consequences. Patient was discharged on oral aspirin with follow up in neurology and metabolic clinics.
Two years later a follow up evaluation in the neurology clinic, the patient continued to complain of infrequent bouts of headaches that were relieved by Aspirin. Neurological exam was normal at that time. So a follow up MRI head was requested, it showed stationary course with no newly developed lesion (Figure 5). Echocardiogram showed mild mitral valve prolapse with mild mitral regurgitation.
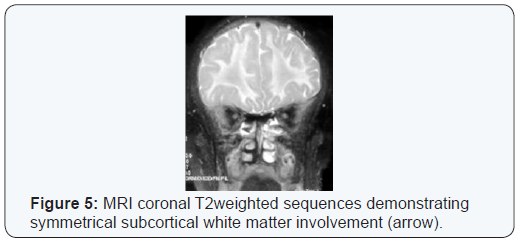
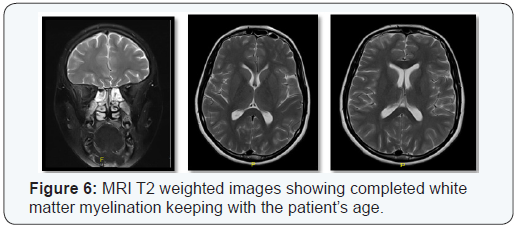
In 2008, sleep and a wake EEG was normal. A repeated MRI head in 2009 was reported as no new changes from previous study. Latest MRI brain done at age of 15 years showed no significant white matter abnormality visualized, no evidence of significant brain atrophy and with resolution of the previously reported white matter changes (Figure 6).
Case 3
A 15 years old girl was diagnosed at age of 3 years and 10 months as a case of classical homocystinuria due to Cystathionine Beta-synthase deficiency in early childhood, based on blood and genetic work up. Later the diagnosis of CHU pyridoxine nonresponsive type was confirmed by genetic test that identified c.1006C>T (p.R336C) CBS gene mutation. Family history revel no parental consanguinity. She was started on medication (Betaine powder 4gm Po twice daily, folic acid 5mg Po twice daily, vitamin B6 100mg Po twice daily, vitamin B12 injection 1mg IV once every 2 weeks, Aspirin 100mg Po once daily) and diet restriction, but had poor compliance. EEG was done routinely and showed abnormal bioccipital foci of spikes without clinically evident seizures.
Further EEG studies at age 6 and 8 years showed no changes.
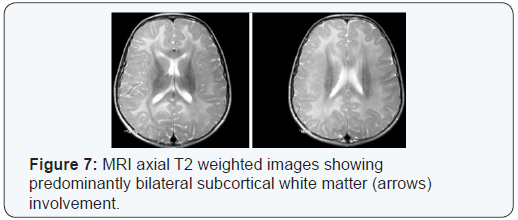
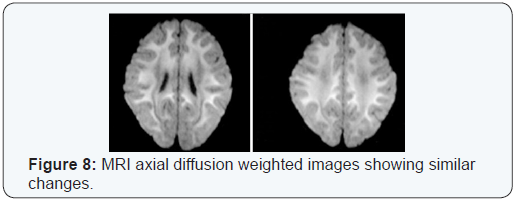
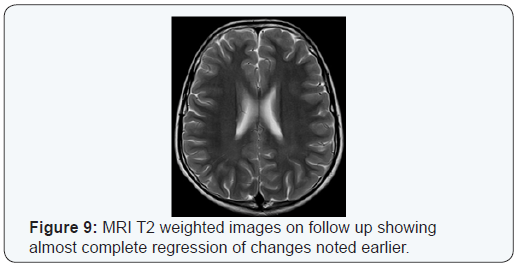
MRI brain at the age of 5 years and reported as normal. At age of 8 years, a follow up brain MRI was reported abnormal and showed diffuse white matter abnormality with predominant per ventricular distribution (Figure 7). One year later, a repeated brain MRI showed bilateral symmetrical white matter hyperintense signals in T2 sequences mainly involving the subcortical white matter and Centrum semi vale region (Figure 8). A follow up brain MRI at 13 years of age revealed subtle high T2 signal noted bilaterally in the frontal and parietal white matter there were no gross white matter changes seen, there was no evidence of cerebral or cerebellar atrophy (Figure 9).
The patient also had regular follow up with ophthalmologist for myopia and was prescribed glasses. She was followed up by orthopedics for thoracolumbar scoliosis. Hearing assessment was normal. Cognitive assessment (I.Q) test score was 82, which is in the low average level.
At the age of 14 years, the patient had a visit to Accident and Emergency complaining of recurrent morning headache; however, her neurological examination was reported normal as well as the rest of her systemic evaluation.
Case 3
A 32 year old male, diagnosed with classical homocystinuria at the age of 10 years. He was born by vaginal delivery after uneventful pregnancy with a birth weight of 3.5 Kg. The patient had history of failure to thrive, and developmental delay mainly in speech and language skills. At 4 years of age his weight was below the 5th percentile for age and sex. There was also a history of bilateral inguinal hernias that were surgically corrected.
At the age of 6 years he presented with severe left eye pain, vomiting, and poor vision. Examination of the left eye showed dislocated lens in the anterior chamber, small pupils, high intraocular pressure >40 mmHg as well as dislocated lens of the right eye. Based on clinical findings of long face, high arched plate pectus carinatum and skin abnormalities he was diagnosed as: Marfa syndrome with recurrent attacks of bilateral dislocation of the lenses of both eyes and severe secondary left eye glaucoma leading to vomiting.
At the age of 10 years the patient traveled to USA for further ophthalmological assessment and management, where the diagnosis of classical homocystinuria was made based on a positive Cyanide Nitroprusside Test and serum amino acids test. Initially he was started on low protein diet and pyridoxine supplement. As he had pyridoxine non-responsive CHU, folic acid, Vitamin B12 injections, and Betaine were added, but he had poor compliance. In 2003, at the age of 20 years, c. 1006C>T (p.R336C) CBS gene mutation was identified.
The patient attended schools for children with special needs and he required educational support. When he was 16 years old, he underwent bilateral lens implantation, surgical correction of large left testicular hydrocele. He was also suffering from joint stiffness and kyphoscoliosis. The patient did not have previous MRI images as part of his diagnostic evaluation, however, at the age of 22 years, he was involved in a road traffic accident where he was a pedestrian. The collision resulted in head injury, chest trauma, right femoral shaft fracture complicated by fat embolism which required open reduction and internal fixation with three weeks of mechanical ventilation. During his hospitalization head MRI showed scattered white matter disease in a linear fashion along the distribution between the anterior and the middle cerebral arteries and scattered foci in the basal ganglia, the frontal white matter and few peripheral areas in the gray-white matter junction. The appearance is not specific. The history and the conspicuity of lesions on the diffusion-weighted images raised the possibility of fat embolism; the differential diagnosis includes ischemic white matter changes secondary to either homocystinuria or even as a complication of hypoxic injury.
The patient was discharged ambulating in good general condition, and Aspirin was added to his medications. He had a cardiology team follow up for trivial mitral regurgitation and mild aortic insufficiency. Laparoscopic cholecystectomy for gallstones and acute cholecystitis was done at 25 years old.
Discussion
In this report, cerebral white matter changes were detected by brain MRI in all the four patients with pyridoxine non-responsive classical homocystinuria whom had poor compliance to treatment and diet restriction. In case
- The brain MRI was requested after he showed signs of upper motor neuron lesion.
- In case had MRI due to frequent bouts of headaches.
- MRI brain was requested as a follow up for abnormal EEG findings.
- It was done to investigate the complications of a road traffic accident. The follow up MRIs images for case (2) and (3) showed resolution of the white matter changes.
Although brain white matter lesions unrelated to thromboembolic events are not classical manifestations of CHU, there is increasing evidence supporting that the presence of cerebral white matter changes might actually be part of the natural history of this rare disease. A 10 years Iranian experience of 20 patients with homocystinuria, neuro-imaging data showed that 13 (65%) patients had white matter involvement [10]. In 2002 Yaghmai et al. [11] reported massive cerebral edema without evidence of thrombosis occurred in a patient with pyridoxine non-responsive classical homocystinuria who was on Betaine treatment, the patient had poor compliance to diet restriction and very high methionine levels reached 3000umol/L [11]. Another study in 2004 described white matter edema after 4-6 weeks of Betaine treatment in a child with cystathionine beta-syntheses deficiency [12]. Reversible white matter changes were also reported by Vatanavicharn and Brenton et al. [7,13]. Regular MRI follow up in a 21 years old male with pyridoxine-nonresponsive CBS deficiency was used to document reversible cerebral white matter lesions in correlation to methionine levels [14].
The underlying cause of cerebral white matter changes in patients with classical homocystinuria is still not well understood whether it is a complication of the condition or as a side effect of the medications. Even though Betaine therapy was claimed to be the culprit by some authors [11,12,15], white matter changes were reported in a 4 year old boy just prior to the diagnosis of classical homocystinuria and starting treatment. His MRI changes were normalized a year after starting treatment including Betain [13]. In addition hypermethioninemia with levels as high as 1000umol/L or higher was a consistent finding in all reported cerebral white matter changes in patients with CHU [7,11-14] regardless of Betaine use. As Betaine acts on conversion of homocysteine to methionine through an alternative pathway (Figure 1), it will worsen the hyper methioninemia especially in poorly compliant patients to methionine dietary restriction. Another controversy is whether the underlying pathophysiology of the white matter changes is due to demyelination process [7] or intramyelinic edema [14,15].
Leukodystrophies have been documented in different amino acids disorders and those are due to many different mutations. An in-depth analysis of the mutations involved in previous and future case studies of white matter changes related to homocystinuria could shed light on another way where neurological deficits in CHU patients can be predicted and perhaps prevented. Linking different CBS gene mutations and the degree of pyridoxine responsiveness to the severity and the prevalence of cerebral white matter changes can be worthwhile.
Summary
In summary, our report confirms the finding of cerebral white matter changes in poorly controlled patients with classical homocystinuria that is most likely due to hypermethioninemia. Additionally we speculated that such complication is more likely to occur in patients with the Pyridoxine non-responsive type, supported by previously reported cases [7,11,12,14]. Unlike other leukodystrophies white matter changes in most classical homocystinuria cases are reversible, so early detection is essential as it may prevent serious complications such as brain edema.
Conclusion
Our report supports the hypothesis of hypermethioninemia rather than Betaine treatment as the cause of the white matter changes. Hypermethioninemia was documented in all our patients with levels ranging between 600->1000umol/L. The high methionine levels are most likely due poor compliance with treatment, especially the diet. We observed in two of our patients that when blood methionine levels dropped because of better compliance, previously seen brain white matter changes were reversible.
Our study opens the door for further research for better understanding of the natural disease progression and the longterm sequel (e.g. cognitive and learning abilities) of white matter involvement especially in non-treated or missed cases. Moreover, studies should be conducted to determine if methionine levels correspond to the appearance of cerebral white matter changes, their progression and resolution./p>
We stress on the importance of family/patient education to improve compliance with medications and follow the dietary restriction. Clinicians should consider performing brain MRI images at diagnosis and follow up studies for patients with classical homocystinuria who have poor adherence to diet restriction with rising methionine levels even in the absence of new neurological findings.
References
- Cusworth DC, Dent CE (1969) Homocystinuria. Biochemical Journal 111(3): 1P-2P.
- Pagon RA, Adam MP, Ardinger HH, Wallace SE, Amemiya A, et al. (1993) Homocystinuria Caused by Cystathionine Beta-Synthase Deficiency Gene Reviews (R). University of Washington, Seattle University of Washington, Seattle, USA.
- Gan-Schreier H, Kebbewar M, Fang-Hoffmann J, Wilrich J, Abdoh G, et al. (2010) Newborn population screening for classic homocystinuria by determination of total homocysteine from Guthrie cards. J Pediatr 156(3): 427-432.
- El-Said MF, Badii R, Bessisso MS, Shahbek N, El-Ali MG, et al. (2006) A common mutation in the CBS gene explains a high incidence of homocystinuria in the Qatari population. Hum Mutat 27(7): 719.
- El Bashir H, Dekair L, Mahmoud Y, Ben-Omran T (2015) Neurodevelopmental and Cognitive Outcomes of Classical Homocystinuria: Experience from Qatar. JIMD Rep 21: 89-95.
- Mudd SH, Skovby F, Levy HL, Pettigrew KD, Wilcken B, et al. (1985) The natural history of homocystinuria due to cystathionine beta-synthase deficiency. Am J Hum Genet 37(1): 1-31.
- Vatanavicharn N, Pressman BD, Wilcox WR (2008) Reversible leukoencephalopathy with acute neurological deterioration and permanent residua in classical homocystinuria: A case report. J Inherit Metab 31(3): 477-481.
- Yap S, Naughten ER, Wilcken B, Wilcken DE, Boers GH (2000) Vascular complications of severe hyperhomocysteinemia in patients with homocystinuria due to cystathionine beta-synthase deficiency: effects of homocysteine-lowering therapy. Semin Thromb Hemost 26(3): 335- 340.
- Yap S, Rushe H, Howard PM, Naughten ER (2001) The intellectual abilities of early-treated individuals with pyridoxine-nonresponsive homocystinuria due to cystathionine beta-synthase deficiency. J Inherit Metab Dis 24(4): 437-447.
- Karimzadeh P, Jafari N, Alai M, Jabbehdari S, Nejad Biglari H (2015) Homocystinuria: Diagnosis and Neuroimaging Findings of Iranian Pediatric patients. Iran J Child Neurol 9(1): 94-98.
- Yaghmai R, Kashani AH, Geraghty MT, Okoh J, Pomper M, et al. (2002) Progressive cerebral edema associated with high methionine levels and betaine therapy in a patient with cystathionine beta-synthase (CBS) deficiency. Am J Med Genet 108(1): 57-63.
- Devlin AM, Hajipour L, Gholkar A, Fernandes H, Ramesh V, et al. (2004) Cerebral edema associated with betaine treatment in classical homocystinuria. J Pediatr 144(4): 545-548.
- Brenton JN, Matsumoto JA, Rust RS, Wilson WG (2014) White matter changes in an untreated, newly diagnosed case of classical homocystinuria. J Child Neurol 29(1): 88-92.
- Sasai H, Shimozawa N, Asano T, Kawamoto N, Yamamoto T, et al. (2015) Successive MRI Findings of Reversible Cerebral White Matter Lesions in a Patient with Cystathionine β-Synthase Deficiency. Tohoku J Exp Med 237(4): 323-327.
- Tada H, Takanashi J, Barkovich AJ, Yamamoto S, Kohno Y, et al. (2004) Reversible white matter lesion in methionine adenosyltransferase I/III deficiency Early-onset combined methylmalonic aciduria and homocystinuria: neuroradiologic findings. AJNR Am J Neuroradiol 25 (10): 1843-1845.






























