Physicochemical Studies on Some Coordination Biopolymer Compounds: Kinetics of Thermal Degradation and Heterogeneous Chemical Equilibrium of Ion Exchange for Coordination Biopolymer Zirconium (IV)- Alginate Complex
Refat M Hassan*
Department of Pharmaceutics, Delhi Pharmaceutical Sciences and Research University, India
Submission: October 14, 2019; Published: November 26, 2019
*Corresponding author: Refat M Hassan, Chemistry Department, Faculty of Sciences, Assiut University, Assiut, 71516 Egypt
How to cite this article: Refat M Hassan. Physicochemical Studies on Some Coordination Biopolymer Compounds: Kinetics of Thermal Degradation and Heterogeneous Chemical Equilibrium of Ion Exchange for Coordination Biopolymer Zirconium (IV)- Alginate Complex. Academ J Polym Sci. 2019; 3(3): 555612. DOI: 10.19080/AJOP.2019.03.555612
Abstract
Thermogravimetry (TG) and differential thermogravimetry (DTG) techniques have been applied for studying the kinetics of thermal degradation of coordination biopolymer zirconium (IV)- alginate complex in static air. The DTG curves indicated the presence of a series of thermal peaks associated with three distinct stages of TG curves, i.e. three stages of degradation were observed. The first stage of degradation was corresponding to the dehydration of four coordinated water molecules in the first step, followed by degradation of the formed dehydrated compound in the other two following stages. The kinetic parameters were computed by a set of different models and a tentative degradation mechanism consistent with the kinetic observations is discussed. In addition, the heterogeneous chemical equilibrium for ion exchange process of Zr4+ counter ions chelated in the biopolymer coordination complex by exchangeable H+ ions of acid electrolyte have been investigated by using both the complexometric and titrimetric techniques. The kinetic and thermodynamic parameters have been evaluated and discussed in terms of the complex stability related to the strength of chelation and geometrical structure of coordination complex.
Keywords: Macro Molecules; Nanoparticles; Suspension; Emulsions; Polymers; Toxicity
Abbreviations: NCPs: Cationic Polymers; DEX: Cationic Dextran; PLL: Poly-L-lysine; WHO: World Health Organization; PEG: Polyethylene Glycol; PDMAEMA: Poly(2-N,N-dimethylaminoethylmethacrylate)
Introduction
Alginate polysaccharide is a water-soluble natural polymer comprising of a D-mannuronic and some L-guluronic acid units linked through β (1-4) position in a linear block copolymer structure [1,2]. Alginate has a high affinity for chelation with silver (I), divalent and polyvalent metal ions through formation of partially coordinate and partially ionic bonds between the metal cations and the carboxylate and hydroxyl functional groups of alginate macromolecular chains, respectively, forming the corresponding metal alginate coordination biopolymer complexes of an egg-carton like structure [3]. Alginate and its derivatives have wide applications in the pharmaceutics and biomedicine industries such as drug delivery and tissues engineering especially in cell encapsulation for tissue organ replacement as well as in food industry owing to its non-toxicity, biocompatibility, biodegradability and hydrophilivity nature [4]. Recently, it is a promising inhibitor for inhibition of metal corrosion [5]. In order to deduce the decomposition mechanism and the construction of the kinetic models from the obtained kinetic parameters, extensive knowledge is necessary from previous studies on similar tetravalent coordination biopolymer compounds. However, there are a few reports on the kinetics of thermal decomposition of some cross-linked monovalent-, divalent- and hexavalent- metal alginate complexes [6-9], it seems that a little attention has been focused to study metal-alginate complexes involving tetravalent metal ions [6]. Moreover, the detailed information on the chemical equilibria for exchange in studied complexes still remains unclear and incomplete [10,11]. In view of the above argument, we have prompted to undertake the present work with a view at shedding some light on the stability of this complex in terms of strength of chelation and model structures. Moreover, the present study aims to relate the composition, structure for selection the complexes which may be applied in electronic devices as semiconducting materials [12-15]..
Experimental
Materials
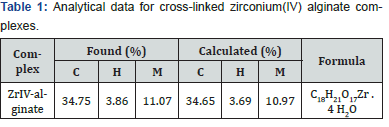
All materials used were of Analar quality (BDH). The degree of substitution of sodium alginate used was found to be 4.34 mmol g-1 (0.95 mol mol-1). Doubly distilled conductivity water was used in all preparations. Preparation of metal-alginate coordination biopolymer complexes Coordination biopolymer Zr (IV)-alginate complex in the form of granules form was prepared by replacement of Na+ counter ions of alginate macromolecule by Zr4+ ions. This process was carried out by stepwise addition of the alginate powder reagent to an electrolyte solutions of Zr(IV) metal ion (Ca. 0.5- 1.0 M) whilst stirring the solutions vigorously in order to avoid the formation of lumpy gelatinous precipitate of metal- alginate jellies, which swell with difficulty. After completion of exchange processes, these formed metal-alginate granules were washed with deionized water several times, then with doubly distilled water until the washings become free from Zr+4 metal ions. The sample complexes were dried at about 105 °C under vacuum over anhydrous CaCl2 or P2O5. FTIR spectra were recorded on a Pye unicam Sp 3100 spectrophotometer using the KBr disc technique (4000-200 cm-1). The elemental analysis for the cited cross linked complex is listed in (Table 1).
Thermal Decompositions Measurements
Dynamic thermogravimetry of non-isothermal method was used to study the thermal decomposition of the present work rather than by conventional isothermal studies, owing to the advantages of the former one in our case [6]. Thermogravimetry (TG) and differential thermogravimetry (DTG) analyses were carried out using a Mettler TA 3000 thermal analyzer in static air. Three heating rates were used 5, 10 and 20 K/min. A composite program was used for best fit analyses.
Chemical Equilibrium Measurements
Mixtures containing stoichiometric ratios of the metal alginate complex and aqueous HClO4 of known concentrations were thermally equilibrated in a constant temperature water-bath at the desired temperature within ± 0.1 o C with continuous stirring using a magnetic stirrer. After equilibrium had been attained (24h), clear solutions containing both metal and hydrogen ions were pipetted out and the concentrations of the metal ion and hydrogen ions were determined by both complexometric and titrimetric techniques, respectively [11,12]. All cited data were an average of five experimental runs. The ionic strength of the mixture was maintained constant at 0.1 mol dm-3 by adding NaClO4 as non-complexing agent. Some experimental runs were performed using spectrophotometric and pH-metric techniques to check the producibility of the data obtained from the complexometric and titrimetric methods, respectively. The results obtained were found to be in good agreement with each other within the experimental errors (± 6%). This result indicates the reproducibility of the data obtained by the applied cited techniques. In order to examine the temperature-dependency of the equilibrium constants and to evaluate the thermodynamic parameters, some experimental runs were performed at various temperatures with keeping all other reagents concentration constant.
Result and Discussion
The replacement of sodium counter ions, Na+, of alginate macromolecule by tetravalent metal ion of the electrolyte solutions can be expressed by the following exchange process
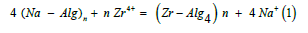
powder electrolyte complex electrolyte
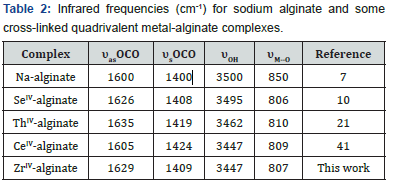
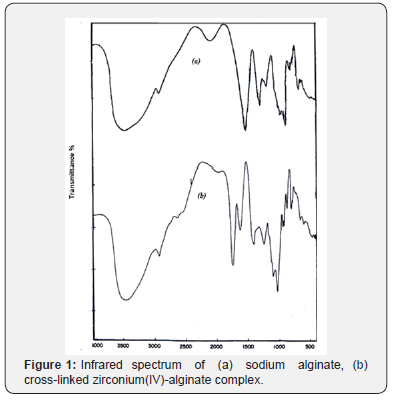
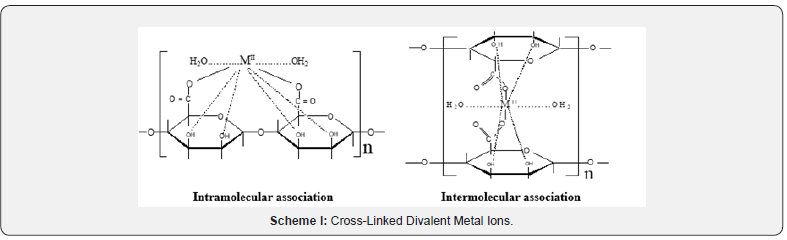
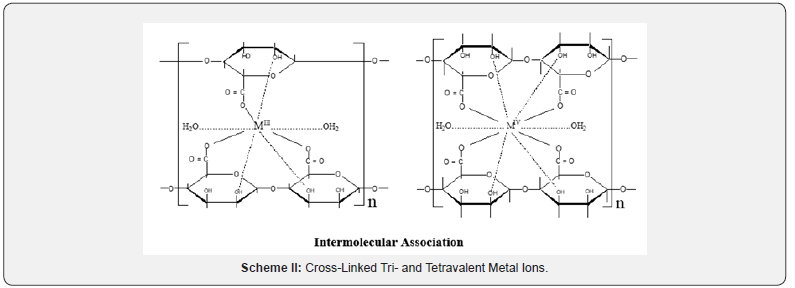
This exchange process is inherently a stoichiometric process [16]. Any Na+ counter ions which leave the alginate macromolecular chains must be compensated by an equivalent amount of the metal ions, even if the nobilities and valences of the two exchangeable counter ions are quite different such as in our case [17,18]. This is the necessary demands of electroneutrality. Infrared spectrum of the present coordination biopolymer complex has been measured and the vibration assignments of the bands are cited in (Table 2). It is well known that the bands correspond to the symmetry stretching vibrations of the –OCO- (υs) in sodium alginate lie at 1400 cm-1 and that of the asymmetric stretching vibrations (υas) lie at 1600 cm-1, respectively [7,19]. Again, a broad band located near 3500 cm-1 is owing to the stretching vibration of the hydrogen bond of water or the hydroxyl group. The free ligand has a strong band in the 1735 cm-1. As shown in (Figure 1), the band of the stretching vibrations of -OCO- group is shifted from 1400 cm-1 (υs) to 1409 cm-1 whereas the band at 1600 cm-1 (υas) is shifted to 1629 cm-1, respectively. This result indicates the participation of- OCO- groups in the coordination. While, the enhancement in the υOH band to lower frequencies which became more broader than that of sodium alginate may also indicate the sharing of OH groups in the chelation process. Furthermore, the geometry of the model structure of coordination may play a role in the stability of the formed metal alginate complex. It is reported [20] that polyvalent metal ions chelate the functional groups, -COO- and OH, of alginate macromolecule via either intra- or intermolecular association mechanisms depending on the nature, valance and coordination number of the chelated metal ions that allow the complex to be of more stability. Divalent metal ions may use either inter- or intramolecular association mechanism in chelation of two carboxylate groups and one or more pairs of hydroxyl groups depending on the coordination number of the chelated metal ion (Scheme I). On the other hand, trivalent and tetravalent metal ions are restricted to use only intermolecular association through chelation of three or four carboxylates and one or more pairs of the hydroxyl groups for geometrical reasons (Scheme II). This fact may be explained by the tendency of the trivalent and tetravalent metal ions to decrease the bond stretching resulting from elongation of metal-oxygen bonds in case of intramolecular association.
The thermal decomposition curve of the studied complex is shown in (Figure 2). The TG curves exhibit three stages of weightloss, the DTG curve indicate the presence of a series of thermal decomposition associated with the TG curve. This behavior was found to be quite different from that of sodium alginate macromolecule [6,7]. This difference can be interpreted by the difference in the strength of chelation of the partially ionic bonds formed between the zirconium ion and carboxylate groups in the complex and that of sodium ion in case of sodium alginate form. In addition, the presence of partially coordinate bonds between zirconium ion and the hydroxyl groups of the macromolecular chains in case of metal alginate complex; while it is not in case of sodium alginate. The essential features of solid-state decomposition are the destruction of the crystal lattice of the reactants, the breaking or redistribution of the chemical bonds and the formation of the crystal lattice of the decomposition products. In view of these aspects and our experimental observations, a tentative mechanism in good consistent with the thermal decomposition analysis applied for selenium (IV) [11] and thorium (IV) [21] complexes may be suggested. The weight loss observed in the first stage in the TG curves (Table 3) can be explained by the evolution of four coordinated water molecules as follows,
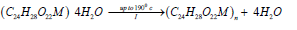
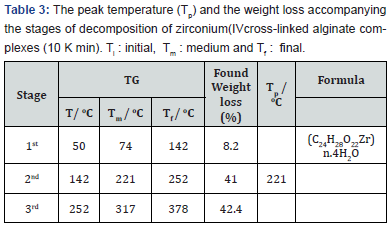
The weight losses associated with the second and third stages of the thermal decomposition (Table 3) may indicate the formation of transient intermediate fragments, followed by fast degradation of the transient species to form the carbonates intermediate fragments in both for ZrIV-alginate complex as follows.
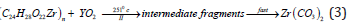
where C24H28O22 corresponds to four monomers of alginate macromolecule chelated with the Zr (IV) metal ion. The formation of carbonate intermediate was also followed by a fast decomposition as the final stage to give rise to the corresponding metal oxide as final product as follows,
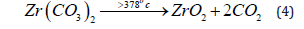
Dealing with the non-isothermal kinetics for the TG curves, one can predict the kinetic mechanism by which the decomposition reaction takes place. Hence, application of thermo analytical techniques for coordinated biopolymeric complexes such as is the present work is an example to study of the bonding and mechanism of decomposition, while the thermogravimetry determines the kinetics. There are different methods for kinetic analysis of thermogravimetric data which can be carried out by the thermal and dynamic methods [22-33]. The kinetic model functions g(α) were examined here for each decomposition step using Sharp method [32]. In this non-isothermal method, the reactions proceed in infinitesimal time intervals and, hence, the rates can be expressed by the Arrhenius type equation

where Z is the frequency factor, t is the time, α is the decomposition fraction and F(α) depends on the mechanism of the process. Sharp and coworkers [32] suggested a reduced time theoretical method in order to distinguish between the different methods where the time scale in the kinetic equation is
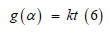
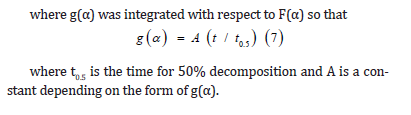
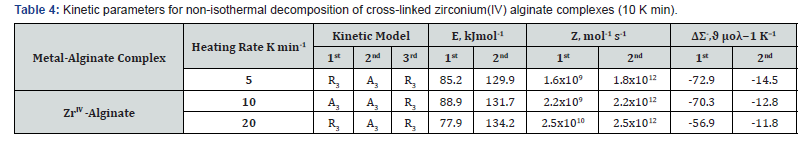
The t0.5 values corresponding to α = 0.5 were determined from the experimental data to curves of the form α (t / t0.5) plots. The results data were analyzed by linear regression analysis for various kinetic model functions [28], The best fitting models for the decomposition steps for the investigated samples are summarized in (Table 4). The table shows that both the phase boundary (R3) and the random nucleation (A3) give the best fit for the first dehydration and second decomposition steps with comparable correlation coefficients of first-order kinetics. The A3- function indicated that the nucleation of the thermal decomposition compound is a random process followed by a rapid surface growth. As nuclei grow larger, they may eventually impinge on one another, so that growth ceases where they touch. On the other hand, R3-function showed that the reaction is controlled by movement of interface at constant velocity and, hence, the phase decomposition is the phase-boundary controlled reactions. In general, the different kinetic mechanisms obtained indicates that the nature of chelated metal ions and the geometrical model structure of the complex may control the kinetic parameters of decomposition for the given alginate complex. However, several methods can be used for calculation of the kinetic parameters from non-isothermal TG data; the modified Coasts-Redform method [33] was generally used from the viewpoints of computation. This method is the most convenient one and seems to be less tedious and has the advantages of leading for more accurate values of the kinetic parameters. According to this method, three heating rates 5, 10 and 20 K/min were employed using a small mass (~ 10 mg) to obtain reliable kinetic parameters. The activation energy (Ea) can be calculated from the following relationship)
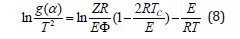
where Z denotes the frequency factor, Φ is the heating rate, T is the absolute temperature, Tc is the maximal temperature decomposition rate, and R is the gas constant. For correct kinetic rate equation, plots of in g(α)/T2 against 1/T should be linear as was observed experimentally. The activation energy (Ea) and frequency factor (Z) can be determined from the slopes and intercepts of such plots, respectively. These values were calculated by using the method of least-squares and are summarized in (Table 4). Again, the entropies of activations, ΔS*, were calculated from the following relationship
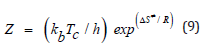
where kb is the Boltzmann’s constant and h is the Plank’s constant [29]. The results are summarized in (Table 4). The negative values of entropy of activations at the initial dehydration step indicate that the reactants are less ordered than that of the activated complex formed during the dehydration processes and vice versa for complexes of positive Δs≠ values. Hydrogen ions were used for exchange purpose in the present study owing to the exchange simplicity and in order to avoid the complications of the suggested equations in particularly in calculations. The exchange equilibria between tetravalent metal ions and hydrogen ions conform to the following equation
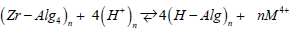
solid aqueous solid aqueous Applying he law of mass-action for such heterogeneous systems, one concludes that
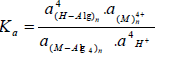
where a`s are the activities of the constituents and Ka is the true thermodynamic equilibrium constant, respectively. Eisenman, Izmailav and Mushinskaya [34,35] postulated an approximate equation to calculate the thermodynamic equilibrium constant, Ka, which varies with the composition of the solid phase. Using the Gibbs-Duhem relationship for ions in the solid phase and considering that the activity of water in the solid phase remains constant, the following equation is obtained.
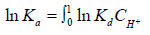
where C is the concentration of the counter ion in appropriate units. Plotting ln K against CH+ and extrapolated to CH+ = 0, gives ln K can be obtained. Since our experimental observations showed no appreciable variations in equilibrium constant values within variation of hydrogen ion concentration, no practical advantage on using such treatment [12,36,37]. Consequently, assuming that the mass action constant is not varied with the concentration of the interacting ions in order to avoid the complexity in selection of suitable procedure for evaluating the thermodynamic equilibrium constant, and considering that the activities of the solid phases are unity [38], Eq. (11) can be rewritten in the form,
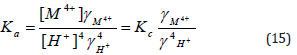
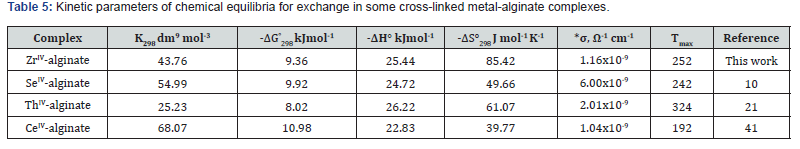
Since the activity coefficients were not available at various temperatures, the thermodynamic chemical equilibria, Ka, could not be evaluated. Then, assuming that the activity coefficients under our experimental conditions of lower electrolyte concentrations of counter ions to be unity [38,39] and, hence, the values of Kc will be approximately equal to that of Ka. The values of Kc were calculated using the least-squares method as shown in (Table 5). The thermodynamic parameters of Kc values were evaluated from the temperature dependence of the equilibrium constants and are summarized along with that of other of tetravalent metal-alginate complexes in (Table 5). The magnitude of the equilibrium constants and the maximal temperature decomposition rates (Table 5) may be considered as essential factors for determining the stability of these metal alginate complexes. The observed values reveal that the stability follows the following order
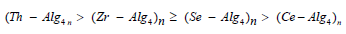
This order agrees with the magnitude of M-O bond energies [40-45], and their coordination geometry [20].
References
- Raina N, Rani R, Khan A, Nagpal K, Gupta M (2019) Interpenetrating polymer network as a pioneer drug delivery system: a review. Polymer Bulletin: 1-24.
- Mody VV (2010) Introduction to polymeric drug delivery. Internet Journal of Medical Update 5(2): 1-2.
- Deb J, Das M, Das A (2017) Excellency of natural polymer in drug delivery system: A Review. International Journal of Pharmaceutical and Biological Science Archive 5(1).
- Vyas SP, Khar RK (2002) Controlled drug delivery concepts and advances. vallabh prakashan 1: 411-47.
- Hiwale P, Lampis S, Conti G, Caddeo C, Murgia S, et al. (2011) In vitro release of lysozyme from gelatin microspheres: effect of cross-linking agents and thermoreversible gel as suspending medium. Biomacromolecules 12(9): 3186-3193.
- Dash M, Chiellini F, Ottenbrite RM, Chiellini E (2011) Chitosan-A versatile semi-synthetic polymer in biomedical applications. Progress in polymer science 36(8): 981-1014.
- Song Y, Zhang L, Gan W, Zhou J, Zhang L (2011) Self-assembled micelles based on hydrophobically modified quaternized cellulose for drug delivery. Colloids and Surfaces B: Biointerfaces 83(2): 313-320.
- Xu FJ, Ping Y, Ma J, Tang GP, Yang WT, et al. (2009) Comb-shaped copolymers composed of hydroxypropyl cellulose backbones and cationic poly ((2-dimethyl amino) ethyl methacrylate) side chains for gene delivery. Bioconjugate chemistry 20(8): 1449-1458.
- Alibolandi M, Mohammadi M, Taghdisi SM, Ramezani M, Abnous K (2017) Fabrication of aptamer decorated dextran coated nano-graphene oxide for targeted drug delivery. Carbohydrate polymers 155: 218-29.
- Ko NR, Cheong J, Noronha A, Wilds CJ, Oh JK (2015) Reductively-sheddable cationic nanocarriers for dual chemotherapy and gene therapy with enhanced release. Colloids and Surfaces B: Biointerfaces 126: 178-187.
- Zauner W, Ogris M, Wagner E (1998) Polylysine-based transfection systems utilizing receptor-mediated delivery. Advanced drug delivery reviews 30(1-3): 97-113.
- Arora A, Mishra A (2018) Antibacterial polymers–a mini review. Materials Today: Proceedings 5(9):17156-17161.
- (2015) WHO. Who, p. 1-44.
- Neu HC (1992) The crisis in antibiotic resistance. Science 257(5073): 1064-1073.
- Yu K, Mei Y, Hadjesfandiari N, Kizhakkedathu JN (2014) Engineering biomaterials surfaces to modulate the host response. Colloids and Surfaces B: Biointerfaces 124: 69-79.
- Li B, Ye Q (2015) Antifouling Surfaces of Self-assembled Thin Layer. In Antifouling Surfaces and Materials, Springer, Berlin, Heidelberg, p. 31-54.
- Xue Y, Xiao H, Zhang Y (2015) Antimicrobial polymeric materials with quaternary ammonium and phosphonium salts. International journal of molecular sciences 16(2): 3626-3655.
- Jain A, Duvvuri LS, Farah S, Beyth N, Domb AJ, et al. (2014) Antimicrobial polymers. Advanced healthcare materials 3(12):1969-85.
- Ghanbarzadeh B, Almasi H (2013) Biodegradable polymers. Biodegradation-life of science. In Tech Publications, Croatia 5:141-186.
- Martins AF, Facchi SP, Follmann HD, Pereira AG, Rubira AF, et al. (2004) Antimicrobial activity of chitosan derivatives containing N-quaternized moieties in its backbone: a review. International Journal of Molecular Sciences 15(11): 20800-20832.
- Siedenbiedel F, Tiller JC (2012) Antimicrobial polymers in solution and on surfaces: overview and functional principles. Polymers 4(1): 46-71.
- Cheng X, Li R, Du J, Sheng J, Ma K, et al. (2015) Antimicrobial activity of hydrophobic cotton coated with N‐halamine. Polymers for Advanced Technologies 26(1): 99-103.
- Li X, Liu Y, Jiang Z, Li R, Ren X, Huang TS (2015) Synthesis of an N-halamine monomer and its application in antimicrobial cellulose via an electron beam irradiation process. Cellulose 22(6): 3609-3617.
- Beyth N, Farah S, Domb AJ, Weiss EI (2014) Antibacterial dental resin composites. Reactive and Functional Polymers 75: 81-88.
- Locock KE, Michl TD, Valentin JD, Vasilev K, Hayball JD, et al. (2013) Guanylated polymethacrylates: a class of potent antimicrobial polymers with low hemolytic activity. Biomacromolecules 14(11): 4021-4031.
- Gizdavic NMR, Bennett JR, Swift S, Easteal AJ, Ambrose M (2011) Broad spectrum antimicrobial activity of functionalized polyanilines. Acta biomaterialia 7(12): 4204-4209.
- Sambhy V, Peterson BR, Sen A (2008) Antibacterial and hemolytic activities of pyridinium polymers as a function of the spatial relationship between the positive charge and the pendant alkyl tail. Angewandte Chemie International Edition 47(7): 1250-1254.
- Ilker MF, Nüsslein K, Tew GN, Coughlin EB (2004) Tuning the hemolytic and antibacterial activities of amphiphilic polynorbornene derivatives. Journal of the American Chemical Society 126(48): 15870-15875.
- Budhathoki Uprety J, Novak BM (2011) Synthesis of alkyne-functionalized helical polycarbodiimides and their ligation to small molecules using ‘click’and sonogashira reactions. Macromolecules 44(15): 5947-5954.
- Palermo EF, Kuroda K (2010) Structural determinants of antimicrobial activity in polymers which mimic host defense peptides. Applied microbiology and biotechnology 87(5): 1605-1615.






























