Synthesis and Radical Copolymerization of Novel Phenyl-Disubstituted Propyl Cyanoacrylates
Christopher Savittieri R, Shechinah Tinsley M, Andrew Diehn J, Fatema Hai, Kara Humanski E, Eric Kempke J, Benjamin Killam Y, Jimmy Kozeny, Evana Makhoul W, Molly Obert C, Anthony Parisi C, Victoria Parrilli C and Gregory Kharas B*
Department of Chemistry, DePaul University, USA
Submission: May 06, 2019; Published: May 13, 2019
*Corresponding author: Gregory Kharas B, Department of Chemistry, DePaul University, Chicago, IL, USA
How to cite this article: Christopher Savittieri R, Shechinah Tinsley M, Andrew Diehn J, Fatema Hai, Kara Humanski E, Gregory Kharas B, et al. Synthesis and Radical Copolymerization of Novel Phenyl-Disubstituted Propyl Cyanoacrylates. Academ J Polym Sci. 2019; 2(5): 555597.DOI: 10.19080/AJOP.2019.02.555597
Abstract
Novel phenyl-disubstituted propyl 3-(R-phenyl)-2-cyanoacrylates, RPhCH=C(CN)CO2C3H7 (where R is 2-fluoro-5-methyl, 3-iodo-4-methoxy, 5-iodo-2-methoxy, 3,5-dichloro, 3,4-difluoro, 3,5-difluoro, 2-chloro-4-fluoro, 2-chloro-6-fluoro, 3-chloro-2-fluoro, and 3-chloro-4-fluoro) were prepared using condensation of substituted benzoic aldehydes and propyl ester of cyanoacetic acid. The ethynyl benzene copolymerization of novel cyanoacrylates was conducted in solution with radical initiation at 70 C. Nitrogen analysis, 1H & 13C NMR was used to analyze composition and the structure. Thermal behavior of the copolymers was analyzed by DSC and TGA.
Keywords: Radical copolymerization; Styrene copolymers; Trisubstituted ethylene’s; Cyanoacrylates
Introduction
Ring–functionalized trisubstituted ethylene’s (TSE), esters of 3-phenyl-2-cyanoacrylates, R1PhCH=C(CN) CO2R2 continue to attract attention as compounds with interesting properties and as comonomers for modification of commercial polymers. 3,4-Difluorophenyl substituted methyl 3-phenyl-2-cyanoacrylate was used in synthesis and studies of histamine H2 agonistic activity [1]. Ethyl 3-phenyl-2-cyanoacrylate was used in studies of stereoselective cascade assembling of (1R*,2S*)-1-cyano-5,7-dialkyl-4,6,8-trioxo-2-aryl-5,7-diazaspiro[2.5]octane-1-carboxylates [2] and analgesic activity of 6-fluoroindan-1-carboxylic acid [3]. Alkyl 2-cyanoacrylates are a family of vinyl monomers renowned for their high reactivity, instant adhesive properties, and wide-ranging applications [4].
We have reported synthesis and ethynylbenzene copolymerization of similar ring-substituted methyl [5], ethyl [6], & butyl 3-phenyl-2-cyanoacrylates [7]. With the objective to design novel structures, that could serve as a spring board for further development of novel materials with new properties and applications, we have prepared halogen ring-disubstituted propyl 3-phenyl-2-cyanoacrylate (PPCA), RPhCH=C(CN)CO2C3H7sub>, where R is 2-fluoro-5-methyl, 3-iodo-4-methoxy, 5-iodo-2-methoxy, 3,5-dichloro, 3,4-difluoro, 3,5-difluoro, 2-chloro-4-fluoro, 2-chloro-6-fluoro, 3-chloro-2-fluoro, and 3-chloro-4-fluoro, and copolymerize them with ethynylbenzene. To the best of our knowledge, there have been no reports on either synthesis of these propyl phenyl cyanoacrylates, nor their copolymerization with ethynylbenzene.
Experimental
All benzoic aldehydes, propyl cyanoacetate, piperidine, ethynylbenzene, 1,1’-azobiscyclohexanecarbonitrile, (ABCN), and toluene supplied from Sigma-Aldrich Co., were used as received. The melting points and Tg, were measured with TA Model Q10 DSC. The thermal stability of the copolymers was measured by TGA TA Model Q50 from ambient temperature to 800 ºC at 20 ºC/min. The MW of the copolymers was determined by GPC using an Altech 426 HPLC pump at an elution rate of 1.0mL/min; Phenogel 5μ Linear column at 25 ºC and Viscotek 302 detector. 1H & 13C NMR spectra were obtained on 10-25% (w/v) monomer or polymer solutions in CDCl3sub> at ambient temperature using Avance 300MHz spectrometer. Elemental analyses were performed by Midwest Microlab, LLC (IN).
Results and Discussion
PPCA synthesis
The ring-substituted propyl 3-phenyl-2-cyanoacrylates (PPCA) were synthesized by Knoevenagel condensation [8] of a ring-substituted benzoic aldehyde with propyl cyanoacetate, catalyzed by base, piperidine (1).
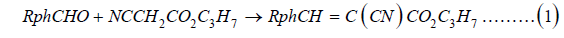
Where R is 2-fluoro-5-methyl, 3-iodo-4-methoxy, 5-iodo- 2-methoxy, 3,5-dichloro, 3,4-difluoro, 3,5-difluoro, 2-chloro- 4-fluoro, 2-chloro-6-fluoro, 3-chloro-2-fluoro, & 3-chloro-4- fluoro. The preparation procedure was essentially the same for all the monomers. In a typical synthesis, equimolar amounts of propyl cyanoacetate and an appropriate ring-substituted benzoic aldehyde were mixed in equimolar ratio in a 20mL vial. A few drops of piperidine were added with stirring. The product of the reaction was isolated by filtration and purified by crystallization from 2-propanol. The condensation reaction proceeded smoothly, yielding products, which were purified by conventional techniques.
Yield 87%; 1H-NMR 8.4 (s, 1H, CH=), 8.2-6.9 (m, 3H, Ph),
4.3 (t, 2H, OCH2sub>), 2.3 (s, 3H, CH3), 1.7 (m, 2H, OCH2CH2sub>), 1.0 (t,
3H, OCH2CH2CH3sub>); 13C-NMR 163 (C=O), 152 (HC=), 138, 135,
130, 121, 115 (Ph), 116 (CN), 104 (C=), 67 (OCH2sub>), 22 (OCH2CH2sub>),
21 (CH3sub>), 10 (OCH2CH2CH3sub>); Anal. Calcd. for C14H14
FNO2: C, 68.00;
H, 5.71; N, 5.66; Found: C, 67.85; H, 5.75; N, 5.86.
Propyl 3-(3-iodo-4-methoxyphenyl)-2-cyanoacrylate
Yield 92%; mp 140°C, 1H-NMR 8.3 (s, 1H, CH=), 8.2 -6.7
(m, 3H, Ph), 4.3 (t, 2H, OCH2sub>), 4.0 (s, 3H, OCH3sub>), 1.8 (m, 2H,
OCH2CH2), 1.0 (t, 3H, OCH2CH2CH3sub>); 13C-NMR 163 (C=O),
154 (HC=), 128, 118, 88 (Ph), 116 (CN), 100 (C=), 67 (OCH2),
56 (OCH3), 22 (OCH2CH2), 10 (OCH2CH2CH3); Anal. Calcd. for
C14H14INO3: C, 45.30; H, 3.80; N, 3.77; Found: C, 44.47; H, 3.72;
N, 3.33.
Propyl 3-(5-iodo-2-methoxyphenyl)-2-cyanoacrylate
Yield 95%; mp 93 °C, 1H-NMR 8.2 (s, 1H, CH=), 7.4-6.7 (m,
3H, Ph), 4.3 (t, 2H, OCH2), 3.9 (s, 3H, OCH3), 1.8 (m, 2H, OCH2CH2sub>),
1.1 (t, 3H, OCH2CH2CH3); 13C-NMR 163 (C=O), 152 (HC=),
122, 115, 80 (Ph), 116 (CN), 111 (C=), 67 (OCH2), 56 (OCH3),
22 (OCH2CH2sub>), 10 (OCH2CH2CH3); Anal. Calcd. for C14H14INO3: C,
45.30; H, 3.80; N, 3.77; Found: C, 44.78; H, 3.87; N, 4.05.
Propyl 3-(3,4-difluorophenyl)-2-cyanoacrylate
Yield 46%; mp 74 °C, 1H-NMR 8.1 (s, 1H, CH=), 7.9-7.0
(m, 3H, Ph), 4.3 (t, 2H, OCH2), 1.7 (m, 2H, OCH2CH2), 1.0 (t, 3H,
OCH2CH2CH3sub>); 13C-NMR 163 (C=O), 153 (HC=), 150, 130,
125, 118 (Ph), 116 (CN), 109 (C=), 67 (OCH2), 22 (OCH2CH2sub>), 10
(OCH2CH2CH3sub>); Anal. Calcd. for C13H11F2NO2: C, 62.15; H, 4.41; N,
5.58; Found: C, 61.97; H, 4.62; N, 5.54.
Propyl 3-(3,5-difluorophenyl)-2-cyanoacrylate
Yield 82%; mp 76 °C, 1H-NMR 8.1 (s, 1H, CH=), 7.7-6.7
(m, 3H, Ph), 4.3 (t, 2H, OCH2), 1.8 (m, 2H, OCH2CH2), 1.0 (t, 3H,
OCH2CH2CH3sub>); 13C-NMR 162 (C=O), 153 (HC=), 163, 145,
135 (Ph), 115 (CN), 103 (C=), 67 (OCH2sub>), 22 (OCH2CH2), 10
(OCH2CH2CH3); Anal. Calcd. for C13H11F2NO2: C, 62.15; H, 4.41; N,
5.58; Found: C, 61.25; H, 4.40; N, 5.79.
Propyl 3-(2-chloro-4-fluorophenyl)-2-cyanoacrylate
Yield 88%; mp 80°C, 1H-NMR 8.1 (s, 1H, CH=), 7.9-6.7
(m, 3H, Ph), 4.3 (t, 2H, OCH2sub>), 1.7 (m, 2H, OCH2CH2), 1.0 (t, 3H,
OCH2CH2CH3); 13C-NMR 164 (C=O), 152 (HC=), 163, 156,
132, 113 (Ph), 115 (CN), 99 (C=), 67 (OCH2), 21 (OCH2CH2), 10
(OCH2CH2CH3sub>); Anal. Calcd. for C13H11ClFNO2sub>: C, 58.33; H, 4.14; N,
5.23; Found: C, 59.46; H, 4.35; N, 5.58.
Propyl 3-(3-chloro-2-fluorophenyl)-2-cyanoacrylate
Yield 73%; mp 53 °C, 1H-NMR 8.5 (s, 1H, CH=), 8.3-7.2
(m, 3H, Ph), 4.3 (t, 2H, OCH2), 1.7 (m, 2H, OCH2CH2), 1.0 (t, 3H,
OCH2CH2CH3); 13C-NMR 163 (C=O), 152 (HC=), 162, 153, 130,
126, 119, 116 (Ph), 115 (CN), 92 (C=), 67 (OCH2sub>), 21 (OCH2CH2),
10 (OCH2CH2CH3); Anal. Calcd. for C13H11ClFNO2: C, 58.33; H,
4.14; N, 5.23; Found: C, 58.17; H, 4.21; N, 5.25.
Propyl 3-(3-chloro-4-fluorophenyl)-2-cyanoacrylate
Yield 84%; mp 118°C, 1H-NMR 8.2 (s, 1H, CH=), 8.1-7.2
(m, 3H, Ph), 4.3 (t, 2H, OCH2), 1.7 (m, 2H, OCH2CH2), 1.0 (t, 3H,
OCH2CH2CH3sub>); 13C-NMR 163 (C=O), 154 (HC=), 153, 130, 128,
120, 117 (Ph), 115 (CN), 92 (C=), 67 (OCH2), 21 (OCH2CH2), 10
(OCH2CH2CH3); Anal. Calcd. for C13H11ClFNO2: C, 58.33; H, 4.14; N,
5.23; Found: C, 58.18; H, 4.12; N, 5.10.
Copolymerization
The PPCA monomers when mixed with ethynylbenzene,
EB at EB/PPCA = 3 (mol) formed copolymers as indicated by
white flaky precipitates in high excess of methyl alcohol. The
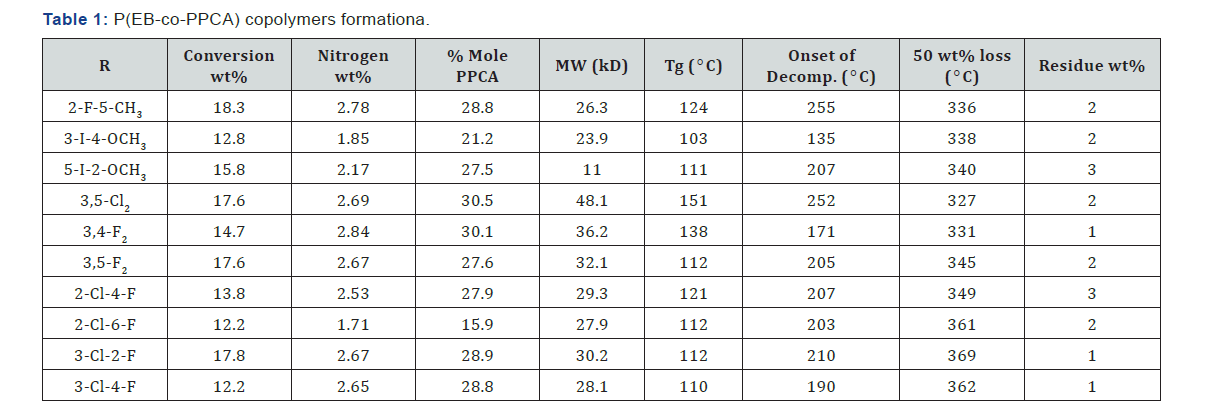
conversion of the EB-PPCA copolymers was maintained 10 - 20%
to decrease compositional drift (Table 1). The PPCA content 8.4-
25.8 mol% in the copolymers indicated relatively high reactivity
of the PPCA monomers towards EB radical Figure 1.
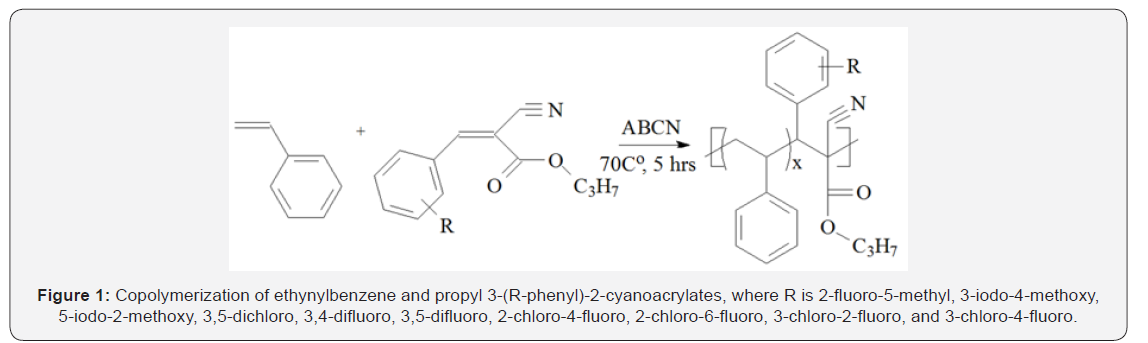
Note: aEB/PPCA: 3 (mol) / methyl benzene / 70C / 5hrs. bby GPC.
The EB-PPCA copolymers were soluble in tetrahydrofuran,
ethyl acetate, chloroform and dimethylformamide, and insoluble
in methyl alcohol, ethyl and petroleum ethers. GPC analysis the
copolymers indicated weight-average molecular masses 10.7-
27.8kD (Table 1). According to elemental analysis, between 18.3
& 42.6 mol% of PPCA monomer is present in the copolymers
prepared at EB/PPCA = 3 (mol), which is indicative of relatively
high reactivity of the monomers towards EB. Results of DSC and
TGA analyses are presented in Table 1.
The FTIR spectra of the monomers were compared with
those of copolymers and poly(ethynylbenzene) thus providing
evidence that the reaction between the PPCA monomers and
EB is a copolymerization. The spectra of the copolymers show
overlapping bands in 3200-2820cm-1 region corresponding to
C-H stretch vibrations. The bands for the PPCA monomer unit
are 2246-2238 (w, CN), 1752-1733 (s, C=O), and 1252-1226cm-1
(m, C-O). Benzene rings of both monomers show ring stretching
bands at 1500-1400cm-1 as well as a doublet 824-715cm-1,
associated with C-H out of plane deformations. These bands can
be readily identified in ethynylbenzene copolymers with TSE
monomers containing cyano and carbonyl electron withdrawing
groups.
EB-PPCA copolymers microstructure analysis is based on
1H and 13C NMR spectroscopy (DEPT, HETCOR, NOESY and
JMODXH) of EB copolymers with 2-phenyl-1,1-dicyanoethene
[9] which showed the formation of both head-to-tail and headto-
head alternating monomer structures, as well as short EB
sequences.
Conclusion
Novel phenyl-disubstituted propyl 2-cyano-3-phenyl-2-propenoates
were prepared and copolymerized with ethynylbenzene.
The compositions of the copolymers were calculated from
nitrogen analysis and the structures were analyzed by IR, H1 &
13C-NMR. The thermal gravimetric analysis indicated that the
copolymers decompose in in two steps, first in the 200-500C
range with residue (1-3%wt), which then decomposed in the
500-800 ºC range.
References
- Buschauer, Friese Kimmel A, Baumann G, Schunack W (1992) Synthesis
and histamine H2 agonistic activity of arpromidine analogues:
replacement of the pheniramine-like moiety by non-heterocyclic groups.
European Journal of Medicinal Chemistry 27(4): 321-330.
- Elinson MN, Vereshchagin AN, Korshunov AD, Ryzhkov FV, Egorov MP
(2017) Heterocyclic Communications 23: 85.
- Das S, Yasmin H, Mehedi MM, Roy SC, Nahar L, et al. (2008) Total synthesis
and analgesic activity of 6-fluoroindan-1-carboxylic acid. Tetrahedron
64(37): 8642-8645.
- Duffy C, Zetterlund PB, Aldabbagh F (2018) Radical Polymerization of
Alkyl 2-Cyanoacrylates. Molecules 23(2): 465
- Kharas GB, Chavez SE, Luna AN, Lusk EE, Mendez DP, et al. (2014) J
Macromol Sci Part A: Pure and Applied Chemistry 51: 1.
- Kharas GB, Molina ES, Fitzpatrick BM, Francis MC, Gallardo C, et al. (2013)
J Macromol Sci Part A: Pure and Applied Chemistry, 50: 370.
- Kharas GB, Schjerven WS, Maurer LM, McGee GC, McGovern ME, et al.
(2016) J Macromol Sci Part A: Pure and Applied Chemistry 53: 258.
- Smith MB, March J (2001) Addition to Carbon-Hetero Multiple Bonds,
In March’s Advanced Organic Chemistry J Wiley & Sons: New York,
Ch16, p. 1225.
- Kharas GB, Murau PA, Watson K, Harwood HJ (1992) Microstructure
of styrene copolymers with 2‐phenyl‐1,1‐dicyanoethene. Polym Int
28(1): 67-74.
































