Polyhydroxyalkanoates (Pha) And Polylactic Acid (Pla) Biodegradation by Anaerobic Digestion in Mesophilic Conditions
Janette Calvillo Solis1, Jérémie Vuong2 and Joseph Greene1*
1Department of Mechanical and Mechatronic Engineering and Sustainable Manufacturing, California State University, USA
2University of Technology of Compiegne, France
Submission: March 12, 2019; Published: May 07, 2019
*Corresponding author: Joseph Greene, Department of Mechanical and Mechatronic Engineering and Sustainable Manufacturing, California State University, Chico, 95926, California, USA
How to cite this article: Janette C S, Jérémie V, Joseph G. Polyhydroxyalkanoates (Pha) And Polylactic Acid (Pla) Biodegradation by Anaerobic Digestion in Mesophilic Conditions. Academ J Polym Sci. 2019; 2(4): 555595. DOI: 10.19080/AJOP.2019.02.555595
Keywords: Polyhydroxyalkanoates; Anaerobic digestion; Biodegradation; Biogas
Introduction
Plastics are a polymeric synthetic or semi-synthetic material polymeric material derived from carbon sources. Its production has incredibly risen since it has been invented because it is a cheap material with good mechanical and physical properties and is relatively easy to produce. The different properties of this compound made it a first-choice material for various utilizations and even replaced wood, cotton or metal for some use. However, its high biological resistance to degradation causes serious environmental problems [1]. More than 5.25 trillion of plastic materials weighting about 270,000 tons in total are dispersed into the oceans [2] while only 9% of the 32 million tons of plastic waste produced in the US every year are recycled [3].
However, there has been progress in the creation of biodegradable plastics, known as ecological plastics, however, this is still harmful for our atmosphere. The biodegradation of bioplastics must be controlled since it produces methane and carbon dioxide. Keeping this in mind, polyhydroxyalkanoates (PHA) and polylactic acid (PLA) fall under the same category of bioplastics. Therefore, both PHA and PLA can partially biodegrade under controlled conditions, which may prove itself useful in further studies [4-6].
Many progresses have been made by producing more ecological plastics able to degrade more easily. But the plastic biodegradation must be controlled because it produces methane which greenhouse effect is 21 times bigger than carbon dioxide [7]. Keeping this point in mind Polyhydroxyalkanoates (PHA) and Polylactic Acid (PLA) might be useful because they can be partially biodegraded in controlled condition [4-6]. Among those con ditions, anaerobic digestion in mesophilic conditions could allow PHA to biodegrade. Anaerobic digestion is a biological process in which biodegradable organic matter is broken down by microorganisms in the absence of oxygen into biogas. This process may contribute significantly to a sustainable bio-based society [8].
Polylactic acid is a viable alternative to petrochemical-based plastics for many applications. It is produced from renewable resources and is biodegradable, decomposing to give H2O, CO2, and humus, the black material in soil. PLA can biodegrade under composting condition at 58°C [5,6]. Polyhydroxyalkanoates (PHA) are polyesters of hydroxyalkanoates synthesized by numerous bacteria as intracellular carbon and energy storage compounds [9]. PHA can biodegrade in marine environment [10]. It is a very promising polymer for a wide range of applications [11]. For example, it has good barrier properties [12] like PVC and PET and can be used in the packaging industry [13]. But its commercialization is dissuaded by a high cost of production [11]. It could be reduced by the scientific community by undertaking more investigations about it [14].
Materials and methods
Laboratory experiment was conducted by mixing PHA with an inoculum coming from an industrial digester to study its biodegradability. One blank (only inoculum), a positive control, and a negative control were used for this experiment.
Materials
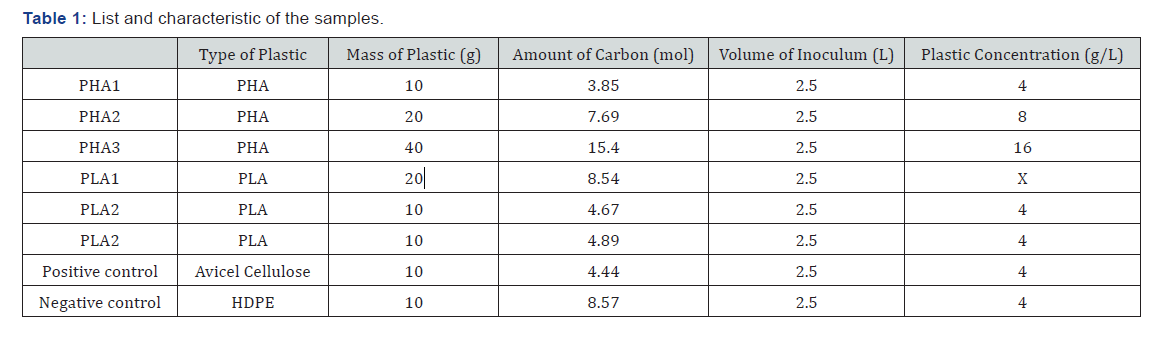
Material used was Polyhydroxyalkanotates (PHA) Mirel-4100 supplied by Metabolix (USA) and Polyactic acid (PLA) produced by Natureworks LLC (Blair, Nebraska). The carbon content in this PHA was 46.15%. Avicel PH-101 micro cellulose was used as a positive control and HDPE powder was used as a negative control. PHA and PLA were blended into 1-2mm wide pellets using a regular kitchen blender.
Inoculum
The inoculum (pH = 7.76; FOS = 1.08g/L; TAC = 6.49g/L [15] was stored one week at 37°C before the beginning of the experiment to let it degas. Methanogen archaea in this mixture are mainly Methanosarcina and Methanobrevibacter. The inoculum came from a two-stage mesophilic semi-continuous anaerobic digester owned by North State Rendering (Oroville, California). The plant was composed of two hydrolysis tanks and one digester. The inoculum was withdrawn on the bottom of the digester. The main feeding of this installation was cheese waste, olive waste and food waste.
Equipment’s
To run this experiment, we used:
a) six glass bottles (2.75 L each)
b) An incubator with an opening on the top to connect the bottles to an external sensor
c) A thermocouple (OMEGA, MODEL 660)
d) six PVC pipe closed with a PVC cap (2” wide & 2’6” long; Volume = 1.6L)
e) Gas Chromatograph SRI310
f) A pH meters
g) An electronic scale
h) Standard gas (15% Nitrogen, 35% Carbon dioxide, 50% Methane)
i) Biogas titration manager (HACH)
Biodegradation under mesophilic anaerobic digestion
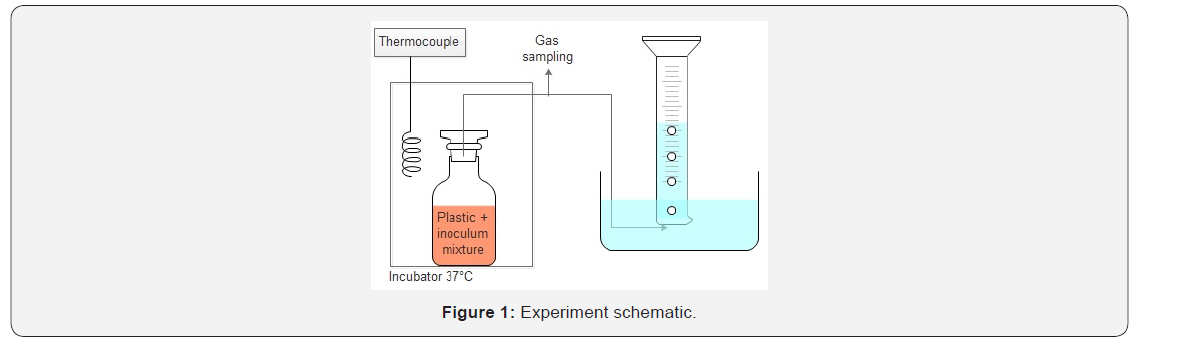
The method used was based on the Standard Test Method for Determining Anaerobic biodegradation of Plastic Materials Under High-Solids Anaerobic-Digestion Conditions ASTM 5511-02 [16]. The system temperature was set to 37°C (±2°C). To match the usual feeding rate at the North State Rendering plant the plastic concentration was chosen between 4g/L-16g/L. The usual concentration is 4g/L, but various concentrations were tested to observe the effect its effect on the degradation. The laboratory scale batch reactors were connected to a plastic pipe designed to collect the gas produced. The plastic pipes were sealed on one side and put upside down in a bucket filled with water. The pipe was filled with water every time it was full of gas. Hydrochloric acid was added to the water to keep the pH under 2.0 to avoid the carbon dioxide to dissolve into it. At the beginning of the experiment, atmospheric oxygen was present inside the headspace of the reactor. This oxygen was rapidly consumed during the initial stages of degradation and the reactor quickly became anaerobic. Moreover, the experiment has been designed so the headspace volume was small (0.25L). The bottles were all closed with rubber tape and silicon to ensure that they were gas tight. The experiment was run until the gas production stopped (21 days). The gas production (volume) and gas composition were daily analyzed (Figure 1 & Table 1).
Biodegradation calculation
The biodegradation rate was determined by the carbon loss and according to the reactions equations (below) the carbon loss corresponds to the gas produced. The volume of gas produced has been measured all along the experiment. The biogas produced by anaerobic digestion is mostly composed of methane and carbon dioxide [17]. This calculation was driven by the hypothesis that the gas produced was only composed by methane and carbon dioxide. Each mole of gas produced contained one mole of carbon. Those two reactions do not represent the biodegradation reactions but were used to calculate the material balance.
C+2H2→CH4
C+O2→CO2

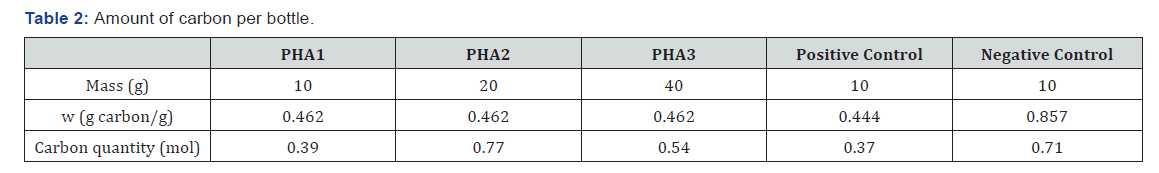
The gas produced by the anaerobic digestion was determined by the difference between the gas measured in the tested bottle and the gas produced by the blank bottle with no plastic (Table 2).
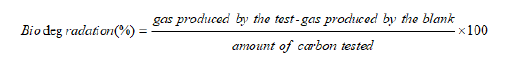
Gas chromatography
The methane production from each reactor was measured every day by using a 500μL gas tight syringe (Hamilton Company, Reno, Nevada). The gas collected was injected into a gas chromatograph (SRI Model 310) to analyze its methane and carbon dioxide content. These measurements were run according to the standard ASTM E 260. The gas chromatograph was equipped with a thermal conductivity detector and helium gas was used as a carrier. The oven temperature was 85°C and the inlet pressure was 50psi.
The gas was calibrated before each use by doing a calibration with a standard gas mixture (15% Nitrogen, 35% Carbon dioxide, and 50% Methane). The gas produced was calculated considering that it was only composed of methane and carbon dioxide because the other gas produced do not represent a big part of the all gas production (e.g. ammonia) [17]. It was chosen to ignore the other gases produced to make the gas composition calculation easier (e.g.: the gas measured is 20% nitrogen, 40% methane and 40% carbon dioxide; the gas produced is considered as 50% methane & 50% carbon dioxide). This method of calculation has been used to consider that a certain amount of nitrogen present in the headspace stayed in it during the all experiment, and the gas was sampled in the headspace.
FOS/TAC measurement
FOS/TAC is a twostep titration process developed to monitor the stability in an anaerobic digestion system. The first stage of titration has an endpoint of pH 5 and determines the inorganic carbonate buffer in solution (TAC). The second stage has an endpoint of pH 4.4 and determines the acetic acid equivalency (FOS). Each of these numbers, as well as the ratio between them (FOS/ TAC), give an indication of the capability of the anaerobic digestions system’s ability to handle greater organic input. The volatile fatty acids/alkalinity ratio (FOS/TAC ratio) was performed before the beginning of the experiment and after the end of the experiment with the Biogas Titration Manager from HACH. The FOS/ TAC analysis is a recognized test used to monitor biodegradation performance in biogas plants [15].
Mass loss
The reactors mass has been measured at the beginning of the experiment and at the end of the experiment to determine the mass lost during the degradation process. The “effective mass loss” is mass lost measured mass loss of the blank.
The expected mass loss was calculated using the gas composition measured with the GC and the volume of gas produced. This data allowed calculating the amount of methane and carbon dioxide produced by each sample. According to the mass balance of this system, the expected mass loss corresponded to the mass of methane and carbon dioxide produced. This “expected mass loss” is expected to match the “effective mass losses to validate the hypothesis that the reactions producing the methane and the carbon dioxide are the only reaction happening during the degradation.
Results and Discussion
The volume produced by the sample PHA3 has not been recorded because of a leak. However, this sample was still at atmospheric pressure. So, the other parameters (gas composition, FOS/ TAC, mass loss) have been measured. The results in the following lists the biodegradation results for PHA, PLA, Cellulose, and HDPE.
Biodegradation
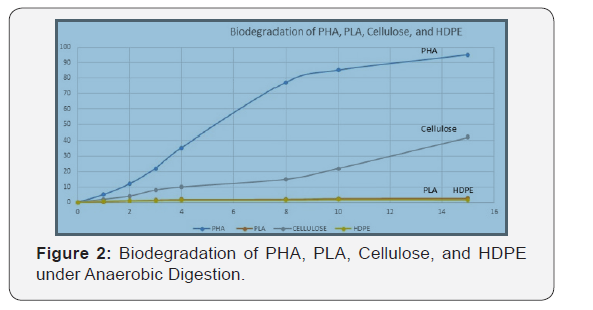
PHA and cellulose biodegradation started after two days. This time corresponds to how long it takes for the material to hydrolyze. Oxygen is used up during the breakdown of organic material; once it is used up the reaction becomes anaerobic (Figure 2).
Both negative control and positive control showed that the experiment was working. However, the cellulose (positive control) biodegradation slowed down after 5 days. Nothing in particular was changed at this moment. PHA1 and PHA2 were close to be totally degraded (102% & 95%). The fact that the PHA1 biodegradation is above 100% shows that the methane and carbon dioxide production are not the only reaction happening in the reactor or could come from a measure uncertainty.
It is important to notice that most of the biodegradation reaction happens in the first 12 days. In fact, 90% of biodegradation is achieved by 11 days (PHA1) and 12 days (PHA2). The reaction’s maximum speed was reached on the 4th day for PHA1 (101mL/g.day in standard conditions) and on the 7th day for PHA2 (95.4mL/g.day in standard conditions). By the end of the experiment, the PHA1 sample produced 8.95 L of gas (standard conditions) and the PHA2 sample produced 15.1L of gas (standard conditions).
Gas composition
The gas composition of the blank, the positive control and the negative control were measured but they do not appear on the next graph because they are not very relevant. Furthermore, the blank and the negative control barely produced gas (Figure 3).
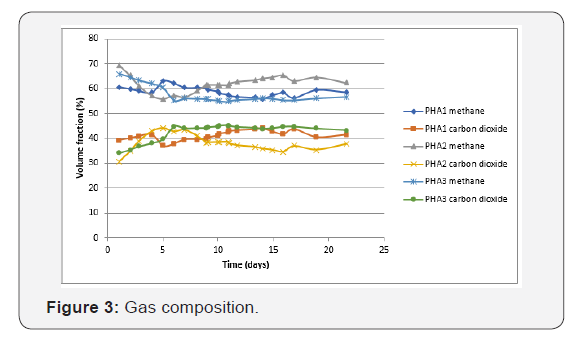
The gas composition was almost the same for the three samples. The average gas composition of the three tests was 60% methane & 40% carbon dioxide. During the last days of the experiment the signal of nitrogen acquired by the gas chromatograph rose for PHA1, PHA2, PHA3 and the positive control. It did not increase for the blank and the negative control. But it does not mean that nitrogen was produced at this moment. It could have been another molecule producing a signal as the same time as nitrogen (e.g. oxygen or ammonia). It shows that a secondary reaction can occur once the biodegradation is close to the end.
FOS/TAC analysis
The FOS/TAC analysis has been done before and after the experiment: (Table 3)
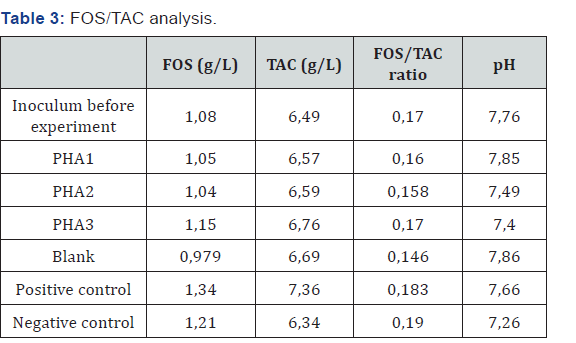
PHA1 and PHA2 had lower FOS than the original inoculum prior to the experiment. This suggests complete degradation of the material. PHA3 only had slightly higher FOS (acetic acid equivalence) indicating good degradation even at higher loading rates. All PHA samples ended with lower FOS than the positive control which suggests a better biodegradation rate of the PHA samples. Moreover, negative control may have had some incomplete degradation of substrate (lower pH, some increase in FOS). The positive control degradation is incomplete; it is possible that there is an alternate hydrolysis pathway leading to other organic acids (butyric, propionic), which are more difficult to degrade.
Mass loss
The mass of each bottle has been measured before and after the experiment. It allowed determining the mass loss of every sample: (Table 4)
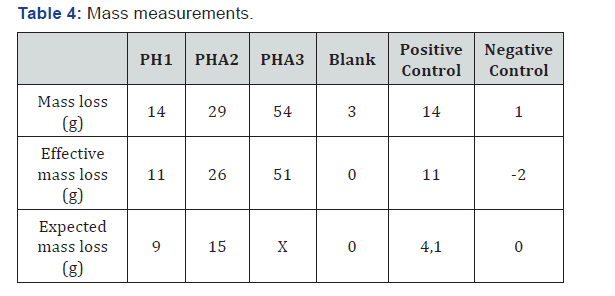
The expected mass loss of PHA3 was not calculated as the volume of gas produced by this sample was not measured. According to the difference between the effective mass loss and the expected mass loss, other reaction(s) happened into the reactor during the degradation. The mass loss measurement does not confirm the results for the gas composition and the volume of gas produced.
Conclusion
This experiment showed that PHA could be biodegraded by anaerobic digestion in mesophilic condition. Its biodegradation is above 90% after 11-12 days and can reach 95%-100%. According to the gas composition analysis the gas composition is about 60% methane and 40% carbon dioxide. Even if the volume measurement showed that the degradation reaction was happening, some points have to be elucidated. Cellulose biodegraded approximately 40% after 15 days. PLA and HDPE did not biodegrade in the anaerobic digestion environment.
Acknowledgement
The authors thank the California State University of Chico departments of Mechanical and Mechatronic Engineering and Sustainable Manufacturing, Chemistry, and Biology for support to the main author during this project. We would also like to thank North State Rendering for providing the inoculum and helping with the FOS/TAC analysis.
References
- Chum H (1991) Polymer from bio-based materials. Park Ridge, NJ: Noyes Data Corp.
- Eriksen M, Lebreton LC, Carson HS, Thiel M, Moore CJ (2014) Plastic Pollution in the World’s Oceans: More than 5 Trillion Plastic Pieces Weighing over 250,000 Tons Afloat at Sea. PLoS ONE 9(12): e111913.
- United States Environmental Protection Agency (2015) Advancing Sustainable Materials Management: Facts and Figures Report.
- Gómez EF, Michel FC (2013) Biodegradability of conventional and biobased plastics and natural. Polymer degradation and stability 98(12): 2583-2591.
- Husárová L (2014) Identification of important abiotic and biotic factors in thebiodegradation of poly (l-lactic acid). International Journal of Biological Macromolecules 71: 155-162.
- Greene J (2007) Biodegradation of Compostable Plastics in Green Yard-Waste Compost Environment. Journal of Polymers and the Environment 15(4): 269-273.
- Wuebbles D, Hayhoe K (2002) Atmospheric methane and global change. Earth-Science reviews 57(3-4): 177-210.
- Kleerebezem R, Joosse B, Rozendal R, Mark CM, Van Loosdrecht, (2015) Anaerobic digestion without biogas? Environmental Science and Bio/ Technology 14(4): 787-801.
- Lee SY (1996) Bacterial polyhydroxyalkanoates. Biotechnology and Bioengineering 49(1): 1-14.
- Greene J (2011) “Marine Biodegradation of PLA, PHA, and Bio-additive Polyethylene Based on ASTM D7081,” Proceedings of the SPE Global Plastics Environmental Conference.
- Bugnicourt E, Cinelli P, Lazzeri A, Alvarez V (2014) Polyhydroxyalkanoate (PHA): Review of synthesis, Express Polymer Letters 8(11): 791-808.
- Chen GQ (2010) Biofunctionalization of Polymers and Their Applications. Advances in Biochemical Engineering / Biotechnology 29-45.
- Kosior E, Messias R, Fowler P (2006) Light weight compostable packaging: Literature review. Banbury: The Waste & Resources Action Programme.
- Bhatt R, Patel K, Trivedi U (2011) Biodegradation of Poly(3- hydroxyalkanoates). In K. Sharma, & A. Mudhoo, A Handbook of Applied Biopolymer Technology: Synthesis, Degradation and Applications. Royal Society of Chemistry.
- Lossie U (2008) Targeted control of biogas plants with the help of FOS/ TAC. Practice Report Hach-Lange
- ASTM International (2012) ASTM D5511-12, Standard Test Method for Determining Anaerobic Biodegradation of Plastic Materials Under High-Solids Anaerobic-Digestion Conditions. West Conshohocken, PA.
- Rasi S, Veijanen A, Rintala J (2007) Trace compounds of biogas from different biogas production plants. Energy 32(8): 1375-1380.
- Rasi S (2009) Biogas composition and upgrading to biomethane. Jyväskylä: Department of Biological and Environmental Science, University of Jyväskylä.






























