Ventricular Rupture during Core Valve® Recapture- The Importance of TAVR-Dedicated Guide Wires
Stephane Leung Wai Sang*, William Merhi and John Heiser
Meijer Heart and Vascular Institute, USA
Received: April 16, 2018; Published: April 26, 2018
*Corresponding author: Stephane Leung Wai Sang, Cardiothoracic Surgery, Spectrum Health Clinical Assistant Professor, Michigan State University, USA, Tel: 616-391-9415; Fax: 616-391-3830; Email: leungwa3@msu.edu
How to cite this article: Stephane Leung Wai Sang, William Merhi, John Heiser. Ventricular Rupture during Core Valve� Recapture-The Importance of TAVR-Dedicated Guide Wires. Open Access J Surg. 2018; 8(5): 555750. DOI: 10.19080/OAJS.2018.08.555750
Abstract
The Core Valve® Evolut R (Medtronic, Minneapolis, MN) is the latest generation self-expanding transcatheter aortic valve with several advantages over the previous iteration, including an extended sealing skirt to reduce paravalvular leak, and the ability to fully recapture and reposition the bioprosthesis after 80% deployment. Little is known regarding the potential implications of valve retrieval, and there is also no consensus to the optimal guide wire necessary for such valves. We present here 3 cases in which valve recapture lead to ventricular rupture. Changing our technique to using TAVR-dedicated guide wires rather than a pre-shaped stiff wire seems to have eliminated this complication.
Keywords: Transcatheter valve implantation; Aortic valve disease; Percutaneous intervention; Aortic repair; Endovascular; Percutaneous valve therapy
Introduction
Transcatheter aortic valve replacement (TAVR) is now an accepted therapy for patients with severe symptomatic aortic stenos is who are at increased risk for open-heart surgery [1,2]. The Core Valve® Evolut R (CER) (Medtronic, Minneapolis, MN), is the latest generation self-expanding transcatheter aortic valve (TAV) with several advantages over the previous iteration, including the ability to fully recapture and reposition the bioprosthesis. To date, our center has implanted over 900 transcatheter aortic valves, the majority self-expanding. We present here three cases in which left ventricular (LV) rupture occurred during CER recapture.
Case 1
Patient #1 was a 74 year-old female with prior aortic valve replacement (AVR) in 2006 with a 23mm Medtronic Mosaic™ bioprosthesis. She presented with New York Heart Association (NYHA) class IV heart failure and was found to have severe aortic regurgitation (AR) due to a flail leaflet. Society of Thoracic Surgeons' predicted risk of mortality (STS PROM) score was 6.5. She met greater than 2 frailty criteria and was deemed high risk for reoperative AVR.
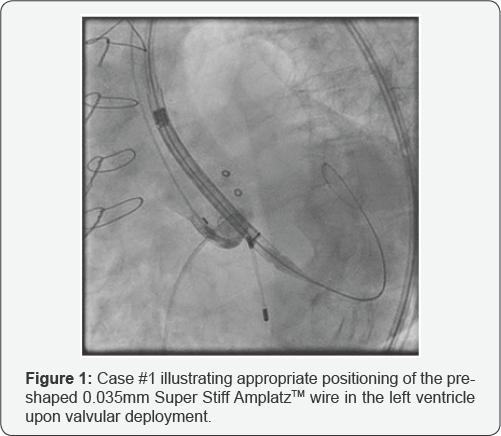
Transfemoral placement of a 26mm CER valve was performed across the previous prosthesis using a pre-shaped 0.035mm Super Stiff Amplatz™ (Boston Scientific, Oakland, NJ) wire with a 1cm soft-tip as a rail (Figure 1). The TAV was deployed while pacing at 120 beats/min with an initial implant depth of 10mm and significant paravalvular leak. The CER was subsequently recaptured fully and redeployed at a depth of 7mm with resolution of leak. At this point, the patient suffered cardiac arrest. Emergent sternal re-entry revealed an LV tear from the wire lateral to the left anterior descending artery. The patient expired after several failed attempts at repairing the myocardium.
Case 2
Patient #2 was an 85 year-old female with history of pericarditis, and severe symptomatic aortic stenosis. Her STS PROM score was 5.8 and TAVR was offered as part of the Surgical Replacement and Transcatheter Aortic Valve Implantation Trial. Preoperative electrocardiogram-gated computed tomography (CT) showed an aortic annular perimeter of 73.5mm and derived diameter of 23.4mm. This patient underwent transfemoral TAVR with a 29mm CER. Again, a pre-shaped 0.035mm Super Stiff Amplatz™ wire was used. Initial deployment was too high, and therefore, the valve was recaptured and during repositioning the patient developed refractory hypotension with subsequent cardiac arrest. Upon emergent sternotomy, dense adhesions were met, likely from the history of pericarditis. Once the heart was mobilized a linear tear could be found along the lateral LV wall. Uncontrollable massive blood loss lead to intraoperative mortality.
Case 3
Patient #3 was a 79 year-old male with a history of prior aortic root replacement using a 27mm Medtronic Freestyle™ bioprosthesis in 2001. The patient developed severe symptomatic prosthetic AR. His STS PROM score was 3.2, but was deemed high-risk for reoperative AVR based on frailty, and porcelain aorta. The aortic annular perimeter measured 79.7mm with a derived diameter of 25.4mm by CT. Initial TAV deployment with a 29 CER, using the Super Stiff Amplatz™ wire, was too low necessitating recapture. However, this was met with endcap separation requiring a 2nd CER delivery system. Placement of the second prosthesis leads to refractory hypotension and a new pericardial effusion (Figure 2). The patient was placed on femoral cardiopulmonary bypass with attempt at redo- sternotomy, however, without success, and the patient expired.

Discussion
The incidence of LV perforation during TAVR is low and in the Core Valve® trial was only 1.3% [1]. Avoidance of this complication is dependent upon stable positioning of a curved stiff wire with a J-curve at the tip placed into the left ventricle which is used as a rail to position and deploy the TAV [3]. Traditionally, a pre-shaped Super Stiff AmplatzTM wire has been used for this purpose. The latest generation CER, has several advantages, including a self-centering valve deployment, an extended sealing skirt to reduce paravalvular leak, and an option to fully recapture and reposition the bioprosthesis. However, as we have shown here recapturing and re-deployment in the setting of a pre-shaped stiff wire may be prone to LV rupture. Great care should be taken upon recapturing the CER minimizing forward tension on the wire.
We postulate that recapturing the TAV leads to excessive tension on the left ventricle transmitted by the rigid wire. For this reason, we have since changed our practice to using TAVR- dedicated guide wires such as the Safari™ (Boston Scientific, Oakland, NJ) wire, or the Confida™ (Medtronic, Minneapolis, MN). These wires are designed to minimize ventricular perforation, while providing stability during TAV positioning. We have had no further ventricular perforations in the last 200 cases as a result of this change.
Conclusion
Our group has performed over 900 TAVRs to date, with extensive experience using self-expanding bioprostheses. This is the first series illustrating a potential devastating complication when using pre-shaped stiff wires not designed specifically for TAVR placement, particularly, in the setting of re-capturing the latest generation CER. We advocate the use of TAVR-dedicated wires for placement of such valves with caution on re-capturing.
Acknowledgement
We would like to acknowledge Tiffany Turner, Rachel Rogers, Sarah McCalley, and Tarah Moelker for their continued support and dedication to the TAVR program.
References
- Adams DH, Popma JJ, Reardon MJ, Yakubov SJ, Coselli JS, et al. (2014) Transcatheter aortic-valve replacement with a self-expanding prosthesis. New Engl J Med 370(19): 1790-1798.
- Reardon MJ, Van Mieghem NM, Popma JJ, Kleiman NS, S0ndergaard L, et al. (2017) Surgical or transcatheter aortic-valve replacement in intermediate-risk patients. New Engl J Med 376(14): 1321-1331
- Masson JB, Kovac J, Schuler G, Ye J, Cheung A, et al. (2009) Transcatheter aortic valve implantation: Review of the nature, management, and avoidance of procedural complications. JACC Cardiovasc Interv 2(9): 811-820.






























