Effects of Low-Level Laser Therapy on Clinical Characteristics and Laboratory Profiles in Synovitis in patients with Rheumatoid Arthritis
Kazuo Yudoh* and Rie Karasawa
Department of Frontier Medicine, Institute of Medical Science, St. Marianna University School of Medicine, Japan
Submission: March 20, 2018; Published: April 04, 2018
*Corresponding author: Kazuo Yudoh, Institute of Medical Science, St. Marianna University School of Medicine, Kawasaki City, Japan.
How to cite this article: Kazuo Y, Rie K. Effects of Low-Level Laser Therapy on Clinical Characteristics and Laboratory Profiles in Synovitis in patients with Rheumatoid Arthritis. Nov Tech Arthritis Bone Res. 2018; 2(5) : 555596. DOI: 10.19080/NTAB.2018.02.555596
Abstract
Rheumatoid arthritis (RA) is a chronic disease that requires long-term administration of immunomodulatory drugs, resulting in a greater risk of side effects such as serious infections, malignancies, hepatic or renal dysfunction and cardiovascular diseases. The main treatment for RA is drug therapy and its therapeutic effect has steadily improved. However, problems including side effects such as infection due to suppression of the immune system and off-target effects at other locations such as the liver still exist because it is a systemic rather than a topical therapy.
Despite the effectiveness of anti-rheumatic drugs, further studies are needed to reduce the risk of side effects. It has been revealed that light therapy by laser irradiation could accelerate wound healing and tissue repair, as well as provide relief of pain and inflammation. The purpose of this study was to clarify whether laser irradiation treatment has a therapeutic potential as an adjunctive therapy against RA synovitis. We recruited 15 patients with RA (mean age: 64.9 years old, mean duration of follow-up: 8.5 years). A laser therapy device (Pulse 10, Panasonic Healthcare Inc.) was used for diode laser treatment. The laser was applied at one point over the swollen or tender joint for 5 minutes, over a period of 4 weeks, once a week for a total of 5 sessions. Outcome measures included arthralgia, patients’ global assessment, physician’s global assessment, the levels of swelling and tenderness of arthritic joints and disability. Also, in addition to laboratory blood tests, serum concentrations of pro inflammatory cytokines were measured.
A total of 26 joints were treated. After the third or 4th treatment with laser irradiation, there was a tendency for the number of swollen and tender joints to decrease. After the 4th treatment, the ADL and VAS scores showed a tendency to decrease significantly. Although no significant difference was observed in the C-reactive protein value, serum concentrations of a matrix degrading enzyme (metalloprotease-3) and a pro inflammatory cytokine (interleukin-6) were significantly reduced after the 5th laser irradiation treatment session. Our results indicate that treatment with laser irradiation may have therapeutic potential as an adjunctive therapy against RA synovitis.
Keywords: Laser Therapy; Rheumatoid Arthritis; Arthralgia; Tumor Necrosis Factor-; Interleukin 1; Interleukin-6
Introduction
Rheumatoid arthritis (RA) is characterized by synovial hyperplasia, neoangiogenesis and infiltration of lymphocytes and macrophages into the RA synovium [1-3]. Numerous studies have already demonstrated that synovitis is closely involved in the pathogenesis of RA [4-6]. In RA synovium, large numbers of infiltrating T cells, macrophages, osteoclast precursor cells and synovial fibroblasts can be seen. It has been demonstrated that infiltrating lymphocytes and macrophages in RA synovial tissue produce excessive levels of pro inflammatory cytokines, such as tumor necrosis factor (TNF)-, interleukin (IL)-1 and IL-6 [4-13]. These cytokines form a network and are key participants in the pathogenesis of arthritis. An imbalance of pro inflammatory and inhibitory cytokines may, at least in part, participate in the development of arthritis [13]. In addition, periarticular osteopenia can be observed in the early stage of the disease before the erosion of bone becomes evident [14-18]. Previously, we have demonstrated that infiltrating inflammatory cells in RA synovium may modulate not only inflammation but also bone and cartilage destruction [11]. A therapeutic strategy against synovitis may have the potential to inhibit inflammation, joint pain (arthralgia), and the degeneration of bone and cartilage tissues [9-10]. In this study, we focused on the synovitis to develop a new therapeutic strategy for RA.
In general, currently-available anti-rheumatic drugs to inhibit RA synovitis include non-steroidal anti-inflammatory drugs (NSAIDs), steroids, disease-modifying anti rheumatic drugs (DMARDs), immune suppressants, or antibiotics. Antirheumatic drugs, such as DMARDs, immune suppressants, and biologics, exert their therapeutic effects via cellular and molecular responses of target cells, such as T cells, B cells, macrophages and synovial fibroblasts [19-22]. In addition to therapeutic drug responses, these anti-RA drugs are likely to induce a variety of side effects, such as anemia, vasculitis, pneumonitis, infection, or liver disorders. Consequently, it is important to consider the balance between therapeutic effects and side effects in anti-rheumatic drugs [21,22]. Novel lowinvasive therapies are required to further improve the symptoms and clinical outcome of RA.
Numerous reports and systemic reviews with meta-analysis have already demonstrated that laser therapy exhibits an anti-inflammatory effect in a variety of diseases [23-28]. It is suggested that laser therapy showed an anti-inflammatory effect through the mechanism involving the control of pro inflammatory cytokine secretion [23-27]. In RA, synovitis induces joint destructions as well as acute and chronic arthralgia. Treatment with laser illumination may be expected as an adjunctive therapy for RA synovitis, although further studies are needed to clarify possible mechanisms for the therapeutic effect of laser therapy on synovitis.
Based on the results of our previous in vitro and in vivo studies, we postulated that light therapy may have potential as an adjunctive and low-invasive therapy for RA. The purpose of our clinical study was to clarify whether or not treatment with laser irradiation has therapeutic potential as an adjunctive therapy against RA synovitis. We hypothesized that laser irradiation would have anti-inflammatory and anti-pain effects against the synovitis in RA. If the light therapy proves effective, it would enable the doses of NSAIDs, steroids or immune suppressants to be reduced, resulting in a greatly decreased risk of side effects such as gastric ulcers, osteoporosis or infection.
Patients
Fifteen patients with RA were recruited and received light therapy by laser irradiation against rheumatoid arthritic joints. The protocol of this study was approved by the ethical committee of St. Marianna University School of Medicine (permission number: 1678) and was conducted in accordance with the 2011 Helsinki declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in the study. Table 1 shows the criteria of patient selection for this study. The mean patient age was 64.9 years old, with a range from 48 to 78 years. The mean duration of follow-up was 8.5 years. Six patients showed lower disease activity as defined by a disease activity score (DAS) score of less than 2.7 [29]. Nine patients had active disease as defined by a DAS score of more than 2.7. Table 2 shows patients’ profiles in this clinical study. All patients had to have been receiving a stable dose of NSAIDs, steroids, immune suppressants or antibiotics for at least 6 months to participate in the study (Table 3).


DAS (disease activity score)

*All patients in the present study had been treated with nonsteroidal anti-inflammatory drugs.
TNF (tumor necrosis factor)
Laser Irradiation
All laser irradiation treatments were applied over a period of 4 weeks, once a week for a total of five sessions. A laser therapy device (Pulse 10, Panasonic Healthcare Inc., Japan) was used for treatment with a diode laser [30]. The wave length of this machine was 830 nm, peak output power was 10 W, and power density was 6–7W/cm2. The product used in the current study can produce the pulsed laser light with peak output power of 10W (20 milliseconds, irradiation energy 1J/second) by intervals of 180 seconds, which enables to suppress the strong thermal effect and to transmit the laser light into the deeper tissue at the same time. The laser was applied at one point over the swollen or tender joint for 5 minutes. Outcome measures included arthralgia, patients global assessment, physicians global assessment, the levels of swelling and tenderness of arthritic joints and disability. The level of joint pain was evaluated by visual analog scale (VAS). The VAS is a standard 10- cm horizontal scale. The patient indicates the severity of pain by placing a mark between terminal points designated “No pain” and “Pain as bad as it could be.” The VAS is scored from 0 (no pain) to 10 (pain as bad as it could be).
In general, it is well known that the VAS found in the Stanford Health Assessment Questionnaire was found to be the most useful available measure for routine clinical use. However, some patients could not complete this scale without assistance. Therefore, it has been developed a brief pain scale for regular use, based on the frequency of pain in the same 8 activities of daily living (ADL) used to assess difficulty, dissatisfaction, and change in status [31].
The patient was asked: “How often is it painful for you to . . .” for each of the 8 ADL (a, b, c, d, e, f, g, h) described below:
a) Dress yourself. including tying shoelaces?
b) Get in and out of bed?
c) Lift a full cup or glass to your mouth?
d) Walk outdoors on flat ground?
e) Wash and dry your entire body?
f) Bend down to pick up clothing from the floor?
g) Turn regular faucets on and off?
h) Get in and out of a car?
And then, four response options were presented and were scored as follows: never = 0, sometimes = 1, most of the time = 2, and always = 3. The total ADL pain score was the mean score for the 8 responses [31,32].
In addition to laboratory blood tests, serum concentrations of biomarkers for inflammation [C-reactive protein (CRP), pro inflammatory cytokines (TNF-, IL-1, IL-6)] and matrix degrading enzyme [matrix metalloprotease (MMP)-3] were measured. The schedule of analysis is summarized in (Table 4).

○Before irradiation, ● After irradiation, △ Before irradiation as necessary, ▲ After irradiation as necessary CRP (C reactive protein), ESR (erythrocyte sedimentation rate), MMP (matrix metalloprotease-3), IL (interleukin), TNF (tumor necrosis factor)
Results
A total of 26 joints were treated (Table 5). Relatively more PIP finger and wrist joints were involved in comparison to large joints.

CM (carpometacarpar)
PIP (proximal interphalangeal)
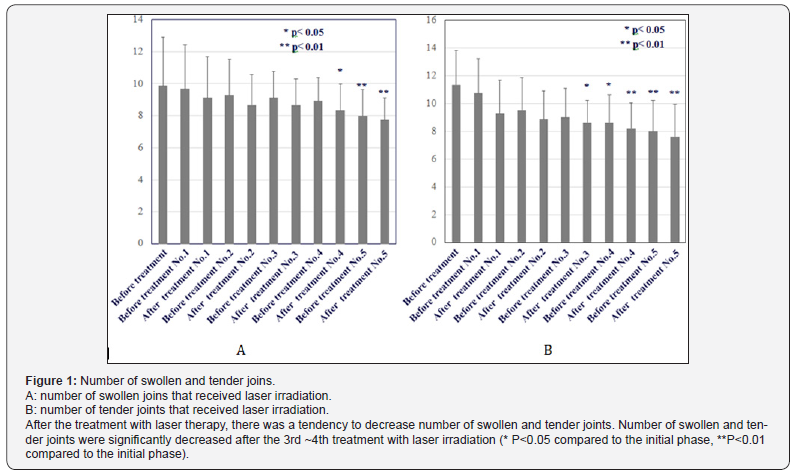
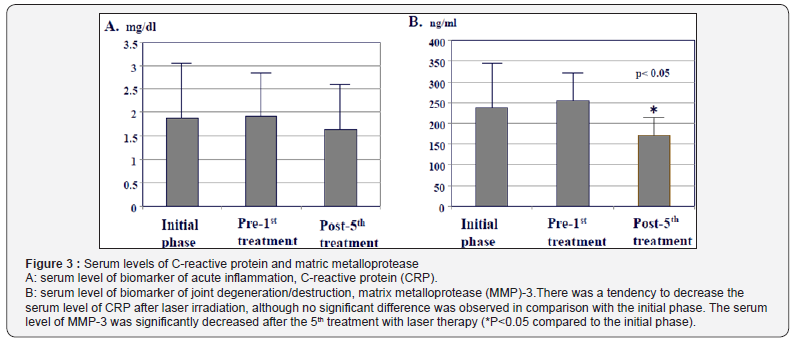
After the 3rd or 4th treatment with laser irradiation, there was a tendency for the number of swollen and tender joints to decrease (Figures 1A & 1B). Regarding ADL (pain) score and VAS of pain, after the 4th treatment, the scores showed a tendency to decrease significantly, suggesting a therapeutic effect of laser irradiation on the rheumatoid synovitis (Figures 2A & 2B). Although no significant difference was observed in the CRP value, serum concentration of matrix metalloprotease MMP-3 was significantly decreased after the 5th laser irradiation treatment (Figures 3A & 3B)
We observed that serum concentrations of pro inflammatory cytokines TNF- and IL-6, but not IL-1, were decreased after the 5th laser irradiation (Figures 4A-4C). These pro inflammatory cytokines are mainly produced by synovial fibroblasts, infiltrating T cells and macrophages. We conclude that the laser irradiation against the arthritic joints directly inhibits the production of pro inflammatory cytokines from synovial tissue.


Discussion
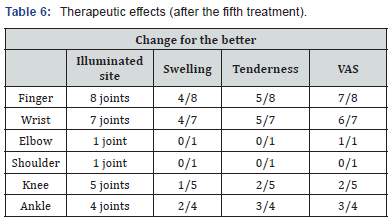
In the present study, we carried out a clinical trial with a device for laser therapy to clarify whether or not treatment with laser irradiation has therapeutic potential for RA patients. Our present study indicates that there is a tendency for the number of swollen and tender joints to decrease after laser irradiation treatment. The arthralgia was also improved by the treatment. Interestingly, we observed that the laser therapy was more effective in fingers and wrist joints than in other large joints such as shoulder, elbow and knee joints (Table 6), although it still unknown why there was difference in therapeutic effects between small joint and large joints.
VAS (visual analog scale)
In the current study, we used two different pain scores, the VAS and ADL pain score. In general, it is well known that the visual analog pain scale found in the Stanford Health Assessment Questionnaire was found to be the most useful available measure for routine clinical use. However, some patients could not complete this scale without assistance. Therefore, it has been developed a brief pain scale for regular use, based on the frequency of pain in the same 8 ADL used to assess difficulty, dissatisfaction, and change in status [31]. Previous study demonstrated that the ADL pain scale met psychometric criteria for validity and reliability. Scores on the 2 pain scales were correlated significantly with one another and with other measures of disease status, including joint count, grip strength, walking time, button test, morning stiffness, erythrocyte sedimentation rate, global self-assessment, ADL difficulty, and ADL dissatisfaction scales [32]. They mentioned that the ADL pain scale was more sensitive to problems in ADL than were the ADL scales for difficulty and dissatisfaction. It has been reported that the ADL and visual analog pain scales appear to provide useful data for quantitative assessment of pain in the routine care of rheumatoid arthritis patients [32].
It is well known that laser irradiation has an anti-inflammatory effect through the mechanism involving the inhibition of pro inflammatory cytokine secretion [23-26,28]. Also, laser therapy shows an anti-pain effect via the suppression of the exited sensory nerve and sympathetic nerves and the improvement of blood flow due to vasodilating action in a feedback system of pain [33,34]. The laser therapy also shows an anti-pain effect via the suppression of the exited sensory nerve and sympathetic nerves and the improvement of blood flow due to vasodilating action in a feedback system of pain [35,36]. Generally, there is a feedback system of pain, consisting of the excitation of sensory and sympathetic nerves and narrowing of blood vessels [36,37]. Previous reports revealed that light therapy could suppress these three factors forming the negative spiral of pain [33,34]. In addition, cellular responsiveness against light therapy had been also been evaluated. Previous studies have revealed that light irradiation can induce activation of the cellular respiratory chain, resulting in an increase of cellular energy [production of adenosine triphosphate (ATP)] and a variety of cellular responses such as gene expression, protein synthesis, collagen synthesis, cell proliferation, cell-cell communication, activation of immune cells and intracellular signal transductions [38-40]. As a photo biological response, light irradiation is thought to accelerate wound healing, tissue repair, and the relief of pain and tissue edema [23,41]. As these photobiological responses, the light irradiation is thought to accelerate the wound healing, tissue repair, tissue edema and the relief of pain.
In our study, scores of joint symptoms (number of swollen joints and tender joints), pain score, and VAS gradually decreased with the advance of laser treatment and showed a tendency to decrease significantly after the 4th treatment. This suggests that the laser irradiation to the arthritic joint may reduce the level of synovitis, such as synovial hyperplasia and infiltration of inflammatory cells into synovial tissue, in rheumatoid arthritic joint. The laser treatment may have a potential to decrease the level of inflammation in RA joints. Anti-inflammatory effect of laser irradiation may accumulate according to increase in the laser irradiation number of times, and then joint symptoms may show a tendency to decrease significantly after the 4th treatment. Although further studies are needed to clarify the issue, we would like to conclude that the laser irradiation have an anti-inflammatory effect against rheumatoid synovitis.
Because the laser light spreads rapidly within the human body, a greater output of laser light is required in order to deliver the light to deeper biological tissue. For laser therapy, the device (Gallium-Alminium-Arsenide Diode) we used in the current study can produce pulsed laser light with a peak output power of 10 W, which simultaneously reduces the strong thermal effect and allows transmission of the laser light into deeper tissue. The device can deliver the laser light more deeply while maintaining average irradiation energy of 1 W which is equivalent to that of conventional laser devices [30]. In deep parts of the body, the intensity of the laser light becomes very low due to diffusion. Sufficient laser light intensity and irradiation energy are achieved by fast switching of the 10 W high-power laser element. By irradiating the high-power laser light periodically, it is possible to transmit laser light into deeper tissue while providing enough cooling time to suppress unnecessary heat generation. Therefore, we believe that the device we used for laser therapy can be safely used to treat synovitis in RA.
Our results showed that the serum concentration of MMP- 3, but not CRP, was significantly decreased after the laser irradiation treatments. CRP is produced by the liver in response to inflammation/immune reaction. MMP-3, that is a highlysensitive marker of joint destruction, is mainly produced by synovial tissue. Laser irradiation against the arthritic joint may directly influence the production of MMP-3 from the inflammatory synovium, but not the production of CRP from liver tissue. We believe this is the reason why the decrease caused by light irradiation was observed in the serum level of MMP-3, but not the CRP value.
Our study also showed that the serum level of pro inflammatory cytokine IL-6 was significantly decreased after the 5th treatment with laser therapy. In addition, the serum level of pro inflammatory cytokine TNF- trended to decrease after laser irradiation, although no significant difference was observed in comparison with the initial phase. These findings suggest that the laser irradiation may have an anti-inflammatory effect in RA synovitis. In RA, it is well known that infiltrating lymphocytes and macrophages in RA synovial tissue produce excessive levels of pro inflammatory cytokines, such as TNF-, IL-1 and IL-6. These cytokines form a network and are key participants in the pathogenesis of arthritis. The laser irradiation may influence the production of pro inflammatory cytokines from infiltrating lymphocytes and macrophages. In contrast, no significant difference in the serum level of IL-1 was observed by laser irradiation in our present study. Among these pro inflammatory cytokines, TNF- and IL-6 seems to be stronger in participation as a pathogenic factor than IL-1 in RA. Indeed, biologics targeting TNF- and IL-6 are widely used to treat in patients with RA.
In conclusion, from the results of the current study, we conclude that treatment with laser irradiation may have therapeutic potential as an adjunctive therapy against RA synovitis. Future developments in light therapy may be able to provide a new therapeutic choice to a patient with RA. This method may show promise as an adjunctive therapy with no side effects over the short term in inpatients and which could allow outpatients to carry out treatment at home. Further studies are needed to clarify the exact therapeutic effect of laser irradiation on rheumatoid synovitis. We are now trying to prepare for a new double-blind randomized controlled trial to analyze the effect of light irradiation therapy on synovitis in patients with rheumatoid arthritis.
Acknowledgement
We would like to thank M. Suzuki, S. Mogi, M. Tamaki, and J. Tamate for excellent technical assistance.
Compliance with ethical standards
a. Ethical approval
This study was independently reviewed and approved by the ethics board of St. Marianna University School of Medicine and was conducted in accordance with the 2011 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent was obtained from all individual participants included in the study.
Conflict of Interest / Funding
This study was supported by Panasonic Healthcare co. ltd, Japan. The sponsor had no control over the interpretation, writing, or publication of this work. The corresponding author had full access to all the data in the study and had final responsibility for the decision to submit for publication.
References
- Angelotti F, Parma A, Cafaro G, Capecchi R, Alunno A, et al. (2017) pathogenesis of rheumatoid arthritis. Clin Exp Rheumatol 35(3): 368- 378.
- Elshabrawy HA, Chen Z, Volin MV, Ravella S, Virupannavar S, et al. (2015) The pathogenic role of angiogenesis in rheumatoid arthritis. Angiogenesis 18(4): 433-48.
- van Steenbergen HW, Aletaha D, Beaart van de Voorde LJ, Brouwer E, Codreanu C, et al. (2017) EULAR definition of arthralgia suspicious for progression to rheumatoid arthritis. Ann Rheum Dis 76(3): 491-496.
- Firestein GS, McInnes IB (2017) Immunopathogenesis of Rheumatoid Arthritis. Immunity 46(2): 183-196.
- van de Sande MG, Baeten DL (2016) Immunopathology of synovitis: from histology to molecular pathways. Rheumatology 55(4): 599-606.
- Humby F, Mahto A, Ahmed M, Barr A, Kelly S, et al. (2017) The Relationship Between Synovial Pathobiology and Magnetic Resonance Imaging Abnormalities in Rheumatoid Arthritis: A Systematic Review. J Rheumatol 44(9): 1311-1324.
- Bernardini G, Benigni G, Scrivo R, Valesini G, Santoni A (2017) The Multifunctional Role of the Chemokine System in Arthritogenic Processes. Curr Rheumatol Rep 19(3): 11.
- Mori M, Hashimoto M, Matsuo T, Fujii T, Furu M, et al. (2017) Cellcontact- dependent activation of CD4+ T cells by adhesion molecules on synovial fibroblasts. Mod Rheumatol 27(3): 448-456.
- Buch MH, Hensor EM, Rakieh C, Freeston JE, Middleton E, et al. (2017) Abatacept reduces disease activity and ultrasound power Doppler in ACPA-negative undifferentiated arthritis: a proof-of-concept clinical and imaging study. Rheumatology 56(1): 58-67.
- Kavanaugh A, van Vollenhoven R, Fleischmann RM, Emery P, Sainsbury I, et al. (2017) Testing treat-to-target outcomes with initial methotrexate monotherapy compared with initial tumour necrosis factor inhibitor (adalimumab) plus methotrexate in early rheumatoid arthritis. Ann Rheum Dis pii: annrheumdis p. 1-4.
- Dai SM, Matsuno H, Nakamura H, Nishioka K, Yudoh K (2004) Interleukin-18 enhances monocyte tumor necrosis factor alpha and interleukin-1beta production induced by direct contact with T lymphocytes: implications in rheumatoid arthritis. Arthritis Rheum 50(2): 432-443.
- McInnes IB, Gracie JA (2004) Interleukin-15: a new cytokine target for the treatment of inflammatory diseases. Curr Opin Pharmacol 4(4): 392-397.
- Miranda-Carus ME, Balsa A, Benito-Miguel M, Perez de Ayala C, Martin Mola E (2004) IL-15 and the initiation of cell contact-dependent synovial fibroblast-T lymphocyte cross-talk in rheumatoid arthritis: effect of methotrexate. J Immunol 15 173(2): 1463-1476.
- Gravallese EM, Manning C, Tsay A, Naito A, Pan C, et al. (2000) Synovial tissue in rheumatoid arthritis is a source of osteoclast differentiation factor. Arthritis Rheum 43: 250-258.
- Morony S, Capparelli C, Lee R, Shimamoto G, Boone T, Lacey DL, et al. (1999) A chimeric form of osteoprotegerin inhibits hypercalcemia and bone resorption induced by IL-1, TNF-α, PTH, PTHrP and 1,25(OH)2D3. J Bone Miner Res 14(9): 1478-1485
- Nakashima T, Kobayashi Y, Yamasaki S, Kawakami A, Eguchi K, et al (2000) Protein expression and functional difference of membranebound and soluble receptor activator of NF-B ligand: modulation of the expression by osteotropic factors and cytokines. Biochem Biophys Res Commun275: 768-775.
- Gravallese EM, Harada Y, Wang JT, Gorn AH, Thornhill TS, et al. (1998) Identification of cell types responsible for bone resorption in rheumatoid arthritis and juvenile rheumatoid arthritis. Am J Pathol 152(4): 943-951.
- Yasuda H, Shima N, Nakagawa N, Yamaguchi K, Kinosaki M, et al (1998) Osteoclast differentiation factor is a ligand for osteoprotegerin/ osteoclastogenesis-inhibitory factor and is identical to TRANCE/ RANKL. Proc Natl Acad Sci USA 95(7): 3597-3602.
- Tanaka Y, Hirata S (2014) Intensive intervention can lead to a treatment holiday from biological DMARDs in patients with rheumatoid arthritis. Drugs 74(18): 2129-2139.
- Miwa Y, Saito M, Furuya H, Yanai R, Ikari Y, et al. (2017) Clinical Characteristics of Rheumatoid Arthritis Patients Achieving Functional Remission after Six Months of Non-tumor Necrosis Factor Biological Disease-Modifying Antirheumatic Drugs (DMARDs) Treatment. Intern Med 56(17): 2271-2275.
- Singh JA, Hossain A, Tanjong Ghogomu E, Mudano AS, Maxwell LJ, Buchbinder R, et al. (2017) Biologics or tofacitinib for people with rheumatoid arthritis unsuccessfully treated with biologics: a systematic review and network meta-analysis. Cochrane Database Syst Rev 3.
- Singh JA, Hossain A, Tanjong Ghogomu E, Mudano AS, Tugwell P, et al. (2016) Biologic or tofacitinib monotherapy for rheumatoid arthritis in people with traditional disease-modifying anti-rheumatic drug (DMARD) failure: a Cochrane Systematic Review and network metaanalysis (NMA). Cochrane Database Syst Rev 11.
- Santinoni CD, Oliveira HF, Batista VE, Lemos CA, Verri FR (2017) Influence of low-level laser therapy on the healing of human bone maxillofacial defects: A systematic review. J Photochem Photobiol B 169: 83-89.
- Lee JH, Chiang MH, Chen PH, Ho ML, Lee HE, et al. (2017) Antiinflammatory effects of low-level laser therapy on human periodontal ligament cells: in vitro study. Lasers Med Sci 33(3): 469-477
- Alayat MS, Aly TH, Elsayed AE, Fadil AS (2017) Efficacy of pulsed Nd:YAG laser in the treatment of patients with knee osteoarthritis: a randomized controlled trial. Lasers Med Sci 32(3): 503-511.
- Alves AN, Fernandes KP, Deana AM, Bussadori SK, Mesquita-Ferrari RA (2014) Effects of low-level laser therapy on skeletal muscle repair: a systematic review. Am J Phys Med Rehabil 93(12): 1073-85.
- Huang Z, Ma J, Chen J, Shen B, Pei F, et al. (2015) The effectiveness of low-level laser therapy for nonspecific chronic low back pain: a systematic review and meta-analysis. Arthritis Res Ther 17: 360
- Huang Z, Chen J, Ma J, Shen B, Pei F, et al. (2015) Effectiveness of lowlevel laser therapy in patients with knee osteoarthritis: a systematic review and meta-analysis. Osteoarthritis Cartilage 23(9): 1437-1444.
- http://www.das-score.nl/das28/en/
- Morimoto Y, Saito A, Tokuhashi Y (2013) Low level laser therapy for sports injuries. Laser Ther 22(1): 17-20.
- Pincus T, Summey JA, Soraci SA Jr, Wallston KA, Hummon NP (1983) Assessment of patient satisfaction in activities of daily living using a modified Stanford Health Assessment Questionnaire. Arthritis Rheum 26(11): 1346-1353.
- Callahan LF, Brooks RH, Summey JA, Pincus T (1987) Quantitative pain assessment for routine care of rheumatoid arthritis patients, using a pain scale based on activities of daily living and a visual analog pain scale. Arthritis Rheum 30(6): 630-636.
- Holanda VM, Chavantes MC, Wu X, Anders JJ (2017) The mechanistic basis for photobiomodulation therapy of neuropathic pain by near infrared laser light. Lasers Surg Med 49(5): 516-524.
- Anders JJ1, Moges H, Wu X, Erbele ID, Alberico SL, et al. (2014) In vitro and in vivo optimization of infrared laser treatment for injured peripheral nerves. Lasers Surg Med 46(1): 34-45.
- Roosink M, Mercier C (2014) Virtual feedback for motor and pain rehabilitation after spinal cord injury. Spinal Cord 201 52(12): 860- 866.
- Uhl RL, Roberts TT, Papaliodis DN, Mulligan MT, Dubin AH (2014) Management of chronic musculoskeletal pain. J Am Acad Orthop Surg 22(2): 101-110.
- Arslan H, Doğanay E, Karataş E, Ünlü MA, Ahmed HMA (2017) Effect of Low-level Laser Therapy on Postoperative Pain after Root Canal Retreatment: A Preliminary Placebo-controlled, Triple-blind, Randomized Clinical Trial. J Endod 43(11): 1765-1769.
- Spoto G, De Iuliis V, Petrini M, Flati V, Di Gregorio J, et al. (2016) Effect of low energy light irradiation by light emitting diode on U937 cells. J Biol Regul Homeost Agents 30(4): 997-1007.
- de Oliveira LSS, de Araújo AA, de Araújo Júnior RF, Barboza CAG, Borges BCD, et al. (2017) Low-level laser therapy (780 nm) combined with collagen sponge scaffold promotes repair of rat cranial criticalsize defects and increases TGF-β, FGF-2, OPG/RANK and osteocalcin expression. Int J Exp Pathol 98(2): 75-85.
- Constantin A, Dumitrescu M, Mihai Corotchi MC, Jianu D, Simionescu M (2017) CO2 laser increases the regenerative capacity of human adipose-derived stem cells by a mechanism involving the redox state and enhanced secretion of pro-angiogenic molecules. Lasers Med Sci 32(1): 117-127.
- França CM, de Loura Santana C, Takahashi CB, Alves AN, De Souza Mernick AP, et al. (2013) Effect of laser therapy on skeletal muscle repair process in diabetic rats. Lasers Med Sci 28(5): 1331-1338.






























