Linear Interstitial Keratitis - Assessment of a Poorly Explored Clinical Entity
Frank Blaser*, Daniel Rudolf Muth, Daniel Barthelmes, Anahita Bajka, Maximilian Robert Justus Wiest, Sandrine Anne Zweifel and Sadiq Said
Department of Ophthalmology, University Hospital Zurich, University of Zurich, Zurich, 8091, Switzerland
Submission: March 05, 2024; Published: March 13, 2024
*Corresponding author: Blaser, Department of Ophthalmology, University Hospital Zurich, University of Zurich, Raemistrasse 100, 8091 Zurich, Switzerland
How to cite this article: Frank Blaser*, Daniel Rudolf Muth, Daniel Barthelmes, Anahita Bajka, Maximilian Robert Justus Wiest, et al. Linear Interstitial Keratitis - Assessment of a Poorly Explored Clinical Entity. JOJ Ophthalmol. 2024; 10(4): 555795. DOI: 10.19080/JOJO.2024.10.555795
Abstract
Purpose: This retrospective, single-center case series included four patients clinically diagnosed with linear interstitial keratitis at the University Hospital Zurich, Switzerland. We aimed to expand the existing knowledge on this rare clinical entity.
Observations: We obtained data on patient demographics, laboratory assessments, medical and surgical treatment, and patient outcomes. All four patients aged 21 and younger reported a sudden onset of painful red eye and were referred to our tertiary care hospital because of suspected unilateral infectious keratitis. For each patient, we performed diagnostic corneal swabs for common bacteria, fungi, and viruses, all of which were inconclusive. We further assessed autoimmune serological markers and screened for HIV, hepatitis B and C, and syphilis. In Case 2, we performed in-vivo confocal microscopy. While infectious screening was negative, we detected antinuclear antibodies in two out of four patients (Cases 2 and 3). Rheumatology consultation found no underlying autoimmune disease in all patients. Treatment mainly included topical antibiotics, corticosteroid eye drops and lubrication, and additional systemic valaciclovir coverage in two patients (Cases 1 and 4). The outcomes depended on the severity of the initial presentation as well as the management of the recurrences.
Conclusions and Importance: Linear interstitial keratitis remains a poorly explored entity, preferably affecting adolescent patients. We recommend that future investigations include autoimmune workup to reveal unknown causal relationships. Long-term patient follow-up is paramount to detect disease recurrence and activity, finally minimizing the risk for corneal perforation.
Keywords: Linear Interstitial Keratitis; Autoimmune Keratitis; Infectious Keratitis; Interstitial Keratitis; Keratitis
Abbreviations: AS-OCT: Anterior Segment Optical Coherence Tomography; CNS: Coagulase-Negative Staphylococci; VA: Visual Acuity; HSV: Herpes Simplex Virus; PCR: Polymerase Chain Reaction; VZV: Varicella Zoster Virus; ANA: Antinuclear Antibodies; HIV: Human Immunodeficiency Virus; TPPA: Treponema Pallidum Particle Agglutination; IOP: Intraocular Pressure; VDRL: Venereal Disease Research Laboratory; FTA-ABS: Fluorescent Treponemal Antibody Absorbed
Introduction
The disease entity interstitial keratitis refers to an immune reaction confined to the corneal stroma, conveniently diagnosed by slit lamp examination [1,2]. Characteristically, the inflammatory reaction does not primarily involve the corneal epithelium or endothelium. The underlying causes are diverse and range from infectious diseases to systemic conditions such as Cogan’s syndrome, sarcoidosis, or Kaposi’s sarcoma [3]. While congenital syphilis remains the leading global cause of interstitial keratitis, viral etiologies, most notably herpes simplex virus (HSV), play the most significant role in Western countries[3]. Linear interstitial keratitis with its unilateral, horizontal, sharply demarcated, linear stromal opacities represents a rare clinical variant of interstitial keratitis. The etiology of the mentioned corneal disease remains unknown, although some authors postulate syphilitic or autoimmune causes [4-6]. However, the current literature on linear interstitial keratitis is scarce, with only a few reports describing this disease entity. This retrospective chart review aims to expand the existing knowledge on this rare finding.
Methods
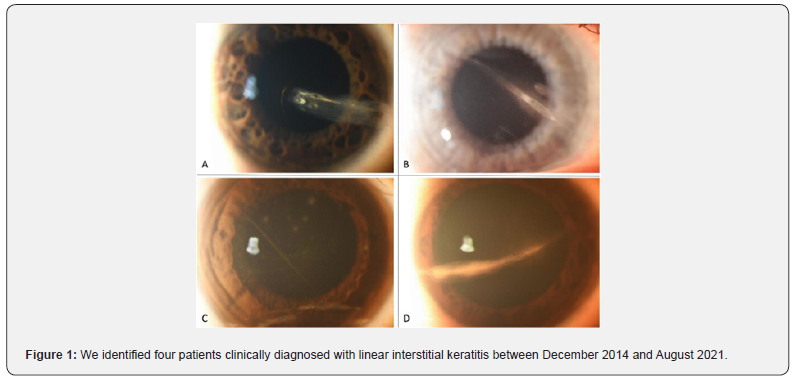
This is an investigator-initiated, retrospective case series conducted at the Department of Ophthalmology at the University Hospital Zurich in Switzerland. Thoroughly reviewing our medical charts for the term linear interstitial keratitis between 2005 and 2022, we identified four patients clinically diagnosed with linear interstitial keratitis between December 2014 and August 2021 (Figure 1). We collected demographic data (age and gender), laboratory values, presenting ophthalmological features, medical or surgical management, and patient outcomes. The leading ethics committee in Zurich waived this study as it does not fall within the scope of the human research act (BASEC-Nr. Req-2023-00109). Nevertheless, all patients signed an informed consent form for the further use of their clinical data for research purposes. We handled all data according to Good Clinical Practice guidelines.
Results
Case 1:
A ten-year-old boy was referred due to a newly discovered corneal scar of unknown origin on the right eye (OD). The patient reported recurrent episodes of a painful red eye and photophobia strictly confined to the OD. Ocular history revealed trauma from a branch in the affected eye one year ago. There was no underlying rheumatologic or autoimmune disease in personal and family history. Polymerase chain reaction (PCR) of corneal swabs were negative for HSV-1, HSV-2, and varicella zoster virus (VZV). Fungal cultures of the corneal scrapings were negative, whereas isolated coagulase-negative staphylococci (CNS) were detected. The unremarkable systemic autoimmune and infective workup included antinuclear antibodies (ANA), anti-Sjogren’s-syndrome-related antigen (anti-SS)-A and B, anti-cardiolipin immunoglobin (Ig)-G and IgM, rheumatoid factor, anti-cyclic citrullinated peptides (anti-CCP), anti-myeloperoxidase (anti-MPO), anti-proteinase 3 (anti-PR3), hepatitis B and C, human immunodeficiency virus (HIV), and treponema pallidum particle agglutination (TPPA) test for syphilis.
At the initial examination, corrected Snellen visual acuity (VA) was 0.8. We found a linear stromal corneal opacity with beginning corneal neovascularization, intact epithelium, normal corneal sensitivity, and an unremarkable anterior chamber. The linear lesion did not extend from corneoscleral limbus to limbus. Figure 2 displays the initial presentation. Corneal topography showed irregular astigmatism and corneal thinning over the lesion. Anterior segment optical coherence tomography (AS-OCT) at a six-year follow-up examination showed pan-stromal involvement.
We treated acute keratitis with topical acyclovir ointment, ceftazidime-ofloxacin eye drops, and systemic valaciclovir coverage. As the corneal epithelium defect closed, we started corticosteroid-containing eye drops such as dexamethasone 0.1% or fluorometholone 1%. Recurrent keratitis under no treatment ultimately led to corneal perforation twice, once after mild ocular trauma by a jacket sleeve and once as a spontaneous perforation without external force. Both perforations could be successfully treated conservatively with bandage contact lenses under topical and systemic antibiotic coverage. At the last follow-up twelve years after the initial presentation, corrected Snellen VA deteriorated from 0.8 to 0.5.
Case 2:
An 18-year-old patient was referred due to suspected infectious keratitis on the OD. The patient reported his third episode of an unprovoked painful red eye with photophobia strictly limited to the OD. His personal and family history was unremarkable regarding rheumatologic or autoimmune conditions. Corneal scrapings were negative for HSV-1, HSV-2, VZV, and acanthamoeba in PCR and fungi and bacteria in cultures. The same autoimmune and infective screen workup as in Case 1 found an ANA titer of 1:320, which led to further ANA differentiation (anti-dfs70, anti-dsDNA, anti-histone, anti-chromatin, and anti-SmD). We detected elevated anti-dfs70 at a level of 101.0.
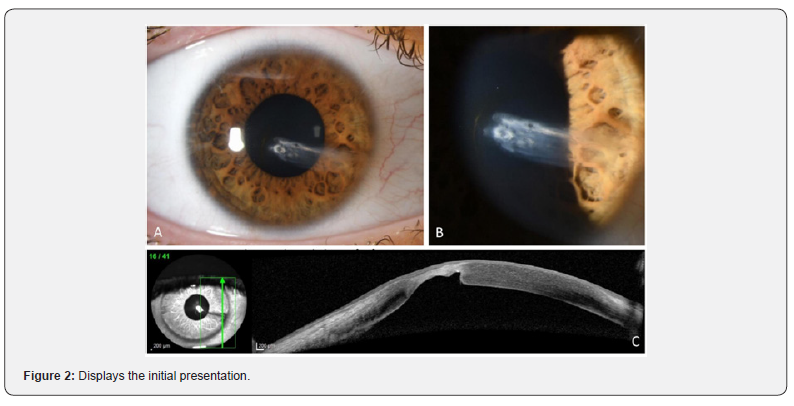
Initial examination of the OD showed uncorrected Snellen VA of 1.0. We found conjunctival hyperemia with limbal injection, a central linear corneal stromal opacity with associated stromal edema, and no corneal neovascularization. The lesion did not extend from corneoscleral limbus to limbus. There was mild anterior chamber reaction with cells 1+. The corneal sensitivity and intraocular pressure (IOP) were normal. No keratic precipitates, synechiae, or iris atrophy were found. Figure 3 displays the initial presentation. AS-OCT revealed a reflective line confined to the anterior corneal stroma. In-vivo confocal microscopy, however, showed pan-stromal inflammation.
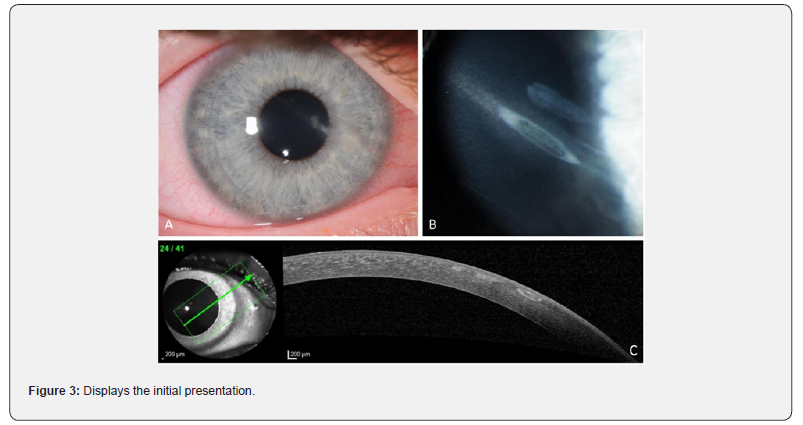
Treatment included topical ceftazidime-ofloxacin, lubrication, and dexamethasone 0.1% containing eye drops. At a follow-up consultation 7.5 years after the initial presentation, corrected Snellen VA remained at 1.0.
Case 3:
A 20-year-old male was referred due to suspected contact lens-associated infectious keratitis on the OD. The patient reported his first episode of a painful OD. Personal and family history were unremarkable for rheumatologic or autoimmune diseases. Corneal swabs were negative for HSV-1, HSV-2, VZV, and acanthamoeba in PCR. Fungal cultures came back negative. Only isolated CNS were detected. We performed the same autoimmune and infective screen workup as in Case 1, detecting a nonspecific ANA titer of 1:320.
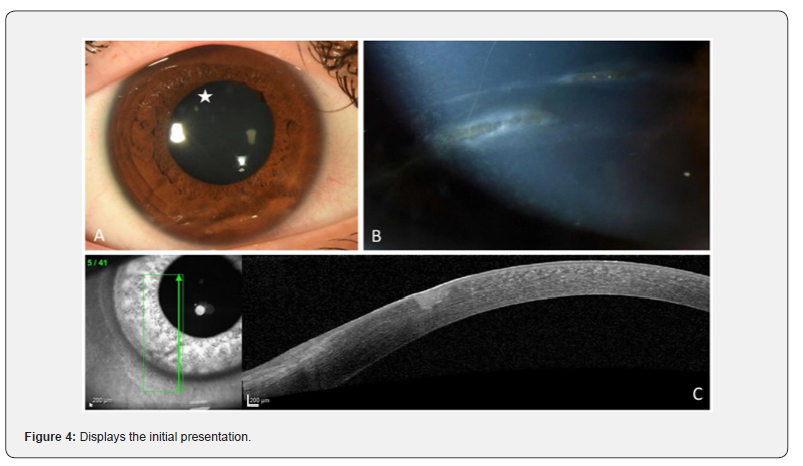
The initial corrected Snellen VA was 0.5. We found mild limbal injection, central nummular keratitis, and paracentral inferior two parallel linear, stromal opacities reaching from corneoscleral limbus to limbus with overlying punctate epithelial erosions. The corneal sensitivity and anterior chamber were normal. Figure 4 displays the initial presentation. On AS-OCT, we found hyperreflectivity confined to the anterior corneal stroma.
Initial treatment included topical ceftazidime-ofloxacin and lubricating eye drops. Five days later, dexamethasone 0.1% containing eye drops were added. At a follow-up consultation after 19 months, corrected Snellen VA improved from 0.5 to 1.0.
Case 4:
A 21-year-old female was referred due to suspected infectious keratitis in the left eye (OS). She reported her first episode of a painful red eye, with no contact lens wear and no history of ocular trauma. Her personal history was unremarkable for rheumatologic or autoimmune diseases. However, her family history revealed that her grandfather suffered from granulomatosis with polyangiitis. We performed corneal swabs negative for HSV-1, HSV-2, and VZV by PCR and negative for bacteria by culture. We detected Penicillium species, an ascomycetous fungus, in the corneal scraping culture. The same autoimmune and infective screen workup as in Case 1 detected a nonspecific ANA titer of 1:1280 and anticytoplasmic antibodies with a titer of 1:640. Rheumatology consultation found no humoral inflammatory activity by laboratory analysis, and the differentiation of ANA antibodies with nuclear fine granular patterns (anti-dfs70, anti-dsDNA, anti-histone, and anti-chromatin) was all negative. In addition, a chest X-ray did not reveal hilar lymphadenopathy.
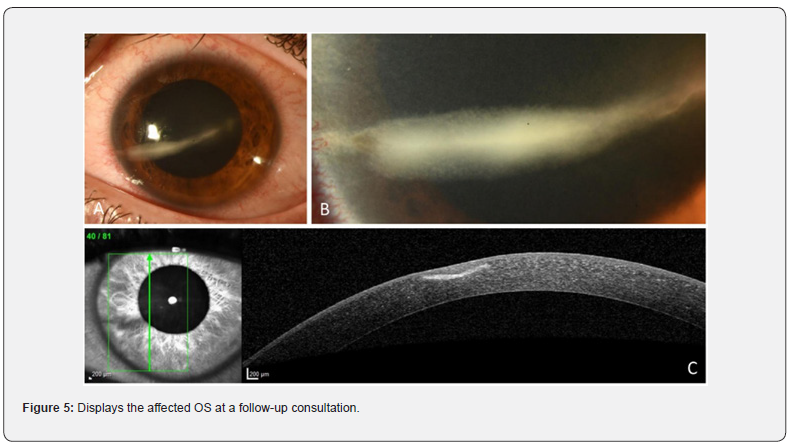
The initial corrected Snellen VA was 0.25. We found conjunctival injection, a linear stromal opacity almost reaching from corneoscleral limbus to limbus with an overlying defect of the corneal epithelium. The anterior chamber was clear. Figure 5 displays the affected OS at a follow-up consultation. AS-OCT revealed hyperreflectivity confined to the anterior stroma and stromal edema with Descemet folds. We initially hospitalized the patient for a total of eight days. Treatment included topical ceftazidime-ofloxacin and dexamethasone 0.1% with systemic valacyclovir coverage, the latter for a total of one month. At a follow-up consultation after four months, corrected Snellen VA improved from 0.25 to 1.0.
Discussion
This retrospective case series included four otherwise healthy patients diagnosed with linear interstitial keratitis at the University Hospital Zurich in Switzerland. To our best knowledge, this represents the largest case series regarding this rare clinical entity. All patients were in their middle childhood to adolescence at the initial diagnosis and were referred due to unilateral, linear-shaped stromal keratitis of unknown cause. Despite extensive local as well as systemic workup, we could not identify a definite underlying cause for the keratitis. Treatment mainly included topical antibiotics, corticosteroid eye drops and lubrication, and additional systemic valaciclovir coverage in two patients. The outcomes depended on the severity of the initial presentation as well as the management of the recurrences.
Autoimmune diseases are conditions with excessive immune responses without eliciting pathogens or other exposure [7]. The association between autoimmune diseases and keratitis is well accepted, although the exact pathogenesis mostly remains unclear. However, ocular manifestations may provide the first symptom or a reflection of disease activity of a systemic condition [7,8]. In this case series, we detected unspecific elevations of ANA in three out of four patients. Rheumatology consultation with clinical and laboratory examination did not reveal an underlying inflammatory disease. They concluded that the elevation of ANA is diagnostically irrelevant at this time. ANAs remain the most common biomarker for autoimmune conditions but may also occur in the general population at approximately 16% [9]. Long-term follow-up of patients suffering from linear interstitial keratitis may reveal a now subclinical autoimmune cause.
Literature initially postulated syphilitic origin as the etiology of linear interstitial keratitis [5,10]. Indeed, syphilis can manifest in all eye segments, including the visual and ocular motor pathways. Characteristically, syphilitic keratitis results in unilateral or bilateral non-ulcerative and non-suppurative interstitial keratitis [11]. However, nontreponemal serologic tests may yield false-positive results for various reasons, including autoimmune disease, pregnant patients, infectious mononucleosis, HIV infection, or tuberculosis [12]. In our tertiary care referral hospital, we use the treponemal test TPPA to screen for syphilis, the fluorescent treponemal antibody absorbed (FTA-ABS) test to confirm the diagnosis, and the non-treponemal Venereal Disease Research Laboratory (VDRL) to monitor disease activity. In this case series, no TPPA reacted positively, and no patient was symptomatic of syphilis. This further strengthens the current understanding that linear interstitial keratitis may not be linked to syphilis.
Regarding our therapeutic measures, we treated all patients with topical broad-spectrum antibiotics, corticosteroid eye drops, and lubrication due to initially suspected infectious keratitis. We additionally used systemic valaciclovir in two out of four patients to cover for possible herpetic keratitis. The outcome regarding VA depended on the severity of the initial presentation (i.e., the extent of the finding, opacity within the visual axis, or corneal thinning-induced astigmatism) and the management of the recurrences and complications (i.e., corneal scarring, or secondary complications such as corneal perforation, cataract, or glaucoma). For example, one patient suffered from a corneal perforation after a minor injury by a jacket sleeve, and about five years later, another spontaneous perforation without excessive external force. Another patient showed a panstromal inflammatory involvement on in-vivo confocal microscopy after having multiple recurrences. We hypothesize that linear interstitial keratitis may initially be confined to the anterior stroma, with the entire stromal thickness becoming affected as the disease progresses. Hence, we believe that regular follow-up and comprehensive patient education about the signs of disease activity are of utmost importance to prevent severe corneal complications.
The strengths of this study included the comparably broad diagnostic workup and the number of cases of this rarely described disease entity. However, there are several limitations. First, a retrospective chart review is inherently prone to selection bias and missing data. We nevertheless consider our data quality to be high, given that all patients received a targeted personal and family history, comprehensive infection and autoimmune screening, and corneal scrapings. Second, the small number of cases makes it difficult to find true causal relationships. Finally, all patients were of European descent. The disease’s prevalence, initial presentation, and course may differ elsewhere in the world.
Conclusion
In conclusion, linear interstitial keratitis is a rare condition with an unknown cause. It seems to affect young patients. Future research should include anterior segment imaging techniques and autoimmune workup to reveal unknown causal relationships. Long-term patient follow-up and patient education are paramount to detect disease recurrence and activity, finally minimizing the risk for corneal perforation.
Acknowledgments and Disclosures
The authors are very thankful to all study participants for the retrospective use of their data.
Authorship: All authors attest that they meet the current ICMJE criteria for authorship.
Availability of data: Data will be made available upon request to the corresponding author.
Conflict of Interest
DB is a consultant and speaker for Novartis and Bayer, and a consultant for Alcon. SS, DRM, TH, AB, MRJW, SAZ, and FB declare no conflicts of interest related to the topic.
Patient Consent: Informed consent was obtained from all subjects involved in the study.
References
- Knox CM, Holsclaw DS (1998) Interstitial keratitis. Int Ophthalmol Clin 38(4): 183-195.
- Schwartz GS, Harrison AR, Holland EJ (1998) Etiology of immune stromal (interstitial) keratitis. Cornea 17(3): 278-281.
- Gauthier AS, Noureddine S, Delbosc B (2019) Interstitial keratitis diagnosis and treatment. J Fr Ophtalmol 42(6): e229-e237.
- Vejdovsky V (1952) Unusual course of deep corneal inflammation possible of congenital origin. Lek List 7(11): 286-288.
- Calvo CM, Sikder S, Mamalis N, Mifflin MD (2012) Linear interstitial keratitis: a distinct clinical entity revisited. Cornea 31(12): 1500-1503.
- Petrovic A, Hashemi K, Blaser F, Wild W, Kymionis G (2018) Characteristics of Linear Interstitial Keratitis by In Vivo Confocal Microscopy and Anterior Segment Optical Coherence Tomography. Cornea 37(6): 785-788.
- Baquero-Ospina P, Paquentin-Jimenez R, Hubbe-Tena C, Concha-Del-Rio LE (2023) Combined Ophthalmology and Rheumatology Evaluation in Systemic Autoimmune Diseases with Ocular Involvement: A Third Level Reference Center in Mexico. Ocul Immunol Inflamm 31(5): 989-995.
- Artifoni M, Rothschild PR, Brezin A, Guillevin L, Puechal X (2014) Ocular inflammatory diseases associated with rheumatoid arthritis. Nat Rev Rheumatol 10(2): 108-116.
- Dinse GE, Parks CG, Weinberg CR, Co CA, Wilkerson J, et al. (2020) Increasing Prevalence of Antinuclear Antibodies in the United States. Arthritis Rheumatol 72(6): 1026-1635.
- Wright MH (1963) Keratitis Linearis Migrans. Br J Ophthalmol 47(8): 504-506.
- Kiss S, Damico FM, Young LH (2005) Ocular manifestations and treatment of syphilis. Semin Ophthalmol 20(3): 161-167.
- Golden MR, Marra CM, Holmes KK (2003) Update on syphilis: resurgence of an old problem. JAMA 290(11): 1510-1514.






























