Central Serous Chorioretinopathy in Course of Polycystic Ovary Syndrome - Literature Review
Anna Ochwał-Brzezinka1, Grzegorz Franik2, Marcin Leśniewski3, Mariusz Wójtowicz4, Dominika Gałczyńska2* and Paweł Madej2
1Department of Ophthalmology, Regional Specialist Hospital No.4 in Bytom, Poland
2Department of Endocrinological Gynecology, Medical University of Silesia, Katowice, Poland
3Department of Gynecology and Obstetrics, District Hospital, 43-200 Pszczyna, Poland
4Department of Gynecology and Obstetrics, Regional Specialist Hospital, 41-803 Zabrze, Poland
Submission: November 14, 2022;Published: December 19, 2022
*Corresponding author: Dominika Gałczyńska, Department of Endocrinological Gynecology, Medical University of Silesia, Katowice, Poland
How to cite this article: FAnna O-B, Grzegorz F, Marcin L, Mariusz W, Dominika G, et al. Central Serous Chorioretinopathy in Course of Polycystic Ovary Syndrome - Literature Review. JOJ Ophthalmol. 2022; 9(3): 555763. DOI: 10.19080/JOJO.2022.09.555763
Abstract
Objective: Central serous chorioretinopathy (CSCR) is one of several retinal disorders that may cause visual loss. It is characterized by serous detachment of the neurosensory retina, often associated with focal detachments of an altered retinal pigment epithelium, mostly confined to the macular area. As CSCR is considered to be correlated with an increased level of testosterone, it was of the interest if it is also linked to the occurrence of polycystic ovary syndrome (PCOS).
Materials And Methods: A systematic literature review in PubMed, Google Scholar and Cochrane library of PCOS- and CSCR-related articles in both Polish and English, published between January 2007 and January 2017.
Results: Most of the authors are consentaneous on the importance of testosterone, what would explain why CSCR is usually attributed to young males, even though testosterone level in this group, in most studies, is within normal range. In case of females, a key factor to the development of CSCR, may be the elevation of testosterone levels, what is one of the most important disturbances in the course of PCOS. Ochwal-Brzezinka in her study suggests the significant link between PCOS and CSCR. She also points out testosterone levels as a main contributor to the development of CSCR during PCOS.
Conclusions: There is an urgent need for further studies, focused on direct link between PCOS and CSCR.
Keywords: Polycystic Ovary Syndrome; Central Serous Chorioretinopathy; Age-Related Macular Degeneration; Testosterone; Dehydroepiandrosterone Sulphate
Abbreviations: CSCR: Central Serous Chorioretinopathy; AMD: Age-Related Macular Degeneration; CNV: Choroidal Neovascularization; RPE: Retinal Pigment Epithelium
Introduction
Central serous chorioretinopathy (CSCR) is one of several retinal disorders that may cause visual loss [1]. CSCR ranks the fourth most common retinopathy after age-related macular degeneration (AMD), diabetic retinopathy and retinal vein occlusion [2]. It is characterized by serous circumscribed detachment of the neurosensory retina often associated with focal detachments of an altered retinal pigment epithelium, mostly confined to the macular area [1]. Major symptoms are blurred vision typically perceived as a dark spot in the middle of the visual field, usually in one eye only, which may coexist with metamorphopsia, dyschromatopsia, micropsia and reduced contrast sensitivity. Best-corrected visual acuity ranges from 20/20 to 20/200 [3]. CSCR is usually classified based on its clinical manifestation, into chronic (more than 3 to 6 months), acute and asymptomatic [1,4].
In acute CSCR visual disturbance resolves spontaneously within 6 months in 80-90% of cases, however up to 50% of cases recurrent [5]. Although this condition is well-known and widely described among men, it’s occurrence in females remains under-diagnosed. This may be related to either differences in clinical manifestation and severity, or significant difference in prevalence of CSCR between genders. Its incidence rate is approximately six times higher in men than women in mid-life. The estimated annual rate reaches 9.9 per 100 000 for men and 1.7 for women [3,6]. In females, CSCR clinical presentation, significantly varies when compared to males, and as previous studies suggest average age of women with CSCR is higher than in men [4,6,7]. While the acute form of CSCR is easy to diagnose for an experienced ophthalmologist, a chronic form, especially in older patients, bears significant resemblance to AMD or can be complicated by choroidal neovascularization (CNV), what may lead to underdiagnosis of CSCR itself [4,8,9,].
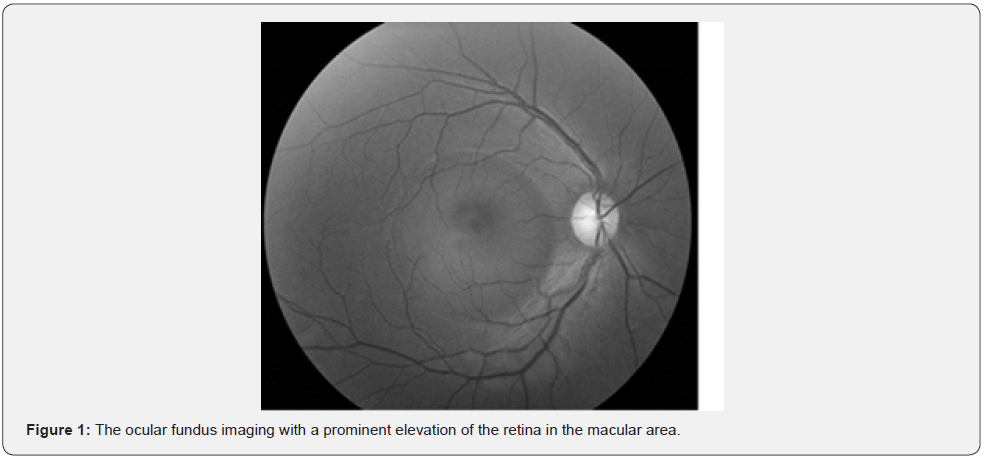
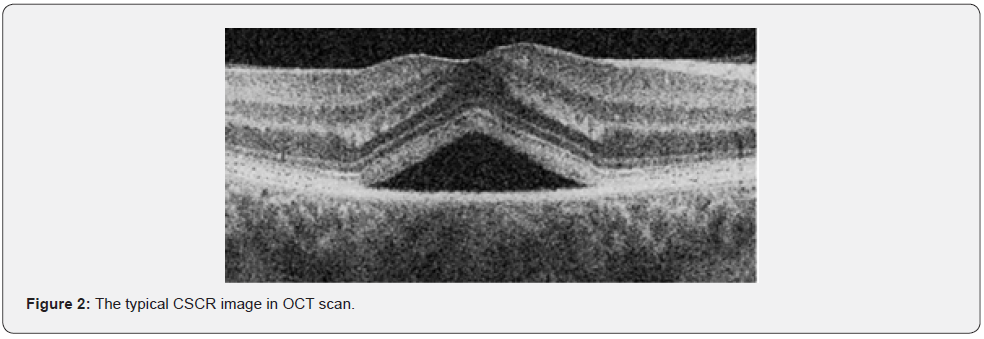
AMD is considered to be the leading cause of blindness in Europe. It usually affects people between 50-60 years old, with a high risk of progression into an irreversible blindness in case of the advanced stages [10]. The advanced type of AMD is usually divided into exudative and dry form, with the dry type in almost 90% of cases leading to the rapid central vision loss [11]. The hallmark signs of development of AMD are druses and distortions of retinal pigment epithelium (RPE) [10]. Although CSCR, has been repeatedly linked to elevated levels of corticosteroids (GCS) [12], this correlation remains controversial due to high complexity of GCS regulation and their influence on retina [4]. Nevertheless, gender tendency present in CSCR epidemiology, imply the crucial role of hormones in its pathophysiology. In spite of that the exact etiopathology of disease is still unclearly understood, numerous risk factors have been associated with CSCR such as type A personality, emotional distress, pregnancy and delivery, H.
Pylori infection, Cushing syndrome, sleep disturbance, allergic respiratory diseases, systemic hypertension, smoking, alcohol consumption, systemic corticosteroids, sympathomimetic agents, PDE-5 inhibitors (Sildenafil, Tadalafil) and a few herbal medicines [2,5,13,14]. The potential role of testosterone level was found in several studies, both as an endogenic and exogenic factor. Testosterone is a vasoactive hormone, that may lead to the elevation of a choroidal blood flow, partially due to vasodilatation of a vascular bed [15]. Additionally, it increases veins’ permeability, which results in a damage to the overlying retinal pigment epithelium (RPE) and to its consecutive detachment [15,16,17].
Although reports on elevated levels of testosterone in course of CSCR are unequivocal [18,19,20] studies reporting on sudden recoveries after stopping of the testosterone treatment, proven gender tendency in CSCR’s occurrence and coexistence of CSCR’s risk factors with hyperandrogenism, seems to validate testosterone’s crucial role in a pathophysiology of at least fraction of CSCR cases, especially in women and in iatrogenic CSCR [4,21]. Additionally, in type A personality, psychological stress and pregnancy are documented risk factors for CSCR occurrence. A significantly increased levels of either serum or urinary testosterone was found in few studies [22,23,24]. Another disease, with a proven elevation of a serum testosterone is PCOS [25,26,27,28].
PCOS is a heterogeneous disorder of reproduction and metabolism, that affects around 12% of an adult female population. It is characterized by a variety of symptoms, including the presence of at least two of the following three criteria: chronic anovulation, androgen excess (clinical or biological) and polycystic ovaries in gynecological ultrasound examination [26]. According to these signs, patients may present four different phenotypes. In fact, the first three including clinical or biochemical hyperandrogenism, represent the majority [29,30]. Insulin resistance plays a crucial role in the pathogenesis of PCOS through hyperinsulinemia stimulating ovarian androgen secretion [28].
PCOS is considered to be one of the most common causes of hyperandrogenism in reproductive-aged women [27,28]. Increased concentrations of circulating androgens are present in as many as 80% of patients with PCOS according to literature [31], from which free testosterone level should be measured [27]. In about 30 to 35 % of patients with PCOS levels of dehydroepiandrosterone sulfate (DHEA-S) [25, 26] which is the precursor of testosterone, are increased. Additionally, leading laboratory abnormality in the course of it, is an elevated level of free testosterone, and also DHEA-S [12], reported to be elevated in patients with CSCR in at least one study [32].
Significantly higher occurrence of CSCR in young males, and gradual decrease in its frequency with age, as well as gender tendency in its incidence implies crucial role of endocrinological homeostasis in its pathophysiology. Role of androgens, especially testosterone, in the etiology of CSCR seems to be an interesting issue, although still remains unequivocal. In that respect, it was of the interest if PCOS, a leading cause of hyperandrogenism in women, is associated with frequency of CSCR.
Methods
The following study is a systematic review of literature published between January 2007 to February 2017 concerning occurrence of the CSCR in the course of PCOS and its relation to increased testosterone concentrations. During analysis, PubMed, Google Scholar, Medline and Cochrane databases were utilized. Additionally, selected records from bibliographies of analyzed studies, were included into further review.
Results
The available literature does not provide unequivocal information on coexistence of PCOS and CSCR. All available data, origins from small, methodologically flawed reports or case studies, and single PhD dissertation by Ochwał-Brzezinka [20]. This dissertation, included 205 female patients diagnosed with PCOS, divided into three groups based on their testosterone levels (Tl) and presence of CSCR signs. Group A- with Tl<5,27 pg/ml (n=101), B- with Tl>5,27 pg/ml (n=100) and group C with Tl>5,27 and coexsisting CSCR symptoms (n=4). Mean age equaled 24,5 years, and women with confirmed H. Pylori infection were excluded. Logistic regression model utilized for sake of a statistical analysis of obtained data, revealed a significant link between CSCR and levels of testosterone (OR=1,2; 95%CI: 1,01- 1,41; p=0,03), and increase in Tl of 1pg/ml resulted in increase of CSCR risk 1,2 times. Sole available report on occurrence of CSCR in females, despite dissertation by Ochwal-Brzezinka [20], was published by Quillen et al. [7], although it did not include PCOS as a risk factor.
Outcomes obtained by Ochwał-Brzezinka [20] are supported by an analysis of four cases of females with coexisting PCOS and CSCR made by Witmer et al [12]. Fifth case analyzed by Witmer et al. was a female with a first-degree relative with history of PCOS, although with no personal diagnosis of PCOS. However, she had never been clinically evaluated. Selected cases did not have any other known risk factors for CSCR. Witmer et al. concluded that although there is a correlation between CSR and PCOS, presented cases did not prove existence of causation between them. Furthermore, Nudelman et al. [15], reported nine patients with confirmed CSCR consequent to the exogenous testosterone therapy. All patients were males and did not have any other known risk factors for CSCR.
Two patients exhibited resolution of symptoms and retinal detachment after administration of testosterone stopped. Similar cases were described by Ahad et al. (52-year-old man with acromegaly who developed CSCR after 1 month of testosterone therapy) [33], Grieshaber et al. (45-year-old female, treated for general fatigue who developed CSCR after 5 weeks of testosterone therapy) [21], Ahad et al. (52-year-old man with hypogonadism, who developed CSCR after 4 years of testosterone therapy) and Hsin-Ying L et al. (52-year-old male with hypogonadism, who developed bilateral, chronic CSCR after 10 years of testosterone therapy) [34]. All of those case-studies describe resolution of CSCR and retinal detachment shortly after end of testosterone administration and decrease in testosterone concentrations.
Outcomes contradicting aforementioned reports, were obtained by Zakir et al. [35,36], who were the first to analyze testosterone levels in 22 males and one female with CSCR, and compare them with matched control, with sudden loss of vision lasting less than month. The mean serum testosterone levels were 3,85±1,81 ng/ml in cases and 4,23±1,89 ng/ml in controls, although difference was not statistically significant (p=0,58). Mean testosterone concentrations in both groups were within normal range. Similar outcomes were obtained by Tufan et al. [31], who described 6 male patients with chronic CSCR. Median value of serum testosterone concentration equaled 549,5ng/ml and was within normal range. Natung et al. [18] reported 30 patients with either acute or chronic CSCR, with the mean age of 42,4±6,37 year. Mean testosterone concentration equaled 5,88±1,57 ng/dL and was within normal range.
The last report, from Haimovici et al [19], described group of 24 patients with diagnosed CSCR, 19 males and 5 females. Average age equaled 43±10,3 years, and only two patients did not have any additional known risk factor for CSCR. Serum testosterone concentrations were within normal range for all but one patient. Although all aforementioned reports seem to agree on normal levels of testosterone in patients with CSCR, we must remember that study groups consisted mostly of males. Baseline testosterone concentration in men is significantly higher than in women, therefore level of free testosterone, required for occurrence of CSCR, may be within normal range. Furthermore, all studies lack proper healthy control, and are mostly constructed as case-series or case-reports. Therefore, their outcomes should be analyzed with caution, and there is a need for a proper cohort or case-control studies on this issue, focusing on a difference in CSCR between male and female patients.
There is an interesting way of explaining inconsistency in available literature on an issue of links between PCOS and CSCR. It also may justify why in dissertation by Ochwal-Brzezinka [20], despite 104 participants with elevated testosterone concentrations, only 4 patients developed CSCR. Türkcü et al. [37], on a group of 46 patients, demonstrated that in the course of a CSCR antioxidant defense system may be inadequate, due to significant decrease in concentrations of antioxidant parameters and DHEA-S, what seem to be an especially interesting finding, taking into account a crucial role of an oxidative damage in patomechanism of an early stage of CSCR. Bucolo et al. [38], reported an in vitro reduction in an oxidative damage in RPE, after administration of DHEA-S. Concentrations of DHEA-S in humans decline with advancing age [22] and average age of CSCR development in women is significantly higher than in men [7].
Therefore, it is possible, that in patients with PCOS, elevated levels of testosterone, predispose them to the occurrence of the CSCR (OR=1,2), however concentrations of DHEA-S, also increased in course of PCOS, protect them. With advancing age, both levels of DHEA-S and testosterone decline, leading to distortion of a balance between them and increased risk of CSCR occurrence in older females. Clinical manifestation of CSCR in this group, may be mistaken for age-related macular degeneration, as those two disorders bear strong resemblance to each other [7]. According to Klein et al. [10] AMD occurrence rate in females is 2,2 times higher than in males. Therefore, limited number of patients in group C of Ochwal-Brzezinka’s dissertation, might be explained by targeting “too young” women with PCOS. Furthermore, inconsistency in available literature on this issue, might be caused by underdiagnosis of CSCR, caused by its similarities with AMD. Both hypotheses, as well as whole subject, require in-depth research, with a proper cohort or case-control studies, to establish the full etiology of CSCR.
Conclusions
Reports available in literature are usually limited to the occurrence of CSCR in males. Therefore, it is necessary to analyze possibility of link between PCOS and CSCR, through indirect studies, focusing on participation of testosterone and DHEA-S in pathophysiology of CSCR development. Most of the authors are consentaneous on the importance of testosterone, what would explain why CSCR is usually attributed to young males, even though testosterone level in this group, in most studies, is within normal range. In case of females, a key factor to the development of CSCR, may be the elevation of testosterone levels, what is one of the most important disturbances in the course of PCOS. The main difficulty in evaluation of possible correlation between PCOS and CSCR, is a long-term follow-up of the patients, which is crucial, because risk of CSCR in those patients may increase over the time. Additionally, in some cases, coexistence of elevated levels of testosterone with increased concentrations of DHEA-S may result in delay of elicitation of hallmark signs of CSCR, and therefore it may overlap with a time range characteristic for AMD development, and result in under-diagnosis of CSCR.
References
- Wong KH, Lau KP, Chhablani J, Tao Y, Li Q, Wong IY (2016) Central serous chorioretinopathy: what we have learnt so far. Acta Ophthalmol 94(4): 321-325.
- Liew G, Quin G, Gillies M, Fraser Bell S (2013) Central serous chorioretinopathy: a review of epidemiology and pathophysiology. Clin Exp Ophthalmol 41(2): 201-214.
- Islam QU, Hanif MK, Tereen S (2016) Frequency of Systemic Risk Factors in Central Serous Chorioretinopathy. J Coll Physicians Surg Pak 26(8): 692-695.
- Quillen DA, Gass DM, Brod RD, Gardner TW, et al. (1996) Blankenship GWCentral serous chorioretinopathy in women. Ophthalmology 103(1): 72-79.
- Daruich A, Matet A, Dirani A, Bousquet E, Zhao M, et al. (2015) Central serous chorioretinopathy: Recent findings and new physiopathology hypothesis. Prog Retin Eye Res 48: 82-118.
- Fung AT, Yannuzzi LA, Freund KB (2012) Type 1 (sub-retinal pigment epithelial) neovascularization in central serous chorioretinopathy masquerading as neovascular age-related macular degeneration. Retina 32(9): 1829-1837.
- Pang CE, Freund KB (2015) Pachychoroid neovasculopathy. Retina 35(1): 1-9.
- Kitzmann AS, Pulido JS, Diehl NN, Hodge DO, Burke JP (2008) The incidence of central serous chorioretinopathy in Olmsted County, Minnesota, 1980-2002. Ophthalmology 115(1): 169-173.
- Kitzmann AS, Pulido JS, Diehl NN, Hodge DO, Burke JP (2008) The incidence of central serous chorioretinopathy in Olmsted County, Minnesota, 1980-2002. Ophthalmology 115(1): 169-173.
- Klein R, Klein BE, Knudtson MD, Meuer SM, Swift M, et al. (2000) Fifteen-year cumulative incidence of age-related macular degeneration: the Beaver Dam Eye Study. Ophthalmology 107: 1593-1600.
- Jaworski M, Tomczyk T, Gładysz S, Babik A, Wilczynski KM, et al. (2016) Radioterapia w neowaskularnym AMD - spojrzenie w przyszłość. Okulistyka po dyplomie 4(31): 30-36.
- Witmer MT, Klufas MA, Kiss S (2015) Polycystic Ovary Syndrome and Central Serous Chorioretinopathy. Ophthalmic Surg Lasers Imaging Retina 46(6): 684-686.
- Dang Y, Mu Y, Zhao M, Lin L, Guo Y, et al. (2013) The effect of eradicating Helicobacter pylori on idiopathic central serous chorioretinopathy patients. Ther Clin Risk Manag 9: 355-360.
- Eom Y, Oh J, Kim SW, Huh K (2012) Systemic factors associated with central serous chorioretinopathy in Koreans. Korean J Ophthalmol 26: 260-264.
- Nudleman E, Witmer MT, Kiss S, Williams GA, Wolfe JD (2014) Central serous chorioretinopathy in patients receiving exogenous testosterone therapy. Retina 34(10): 2128-2132.
- Gass J (1967) Pathogenesis of disciform detachment of the neuroepithelium. Idiopathic central serous choroidopathy. Am J Ophthalmol 63: 1-139.
- Guyer DR, Yannuzzi LA, Slakter (1994) Digital indocyanine green videoangiography of central serous chorioretinopathy. Arch Ophthalmol 112: 1057-1062.
- Natung T, Keditsu A (2015) Comparison of Serum Cortisol and Testosterone Levels in Acute and Chronic Central Serous Chorioretinopathy. Korean J Ophthalmol 29(6): 382-388.
- Haimovici R, Rumelt S, Melby J (2003) Endocrine abnormalities in patients with central serous chorioretinopathy. Ophthalmology 110: 698-703.
- Madej P, Ochwał Brzezinka A, Kukla U, Chronowska J, et al. The connexion between the central serous choroidoretinopathy and hyperandrogenism in young women.
- Grieshaber MC, Staub JJ, Flammer J (2007) The potential role of testosterone in central serous chorioretinopathy. Br J Ophthalmol 91: 118-119.
- Zumoff B, Rosenfeld RS, Friedman M, Byers SO, Rosenman RH, et al. (1984) Elevated daytime urinary excretion of testosteroneglucuronide in men with type A behavior pattern. Psychosom Med 46(3): 223-225.
- King JA, Rosal MC, Ma Y, Reed GW (2005) Association of stress, hostility and plasma testosterone levels. Neuro Endocrinol Lett 26(4): 355-360.
- Laudat, MH, Guilhaume B, Blot P, Fournier C, Giauque JP, et al. (1987) The hormonal state of pregnancy: modification of cortisol and testosterone. Ann. Endocrinol 48: 334-338.
- Carmina E (2006) Ovarian and adrenal hyperandrogenism. Ann NY Acad Sci 1092: 130-137.
- Goodarzi MO, Carmina E, Azziz R (2015) DHEA, DHEAS and PCOS. J Steroid Biochem Mol Biol 145: 213-225.
- Goodman NF, Cobin RH, Futterweit W, Glueck JS, Legro RS, et al. (2015) American Association of Clinical Endocrinologists, American College of Endocrinology, and Androgen Excess and PCOS Society. Disease state clinical review: guide to the best practices in the evaluation and treatment of polycystic ovary syndrome - part 1. Endocr Pract 21(11): 1291-1300.
- Goodman NF, Cobin RH, Futterweit W, Glueck JS, Legro RS, et al. (2015) American Association of Clinical Endocrinologists, American College of Endocrinology, and Androgen Excess and PCOS Society. Disease state clinical review: guide to the best practices in the evaluation and treatment of polycystic ovary syndrome - part 2. Endocr Pract 21(12): 1415-1426.
- Ettore Guastella, Rosa Alba Longo, Enrico Carmina (2010) Clinical and endocrine characteristics of the main polycystic ovary syndrome phenotypes.Fertil Steril 94: 2197-2201.
- Azziz R, Carmina E, Dewailly D, Diamanti Kandarakis E, Escobar-Morreale Hfet al. (2006) Positions statement: criteria for defining polycystic ovary syndrome as a predominantly hyperandrogenic syndrome: an Androgen Excess Society guideline. J Clin Endocrinol Metab 91(11): 4237-4245.
- Forooghian F, Meleth AD, Cukras C, Chew EY, Wong WT, et al. (2011) Finasteride for chronic central serous chorioretinopathy. Retina 31: 766-771.
- Ying L, KaiLing P (2015) Bilateral Chronic Central Serous Chorioretinopathy (CSCR) Induced by Long-Term Testosterone Treatment. J Clin Exp Ophthalmol 6: 6.
- Zakir SM, Shukla M, Simi ZU, Ahmad J, Sajid M (2009) Serum cortisol and testosterone levels in idiopathic central serous chorioretinopathy. Indian J Ophthalmol 57: 419-422.
- Türkcü FM, Yüksel H, Yüksel H, Sahin A, Cinar Y, et al. (2014) Serum dehydroepiandrosterone sulphate, total antioxidant capacity, and total oxidant status in central serous chorioretinopathy. Graefes Arch Clin Exp Ophthalmol 252(1): 17-21.
- Bucolo C, Drago F, Lin LR, Reddy VN (2005) Neuroactive steroids protect retinal pigment epithelium against oxidative stress. Neuroreport 16: 1203-1207.
- Wang M, Munch IC, Hasler PW, Prünte C, Larsen M (2008) Central serous chorioretinopathy. Acta Ophthalmol 86(2): 126-145.
- Salilew Wondim D, Wang Q, Tesfaye D, Schellander K, Hoelker M, et al. (2015) Polycystic ovarian syndrome is accompanied by repression of gene signatures associated with biosynthesis and metabolism of steroids, cholesterol and lipids. J Ovarian Res 8(1): 24.
- Tufan HA, Gencer B, Comez AT (2013) Serum cortisol and testosterone levels in chronic central serous chorioretinopathy. Graefes Arch Clin Exp Ophthalmol 251: 677-680.






























