Efficacy of Amniotic Membrane Transplantation in Refractory Infective Keratitis Leading to Stromal Thinning, Descematocele and Perforations
Shreya Thatte*, Urvija Choudhary and Babita Sharma
Department of Ophthalmology, Sri Aurobindo Medical college, India
Submission: March 19, 2017; Published: June 06, 2017
*Corresponding author: Shreya Thatte, Department of Ophthalmology, Sri Aurobindo Medical college, Chaitanya, 17 Yeshwant Colony, Indore MP, India, Tel: +919302104864; Email: shreyathatte@gmail.com
How to cite this article: Shreya T, Urvija C, Babita S . Efficacy of Amniotic Membrane Transplantation in Refractory Infective Keratitis Leading to Stromal Thinning, Descematocele and Perforations. JOJ Ophthal. 2017; 3(3): 555611. DOI: 10.19080/JOJO.2017.03.555611
Abstract
It is always challenging to manage refractory infectious keratitis. The goal is to achieve complete healing and preventing further complications. A prospective, interventional study was done on 150 patients using single or multilayered amniotic membrane transplantation (AMT) on eyes with different grades of refractory infective keratitis-deep ulcers, descemetocele and corneal perforations. Efficacy was monitored till six months frequent follow-ups on basis of symptomatic comfort, visual acuity improvement, healing and regression of ulcer, formation of anterior chamber depth, epithelialization and improvement in corneal thickness. Successful results were seen in 145 of 150 eyes (96.67%) and failure in 5 cases (3.33%) due to graft failure and subsequent perforation requiring therapeutic penetrating keratoplasty. Supportive secondary surgical interventions were required in 30 cases like washing hypopyon from anterior chamber, air injection to form the shallow anterior chamber, intra cameral antibiotic and re-grafting in a case of graft retraction. Thirty patients were advised subsequent penetrating keratoplasty for leucomatous corneal opacity left after healed ulcer. AMT has come up as a very effective managing technique for refractory ulcers. It aids in permanent healing of the refractory infective keratitis & prepares the cornea for definitive reconstructive procedure if required.
Keywords: Amniotic membrane transplantation; Refractory keratitis; Corneal perforation; descematocele; Infective keratitis; Deep stromal ulcer
Introduction
Diseases affecting the cornea are a major cause of blindness all over the world, second only to cataract in overall importance [1]. One of the commonest corneal causes is Infectious Keratitis. The prevalence of blindness directly resulting from complications of Infective Keratitis is estimated to be 5% [2]. Cases refractory to the medical therapy requires urgent surgical intervention to retrieve the vision and most importantly to salvage eye. Available surgical management in refractory keratitis cases include tissue adhesives, Bandage Contact Lenses (BCL), penetrating or lamellar keratoplasty [3] patch grafts, or conjunctival flaps. Unfortunately, these therapies are associated with a considerable number of complications and address only the tectonic problem, without solving the ongoing infection and inflammation. BCL and conjunctival flaps being a temporary measure does not provide with new collagen to improve corneal thickness and stabilize the cornea. For such situations Penetrating Keratoplasty (PK), Lamellar Keratoplasty (LK) or patch grafts was the only option and is still being used widely. PK and patch grafts performed to seal a corneal perforation may be complicated with synechiae, glaucoma, uveitis, and graft failure in the setting of an inflamed or infected eye [4]. Recurrence of infection in corneal grafts is also challenged. LK being difficult to perform may result in a double chamber between the donor and recipient cornea in some cases. Tissue adhesives may dislodge and are used as a temporary measure, obviating the need for a PK within a few days [5,6].
Preserved human amnion has been successfully used as a biological bandage, promoter of epithelialization, inhibitor of inflammation and angiogenesis, as well as a carrier for ex vivo cultured limbal stem cells [7]. Amniotic Membrane Transplant (AMT) offers the advantage of avoiding potential allograft rejection. Even if corneal transplantation is needed, the success rate is improved if performed on an eye that underwent AMT reducing inflammation [8,9]. Amniotic Membrane (AM) integrates in cornea and thus can be used as a treatment for corneal perforation by restoring corneal stromal thickness so that emergency PK can be avoided, as suitable donor corneal button availability is difficult in every place. Therefore, an alternative management for various stages of infectious keratitis including deep refractory stromal ulcers, descematocele and corneal perforations is reconstruction of the surface with AMT adjuvant with appropriate antimicrobials and supportive medications. In this prospective study AMT in various gravities refractory infective keratitis has been attempted to understand the efficiencies and limitations associated with it.
Methods
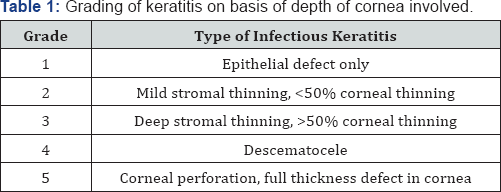
A prospective, interventional study was done on 150 eyes of 150 patients. All patients with refractory (unresponsive to conventional treatments significantly for more than 2 weeks) infective keratitis, advanced infectious keratitis with descematocele and corneal perforation requiring urgent concealment to salvage the eye, were treated with single or multi layered AMT. Patients with non-infective ulcers and perforations were excluded from the study. Corneal ulcer was graded 1-5 according to the depth of corneal involvement on slit lamp biomicroscopy (Table 1). Microbial investigations (staining for bacteria and fungus with culture-sensitivity) were done and antimicrobials started accordingly. B-scan ultra sonography was done in hazy media to rule out involvement of posterior segment. Any systemic (diabetes) or ocular (dacryocystitis) conditions hindering the healing of ulcer or triggering the infection were investigated and managed.
On basis of slit lamp examination at the site of most impact.
Technique
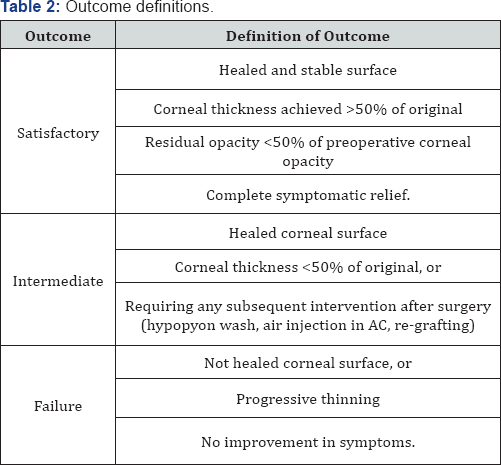
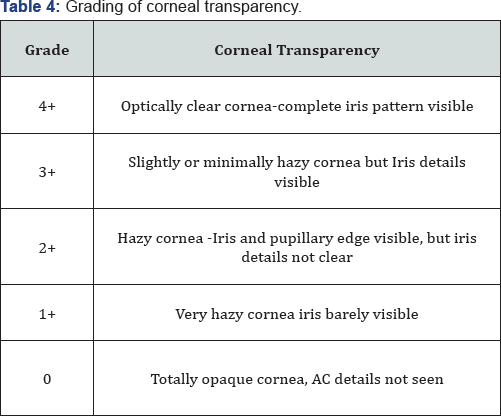
Surgery was performed preferably under sub conjunctival or peribulbar anesthesia. In children or uncooperative patients general anesthesia was used. Debridement of the necrotic tissue was done from and around the ulcer bed. Care was taken to remove the pseudo cornea over the perforation at the end of debridement to prevent leaking of aqueous and thus allowing proper keratectomy. Single layer preserved AM was used in cases of deep stromal ulcer. AM with epithelial side up was spread over the ulcer and trimmed to fit the ulcer. It was secured with continuous or interrupted 10-0 monofilament nylon suture. Descematocele and small corneal perforations up to 4mm were treated with multilayer AMT owing to deep corneal involvement. A sheet of AM, folded over it-self with epithelial side out, filled the ulcer crater and anchored to the healthy ulcer margin with interrupted 10-0 nylon suture. It was covered with a single sheet of AM similarly as in cases of deep stromal ulcers. In large corneal perforations of 4-6mm with extensive surrounding stromal necrosis, margins were not sturdy to hold the suture and there was a risk of cutting-off a corneal bite. In such cases single layer was sutured at limbus to at least provide tectonic support to the eyeball and delaying the need for PK. Side port or paracentasis was made in cases hypopyon and corneal perforation to reform the anterior chamber with air and reposit the prolapsed iris with help of spatula. Anterior synechiae if present were broken to prevent formation of adherent leucoma and thereby secondary glaucoma. Hypopyon if present was washed through the side port and intracameral antibiotic or antifungal was also injected according to sensitivity. At the end a BCL was placed over the cornea to prevent irritation from corneal sutures and maintaining AM in place. Antimicrobial, cycloplegics, ocular hypotensive and lubricating drops were continued along with systemic supportive therapy. Frequent follow-ups were done weekly for 1 month, biweekly till 3 months and monthly till 6 months. Efficacy was monitored on basis of improvement in symptoms and visual acuity, healing of the ulcer by re-epithelization and formation of anterior chamber, achievement of corneal transparency and corneal thickness. Accordingly patient's outcome was described as satisfactory, intermediate and failure (Table 2).
Observation and Results
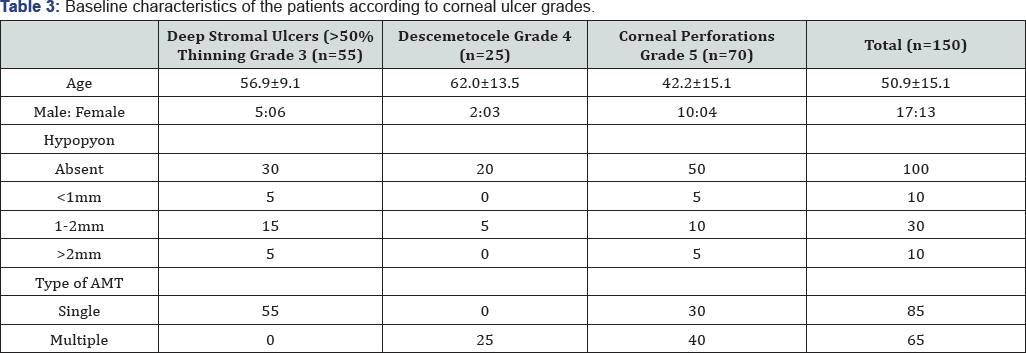
Keratitis was classified (Table 1) according to the depth of the cornea involved into 5 grades. Grades 1 and 2 responded well with medical management, therefore did not require AMT. Grades 3-5 with deeper corneal penetration of infection did not heal merely with medical management, there was an apprehension of corneal thinning and progression of infection, which required AMT. Of the 150 patients who underwent AMT, 55 (36.67%) were deep stromal ulcers, 25 (16.66%) were descematocele and maximum 70 (46.67%) patients were of corneal perforation ranging from 1-6mm. There was no age group or gender preponderance. Symptoms of redness, pain, watering and foreign body sensation (FBS) were collectively present in all the cases. Lid oedema and photophobia were also present in majority of the cases (70.6% and 90% respectively).Presence of discharge was seen in moderately less cases (30%). ranging between 1-2mm and 10 cases (20%) had hypopyon of Hypopyon was present in total 50 (33.3 %) cases where 10 cases >2mm (Table 3).
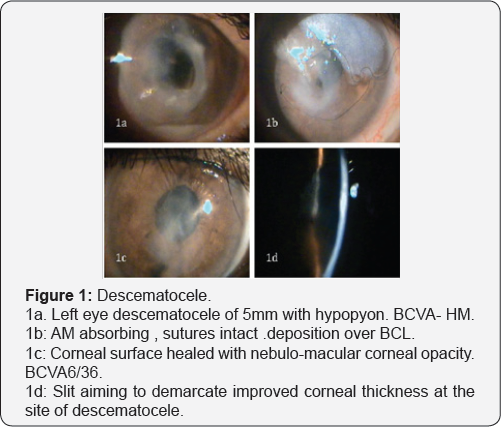
Single layer AMT was done in total 85 cases, all 55 cases of deep stromal ulcer and 30 cases of corneal perforation >4mm with extensive necrosis to provide tectonic support to maintain integrity to eyeball. Roofing with multilayer technique was done in 65 cases, all 25 cases of descematocele and 40 cases of corneal perforation >4mm in largest dimension where neighboring corneal tissue was healthy to hold the corneal sutures (Table 3). Patients were observed in repeated postoperative days. Rapid descent of symptoms was observed after the AMT. There was drastic improvement in pain, lid oedema, FBS and discharge in the first week. Symptoms were barely present in few cases by 1 month, which totally recovered by 3 months in all the cases (Figure 1).
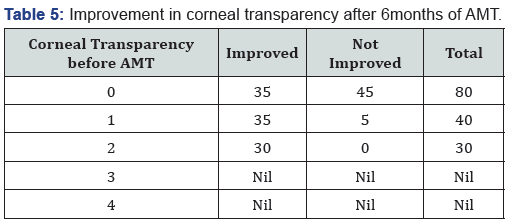
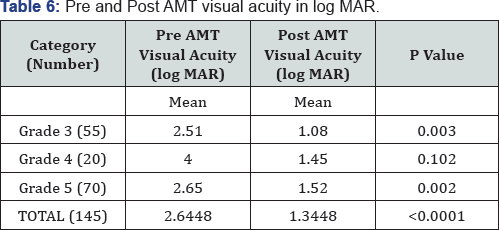
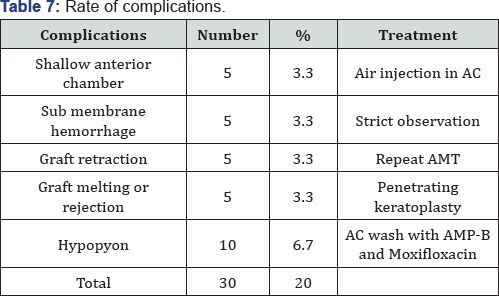
Corneal transparency graded from 0 (leucomatous opacity) to +4 (clear cornea, with no haze) was measured objectively at the site of most impact on slit lamp (Table 4). Improvement was seen in 105 of 150 cases and was statistically significant (p=0.016). However none of the cases improved to +4 transparency that is totally clear cornea (Table 5). Visual acuity was recorded before and after 6 months of treatment in 145 of 150 cases as 5 cases of fungal ulcer failed to heal with AMT (Table 6). Improvement in BCVA when taken collaborate, was extremely significant (p >0.0001). Mild to moderate complications were faced during the entire course of treatment. They were shallow anterior chamber in 5 cases in perforation which was tackled with air injection in anterior chamber and breaking anterior synechiae. Hemorrhage beneath AM in five cases which resolved spontaneously. Graft retraction was seen in five cases for which repeat AMT was done. Hypopyon developed in 10 cases and did not resolve with topical therapy was managed with anterior chamber wash and intracameral moxifloxacin and amphotericin-B respectively (Table 7). Hypopyon did not redevelop in these cases. All the complications were successfully managed with appropriate treatment with no recurrence and good results. Also no re-infection was noted. Graft melting and corneal perforation was seen in 5 cases of fulminant fungal ulcer and required urgent therapeutic PK.
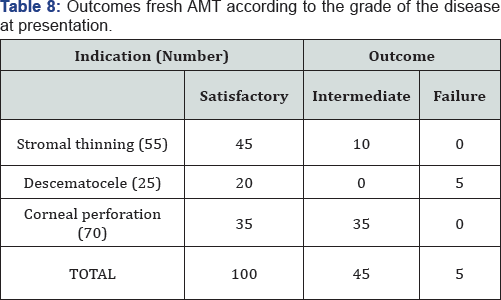
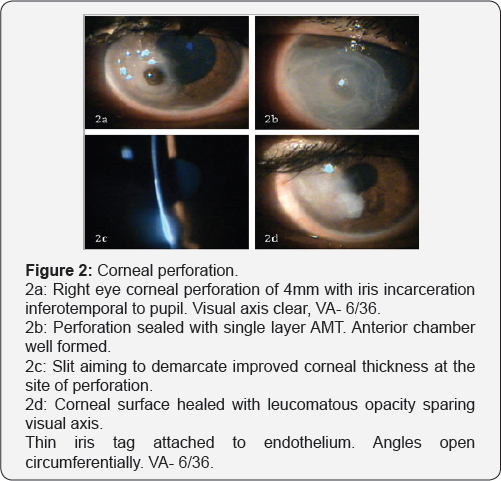
Satisfactory results were seen in 100 of 150 eyes (66.67%), intermediate results seen in 45 cases (23.33%). Failure was noted in 5 cases (3.33%) of fulminant fungal ulcers that showed subsequent corneal perforation requiring Therapeutic PK (Table 8). All the cases in intermediate category which also required subsequent intervention, healed with stable cornea. Thus, successful results were seen in 145 of 150 cases (96.67%) of which in 30 cases subsequent penetrating keratoplasty was done for leucomatous corneal opacity obscuring the visual axis left after healed ulcer (Figure 2).
Discussion
Approximately one-third of cases of infective keratitis require surgical interventions at the acute stage to prevent perforation or spreading of infection [10-14]. Keratoplasty being majorly followed in such situation faces a limitation of availability of good quality donor corneas, mainly in developing countries, recurrence of infection, difficulty in technique and graft rejection. Moreover, for fungal keratitis PK is technique dependent and may also carry a risk of recurrent infection [15].
Thus AMT is sought as an alternative, which has been extensively reported in ophthalmology literature [16-19]. AMT offers the advantage of stimulating re-epithelization, preventing neovascularization and scar formation and avoiding potential allograft rejection. Even if corneal transplantation is needed, the success rate is improved if performed on an eye with reduce infection and inflammation, this can be achieved with AMT [8,9]. In present study complete epithelization was noted in 145 of 150 cases, that is 96.67% success rate. Similar to our study, Chen et al. [20], showed 82.61% success rate, 4 of 23 cases in there study faced AM melting and graft failure requiring therapeutic PK in 3 and delayed healing with vascularization in the other. Kim et al. [21] used multilayer AM in cases of descematocele and corneal perforation. Corneal surface was healed successfully in all cases, and no recurrence of infection or rejection was experienced. Hanada et al. [22] used multiple layers of AM for deeper stromal ulcers down to descemetocele, to restore the normal corneal thickness as well as in corneal perforations from 0.5 to 3mm with or without additional tissue adhesive with high success rates (73-93%). In present study corneal perforations in cases of infectious keratitis up to 6mm have been treated successfully with AMT alone, and 100% corneal epithelization with more than 50% corneal thickness have been achieved in all 70 cases of perforation. In a series by Heiligenhaus et al. [23]. Seven patients with herpes simplex virus or varicella zoster- induced severe ulcerative keratitis, 5 of 7 eyes healed after first AMT [23]. In another study, stromal defect was filled up with multilayer technique proved to be better than monolayer procedure [22,24,25].
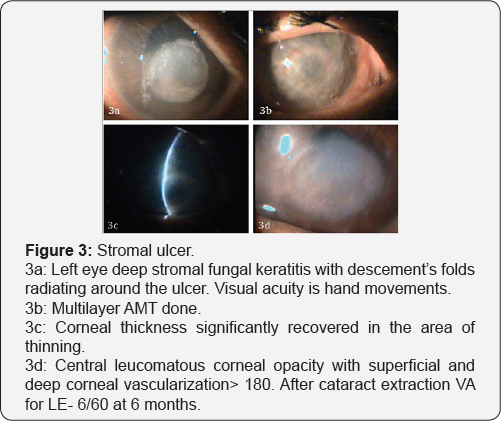
In present study 70% showed significant improvement in corneal transparency and increasing corneal transparency improves the best-corrected visual acuity further emphasizing the healing properties of AMT. Chen et al. [20], preserved useful vision after AMT in cases of fungal keratitis in 52.2% eyes. Kim et al. [21], reported 21 cases of successful AMT in infectious keratitis, in which visual acuity increased except for 5 cases because of irreversible corneal opacity. AMT has come up as a very effective managing technique for refractory ulcers. It aids in permanent healing of the refractory infective keratitis and prepares the cornea for definitive reconstructive procedure if required (Figure 3).
Conclusion
We have found that AMT represents a viable method of treatment to promote healing and prevent progressive melting of refractory infectious keratitis. Besides being cost-effective it’s easy to perform, with a short learning curve. Thus, it might be considered a first-line surgical technique when maximal medical treatment has failed.
References
- Brillant LB, Pokhrel RP, Grasset NE, Lepkowski JM, Kolstrad A, et al. (1985) Epidemiology of blindness in Nepal. Bull World health organ 63(2): 375-86.
- Whitcher J, Srinivansan M (1997) Corneal Ulceration in the developing World. Br J Ophthalmol 81(8): 622-623.
- Leonard PKA, Boruchoff A, Azar DT, Yanoff D, chapter 60 penetrating keratoplasty.
- Markuszewska J, Krzyzanowska P (1999) Amniotic membrane transplantation (AMT) for ocular surface reconstruction. Klin Oczna 101(4): 311-316.
- Solomon A, Tseng SCG (2000) Amniotic membrane transplantation for ocular surface diseases. Harefuah 139: 134-140.
- Kruse FE, Meller D (2001) Amniotic membrane transplantation for reconstruction of the ocular surface. Ophthalmology 98(9): 801-810.
- Sangwan VS, Burman S, Tejwani S, Mahesh SP, Murthy R (2007) Amniotic membrane transplantation: A review of current indications in the management of ophthalmic disorders. Indian J Ophthalmol 55(4): 251-260.
- Liu J, Sheha H, Fu Y, Liang L, Tseng SC (2010) Update on amniotic membrane transplantation. Expert Rev Ophthalmol 5(5): 645-661.
- Solomon A, Meller D, Prabhasawat P, John T, Espana EM, et al. (2002) Amniotic membrane grafts for nontraumatic corneal perforations, descemetoceles, and deep ulcers. Ophthalmology 109(4): 694-703.
- Rosa RH, Miller D, Alfonso EC (1994) The changing spectrum of fungal keratitis in south Florida. Ophthalmology 101(6): 1005-1013.
- Xie L, Shi W, Liu Z, Li S (2002) Lamellar keratoplasty for the treatment of fungal keratitis. Cornea 21(1): 33-37.
- Panda A, Vajpayee RB, Kumar TS (1991) Critical evaluation of therapeutic keratoplasty in cases of keratomycosis. Ann Ophthalmol 23(10): 373-376.
- Chen WL, Wu CY, Hu FR, Wang IJ (2004) Therapeutic penetrating keratoplasty for microbial keratitis in Taiwan from 1987-2001. Am J Ophthalmol 137(4): 736-743.
- Tanure MA, Cohen EJ, Sudesh S, Rapuano CJ, Laibson PR, et al. (2000) Spectrum of fungal keratitis at Wills Eye Hospital, Philadelphia, Pennsylvania. Cornea 19(3): 307-312.
- Yao YF, Zhang YM, Zhou P, Zhang B, Qiu WY, et al. (2003) Therapeutic penetrating keratoplasty in severe fungal keratitis using cryo preserved donor corneas. Br J Ophthalmol 87: 543-547.
- Chen HJ, Pires RTF, Tseng SCG (2000) Amniotic membrane transplantation for severe neurotrophic corneal ulcers. Br J Ophthalmol 84(8): 826-833.
- Lee S, Tseng SC (1997) Amniotic membrane transplantation for persistent epithelial defects with ulceration. Am J Ophthalmol 123(3): 303-312.
- Shimazaki J, Yang HY, Tsubota K (1997) Amniotic membrane transplantation for ocular surface reconstruction in patients with chemical and thermal burns. Ophthalmology 104(12): 2068-2076.
- Ma DH, See LC, Liau SB, Tsai RJ (2000) Amniotic membrane graft for primary pterygium: comparison with conjunctival autograft and topical mitomycin C treatment. Br J Ophthalmol 84(9): 973-978.
- Chen HC, Tan HY, Hsiao CH, Huang SC, Lin KK, et al. (2006) Amniotic Membrane Transplantation for Persistent Corneal Ulcers and Perforations in Acute Fungal Keratitis. Cornea 25(5): 564-572.
- Kim JS, Kim JC, Hahn TW, Park WC (2001) Amniotic membrane transplantation in infectious corneal ulcer. Cornea 20(7):720-726
- Hanada K, Shimazaki J, Shimmura S, Tsubota K (2001) Multilayered amniotic membrane transplantation for severe ulceration of the cornea and sclera. Am J Ophthalmol 131(3): 324-331.
- Heiligenhaus A, Li H, Hernandez Galindo EE, Koch JM, Steuhl KP, et al. (2003) Management of acute ulcerative and necrotizing herpes simpex and zoster keratitis with amniotic membrane transplantation. Br J Ophthalmol 87(10): 1215-1219.
- Kruse FE, Rohrschneider K, Völcker HE (1999) Multilayer membrane transplantation for reconstruction of deep corneal ulcers. amniotic Ophthalmology 106(8): 1504-1511.
- Hu DJ, Basti S, Bryar PJ (2003) Staining characteristics of preserved human amniotic membrane. Cornea 22(1): 37-40.






























