Safety and Efficacy of Ospemifene in Women with A History of Breast Cancer
Nico Bruyniks1*, Lino Del Pup2 and Nicoletta Biglia3
1BrInPhar Ltd, Iver Heath, Buckinghamshire, United Kingdom
2Centro di Riferimento Oncologico, National Cancer Institue, Italy
3Department of Oncological Gynaecology, University of Turin, Italy
Submission: December 21, 2018;Published: January 08, 2019
*Corresponding author: Nico Bruyniks, BrInPhar Ltd, Iver Heath, Buckinghamshire, United Kingdom
How to cite this article: Bruyniks N, Del Pup L, Biglia N. Safety and Efficacy of Ospemifene in Women with A History of Breast Cancer. J Gynecol Women’s Health. 2019: 13(5): 555871. DOI: 10.19080/JGWH.2019.13.555871
Abstract
Background: This post-hoc analysis compares the efficacy and safety data from women with or without a history of breast cancer who had been enrolled in the ospemifene clinical trial program.
Methods: Efficacy was assessed by the co-primary endpoints of most bothersome symptom (MBS; pooled data from two studies) and mean baseline to Week 12 change in vaginal pH and maturation index (pooled data from three studies). Safety was assessed using pooled data from all three studies. Comparisons were made between ospemifene-treated women with a history of breast cancer (≥10 years prior to enrolment; n=11) versus those without (n=1091).
Result: There were no differences in ospemifene-related improvements in symptoms of vulvar and vaginal atrophy (percentage whose MBS severity improved by ≥1 point on the 4-point severity scale) and in physiological parameters (vaginal pH, parabasal cells, and superficial cells) in women with versus without a history of breast cancer. Treatment-emergent adverse events and adverse drug reactions were comparable in the women using ospemifene who had a previous history of breast cancer (64% and 36%, respectively) compared with those that who did not (69% and 47%, respectively).
Conclusion: In this small post-hoc analysis, previous history of breast cancer did not appear to affect the efficacy or safety of ospemifene. Trial registration numbers NCT01585558, NCT01586364, NCT00729469 and NCT00566982.
Keywords: Breast cancer; Dyspareunia; Estrogen agonist/antagonist; Ospemifene; Postmenopausal; Safety; Selective estrogen receptor modulator; Vulvar and vaginal atrophy
Abbreviatations: ADR: Adverse Drug Reaction; ER: Estrogen Receptor; EU: European Union; FDA: Food and Drug Administration; MBS: Most Bothersome Symptom; SD: Standard Deviation; SERM: Selective Estrogen Receptor Modulator; TEAE: Treatment-Emergent Adverse Event; VVA: Vulvar and Vaginal Atrophy
Background
Vulvar and vaginal atrophy (VVA) is a chronic, progressive postmenopausal condition characterized by dyspareunia, vaginal dryness and vaginal irritation, which are a consequence of a decline in endogenous estrogen production [1]. It has previously been shown that women with a history of breast cancer are more likely to have moderate or severe symptoms and signs of VVA than women without breast cancer, particularly if treated with aromatase inhibitors [2] or tamoxifen after high-dose chemotherapy [3]. Due to the risk of breast cancer recurrence if exposed to estrogen [4], all estrogens (even low-dose vaginal estrogens) are contra-indicated in women with known, past, or suspected breast cancer [5].
Ospemifene is an oral non-estrogen treatment that has been approved for the treatment of moderate to severe symptomatic VVA in post-menopausal women who are not candidates for local vaginal estrogen therapy [5,6]. It belongs to the selective estrogen receptor modulator (SERM) class, which also includes treatments indicated for the treatment and/or prevention of certain breast cancers [7-10]. Ospemifene binds selectively to estrogen receptors to exert a tissue-specific effect [11]. In the breast, there are two estrogen receptors (ERα and ERβ), which can bind estrogen or a SERM [12,13]. Preclinical animal data suggest that ospemifene inhibits malignant breast tissue growth [14-18]. In addition, data from the ospemifene clinical trial program in women with VVA (including long-term safety analyses) show that it is well tolerated with neutral/minimal effects in breast tissue as assessed using breast palpation, mammography and breast-related treatment-emergent adverse events (TEAEs) [6,19-21]. Use of ospemifene is indicated in women with a history of breast cancer once treatment of the cancer has been completed [6].
The efficacy and safety of ospemifene was established in three pivotal Phase 3 trials: two 12-week, double-blind placebocontrolled trials [22-24] one of which had a blinded 40-week safety extension in women with an intact uterus [20] and a 52- week open-label extension in women without a uterus [21], and one 52-week, double-blind placebo-controlled safety trial that included a primary efficacy assessment at 12 weeks [19]. This short report focuses on efficacy and safety data from the 19 women with a history of breast cancer who had been included in these pivotal Phase 3 trials.
Methods
Three pivotal Phase 3 studies included 19 women with a history of breast cancer (diagnosis ≥10 years prior to enrolment)-11 who had been randomized to 60 mg ospemifene and 8 women to placebo:
a) In a 12-week, double-blind, placebo-controlled study (15-50310), there were 2 women in the 60 mg ospemifene group and 4 in the placebo group who had a prior history of breast cancer [24]. One woman in each group continued into the 40-week extension period [21].
b) In a second 12-week, double-blind, placebo-controlled study (15-50821), 7 women in the 60mg ospemifene group and 4 in the placebo group had a prior history of breast cancer [22,23].
c) In a 52-week, double-blind, placebo-controlled safety study (15-50718), 2 women in the 60mg ospemifene group had a prior history of breast cancer [19].
Based on the FDA ‘Guidance for Products to Treat Vasomotor Symptoms and Vulvar and Vaginal Atrophy Symptoms [25], the following endpoints were assessed as co-primary endpoints in the two efficacy studies and used in this analysis:
1. Mean change from baseline to Week 12 in the moderate to severe symptom that has been identified by the patient as the most bothersome symptom (MBS), based on the following symptoms (pooled data from studies 15-50310 and 15-50821) [16, 21-23]:
a. Vaginal dryness (none, mild, moderate, or severe)
b. Vaginal and/or vulvar irritation/itching (none, mild, moderate, or severe)
c. Dysuria (none, mild, moderate, or severe)
d. Vaginal pain associated with sexual activity (none, mild, moderate, or severe)
e. Vaginal bleeding associated with sexual activity (presence vs absence)
2. Mean change from baseline to Week 12 in vaginal pH (pooled data from all three studies) [16,19, 21-23]
3. Mean change from baseline to Week 12 in vaginal maturation index (parabasal and superficial cells; pooled data from all three studies) [16,19,21-23].
Due to the small number of women with a history of breast cancer in each of the MBS categories, it was not attempted to identify the effect on each MBS individually, but to analyse if there was a difference in effect on MBS between the women who did and those who did not have a history of breast cancer. Responders were identified as those whose severity of MBS improved by at least one point on the 4-point severity scale.
Safety data from all three studies were pooled for analysis [19,21-23]. No formal statistical testing was carried out, but where possible statistics have been included. Data are mean±SD unless otherwise specified.
Result
Baseline demographics
With the exception of a difference in age (those with a history of breast cancer were older at 65.4±6.9 years (range 54-79) vs 59.4±6.5 years (range 40-80); p=0.0014), there were no other differences between the group with (n=19) and without (n=1870) a history of breast cancer in terms of baseline demographics (data not shown).
Efficacy
Of the 9 subjects in the ospemifene 60mg group in studies 15-50831 and 15-50310, three identified dyspareunia as their MBS and six identified vaginal dryness. The percentage of women showing a reduction in severity of MBS (improvement by at least one point on the 4-point severity scale) was higher in the ospemifene 60 mg groups versus the placebo groups in the total population (74.9% vs 61.8%, respectively), women with a history of breast cancer (66.7% vs 50.0%, respectively) and women with no history of breast cancer (75.0% vs 61.9%, respectively). There were comparable improvements from baseline to Week 12 in the severity of the MBS (vaginal dryness or dyspareunia) for those in the ospemifene groups with (n=9) versus without (n=692) a history of breast cancer (Welch’s T-test, p=0.5492; data not shown).
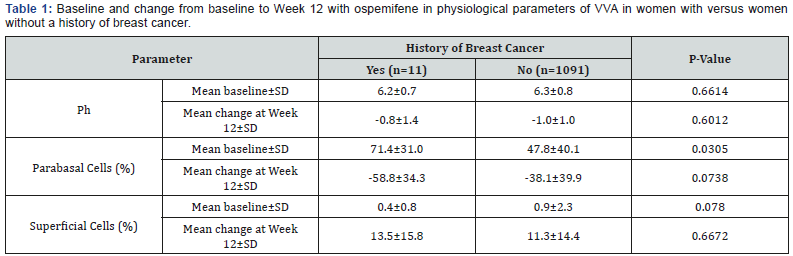
The data from all three studies were combined for the change from baseline to Week 12 for vaginal pH, parabasal cells, and superficial cells. With the exception of a higher percentage of parabasal cells in the group of women with a history of breast cancer, there were no significant differences in physiological parameters at baseline between women with or without a history of breast cancer in the ospemifene groups (Table 1). Ospemifene improved post-menopausal physiological changes in women with a history of breast cancer, with statistically significant mean decreases from baseline in the percentage of parabasal cells and vaginal pH and a statistically significant mean increase from baseline in the percentage of superficial cells (p<0.001 for each; Table 1). Although the decrease in percentage of parabasal cells and the increase in percentage of superficial cells in the women with a history of breast cancer was somewhat larger, and the decrease in vaginal pH somewhat smaller, than in women without a history of breast cancer, the differences were not statistically significant (Table 1).
Safety
In the clinical database of all Phase 2 and 3 studies of ospemifene (5-90mg, 6-64 weeks’ treatment duration; n=1,892), 1291 women (68.2%) reported a TEAE [5]. This was considered by the investigator to be related to the study drug (adverse drug reaction [ADR]) in 604 women (31.9%). There was no clear dose relationship for any TEAE or ADR.
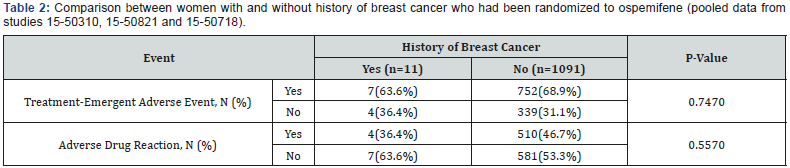
For studies 15-50310, 15-50821 and 15-50718, the incidence of TEAEs and ADRs reported by ospemifene-treated women who had a history of breast cancer was compared with those who did not have history of breast cancer (Table 2). No differences were found between groups. The proportion of women with a history of breast cancer reporting a TEAE was similar to the number of women on ospemifene in the Phase 2 and 3 double-blind, placebo-controlled studies (63.6% and 67.6%, respectively).
No adverse event was reported more than once in the ospemifene-treated women with a history of breast cancer. Three unique TEAEs were reported in this population (mild nasal dryness and sinus operation in one subject and abnormal weight gain in a second subject). All other TEAEs were also reported in the ospemifene population without a previous history of breast cancer and in the placebo population. Interestingly, whilst hot flushes were the most commonly reported TEAE (N=106, 8.5%) and ADR (N=93, 7.5%) in women randomized to ospemifene in the Phase 2 and 3 double-blind, placebo-controlled studies, they were not reported by ospemifene-treated women with a previous history of breast cancer.
Discussion
There are long-term safety concerns (particularly with respect to recurrent breast cancer risk) with systemic and vaginal estrogen therapies that limit their use in postmenopausal women with symptomatic VVA and previous breast cancer. In fact, ospemifene is the only treatment approved in the EU for this patient population, although it should be noted that it is indicated in women who have completed breast cancer therapies and is contraindicated in women who have breast cancer or are undergoing current treatment for breast cancer [5].
It has previously been shown in the ospemifene development program that ospemifene was effective and well tolerated in the management of postmenopausal women with VVA [19,20-24]. Importantly, enrolment of women with a history of breast cancer >10 years previously was permitted in two pivotal efficacy studies (15-50310 and 15-50821) and a 1-year endometrial safety study (15-50718). Here, we provided details of a post-hoc analysis of data from ospemifene-treated women with versus without a history of breast cancer from the three studies.
At baseline, the population of women with a history of breast cancer was not significantly different from the population that did not have such history, with the exception of age (mean difference in age was 8.4 years older in the population with a history of breast cancer). Unsurprisingly, the mean percentage of parabasal cells at baseline was also significantly higher in this population (71.4% vs 47.8% in those without breast cancer). Despite these differences, efficacy as assessed by vaginal dryness or dyspareunia (identified by the women as their MBS) and the improvement in physiology (vaginal pH and percentage of parabasal and superficial vaginal epithelial cells) was comparable between women with and without a history of breast cancer.
Likewise, there was no difference in the safety profile of ospemifene, regardless of whether women had a previous history of breast cancer or not. Three unique TEAEs were reported in women with a history of breast cancer: mild nasal dryness, sinus operation and abnormal weight gain. It is difficult to see how the first two could be related to ospemifene use. The abnormal weight gain was reported from the first day of tablet intake (from 82 to 87.7kg) and is the only increase in weight gain reported as a TEAE among 2471 subjects receiving ospemifene in the clinical trial program. There was no change in median weight in the placebo or 60mg ospemifene population in the 12-month safety study 15-50718 [19]. Interestingly, hot flushes were not reported in the ospemifene-treated women with a history of breast cancer.
The data from this analysis add to previous reports of neutral/beneficial effects of ospemifene in breast tissue from preclinical animal studies [14-18] and long-term safety analyses from clinical trials, which found no significant differences in breast-related adverse events between ospemifene and placebo [6,19-24].
Conclusion
The data do not show any differences in efficacy and safety between ospemifene-treated women with versus without a history of breast cancer. However, this analysis is limited due to its post-hoc nature and the small number of patients with a previous history of breast cancer and will require confirmation in a larger number of patients before firm conclusions can be made.
Declarations
Ethics approval and consent to participate
The three parent studies were performed in accordance with the Declaration of Helsinki current Good Clinical Practice, and local regulatory requirements. All participants provided written informed consent. The protocol for study 15-50821 [22,23] was reviewed and approved by the Copernicus Group Institutional Review Board (Durham, NC, USA) for most clinical sites; local institutional review boards were used for the remaining sites. The protocol, amendments and informed consent forms for study 15-50718 [19] were reviewed and approved by the Independent Ethics Committee (IEC) before study initiation. For study 15-50310, the protocol was reviewed and approved by the institutional review board of each study center.
Availability of data and material
The datasets used and analysed during the current study are available from the corresponding author on reasonable request.
Competing interests
NBr is a consultant to Shionogi Ltd. LDP acted as a consultant and advisory board member for TEVA and Shionogi Ltd. NBi is a member of advisory boards and/or consultant with Gedeon Richter, Italfarmaco, and Shionogi Ltd.
Funding
The three parent studies were funded by QuatRx Pharmaceuticals Company.
Authors’ Contributions
All authors were involved in analysing the data, drafting and/ or critically revising the manuscript, have given final approval for publication, and agreed to be accountable for the content of the manuscript.
Acknowledgement
We thank Susan Crawford, Absolute Healthcare Communications, Limited, who provided unpaid editorial assistance on the manuscript.
References
- Bride MMB, Rhodes DJ, Shuster LT (2010) Vulvovaginal atrophy. Mayo Clin Proc 85(1): 87-94.
- Baumgart J, Nilsson K, Stavreus-Evers A, Kask K, Villman K, et al. (2011) Urogenital disorders in women with adjuvant endocrine therapy after early breast cancer. Am J Obstet Gynecol 204(1): 26.e1-7.
- Mourits MJ, Böckermann I, Vries DEG, Zee VDAG, Hoor TKA, et al. (2002) Tamoxifen effects on subjective and psychosexual well-being, in a randomised breast cancer study comparing high-dose and standarddose chemotherapy. Br J Cancer 86(10): 1546-1550.
- Holmberg L, Iversen OE, Rudenstam CM, Hammar M, Kumpulainen E, et al. (2008) Increased risk of recurrence after hormone replacement therapy in breast cancer survivors. J Natl Cancer Inst 100(7): 475-482.
- Core smpc for hormone replacement therapy products, Heads of Medicines Agencies.
- (2015) Senshio (ospemifene) tablets. Summary of product characteristics. Shionogi Ltd, London, UK.
- Cummings SR, Eckert S, Krueger KA, Grady D, Powles TJ, et al. (1999) The effect of raloxifene on risk of breast cancer in postmenopausal women: results from the MORE randomized trial. Multiple Outcomes of Raloxifene Evaluation. JAMA 281(23): 2189-2197.
- Early Breast Cancer Trialists’ Collaborative Group (1998) Tamoxifen for early breast cancer: an overview of the randomised trials. Lancet 351(9114): 1451-1467.
- Evista [package insert] (2011) Indianapolis, IN: Eli Lilly and Company, USA.
- Nolvadex [package insert] (2002) Wilmington, DE: AstraZeneca, UK.
- Gregorio DMW, Zerbe RL, Wurz GT (2014) Ospemifene: a first-in-class, non-hormonal selective estrogen receptor modulator approved for the treatment of dyspareunia associated with vulvar and vaginal atrophy. Steroids 90: 82-93.
- Jordan VC, O’Malley BW (2007) Selective estrogen-receptor modulators and antihormonal resistance in breast cancer. J Clin Oncol 25(36): 5815-5824.
- Zwart W, Leeuw DR, Rondaij M, Neefjes J, Mancini MA, et al. (2010) The hinge region of the human estrogen receptor determines functional synergy between AF-1 and AF-2 in the quantitative response to estradiol and tamoxifen. J Cell Sci 123(pt 8): 1253-1261.
- Burich RA, McCall JL, Mehta NR, Greenberg BE, Bell KE, et al. (2012) Ospemifene and 4- hydroxyospemifene effectively prevent and treat breast cancer in the MTag.Tg transgenic mouse model. Menopause 19(1): 96-103.
- Namba R, Young LJ, Maglione JE, McGoldrick ET, Liu S, et al. (2005) Selective estrogen receptor modulators inhibit growth and progression of premalignant lesions in a mouse model of ductal carcinoma in situ. Breast Cancer Res 7(6): R881-R889.
- Qu Q, Zheng H, Dahllund J, Laine A, Cockcroft N, et al. (2000) Selective estrogenic effects of a novel triphenylethylene compound, FC1271a, on bone, cholesterol level, and reproductive tissues in intact and ovariectomized rats. Endocrinology 141(2): 809-820.
- Taras TL, Wurz GT, DeGregorio MW (2001) In-vitro and in-vivo biologic effects of ospemifene (FC-1271a) in breast cancer. J Steroid Biochem Mol Biol 77(4-5): 271-279.
- Wurz GT, Read KC, Marchisano-Karpman C, Gregg JP, Beckett LA, et al. (2005) Ospemifene inhibits the growth of dimethylbenzanthraceneinduced mammary tumors in Sencar mice. J Steroid Biochem Mol Biol 97(3): 230-240.
- Goldstein SR, Bachmann GA, Koninckx PR, Lin VH, Portman DJ, et al. (2014) Ospemifene Study Group. Ospemifene 12-month safety and efficacy in postmenopausal women with vulvar and vaginal atrophy. Climacteric 17(2): 173-182.
- Simon J, Portman D, Mabey RG (2014) Ospemifene Study Group. Long-term safety of ospemifene (52-week extension) in the treatment of vulvar and vaginal atrophy in hysterectomized postmenopausal women. Maturitas 77(3): 274-281.
- Simon JA, Lin VH, Radovich C, Bachmann GA (2013) One-year long-term safety extension study of ospemifene for the treatment of vulvar and vaginal atrophy in postmenopausal women with a uterus. Menopause 20(4): 418-427.
- Portman D, Palacios S, Nappi RE, Mueck AO (2014) Ospemifene, a nonoestrogen selective oestrogen receptor modulator for the treatment of vaginal dryness associated with postmenopausal vulvar and vaginal atrophy: A randomised, placebo-controlled, phase III trial. Maturitas 78(2): 91-98.
- Portman DJ, Bachmann GA, Simon JA, Ospemifene (2013) A novel selective estrogen receptor modulator for treating dyspareunia associated with postmenopausal vulvar and vaginal atrophy. Menopause 20(6): 623-630.
- Bachmann GA, Komi JO (2010) Ospemifene Study Group. Ospemifene effectively treats vulvovaginal atrophy in postmenopausal women: results from a pivotal phase 3 study. Menopause 17(3): 480-486.
- (2018) Guidance for Industry Estrogen and Estrogen/Progestin Drug Products to Treat Vasomotor Symptoms and Vulvar and Vaginal Atrophy Symptoms -Recommendations for Clinical Evaluation.






























