Impact of Serum Progesterone Levels on Pregnancy Outcomes in Patients Undergoing Artificial Endometrial Preparation for Embryo Transfer
Diego Tarasconi1,2*, Miguel Ángel Checa1,2,3, Mireia Julià1,2,3 and Juan José Espinós1,2,3
1Fundación Fertty Assisted Reproduction Clinic, Barcelona, Spain
2Universitat Autònoma de Barcelona, Barcelona, Spain
3Barcelona Research Infertility Group, IMIM, Hospital del Mar, Barcelona, Spain
Submission: March 22, 2022; Published: April 11, 2022
*Corresponding author: Diego Tarasconi, Fundación Fertty Assisted Reproduction Clinic, Universitat Autònoma de Barcelona, Barcelona, Spain
How to cite this article: Diego T, Miguel Ángel C, Mireia J, Juan José E. Impact of Serum Progesterone Levels on Pregnancy Outcomes in Patients Undergoing Artificial Endometrial Preparation for Embryo Transfer. J Endocrinol Thyroid Res. 2022; 6(5): 555697. DOI:10.19080/JETR.2022.06.555697
Abstract
In this retrospective cohort study, we examined the impact of measuring serum progesterone (P) levels on pregnancy outcomes in patients undergoing artificial endometrial preparation and embryo transfer (ET). We analyzed 342 ET cycles at the blastocyst stage, either from autologous or donated oocytes, in a private fertility care center. ET cycles were performed after an artificial endometrial preparation with a sequential administration of estradiol valerate followed by vaginal micronized progesterone. In the study population, when serum P levels were lower than 10.0 ng/ml the day prior to ET, a subcutaneous preparation of P was added, attempting to bring its levels to the luteal phase support. Interestingly, in the case of autologous ET, the rates of early pregnancy loss were reduced by measuring and eventually supplementing serum P levels.
Keywords: Serum progesterone; Blastocyst transfer; Early pregnancy loss; Frozen embryo transfer; Endometrial preparation
Abbreviations: P: Progesterone; ET: Embryo transfer; IVF: In vitro fertilization; HRT: Hormone replacement therapy; OPU: Oocyte pick-up; ICSI: Intracytoplasmatic sperm injection
Introduction
Nowadays, after in vitro fertilization (IVF), many embryos are cryopreserved before intrauterine transfer. This is due to the supernumerary embryos that are obtained and vitrified at each cycle. Also, there are cases in which embryo transfer (ET) is deferred for medical or social reasons [1,2]. Improvements in cryopreservation techniques, such as vitrification, resulted in high embryonic survival and viability rates, and increased their application and the number of postponed ET cycles [3]. When performing a postponed ET cycle, the endometrium must be prepared to achieve a secretory pattern and to open an adequate window for implantation. No endometrial preparation protocol for ET is consistently superior to the others in terms of pregnancy outcomes [4]. Therefore, the protocol must be chosen according to the physician and patients convenience. Most centers prefer to use artificial endometrium preparation with hormone replacement therapy (HRT) because it allows to schedule the day of the ET [5].This treatment consists in sequentially administering exogenous estrongens and progesterone (P). It starts with administering estrogens at the beginning of the menstrual cycle. Such hormone stimulates the endometrium to proliferate and achieve a minimal thickness and pattern. Afterwards, the addition of P allows to modify the endometrium into a secretory one, always mimicking the normal menstrual cycle [6]. Regarding the administration route for the different hormones, it has been shown that estrogens can be taken either orally or subcutaneously, without any differences in outcomes [7]. However, the oral route for P is not sufficient to achieve the necessary endometrial transformation, and the administration must be vaginal, intramuscular, or subcutaneous [8]. Vaginal administration is comfortable for patients and represents the most common route [9]. The result is a rapid absorption of P that directly modifies the endometrium. Such event is called “uterine first pass”. Despite of the advantages of this first pass, its correlation with endometrial and serum P levels has not been demonstrated yet. Indeed, several studies failed to prove that the histological changes in the endometrium were related with levels of P in the serum, as it occurs for intramuscular preparation of P [10,11]. Nevertheless, recently, a prospective study demonstrated a significant detrimental effect on pregnancy prognosis when P levels in the serum were lower than 9.2 ng/ml on the day of ET [12]. Similarly, Gaggiotti-Marre et al. showed significantly lower live birth rates and higher miscarriage rates after transfers of euploid embryos when serum P levels were lower than 10.64 ng/ml the day before ET [13]. The aim of this study was to retrospectively examine the impact of serum P levels on pregnancy outcomes in patients undergoing ET, after endometrial preparation cycle with sequential exogenous replacement of estrogens and progesterone. We assessed whether measuring P levels the day before ET and supplementing them in case they were low, could result in better pregnancy outcomes.
Materials and Methods
Study design
A retrospective cohort analysis was approved by Parc de Salut Mar ethics committee (n. 2020/9046) and performed at FERTTY, a private fertility care center in Barcelona, Spain, from March 2018 to January 2020.
Study population
This study included infertile patients undergoing ET at blastocyst stage after artificial endometrial preparation with HRT. We analyzed transfers of embryos obtained from autologous and donated eggs, including fresh and frozen-thawed embryos.
Study protocol
All patients received HRT either through oral administration of 6 mg/day of estradiol valerate (Progyluton® white tablets, Bayer Hispania Barcelona, Spain) or through transdermal patches releasing 150 mg of estradiol valerate every 2 days (Evopad®, Janssen Cilag, Madrid, Spain). Every 3 to 4 days from the onset of this treatment, a vaginal 2D ultrasound examination was performed to check if the endometrium was responding to the therapy, and if the ovaries were quiescent. Patients received estrogens for 10 – 13 days, till the endometrium presented with a triple layer pattern and its thickness was greater than 6.0 mm. At this point, 200 mg of natural micronized P (Utrogestan®, Seid, Barcelona, Spain or Progeffik®, Laboratorios Effik S.A, Madrid, Spain) were administered daily every 8 hours (at 0, 8, 16, and 24 hours). ET was performed after five days from the beginning of P therapy. We divided the population into 2 groups. In the study group, at the fourth day of P therapy, patients were submitted to blood analysis to determine the concentration of P in the serum. When P levels were lower than 10.0 ng/mL, patients received 25 mg/day in a subcutaneous preparation (Prolutex®, IBSA, Barcelona, Spain), starting on the same day. When P levels were higher than 10.0 ng/mL, no supplementation was administered and patients continued with the same HRT doses. The control group consisted of patients undergoing conventional HRT and ET. In case of pregnancy, all types of HRT were maintained until 9-10 weeks.
Blood analysis and sampling
Blood samples were drawn at our clinic between 9 and 11 a.m., after 1 - 3 hours from the last dose of vaginal P. Samples were tested by an electrochemiluminescence immunoassay (Cobas® e-600, Roche diagnostics GmbH, Germany) with a sensitivity and a detection limit of 0.05 ng/mL and 60.0 ng/mL, respectively.
Embryo development and transfer
Oocyte pick-up (OPU) was guided by ultrasound with anesthetic sedation. The semen was analyzed and prepared in the same day. Afterwards, embryos were generated in our laboratory through conventional IVF or intracytoplasmic sperm injection (ICSI), following medical recommendation. In vitro embryo culture and development was monitored in sequential culture medium, and morphologically analyzed and classified according to the Spanish ASEBIR classification [14]. ET was performed at the blastocyst stage in all patients under abdominal ultrasound control [15].
Statistical analysis
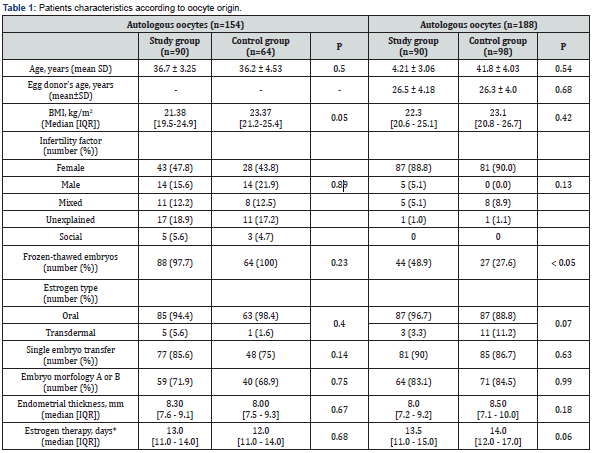
We carried out a descriptive analysis of the sample for all variables (Table 1). Quantitative variables were analyzed for normality through histograms and with the Shapiro-Wilk test. For the non-parametric variables, we calculated the median and the interquartile intervals and performed the Mann-Whitney test. To verify the correlation between variables, we used the Spearman`s correlation test. Ordinal and binary variables were compared using the Chi-square test and the Fisher’s exact test, respectively. We used different logistic regression models to measure the association between serum P levels and the pregnancy outcomes: model 1 (crude), adjusted by oocyte age; model 2, adjusted by BMI; and model 3, adjusted by number and quality of embryos. Differences, correlations, and other parameters for which we obtained a value of p <0.05 were considered statistically significant. All the analyses and comparisons in this study were stratified by the origin of oocytes. This was done to avoid that embryonic quality influenced the results, according to whether or not serum P levels were measured. The analyses were carried out with the R, Rstudio version 3.6.2, and SPSS Statistics software.
Source: *days of exogenous estrogen administration until the first day of the treatment with vaginal progesterone.
Results
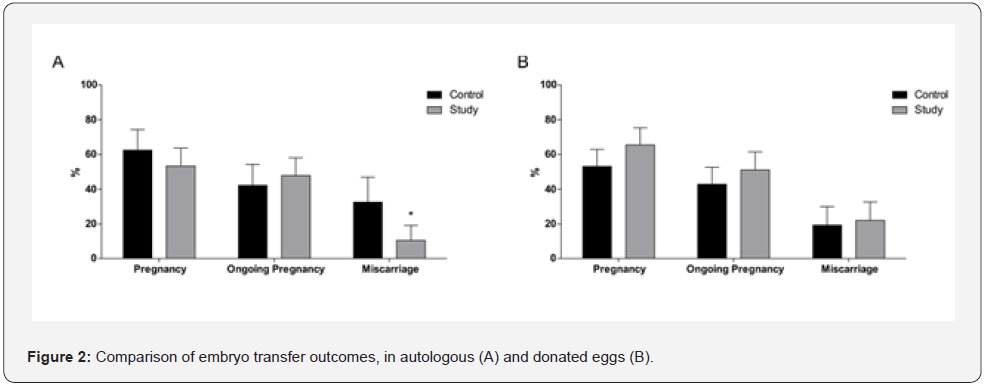
We initially selected a total of 261 women undergoing 359 cycles of ET. In this cohort, 17 cases were excluded from the analysis: 12 cases of modified natural ET cycles; 3 cases of ET at cleavage stage; 1 case of Mullerian abnormality submitted to ET with transmyometrial technique; and another one with a slow embryonic development and ET in a later blastocyst stage (D+7). These were divided in two groups according to the origin of the oocytes: 154 fell into the autologous eggs group, and 188 into the donated eggs group. Each of these two groups was further divided in a control and a study group, as specified in Materials and Methods. There were no significant differences between the study and the control groups in both the autologous eggs group and donated eggs group, considering different factors. Those factors included age; body mass index (BMI), infertility factor; estrogen type; number of embryos transferred; embryo morphology; endometrial thickness and days of estradiol treatment before administering P (Table 1). The prevalence of P levels lower than 10.0 ng/ml was 31.1% for patients in the autologous eggs group, and 37.7% for patients in the donated eggs group. The overall single ET rate was 84.3 % and the rate of newborn twins and ongoing twin pregnancies was 3.8%. To evaluate the effect of P levels, a multivariable logistic model was fitted to analyze ongoing pregnancy and miscarriage rates adjusted by age, BMI and embryo quality. The ongoing pregnancy rate was not significantly different between the study group and the control group in patients undergoing autologous ET (47.8% vs 42.2%, P = 0.51 [OR 1.25 - IC 95% 0.62 – 2.52]). However, importantly, the study group had a significantly lower miscarriage rate compared to the control group (10.4% vs 32.5%, P = 0.015 [OR 0.24 - IC 95% 0.06 – 0.84]) (Figure 1). On the contrary, in patients that underwent the transfer of embryos generated from donated eggs, we did not find any significant difference between study group and control group, either in ongoing pregnancy rates (51.1 % versus 42.9%, P > 0.05 [OR 1.39 - IC 95% 0.75 - 2.58) or in miscarriage rates (22% versus 19.2%, P > 0.05 [OR 0.82 - IC 95% 0.42 - 3.37]) (Figure 2).

Discussion
Our study shows that determining the levels of P in the serum the day before ET and supplementing them when indicated reduce miscarriage rates in autologous ET cycles. Although lower concentrations of P in the serum are sufficient to allow implantation, we speculate that certain levels must be reached to obtain an adequate immunological environment and reduce pregnancy loss. In the case of serum P levels lower than 10.0 ng/ ml, we indicate to supplement with an alternative preparation of subcutaneous P. This protocol is also based on concordant results from previous studies with similar models [12,13]. The authors of these studies could infer that higher P levels were related to better pregnancy outcomes after artificial endometrial preparation and transfer of frozen-thawed blastocysts. On the contrary, in transfer cycles of embryos from donated eggs, we observed the same pregnancy outcomes regardless of P levels measurement and supplementation. A possible reason for this might be the limited number of cases analyzed and the retrospective design of our study. Despite these limitations, our study population was quite consistent with the cases in the daily routine of assisted reproduction centers; as we could not indicate pre-implantation genetic testing for all patients, nor include most of them in oocyte reception programs.
Another limitation that could represent a bias in our analysis was the lack of data on the oocyte status before IVF (frozen vs fresh) and the different ovarian stimulation protocols (antagonist vs progesterone priming). Moreover, not to compromise patients convenience, a measurement of serum P levels after supplementation was not performed. This would have provided a final proof that a higher concentration of P was achieved in the serum of these patients.
Conclusion
More and more cycles of ET are postponed and carried out in non-continuity with ovarian stimulation. Therefore, it is necessary to seek for innovative solutions to refine ET, and create higher consensus on the type of preparation specifically indicated for single patients. This study suggests that, in patients undergoing HRT and autologous ET, the measurement of serum P levels on the day prior to ET and its eventual supplementation reduce the miscarriage rate. This may lead to select patients who would benefit from additional subcutaneous P in the luteal phase. Finally, we believe that our findings represent a step forward in the field of IVF, leading to better patients stratification and pregnancy outcomes.
Acknowledgements
The authors thank the patients of this group, that participate in the study without any financial incentive. This work was performed under the auspices of the Assisted Reproduction Clinic FERTTY, and the Universitat Autònoma de Barcelona, Barcelona, Spain.
References
- Chen ZJ, Shi Y, Sun Y, Zhang B, Liang X, et al. (2016) Fresh versus frozen embryos for infertility in the polycystic ovary syndrome. N Engl J Med 375(6): 523-533.
- Shapiro BS, Daneshmand ST, Garner FC, Aguirre M, Hudson C, et al. (2011) Evidence of impaired endometrial receptivity after ovarian stimulation for in vitro fertilization: a prospective randomized trial comparing fresh and frozen-thawed embryo transfer in normal responders. 96(2): 344-348.
- Rienzi L, Gracia C, Maggiulli R, LaBarbera AR, Kaser DJ, et al. (2017) Oocyte, embryo and blastocyst cryopreservation in ART: systematic review and meta-analysis comparing slow-freezing versus vitrification to produce evidence for the development of global guidance. Hum Reprod Update 23(2): 139-155.
- Burks H, Paulson R (2015) Cryopreserved embryo transfer: Endometrial preparation and timing. Semin Reprod Med 33(2): 145-152.
- Ghobara T, Gelbaya TA, Ayeleke RO (2017) Cycle regimens for frozen-thawed embryo transfer. Cochrane Database of Systematic 7(7): CD003414.
- Lutjen P, Trounson A, Leeton J, Findlay J, Wood C, et al. (2020) The establishment and maintenance of pregnancy using in vitro fertilization and embryo donation in a patient with primary ovarian failure. Nature 307(5947): 174-175.
- Molina PF, Lliso CC, Collado RC, García MM, Bachiller MD, et al. (2018) Oral versus transdermal oestrogen delivery for endometrial preparation before embryo transfer: a prospective, comparative, randomized clinical trial. Reprod Biomed Online 37(6): 693-702.
- Fanchin R, De Ziegler D, Bergeron C, Righini C, Torrisi C, et al. (1997) Transvaginal administration of progesterone. Obstet Gynecol 90(3): 396-401.
- Ziegler D, Seidler L, Schärer E, Bouchard P (1995) Non-oral administration of progesterone: experiences and possibilities of the transvaginal route]. Praxis (Bern 1994) 84(5): 127-133.
- Devroey P, Palermo G, Bourgain C, Van Waesberghe L, Smitz J, et al. (2020) Progesterone administration in patients with absent ovaries. Int J Fertil 34(3): 188-193.
- Bourgain C, Devroey P, Waeshberghe LV, Smitz J, Steirteghem ACV (1990) Effects of natural progesterone on the morphology of the endometrium in patients with primary ovarian failure. Hum Reprod 5(5): 537-543.
- Labarta E, Mariani G, Holtmann N, Celada P, Remohí J, et al. (2017) Low serum progesterone on the day of embryo transfer is associated with a diminished ongoing pregnancy rate in oocyte donation cycles after artificial endometrial preparation: a prospective study. Hum Reprod 32(12): 2437-2442.
- Gaggiotti MS, Martinez F, Coll L, Garcia S, Álvarez M, et al. (2019) Low serum progesterone the day prior to frozen embryo transfer of euploid embryos is associated with significant reduction in live birth rates. Gynecol Endocrinol 35(5): 439-442.
- Pons MC (2015) Criterios ASEBIR de Valoración morfológica de ovocitos, embriones tempranos y blastocistos humanos. In: Góbalo, Agencia creativa digital, 3rd ed, Madrid, Spain, p. 58-68.
- Coroleu B, Barri PN, Carreras O, Martínez F, Veiga A, et al. (2002) The usefulness of ultrasound guidance in frozen-thawed embryo transfer: a prospective randomized clinical trial. Hum Reprod 17(11): 2885-2890.






























