Nanomedicine May Provide New Hope for Brain Cancer Therapy
Alain L Fymat*
International Institute of Medicine & Science, USA
Submission: November 18, 2018;Published: December 06, 2018
*Corresponding author: Alain L Fymat, International Institute of Medicine & Science, California, USA
How to cite this article: Alain L Fymat. Nanomedicine May Provide New Hope for Brain Cancer Therapy. Glob J Nano. 2018; 4(5): 555647. DOI: 10.19080/GJN.2018.04.555647
Abstract
Cancer cells are notoriously resistant to drugs intended to kill them by rerouting signaling networks responsible for cancer growth, proliferation and survival. They also contribute to drug resistance to conventional treatment and high recurrence rates. For brain cancers (glioblastomas), the brain protective barriers, particularly the blood brain barrier, inhibit or even preclude the entry of many (if not most) therapeutic drugs. Because of this barrier, the several drugs approved by the FDA have had little effect. Fortunately, nanobiotechnology has come to the rescue offering nanoparticles and nanodevices to ferry therapeutic drugs across the barrier and deposit them theoretically at the right location, at the right time, and in the right dosage or dose fractionation. These developments are described below, and comments are made regarding the needed advances in nanobiotechnology as well as the lacking understanding of the deep biology of glioblastomas.
Keywords: Blood brain barrier; Glioblastoma; Nanodevices; Nanoparticles; Nanobiotechnology
Abbrevations: BBB: Blood Brain Barrier; BPB: Blood Protective Barriers; CSF: Cerebro Spinal Fluid; CNS: Central Nervous System; CT: Computed Tomography; FDA: Food and Drug Administration; GB: Glioblastoma; GBM: Glioblastoma Multiform; 3HM: 3-Helix Micelles; LPH: Lipid-Coated Polymeric Hybrid; MDR: Multiple Drug Resistance; NBT: Nanobiotechnology; MRI: Magnetic Resonance Imaging; ND: Nanodevices; NGP: Neural/Glial Progenitors; NM: Nanomedicine; NP: Nanoparticles; NSC: Neural Stem Cells; PET: Positron Emission Tomography; P-gp: P-Glycoprotein; PLGA: Poly Lactic Co-Glycolic Acid; PNP: Polymeric NPs; RCT: Rational Combination Therapy; SERS: Surface-Enhanced Raman Spectroscopy; TAC: Transit Amplifying Cells; TIC: Tumor Initiating Cells
Drugs: Bradykinin; Casomorphion; Mannitol; Osteopontin.
Introduction
Cancer cells are notoriously resistant to drugs intended to kill them. They reroute signaling networks responsible for cancer cell growth, proliferation and survival. A drug may block a signaling pathway but within days (at times minutes), cancer cells rely on alternate pathways to promote their survival. The simultaneous use of several drugs, called “rational combination therapy” (RCT) aims to block pathways (primary and alternate) to pre-emptively block escape routes. Unfortunately, RCT has limited efficacy because drugs have different chemical properties, travel to different parts of the body, and enter cancerous cells at different rates. This illustrates one of the many treatment difficulties for cancer.
The situation is further complicated by the fact that cancer cells with stem cell-like properties reside in a protective niche around arterioles, maintaining a hypoxic environment and extending their tendrils into surrounding tissues. They also contribute to drug resistance to conventional treatment and high recurrence rates. In the case of brain cancers (including glioblastomas, also called glioblastoma multiform, GBM), the situation is compounded by the presence of protective barriers (BPBs), more particularly the blood brain barrier (BBB), that preclude the entry of many (if not most) therapeutic drugs.
Several drugs have been approved by the FDA for brain cancer treatment, all with little impact because the BBB limits their accumulation. Fortunately, drugs encased in nanoparticles (NP) and delivered by nanodevices (NDs) can overcome these limitations. These will be described below including particularly special liposomes (size: 110 nm) and micelles (self- assemblies of peptides and polymers, size: 20nm). However, micelles have so far only been demonstrated in rats using radiolabeling with copper (Cu-64). To evaluate the different BBB responses, and to quantitatively study the behavior of the BBB, a database exists (“Brainpeps”), providing transport information and prioritizing peptide choices. As an example, Casomorphion (a heptapeptide) is able to pass the BBB.
On Glioblastomas
Figure 1 is a schematic representation of the development of a glioblastoma from a neural stem cell. It shows the following processes in GBM formation:
Differentiation
From neural stem cells (NSC) to transit amplifying cells (TAC) to neural/glial progenitors (NGP), to tumor initiating cells (TIC).
Proliferation
From TIC to GBM.
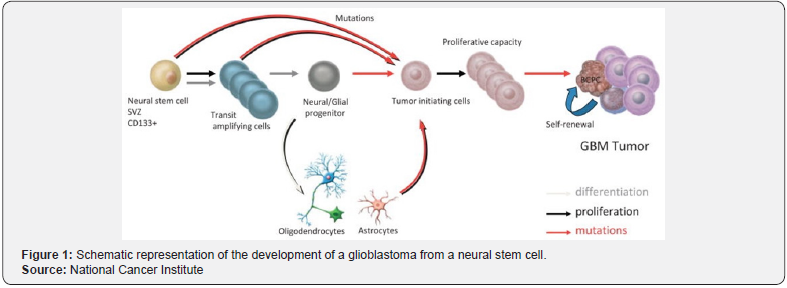
Conversion of NGP to TIC via Oligodendrocytes and Astrocytes
Glioblastomas (GBs) or glioblastomas multiform (GBMs) are the most common, most aggressive primary brain tumors in adults, accounting for ~ 15% of all brain tumors and 3.19 cases/100,000 patients/year. The average age at diagnosis is 64 with a median survival time of ~ 3 months (up to 1-2 years with treatment), the most common being 12-15 mos (3%-5% surviving more than 5 years) (Figure 2 & 3).


The first treatment line for GBMs was defined since 2005, but there is no standardized second treatment line for recurring tumors and no prevention strategy. Whereas several risk factors have been identified, let us recall this author’s admonition that “risk is not cause and risk management is not cure (only palliation)”. Unfortunately, most treatments cannot eradicate most tumor cells.
I have discussed elsewhere the surgical and non-surgical management and treatment of GBMs, including the cases of primary, secondary and recurring tumors Fymat [1-3]. That discussion included their morphology, biological types, risk factors, and prognosis. It also included the various management and treatment approaches including several surgical and non-surgical therapies: symptomatic, palliative, surgery, complementary and alternative, conformal, boron neutron capture, intensity-modulated proton beam, chemotherapy, antiangiogenic, alternating electric, immunotherapy, vaccines and lifestyle changes. Figure 4 & 5 show respectively a surgical total resection of a GBM and an excised brain showing the same.


The most serious impediment in the treatment of GBMs is posed by the brain protective barriers (BPBs) that hinder or preclude the penetration and delivery of most therapeutic drugs as next discussed.
On the Brain Protective Barriers
Being the most delicate organ of the body, the brain is protected against potentially toxic substances by the brain protective barriers (BPBs), four of which under the collective term “blood-brain barrier” (BBB) describe the four main interfaces between the central nervous system (CNS) and the periphery [4]:
The BBB proper
It is formed by complex tight junctions between the endothelial cells of the cerebral vasculature. Its primary manifestation is the impermeability of the capillary wall due to the presence of the junctions and a low endocytic activity;
The blood-cerebro spinal fluid (CSF) barrier
It is formed by tight junctions between epithelial cells of the choroid plexus. Both the BBB proper and the blood-CSF barrier extend down the spinal cord;
The outer CSF-brain barrier
It is formed by tight junctions between endothelial cells of the arachnoid vessels (the pia arachnoid); and
The inner CSF-brain barrier
It is formed by strap junctions between the neuro-ependymal cells lining the ventricular surfaces. It is present only in early development and absent in the adult.
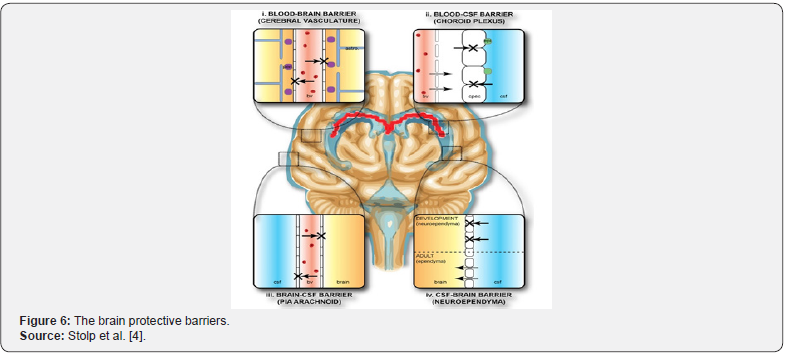
The above barriers and the blood retinal barrier (not studied here) are parts of a whole realm of barriers. We are here essentially interested in the BBB proper (Figure 6).
The BBB is a continuous lipid physical and physiological bilayer, constituting the major obstacle that restricts the entry of most pharmaceuticals and polar and lipid-insoluble substances and drugs into the brain. Therapeutic molecules and antibodies that might otherwise be effective in diagnosis and therapy do not cross the BBB in adequate amounts. Extensive efforts are therefore being made to come up with drug delivery strategies that would enable the passage of therapeutic molecules safely and effectively. Such strategies involve modifying the drug itself or coupling it to a vector for receptor-mediated or adsorptionmediated transcytosis [5,6].
Various attempts have been made to overcome the limited access of drugs to the brain, e.g. chemical modification, development of more hydrophobic analogs or linking an active compound to a specific carrier. Transient opening of the BBB in humans has been achieved by intracarotid infusion of hypertonic mannitol solutions or of bradykinin analogs. Another way to increase or decrease brain delivery of drugs is to modulate the P-glycoprotein (P-gp) whose substrates are actively pumped out of the cell and into the capillary lumen. Many P-gp inhibitors or inducers are available to enhance the therapeutic effects of centrally acting drugs or to decrease central adverse effects of peripherally active drugs. Nonetheless, overcoming the difficulty of delivering therapeutic agents to specific regions of the brain presents a major challenge to the treatment of most brain disorders. We are here mostly interested in the use of nanomedicine technologies, which may provide new hope for brain cancer therapies.
Use of Nanomedicine Technologies
Nanobiotechnology (NBT) can deliver drugs across, around, and beyond the BBB at the right location, the right time, and in the right dosage. Receptor-mediated transport systems enable molecules to cross the BBB in vivo. Although there are FDAapproved therapeutic drugs for the treatment of GBM, these treatments have had little impact on patient survival rate because the BBB has limited the accumulation of therapeutics within the brain.
We shall distinguish between the nanoparticles (NPs) and the nanodevices (NDs) that carry them and transport them across the BBB.
Nanoparticles
Of the multiplicity of NPs that are available, currently only two are of direct application to brain tumors, namely, gelatin NPs for delivering multiple drugs and, even more promisingly, lipid-based surface-engineered PLGA nanoparticles. These are briefly reviewed below. Other NPs that have been applied to other cancers but not yet to GBMs will not be considered here. The latter include: nutshells; microbubbles; the surface-enhanced Raman spectroscopy (SERS) nanotags; platelet-coated NPs; shapeshifting engineered NPs; kinase inhibitors in NP formulation; bioavailability-improved NPs and molecules; lipid-polymers and polymeric hybrid NPs; hybrid nanocrystals; and superparamagnetic iron oxide NPs (for a detailed review, see Fymat [7].
Gelatin nanoparticles for delivering multiple drugs to the brain
Gelatin is biocompatible, biodegradable, and generally recognized as safe by the U.S. Food and Drug Administration (FDA). Gelatin NPs are laced with the drug osteopontin and administered intra-nasally along the olfactory nerve cells – a noninvasive and direct route to the brain, to reduce inflammation and prevent brain cell death. This delivery pathway bypasses the BBB. It can be most effective in delivering drugs that cannot otherwise cross the barrier, and it can deliver therapeutic agents to specific regions of the brain. Once administered, the gelatin NPs target damaged brain tissue thanks to an abundance of gelatin-munching enzymes produced in injured regions. As far as is known, gelatin particles have not yet been used clinically to treat GBMs.
Lipid-based surface engineered PLGA nanoparticles:
Polylactic co-glycolic acid (PLGA)-based nanocarriers are one of the most promising drug delivery system for crossing the BBB. While they offer great promise, they nevertheless present several major challenges and intrinsic drawbacks, and require further engineering for clinical and research applications. These challenges include:
a) Synthetic hydrophobic surface,
b) Low transfection efficiency
c) Short circulation half-life and
d) Nonspecific tissue distribution.
To overcome these problems, numerous engineering strategies have been employed with lipid-based surface functionalization of PLGA NPs showing the following promising results:
i. Enhancement of target specificity of the carrier,
ii. Improvement of its physicochemical properties,
iii. NP-cell associations such as cellular membrane permeability,
iv. Immune responses and
v. Long in vivo circulation half-life [8].
These challenges can be classified in three major categories:
a. First generation NPs involving strategies to facilitate travel from the injection site;
b. Second generation NPs involving BBB pre-transcytosis to enhance passage across the brain endothelial cells; and
c. Third generation NPs to achieve targeting of the impaired system cells (post-transcytosis strategies). A fusion of all or some of these strategies may be required to engineer multi-functional PLGA NPs for treating brain cancers (and also neurological disorders) for which pharmaceutical treatments have been limited due to drug access to the central nervous system (CNS).
Nanodevices for Drug Delivery
Engineered nanoscale devices (ENDs) are minute devices with the potential to be engineered to efficiently and more safely deliver drug treatments directly to the location of diseased cells while helping avoid harm to healthy cells that fall victim to toxic drugs administered by conventional means. Because of their diverse capabilities, nanoscale devices can contain both targeting and therapeutic agents (in both single and multidrug approaches). They can deliver high drug levels in several situations, including anticancer drugs at the tumor site that can increase chemotherapeutic efficacy. They can also offer the opportunity to develop new approaches to therapy, including “smart” nanotherapeutics to “time” the release of any given drug or to deliver multiple drugs sequentially in a timed manner or at several locations in the body.
Miniaturized devices loaded with life-saving drugs may revolutionize chemotherapy, reducing the debilitating side effects of the therapy, making medications more effective, and all the while preserving the healthy living cells. The same systems could likewise be used for delivering clot-busting drugs to the brain. Several “nano-carriages” for drug delivery have been created but many challenges remain, chief among them being how not to let the medicine act before it gets to the right location in the brain. The carriers usually encapsulate drugs through long-range electrostatic interactions wherein the carrier attracts oppositely charged medicines. Other tools are available to trigger the release of drugs, for example, an external magnetic field, ultrasound waves, different pH values, etc. But, in each case, researchers face the problem of efficiency of the drug release. The following nanodevices (NDs) are available.
The Multi-Layered Shell:
It consists of (a) a core vesicle containing both hydrophilic and hydrophobic drugs; (b) a multilayered shell; and (c) an exterior layer. The purpose of the multilayered shell is to stabilize the NPs, prevent drug leakage, target the NPs to the slightly acidic environment of the tumor, and minimize the NPs’ interactions with non-cancerous cells. It can also be used to transport drugs that are not easily stored in the core such as highly charged nucleic acids [9,10].
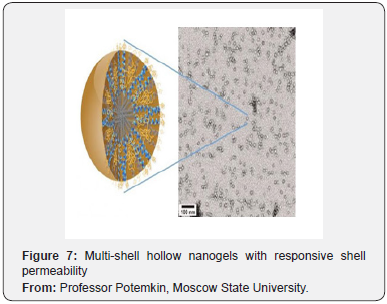
Multi-shell hollow nanogels with responsive shell permeability:
Departing from the multi-layered shell design, and irrespective of any electrostatic force (i.e., whether the medicines are either charged or neutral), gel nano-capsules are filled by the guest molecules, locking the molecules in the cavity and releasing them under temperature control. The carrier is surrounded by two “membranes” (or shells) of different chemical structures around a silica core which, at the end of the synthesis, will be chemically dissolved leaving only the “empty space” (cavity). The outer porous shell plays a protective (stabilizing) role and hinders aggregation of the nano-capsules, while the pores of the inner shell can open and close depending on the temperature. At the time of filling, the pores of both shells are open and the nanogel absorbs the drug molecules like a sponge. Then, the temperature changes and the pores of the inner shell close locking in the cavity and readying the drug for delivery. Subsequently, the pores will open again and the guest molecules will be released only in the places where the temperature allows (Figure 7).
Helix micelles for brain cancer therapy:
Nanobiotechnology (NBT) has a fundamental role to play in the treatment of GBMs. What is desperately needed is a means of effectively transporting therapeutic drugs through the BBB. Typically, GBM-therapeutics are ferried across the BBB in special liposomes (size ~ 110 nm). By contrast, an interesting device is the one presented by a new family of nanocarriers formed from the self-assembly of amphiphilic peptides and polymers, called “3HM” (for coiled-coil 3-helix micelles). At only 20 nm in size, and featuring a unique hierarchical structure, these new nanocarriers meet all the size and stability requirements for effectively delivering therapeutic drugs to GBM tumors. Their smaller size and unique hierarchical structure afford the 3HM nanocarriers much greater access to rat GBM tumors than larger-sized devices including the liposomes (of 110 nm in size). Copper-64 is used to label both 3HM and liposome nanocarriers for systematic PET and MRI studies to find out how a nanocarrier’s size might affect the pharmacokinetics and biodistribution in rats with GBM tumors. The results not only confirmed the effectiveness of 3HM as GBM delivery vessels, they also suggest that PET and MRI imaging of NP distribution and tumor kinetics can be used to improve the future design of NPs for GBM treatment.

Micelles are spherical aggregates of ampiphiles (chemical compounds that feature both hydrophilic and lipophilic properties). They can cross the BBB and accumulate inside GBM tumors at nearly double the concentration rate of current FDAapproved nanocarriers. (Figure 8). They have shown very good attributes for the treatment of brain cancers in terms of long circulation, deep tumor penetration and low accumulation in off-target organs such as the liver and spleen. There also is the possibility that they can be administered intravenously rather than invasively.
Future Prospects and Conclusions
NT will reduce the need for invasive surgery although some devices (implanted catheters and reservoirs) will likely still be needed. Nanomaterials improve the safety/efficacy of NDs. Nanoengineered probes can deliver drugs at the cellular level using nanofluidic channels. Microchips and biodegradable polymeric NP carriers may be more effective therapeutically for brain tumors. Next-generations include H3M micelles, nanoparticle nasal spray, and “sticky” NPs. However, while great advances have been made in developing effective drugs for other types of cancer, we still do not fully understand the deep biology of glioblastomas and have not come up with the corresponding drugs. In the meantime, GBMs continue to progress rapidly in their ineluctable death path. We should dedicate considerably more time on understanding that deep biology!
References
- Fymat AL (2017a) Surgical and non-surgical management and treatment of glioblastoma: I. Primary tumors. Open Access J of Surgery 7(2): 1-8.
- Fymat AL (2017b) Surgical and non-surgical management and treatment of glioblastoma: II. Recurring tumors. Open Access J of Surgery 7(1): 1-7.
- Fymat AL (2017c) Glioblastoma treatments: Where do we stand? Med Plus J of Cancer & Oncology Research 1(1): 1-12.
- Stolp HB, Liddelow SA, Sá-Pereira I, Dziegielewska KM, Saunders NR (2013) Immune Responses at Brain Barriers and Implications for Brain Development and Neurological Function in Later Life. Front Integr Neurosci 7: 61.
- Scherrmann JM (2002) Drug delivery to brain via the blood-brain barrier. Vascul Pharmacol 38(6): 349-354.
- Temsamani J, Rousselle C, Reese AR, Scherrmann JM (2001) Vectormediated drug delivery to the brain. Expert Opin Biol Ther 1(5): 773- 782.
- Fymat AL (2018) Nanobiotechnology Advances in Oncology. Advances in Bioengineering & Biomedical Science Research 1(1): 1-6.
- Bose RJ, Lee SH, Park H (2016) Lipid-Based Surface Engineering of PLGA Nanoparticles for Drug and Gene Delivery Applications. Biomater Res 20: 34.
- Ashton (2016) Accurin Nanoparticles Dutifully Deliver Drugs. Science Translational Medicine 8(325).
- Fymat AL (2017d) Nanomedicine as a precursor to precision medicine for glioblastoma treatment. J of Current Opinions on Neurological Science 1(4): 200-206.






























