Cancer Nanotechnology: The Recent Developments in the Cancer Therapy
Gandhali A Deshpande*
SNDT University, India
Submission: April 28, 2016; Published: May 19, 2016
*Corresponding author: Gandhali A Deshpande, SNDT University, Building no 14/Flat no 24, Vijay Vilas near Vijay garden Ghodbundar Road, Thanewest, Mumbai 400607, India, Tel:9673296800; Email: gandhalideshpande1@gmail.com
How to cite this article: Gandhali AD. Cancer Nanotechnology: The Recent Developments in the Cancer Therapy. Glob J Nano. 2016; 1(1): 555551. DOI: 10.19080/GJN.2016.01.555551
Abstract
The nanotechnology is prominent in medicine for various applications. Nanotechnology is a multidisciplinary field, which covers diverse arrays of devices made using principles of engineering, biology, physics, and chemistry. The increasing number of nanomedicines is approved clinically, used safely, which highlights the important role of nanotechnology in the field of cancer research. The successful application of nanotechnology to the targeted smart drug therapy ISA rapidly growing component of armamentarium against cancer. Nanotechnology is being applied to cancer in two broad areas: the development of nanovectors, such as nanoparticles, which can be loaded with drugs or imaging agents and then targeted to tumors, and high throughput nanosensor devices for detecting the biological signatures of cancer. This advanced technology provides a unique approach and comprehensive technology against cancer through early diagnosis, prediction, prevention, personalized therapy, and medicine. This review focuses on nanotechnology-based clinical approaches in the advancements of cancer therapy.
Keywords: Nanotechnology; Nanosystems; Targeted therapy; Cancer therapy; Nano-oncology
Abbreviations: NPs: Nanoparticles; NM: Nanomedicine; FDA: Food and Drug Administration; HER2: Human Epidermal Growth Factor Receptor 2; ADC: Antibodies-Drug Conjugates; RNAi: RNA Interference; PSMA: Prostate-Specific Membrane Antigen; EPR: Enhanced Permeability and Retention; mAbs: Monoclonal Antibodies
Introduction
Cancer is a leading cause of death worldwide. American cancer society estimates that in 2016, there will be an estimated 1,685,210 new cancer cases diagnosed and 595,690 cancer deaths in the US [1]. About 595,690 Americans are expected to die of cancer in 2016, which translates to about 1,630 people per day. Cancer is the second most common cause of death in the US, exceeded only by heart disease, and accounts for nearly 1 of every 4 deaths. Cancer is a complex disease to understand because it comprises six biological capabilities acquired during the multistep development of human tumors [2,3] they include sustaining proliferative signaling, evading growth suppressors, resisting cell death, enabling replicative immortality, inducing angiogenesis, and activating invasion and metastasis. Underlying these hallmarks is genome instability, which generates the genetic diversity that expedites their acquisition, and inflammation, which fosters multiple hallmark functions. However in the last decade, two emerging hallmarks are reprogramming of energy metabolism and evading immune destruction (Figure 1).

Most human cancers are treated by surgical resection, chemotherapy, and/or radiation. Surgery cures ∼45% of all patients with cancer, whereas chemotherapy and radiation therapy together cure only 5%, and the remainder succumbs to their diseases [4]. Limitations in cancer treatment is a result of current challenges seen in cancer therapies, including lack of early disease detection, nonspecific systemic distribution, inadequate drug concentrations reaching the tumor, and inability to monitor therapeutic responses. Poor drug delivery and residence at the target site leads to significant complications, such as multi-drug resistance [5].
Several challenges noticed and well reported by current cancer therapies include nonspecific systemic distribution of antitumor agents, inadequate drug concentrations reaching the tumor site, intolerable cytotoxicity, limited ability to monitor therapeutic responses and development of multiple drug resistance. The most troublesome issues of current cancer treatment are drug toxicity and tumor resistance. Toxicity can cause major complications, such as low white-blood-cell counts or heart failure that necessitates cessation of treatment [6-8]. Evolution of drug resistance by tumors accounts for the vast majority of cases in which treatment fails. At present, the early recognition and treatment of cancer remain a technological bottleneck. Despite many advances in conventional treatment options such as chemotherapy and radiation, cancer therapy is still far from optimal because it is plagued by some drawbacks. Thus, there is an urgent need and major opportunities to develop new and innovative technologies [9]. To overcome the above challenges associated with cancer treatment safety and efficacy, many scientists have taken tremendous efforts to introduce smart targeted therapy, using advanced nanotechnology to fight against the lethal killer cancer. Nanotechnology-based therapeutics have exhibited clear benefits when compared with unmodified drugs, including improved half-lives, retention, targeting efficiency, and fewer patient side effects (Figure 2 & Table 1).
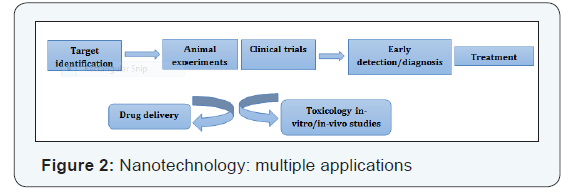
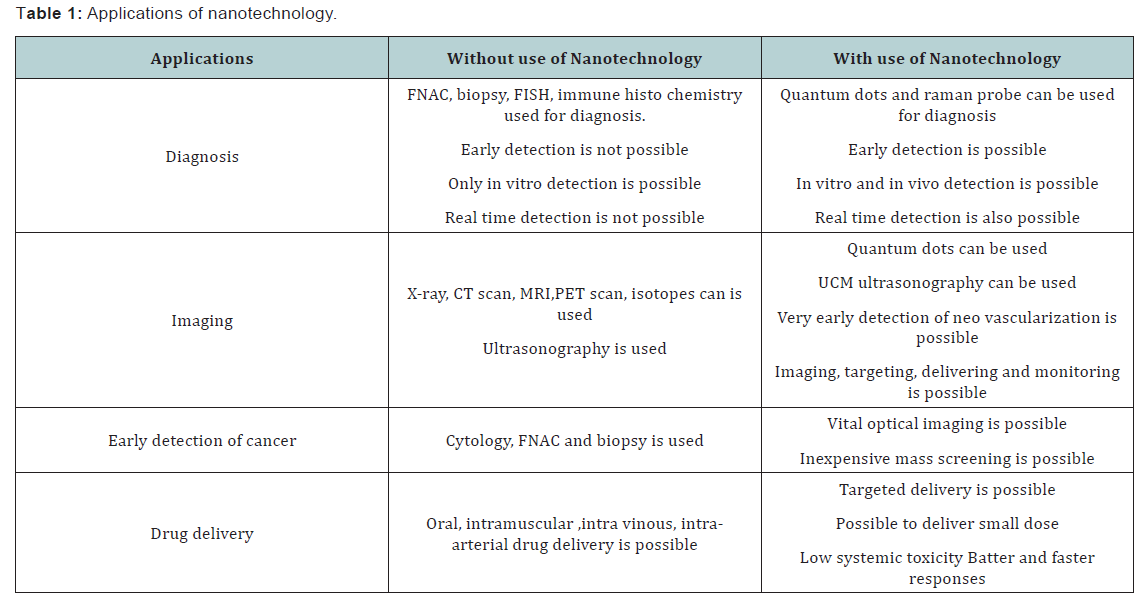
Early detection, imaging, diagnosis and drug delivery with and without use of nanotechnology
Nanomedicines platform, drug targeting strategies
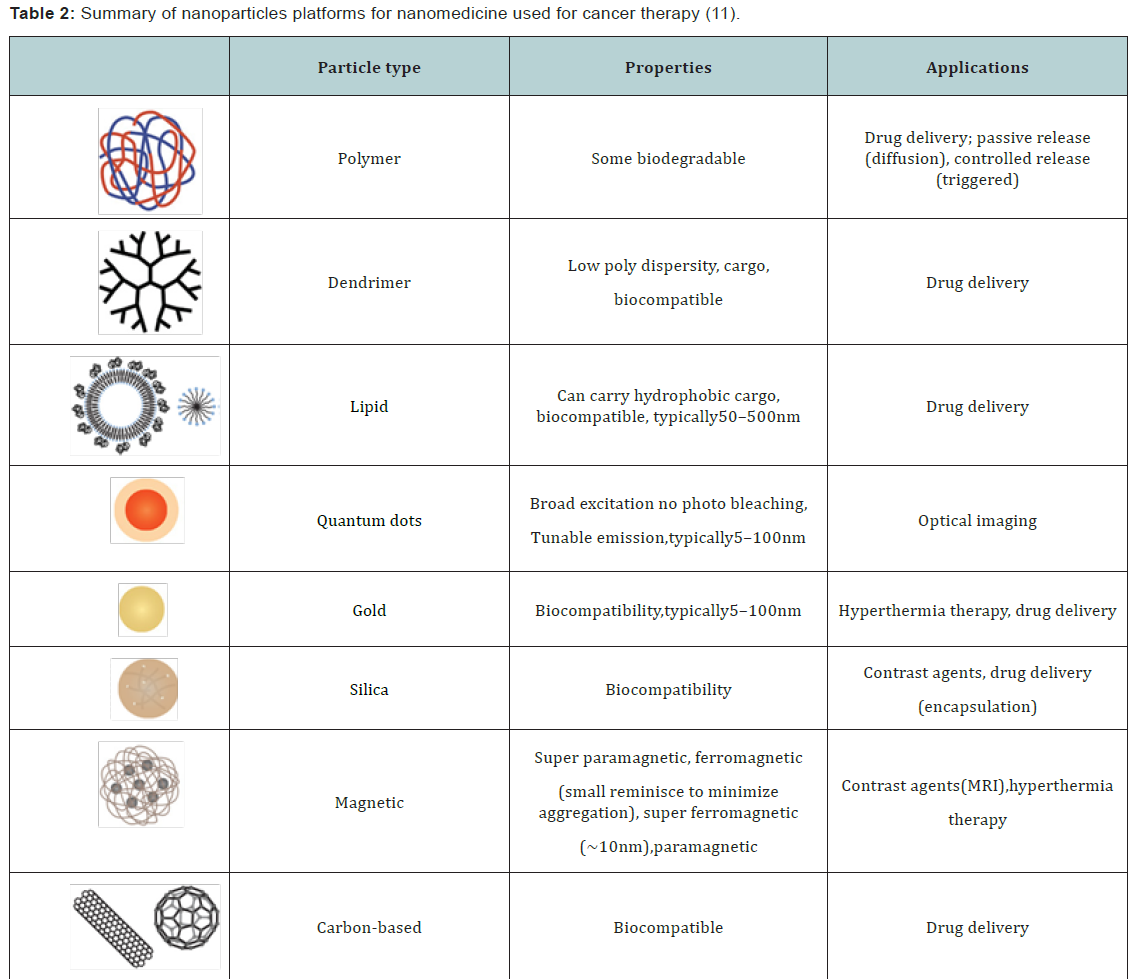
Nanomedicine is a rapidly growing area of medical research that is focused on developing nanoparticles (NPs) for prophylactic, diagnostic, and therapeutic applications. Recently nanocarriers (particularly in the size range of 10 nm to 100 nm) have been an emerging class of therapeutics for cancer treatment. In addition liposomal doxorubicin, albumin-bound and paclitaxel (Abraxane1) is FDA approved Nanosystems used in clinical practice [10]. Nanoparticles platforms can be broadly categorized as organic, inorganic and hybrid (Table 2). Organic nanoparticles have been widely explored for decades, yielding a large variety of materials, formulations, imaging modalities, and targets for cancer therapy [11]. Nanotechnology is gaining importance in biology due to its small size and targeted effects. Nano-devices are 100-10, 000 times smaller than the human cell. Due to the smaller size and larger surface area relative to their volume, Nanosystems can easily interact with the surface of the cell and inside of the cell. By interacting with cellular proteins Nanosystems can have the potential to detect tumor microenvironment and deliver drug treatment [12]. Nanosystems have four unique properties that distinguish them from other cancer therapeutics: (i) the Nanosystems can themselves have therapeutic or diagnostic properties and can be designed to carry a large therapeutic ‘payload’; (ii) Nanosystems can be attached to multi valet targeting legends, which yield high affinity and specificity for target cells; (iii) Nanosystems can be made to accommodate multiple drug molecules that simultaneously enable combinatorial cancer therapy and (iv) Nanosystems can bypass traditional drug resistance mechanisms. By using both passive and active targeting strategies, the nanocarriers can achieve increased intracellular concentration of drugs in cancer cells while minimizing toxicity in normal cells, simultaneously enhancing anti cancer effects and reducing systemic toxicity.
Drug targeting strategies
To achieve the effective drug delivery two basic fundamental principles should be involved first the drug should able to reach the desired tumor site after administration with the minimalloss of their volume and activity in blood circulation. Secondly, drugs should only kill tumor cells without harmful effects to healthy tissue. These requirements may be enabled using two strategies: passive and active targeting of drugs [12] (Figure 3).

Passive targeting systems include liposomes, micelles, gold particles, iron oxide nanoparticles, silica nanoparticles, quantum dots, and hybrid nanoparticles. Passive targeting takes advantage of the unique path physiological characteristics of tumor vessels, enabling nondrug to accumulate in tumor tissues. Typically, tumor vessels are highly disorganized and dilated with a high number of pores, resulting in enlarged gap junctions between endothelial cells and compromised lymphatic drainage. The ‘leaky’ vascularization, which refers to the EPR effect, allows migration of macromolecules up to 400 nm in diameter into the surrounding tumor region. Drug carriers are expected to stay in the blood for a long time, accumulate in pathological sites with affected and leaky vasculature (tumors, inflammations, and infarcted areas) via the enhanced permeability and retention (EPR) effect, and facilitate targeted delivery of specific legendmodified drugs and drug carriers into poorly accessible areas [13]. Although passive targeting approaches form the basis of clinical therapy, they suffer from several limitations like some tumors do not follow EPR phenomena and some tumors are highly unorganized tumor mass. One way to overcome the limitations of passive targeting is to attach high-affinity legends such as aptamers, antibodies, small molecule, or peptides that will only bind to specific cellular receptors. Active targeting is an alternative strategy to overcome the limitations of the passive method. Active method conjugates with target ligand or an antibody to nanoparticles. This ligand-target approach is expected to deliver drug efficiently at the active site [14].
Choice of target receptor
A design of targeted nanoparticles crucially depends on, selection of appropriate receptors or antigens locally expressed on cancer cells. Importantly ideal receptors or antigens are those which are uniquely expressed only on cancer cells and not on normal cells. In addition, these targets should also have a high density on the surface of the target tumor cells. Targeted Nanoconjugate should internalize after binding with ligands to avoid simple diffusion of the drug near the cell surface. However, this drug transport mechanism outside the cell may disperse or redistribute the drug to the surrounding normal tissues, instead of cancer cell mass. Internalization of target receptor viareceptor-mediated endocytosis triggers the drug release pattern specifically into cancer cells [14].
Choice of targeting ligand
Selective and successful drug transport mechanisms to the cancerous tissue are the emerging challenges. The choice of ligands has a great role in triggering receptor-mediated endocytosis. Ligands targeting cell-surface receptors can be natural materials like foliate and growth factors, which have the advantages of lower molecular weight and lower immunogenicity than antibodies. Recent advances in molecular biology and genetic engineering allow modified antibodies to be used as targeting moieties in an active-targeting approach. MAbs or antibody fragments (such as antigen-binding fragments or single-chain variable fragments) are the most frequently used ligands for targeted therapies. Compared with whole mAbs, the use of antibody fragments as a targeting moiety can reduce immunogenicity and improve the pharmacokinetic profiles of nanoparticles. For example, liposome’s coupled with mAb fragments instead of whole antibodies showed decreased clearance rates and increased circulation half-lives, allowing the liposome’s sufficient time to be distributed and bind to the targeted cells. This strategy improved the therapeutic efficacy of immune liposomal DOX targeted against CD19 on human B lymphoma cells in animal models [15, 16].
Nanotechnology-based advanced technologies
In the treatment of cancer, targeted therapy is desirable due to specificity to cancer cells and normal cells are not affected. Nanotechnology-based approaches are very recent and noninvasive strategies. Bioengineered nanoparticles or nanosystems are new armamentaria in cancer therapy which includes photo dynamic therapy (PDT), radiotherapy and radiofrequency therapy, and theranostics (Figure 4).

FDA approved Nanomedicines: An update in cancer therapy
Researchers have already made progress with chemotherapeutic Nanomedicine in the clinic. Nanomedicines have demonstrated the potential for increasing bio availability,enhancing drug solubility, active targeting, and high drug loading [17].
Several compounds that are in various stages of trials or already approved by the US Food and Drug Administration (FDA). For example, Calando Pharmaceuticals has demonstrated the first evidence of nanoparticles-delivered clinical RNA interference (RNAi). BIND Biosciences has shown that nanoparticles combining a chemotherapeutic drug with prostate-specific membrane antigen (PSMA) can reduce lung and tensile lesions with greater efficacy compared with the drug alone, and at substantially lower doses. Furthermore, Celgene’s Abraxane. Doxil, daunoxome, Oncaspar, Depocyt and those in Nano-platforms that have reached an advanced stage of clinical development utilizing liposome’s, lipid nanoparticles, lipoplexes, albumin nanospheres, thermo sensitive devices,micelles and gold nanoparticles [18]. An albumin-functionalized paclitaxel formulation was initially approved by the FDA for sale as a breast cancer therapy, but also recently received approval for the treatment of lung and pancreatic cancers [9]. (Table 3) Additionally, the FDA approved antibodies-drug conjugates (ADC) such as brentuximab, abvedotin, trastuzumab emtansine and Marqibo, Brentuximab, abvedotin and Trastuzumab, emtansine conceptually one of the simplest Nanomedicine conjugated to a targeting molecule. Brentuximab targets the proteinCD30, a glycosylated phosphoprotein expressed by Bcells, including Bcell lymphomas, some leukemia’s, and melanoma cancer stem cells. Trastuzumab targets the human epidermal growth factor receptor 2 (HER2) over expressed in HER2 positive breast cancer. However, there remain many challenges in exploiting advances in nanotechnology and bioengineering to develop systems that will have a significant impact on patient survival rates.

Discussion and Conclusion
Nanomedicine refers to the application of nanotechnology in medicine, and endeavors to diagnose, treat, and/or monitor disease on a nanoscale. Cancer nanotechnology is a quickly evolving field of inter disciplinary research that involves the biomedical application of nanoparticles, which are nanoscale devices that are able to overcome biological barriers, specifically, recognize a single type of cancer cell, and accumulate preferentially in tumors. Medical applications with nanoparticles are growing, as they have the potential to offer novel methods of noninvasive cancer detection, diagnosis, and treatment. Tumor targeting ligands, such as antibodies, peptides, or small molecules, can be attached to nanoparticles for targeting of tumor antigens and vasculatures with high affinity and specificity. In addition, diagnostic agents (i.e. optical, radiolabels, or magnetic) and chemotherapeutic drugs can be integrated into their design for more efficient imaging and treatment of the tumor with fewer side effects. Recent advances in nanomedicine raise exciting possibilities for future nanoparticles applications in personalized cancer therapy. nanotechnology has become an enabling technology for personalized oncology, in which cancer detection, diagnosis, and therapy are tailored to each individual’s tumor molecular profile, and for predictive oncology, in which genetic and/or molecular markers are used to predict disease development, progression and clinical outcomes.
Nanomedicine holds much promise but there are still major areas of both basic and applied research in the area nanotechnology that needs to be explored to solve some of the existing problems. Nanooncology is the promising technology of the new ear but significant obstacles have restricted the clinical translation of the cancer therapy. One of the main reasons for the restricted use of Nanomedicine is the matter of multifunctionality, multidrug resistance, and variable responses to standard therapy, complexities associated with heterogeneous mass of tumor cells, cancer stem cells, complicated therapy, and cross talk between several cellular pathways.
Overall considering potential applications of the Nanomedicine in the health sector, especially with regards to cancer therapy, there is an emerging need for the development of safety guidelines by the government. The emergence of Nanotechnology Research Centers, established in recent years (some of which are funded by the National Institutes of Health and the National Science Foundation), demonstrate the enthusiasm of investigators and granting agencies for the technology. In the next few years, many applications of nanotechnology will become commonplace within medical practice.
References
- Cancer Facts & Figures (2016).
- Douglas Hanahan , Robert A Weinberg (2011) RA Hallmarks of Cancer The Next Generation Cell 144 (4): 646-674.
- Kakde D, Jain D, Shrivastava V, AT Patil (2011) Cancer Therapeutics- Opportunities Challenges and Advances in Drug Delivery Journal of Applied Pharmaceutical Science 1(9): 01-10.
- Singhal S, Nie S, Wanq MD (2010) Nanotechnology Applications in Surgical Oncology Annual Review Medicine 61: 359373
- Ehdaie B (2007) Application of Nanotechnology in Cancer Research: Review of Progress in the National Cancer Institute’s Alliance for Nanotechnology Int J Biol Sci 3(2): 108-110.
- Das M, Mohanty C, Sahoo SK (2009) Ligand-based targeted therapy for cancer tissue Expert Opinion Drug Delivery 6(3): 285-304.
- Parveen D S, Sahoo SK (2006) Nanomedicine clinical applications of polyethylene glycol conjugated proteins and drugs Clinical Pharmacokinetic 45(10): 965-88.
- Parveen D S, Sahoo SK (2008) Polymeric nanoparticles for cancer therapy J Drug Target 16(2 ): 108-123.
- Misro R, Sarbari Acharya, Sanjeeb k (2010) Cancer nanotechnology: application of nanotechnology in cancer therapy Drug Discovery Today 15(19-20): 842-851.
- Bharali DJ, Khalil M, Gurbuz M, Simone TM, Mousa et al. (2009) SA Nanoparticles and cancer therapy: a concise review with emphasis on dendrimers International Journal Nanomedicine 4: 1-7.
- Bamrungsap, Zilong Zhao, Tao Chen, Lin Wang, Chunmei Li, et al. (2016) Therapeutics 7(8): 1253-1271.
- Rajshri M Navalakhe & Tarala D Nandedkar (2007) Application of nanotechnology in biomedicine. Indian journal of experimental biology 45: 160-165.
- Trochilin VP (2010) Passive and active drug targeting: drug delivery to tumors as an example Handbook Experimental Pharmacology (197): 3-53.
- Wang X, Yang L, Chen ZG, Shin DM (2008) Application of Nanotechnology in Cancer Therapy and Imaging. CA Cancer J Clin l58: 97-110.
- Sapra P, Tyagi P, Allen TM (2005) Ligand-targeted liposomes for cancer treatment. Current Drug Delivery 2(4): 369-381.
- Sapra P, Moase EH, Ma J, Allen TM (2004) Improved therapeutic responses in a xenograft model of human B lymphoma (Namalwa) for liposomal vincristine versus liposomal doxorubicin targeted via anti- CD19 IgG2a or Fab fragments. Clin Cancer Res 10(3): 1100-1111.
- Dawidczyk CM, Kim C, Park JH, Russell LM, Lee KH, et al. (187) Stateof- the-art in design rules for drug delivery platforms: lessons learned from FDA-approved nanomedicines. J Controlled Release 187: 133-144.
- Pillai G (2014) Nanomedicines for Cancer Therapy: An Update of FDA Approved and Those under Various Stages of Development. SOJ Pharmacy Pharmaceutical Sciences 1(2): 1-13.






























