Immunotherapy’s Potential Over Whole Brain Radiotherapy in Primary Isolated CNS Marginal Zone Lymphoma: A Case Report and Literature Review
Hadia Fatima*, Abdulrahman Al Hadab and Salem Mohammad Al Shehri
Radiation Oncology Department, King Abdulaziz Medical City, Ministry of National Guards Health Affairs, Riyadh, Saudi Arabia
Submission: December 12, 2023; Published: January 03, 2024
*Corresponding Address: Hadia Fatima, Radiation Oncology Department, Email: hadia.fatima@hotmail.com
How to cite this article: Hadia Fatima*, Abdulrahman Al Hadab and Salem Mohammad Al Shehri. Immunotherapy’s Potential Over Whole Brain Radiotherapy in Primary Isolated CNS Marginal Zone Lymphoma: A Case Report and Literature Review. Canc Therapy & Oncol Int J. 2024; 25(5): 556173. DOI:10.19080/CTOIJ.2024.25.556173
Abstract
Primary CNS Marginal Zone Lymphoma is a rare, slow-growing type of B-cell lymphoma. Due to its diverse nature, it can be challenging to detect and treat. Diagnosis requires a biopsy and thorough immunohistochemical analysis. Treatment typically involves a multimodality approach, which includes maximal safe reaction, low-dose whole-brain irradiation, and a combination of systemic therapies such as chemoimmunotherapy and targeted therapy. We would like to share a case report of a 51-year-old female who was diagnosed with Primary CNS Marginal Zone Lymphoma. She was successfully treated without whole-brain radiotherapy in the adjuvant setting. The patient received a combination of Rituximab and Bendamustine, which provided remarkable local and systemic control.
Keywords: Cancer Immunotherapy; Adjuvant Radiation Therapy; Primary Dural Lymphoma; Marginal Zone Lymphoma mzl; CNS Lymphoma
Introduction
Primary CNS Marginal Zone Lymphoma is a rare but slow-growing condition [1]. Female patients with this tumor usually have a good prognosis, with a survival rate of over 86% within 5 years [1]. Cancer typically starts in the dura but may also impact the epidural and subdural areas [2]. It is difficult to identify and is often misdiagnosed as meningioma or subdural hematoma on diagnostic imaging [3,4]. Patients display a range of neurological symptoms, including chronic headaches, seizures, poor coordination, and stroke [3]. A definitive diagnosis can only be made through histology and immunohistochemical staining [5]. There is no established treatment for this cancer, and various approaches, such as chemotherapy, targeted therapy, low dose whole brain radiotherapy, and excision, are still being studied. Our patient is a 51-year-old woman who initially presented with symptoms of raised intracranial pressure and seizures. She was initially misdiagnosed on imaging as having a parafalcine meningioma, but a definitive diagnosis of Isolated Primary CNS Marginal Zone Lymphoma was made on histopathological review. This type of cancer is extremely rare, and there are only a few reported cases in the literature. Moreover, no cases have been previously reported from the Middle East Asia region.
Case Report
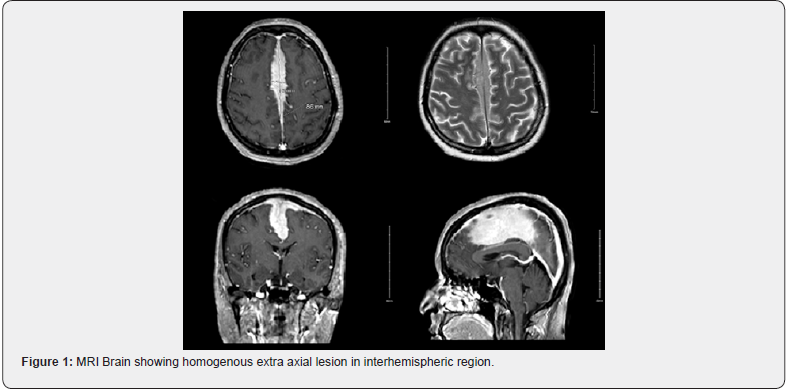
A 51-year-old woman with no history of malignancy or comorbidities presented with sudden-onset headaches that progressed to persistent morning headaches and vomiting over a week. Initial CT scans indicated a possible giant parafalcine meningioma, leading to the initiation of steroid and antiepileptic treatment. She later had two episodes of generalized tonic-clonic seizures, prompting a referral for neurosurgical evaluation. On examination, the patient had a good performance status (ECOG 1), stable vital signs, and no apparent neurological deficits. Systemic examination revealed no abnormalities. A brain MRI revealed a sizable, intensely enhancing extra-axial lesion in the interhemispheric region that measured approximately 8.6 x 1.5 cm. The tumor invaded and caused stenosis of the superior dural sagittal venous sinus, indicative of meningioma. Notably, no signs of acute territorial infarction, intracranial hemorrhage, midline shift, mass effect, or hydrocephalus and posterior fossa structures appeared unremarkable.
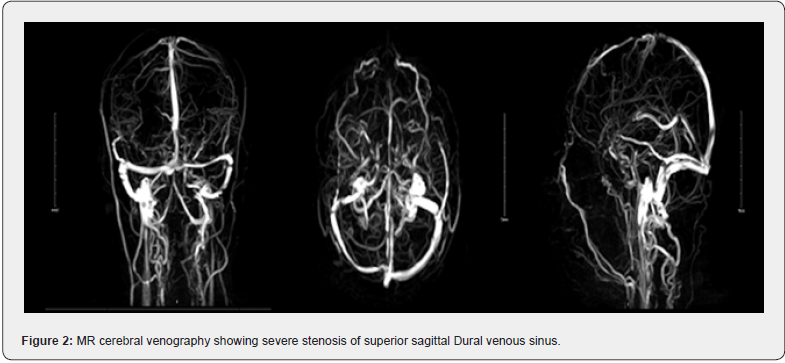
The clinical and imaging findings supported the decision to seek neurosurgical input. The use of steroids and antiepileptics addressed symptom control, and ongoing monitoring was done given the absence of neurological deficits. The superior dural sagittal venous sinus involvement influenced surgical consideration and necessitated a comprehensive discussion of risks and benefits with the patient (Figure 1). An MR cerebral venography exam revealed severe stenosis and interruption in the superior sagittal dural venous sinus. This was mainly due to an invasion and mass effect of an extra-axial enhancing lesion, located in the anterior part. However, the other dural venous sinuses and major cerebral vessels were patent (Figure 2). The patient underwent a bur hole craniotomy to remove the left frontal extra-axial parasagittal dural-based lesion. The histopathology report revealed that the lesion was an Extranodal Marginal Zone Lymphoma with prominent plasma cell differentiation. The microscopic examination of the dural tissue showed heavy lymphoplasmacytic cell infiltration, predominantly small, and admixed with a large number of mature-appearing plasma cells. Perivascular cuffing was evident, and scattered large lymphoid cells with immunoblastic features were seen within the infiltrate.
On immunostaining, the lymphoid cells were positive for CD20 and negative for CD3, CD5, CD10, BCL6, BCL2, MUM1, CD43, and CD23. The plasma cells were positive for CD38, CD138, MUM1, IgG, IgG4 (IgG4: IgG ratio 80%), and kappa, and negative for IgD and lambda (kappa light chain restricted). Ki-67 was expressed by around 10% of cells, and CD21 highlighted a few distorted follicles in the background. The immunoblasts were CD30 positive. The patient’s recovery after surgery was uneventful. Metastatic workup included a PET scan that showed a small residual active lesion in the interhemispheric extra-axial dural-based region. In addition, a 0.7 cm focal hypermetabolic nodule was found in the right lower lobe of the lungs, the nature of which was unknown (Figure 3). The bone marrow biopsy showed reactive bone marrow. The pretreatment evaluation revealed acute deep vein thrombosis (DVT) in the left lower limb and a non-obstructing pulmonary embolism in the left lower lobe with sub-segmental extension (May-Thurner syndrome). Despite this, the ejection fraction was normal (55% on echocardiogram), and viral serology was negative.


The case was discussed in the tumor board with CNS lymphoma experts to determine the best course of action for this rare case, including the controversial role of low-dose radiotherapy versus systemic therapy. Given the lack of clear guidelines for the management of rare primary CNS MZL and the presence of a questionable right lower lobe lung nodule, the patient was planned for systemic combination therapy with Rituximab and Bendamustine due to its low toxicity profile, good CNS penetration, and systemic coverage. Radiation therapy was reserved for salvage. After receiving 3 cycles of interim evaluation with MRI, good radiological response was observed, with postsurgical changes, a reduction in the size of the primary lesion, and regression of vasogenic edema. There was residual enhancement along the falx and nodular enhancement along the surgical tract, with a maximal thickness of 0.57 cm compared to 1.5 cm previously (Figure 4). The patient has completed 6 cycles of combination immunotherapy and is now on maintenance therapy with Rituximab.
Discussion
Marginal zone lymphoma (MZL) is the second most common type of non-Hodgkin’s lymphoma after follicular lymphoma. It is generally slow-growing and indolent [1]. The WHO recognizes three subtypes of MZL: extranodal MZL (EMZL) of mucosaassociated lymphoid tissue (MALT lymphoma), splenic MZL, and nodal MZL [6]. MZL accounts for 6% of all B-cell lymphomas, with the stomach, ocular/adnexal, lung, skin, and salivary glands being the most common extranodal locations [7]. Primary CNS MZBL is a rare subtype that affects middle-aged women with a median age of 59.5 years and has not been conclusively linked to any infectious or autoimmune cause [8]. The exact pathophysiology of primary CNS MZBL is not well understood due to the absence of mucosa-associated lymphoid tissue in the CNS. However, certain chronic viral and autoimmune diseases have been linked to MZBL development in non-CNS extranodal tissue sites [8]. Our patient, a 51-year-old woman with no history of infection or autoimmune disorder, was diagnosed with May-Thurner syndrome after surgery. CNS lymphomas have never been associated with this vascular disease.
Patients with primary CNS MZBL typically present with focal neurological symptoms, such as headaches, seizures, and/ or sensory loss, as well as limb weakness and/or alterations in vision [9]. Most patients have good performance status (ECOG 1) at diagnosis. The median number of days with symptoms before a CNS diagnosis is 105 (interquartile range: 30-420) for the primary disease and 60 (interquartile range: 0-930) for the secondary condition [9]. Our patient’s history of headaches and sudden onset of seizures was brief, and there were no obvious neurological symptoms. Primary CNS MZBL usually develops in the dura mater, epidural, and subdural spaces, sparing the brain parenchyma [4]. Meningioma is the most common primary CNS dural-associated lesion, accounting for 30% of all intracranial malignancies [4,5]. It exhibits essentially comparable clinical and MRI features to dural-based MZBL. On T1-weighted MRI scans, these tumors look isointense and may show enhanced extra-axial lesions and the dural tail sign [4]. T2-weighted imaging can distinguish between dural-based MZBL and vasogenic edema, which is typically seen in tissue next to the tumor. Diffusion-weighted MRI can also be used to diagnose MZBL since it exhibits restricted diffusion due to the high cellularity of these tumors compared to other cancers. Other potential differentials include subdural hematomas, sarcoidosis, aspergillosis, metastases, schwannomas, and chloromas. In our patient, the tumor originated from the left frontal cerebral convexity, causing stenosis of the superior dural sagittal venous sinus. There was no involvement of brain parenchyma, vasogenic edema, or intracranial hemorrhage [4,5]. MZBL tumors are composed of B cells with CD20 positivity and clonal IgH and IgK rearrangements, but not B cells that express CD5, CD10, CD43, Bcl-6, or cyclin D1. CNS MZBL is generally latent and has low proliferation indexes [1,6]. Our patient’s tumor showed heavy infiltration of lymphoid cells admixed with mature appearing plasma cells.
The immunohistochemical profile of the tumor showed positivity for CD20 in lymphoid cells and CD38, CD 138, MUM1, and IgG4: IgG ratio of 80%. The immunoblasts were positive for CD30 with a low proliferation index of 10%. The attending physician decides how to proceed with treatment. Surgery and radiotherapy are the two most used treatment modalities for primary diseases (27%). Patients with parenchymal disease undergo craniotomy and biopsy, while those with duralrestricted disease receive total resection [10]. Rituximab and methotrexate are used to treat patients with primary illness who have previously undergone systemic therapy, either alone or in combination with cyclophosphamide, doxorubicin, vincristine, and prednisolone [RCHOP]. Other treatment options include bendamustine, rituximab, intrathecal cyclophosphamide, and methotrexate. Patients with primary disease are typically not treated with radiation or chemotherapy alone [10,11]. For patients with well-defined CNS-MZL lesions, the International Lymphoma Radiation Oncology Group recommends a radiation therapy dose of 30-36 Gy. However, lower doses ranging from 20- 25.2 Gy have also been used in various case reports [12]. Since no clear guidelines are available on the management of these tumors, our CNS lymphoma experts decided to treat the patient with a combination of bendamustine with rituximab [13]. This was done to avoid neurotoxicity and provide systemic coverage. However, many patients in the literature are treated with low doses of whole-brain radiotherapy. In our case, radiotherapy was not opted for and was reserved for salvage. A study has validated the generally good prognosis of primary CNS MZL in patients with MZL and highlighted a poor prognosis of subsequent CNS relapse [14].
Conclusion and Recommendation
Diagnosing and treating Primary Isolated CNS Marginal Zone Lymphoma can be a challenging task, as it is a rare form of cancer. Radiologists and oncopathologists need to report cases carefully, considering any unusual disease presentations, to determine the best treatment plan through a multidisciplinary approach. Medical boards should consider such cases. For determining the curative radiation dosage and systemic therapy options for Marginal Zone Lymphomas of the CNS, an in-depth systematic review is essential.
Conflict of Interest
The authors declare no conflict of interest.
Declarations
No funding was received in the preparation of this manuscript.
References
- Lopetegui‐Lia N, Delasos L, Asad SD, Manish K, Jonathan SH (2020) Primary central nervous system marginal zone B‐cell lymphoma arising from the dural meninges: A case report and review of literature. Clinical Case Reports 8(3): 491-497.
- Ariani R, Ballas L (2021) Primary CNS Extranodal Marginal Zone B-Cell Lymphoma: A Case Series of 2 Patients Treated with External Beam Radiation Therapy. Case Reports in Oncology 14(2): 725-732.
- Pavlou G, Pal D, Bucur S, Van Hille PT (2006) Intracranial non-Hodgkin’s MALT lymphoma mimicking a large convexity meningioma. Acta Neurochir 148(7): 791-793.
- Smith AB, Horkanyne-Szakaly I, Schroeder JW, Elisabeth JR (2014) From the radiologic pathology archives: mass lesions of the dura: beyond meningioma-radiologic-pathologic correlation. Radiographics 34(2): 295-312.
- Elster AD, Challa VR, Gilbert TH, Richardson DN, Contento JC (1989) Meningiomas: MR and histopathologic features. Radiology 170(3): 857-862.
- Swerdlow SH, Campo E, Pileri SA, Nancy LH, Harald S, et al. (2016) The 2016 revision of the World Health Organization classification of lymphoid neoplasms. Blood 127(20): 2375-2390.
- Tu PH, Giannini C, Judkins AR, Jason MC, Richard B, et al. (2005) Clinicopathologic and genetic profile of intracranial marginal zone lymphoma: a primary low-grade CNS lymphoma that mimics meningioma. J Clin Oncol 23(24): 5718-5727.
- Cerhan JR, Habermann TM (2021) Epidemiology of marginal zone lymphoma. Ann Lymphoma 21037: 20-28.
- Bustoros M, Liechty B, Zagzag D, Cynthia L, Timothy S, et al. (2018) A rare case of composite dural extranodal marginal zone lymphoma and chronic lymphocytic leukemia/small lymphocytic lymphoma. Front Neurol 9: 267.
- Sunderland AJ, Steiner RE, Al Zahrani M, Chelsea CP, Bouthaina SD, et al. (2020) An international multicenter retrospective analysis of patients with extranodal marginal zone lymphoma and histologically confirmed central nervous system and dural involvement. Cancer Med 9(2): 663-670.
- Karschnia P, Batchelor TT, Jordan JT, Brian S, Sebastian SW, et al. (2020) Primary dural lymphomas: clinical presentation, management, and outcome. Cancer 126(12): 2811-2820.
- Sato GE, Ikeda I, Sakoda M, Kiyotomo M, Takahiko U, et al. (2020) Whole brain radiation therapy for primary central nervous system marginal zone lymphoma: a case report. Int Cancer Conf J 1: 31-34.
- Tsutsumi Y, Ito S, Nagai J, Takahiro T, Takanori T (2021) Patients with marginal zone dural lymphoma successfully treated with rituximab and bendamustine: A report of two cases. Mol Clin Oncol 15(4): 1-3.
- Chihara D, Fowler NH, Oki Y, Michille AF, Loretta JN, et al. (2018) Impact of histologic subtypes and treatment modality among patients with primary central nervous system lymphoma: a SEER database analysis. Oncotarget 9(48): 28897.






























