Evaluation of Dysphagia with Laryngo- Pharyngeal Sparing Intensity Modulated Radiotherapy in Head and Neck Cancer Patients
Atul JG*, Sanjay R and Kartik P
Department of Radiation Oncology, Yashoda Hospital, Somajiguda, Hyderabad
Submission:January 30, 2021; Published: March 03, 2021
*Corresponding Address: Atul JG, Department of Radiation Oncology, Yashoda Hospital, Somajiguda, Thiruvananthapuram, Kerala, India
How to cite this article: Atul JG, Sanjay R, Kartik P. Evaluation of Dysphagia with Laryngo-Pharyngeal Sparing Intensity Modulated Radiotherapy in Head and Neck Cancer Patients. Canc Therapy & Oncol Int J. 2021; 18(2): 555983. DOI:10.19080/CTOIJ.2021.18.555983
Abstract
Aims and Objectives: The aim of this study was to determine how the dose received by Dysphagia/Aspiration at risk structures (DARS) affects swallowing in patients with head and neck cancer treated using IMRT. Primary objective of the study was to determine whether limiting the radiation dose to DARS using Do-IMRT improves swallowing compared to S-IMRT in head and neck cancer patients and secondary objectives were to find out a correlation between dysphagia associated Quality of Life (QOL) and radiation dose to DARS.
Materials and methods: This single institution study with patients satisfying all inclusion and exclusion criteria were randomised in 1:1 ratio into Do-IMRT and S-IMRT arms. CT simulations were taken, contouring of target volumes and OARs were done. In Do-IMRT arm, a mean dose constraint of 50Gy was given to Pharyngeal constrictors (PCM). All patients received standard radiation dose alongside weekly cisplatin. Assessment of dysphagia using MD Anderson Dysphagia Inventory (MDADI) score and associated QOL was done pre-RT, three months and six months post-RT.
Results: There is a significant improvement in the MDADI score in the Do-IMRT arm compared to S-IMRT arm at six months after completion of radiation. Patients in Do-IMRT arm have less Ryles tube dependence compared to the S-IMRT arm. There is a negative correlation between mean dose to PCM and mean MDADI score.
Conclusion: All cases of head and neck cancer where swallowing structures are irradiated and late dysphagia is suspected, Do-IMRT technique can be adopted for improvement of dysphagia and quality of life.
Keywords: Cancer; Oncology; Radiation; IMRT; DARS; Swallowing; Head and neck cancer; Radiotherapy; Dysphagia; Quality of life
Abbrevations: 3D-CRT: Three-dimensional conformal radiotherapy; ANCOVA: Analysis of Covariance; ASHA: American Speech-Language-Hearing Association; BT: Brachytherapy; CBK: Cyberknife; CE: Cervical Esophagus; CPI: Cricoid pharyngeal inlet; CRT: Chemoradiation; CT: Computed Tomography; CTCAE: Common Terminology Criteria for Adverse Events; CTV: Clinical Target Volume; DARS: Dysphagia aspiration related structures; DOI: Depth of invasion; do-IMRT: dysphagia optimized Intensity modulated radiation therapy; DS-QOL: Dysphagia-Specific Quality of Life; DVH: Dose-volume Histogram; EBRT: External Beam Radiation Therapy; ECOG: Eastern Cooperative Oncology Group; EORTC: European Organization for Research and Treatment of Cancer; FACT-H&N: Functional Assessment of Cancer Therapy Head and Neck Module; FEES: Flexible Endoscopic Evaluation of Swallowing Safety; GSL: Glottic and supraglottic larynx; GTV: Gross Tumour Volume; HNCI: Head and Neck Cancer Inventory; HNPS: Head and Neck Performance Status Scale; HNSCC: Head and neck squamous cell carcinoma; HPV: Human Papilloma Virus; HR-QOL: Health-related quality of life; IMRT: Intensity modulated radiation therapy; IPC: Inferior Pharyngeal Constrictor; LES: Lower oesophageal sphincter; MACH-NC: Meta-Analysis of Chemotherapy in Head and Neck Cancer; MBSS: Modified Barium Swallow Study; MDADI: MD Anderson Dysphagia Inventory; MPC: Middle Pharyngeal Constrictor; MRI: Magnetic Resonance Imaging; MV: Mega Voltage; MeV: Mega electron Volt; OAR: Organs at Risk; PAS: Penetration-Aspiration Scale; PCM: Pharyngeal constrictor muscles; PTV: Planning Target Volume; RT: Radiation Therapy (Radiotherapy); SIB: Simultaneous Integrated Boost; SMPC: Superior/ Middle Pharyngeal Constrictors; SPC: Superior Pharyngeal Constrictor; SWOAR: Swallowing Organs at Risk; QoL: Quality of life; QUANTEC: Quantitative Analyses of Normal Tissue Effects in the Clinic; RTOG: Radiation Therapy Oncology Group; SCC: Squamous cell carcinoma; SDQ: Swallowing Disturbance Questionnaire; s-IMRT: Standard Intensity modulated radiation therapy; SSW: Sydney Swallow Questionnaire; SWAL-QOL: Swallowing Quality of Life Questionnaire; TGF- β: Transforming growth factor β; TOM: Therapy Outcome Measure; TPS: Treatment Planning System; UOS: Upper oesophageal sphincter; VHNSS: Vanderbilt Head and Neck Symptom Survey
Introduction
Head and Neck cancer is the term used to describe a wide range of malignant tumours originating in the upper aero digestive tract, including the oral cavity, larynx, pharynx, and nasopharynx. The vast majority (90%) of head and neck cancers are squamous cell carcinomas (HNSCC) arising from the epithelial membranes of these regions [1].
Incidence and prevalence patterns of head and neck cancers are higher in developing countries than in developed countries [2]. Overall, 57.5% of global head and neck cancers (excluding oesophageal cancers) occur in Asia, especially in India, for both sexes. Over 200,000 cases of head and neck cancers occur each year in India, consisting of nearly 80,000 oral cancers, 40,000 cases of pharyngeal cancers excluding nasopharyngeal cancers (31% of global cases) and 29,000 cases of laryngeal cancers (18% of global cases) [3].
Cancers arising in the head and neck sites are near several critical structures such as the spinal cord, brainstem, parotid glands, optic apparatus (eyes, optic nerves, and optic chiasm), lacrimal glands, cochlea, and mandible that makes its treatment difficult and challenging. Radical radiotherapy with concurrent platinum-based chemotherapy remains the contemporary standard of care, in the non-surgical management of patients with loco-regionally advanced head and neck squamous cell carcinomas (HNSCC) [4,5].
Conventionally, the endpoints of medical care for head and neck cancer patients usually focused on objective tumour response, overall survival rate, local control rate, or complication rate. These endpoints were usually assessed from the physicians’ points of view. With the increasing use of multimodality treatment particularly in head and neck cancers, and the growing number of survivors, the need to understand patients’ experience, their perceptions of treatment effects and their priorities have to be well recognized. Hence, Health-related quality of life (HR-QOL) and its assessment have become increasingly important [6,7].
Common acute toxicities of head–neck chemo-radiotherapy include mucositis, dysphagia, dysgeusia, and dermatitis that can severely and adversely influence upon Quality of Life [8]. The most common and debilitating late toxicity is xerostomia – gross reduction in salivary output, leading to persistent dryness of mouth, oral discomfort, difficulty in speech and swallowing, impairment of taste, and deterioration of oro-dental hygiene [9,10] The other late effects include subcutaneous fibrosis, hoarseness of voice, and mucosal atrophy resulting in chronic dysphagia and increased risk of aspiration. With increasing use of aggressive combined modality therapy and altered fractionation schedules for head and neck cancers in radical and adjuvant settings, the presence of acute and late effects of radiation treatment has become an area of intensive interest and research [11]. Dysphagia associated with radiation is one problem that has garnered much attention. Acute and late swallowing dysfunction rates of about 15-63%, and 3-21%, respectively, have been documented [12].
Thus, the impact of dysphagia can be such that it can lead to disruption of the normal swallowing function, which may lead to aspiration, laryngeal penetration, and more than normal residue after the swallow and/or reflux. All these issues, along with the disease (head–neck cancer) and its treatment (chemoradiotherapy) can significantly affect Quality of Life domains such as swallowing functions as well as general Quality of Life domains such as physical, mental, and social health [13,14].
Over the years, major technological innovations have resulted in substantial improvements in radiotherapy planning, delivery and verification. The increasing use of computed tomography (CT) imaging for target volume delineation coupled with availability of computer-controlled treatment planning and delivery systems have progressively led to conformation of radiation dose to the target tissues while sparing surrounding normal tissues, i.e., three-dimensional conformal radiotherapy (3D-CRT). The advent of intensity-modulated radiation therapy (IMRT) defined as an advanced form of high-precision radiotherapy that uses nonuniform radiation beam intensities; determined using various computer-based optimizations has ushered in a new paradigm that has completely revolutionized contemporary radiotherapy practice [15]. Looking forward to improving the quality of life, and ways to reduce the incidence of both acute and late dysphagia, we decided to conduct a prospective trial to determine how the dose received to the Dysphagia Aspiration Related Structures (DARS) affects swallowing in patients with malignancies of head and neck cancers, treated using IMRT technique
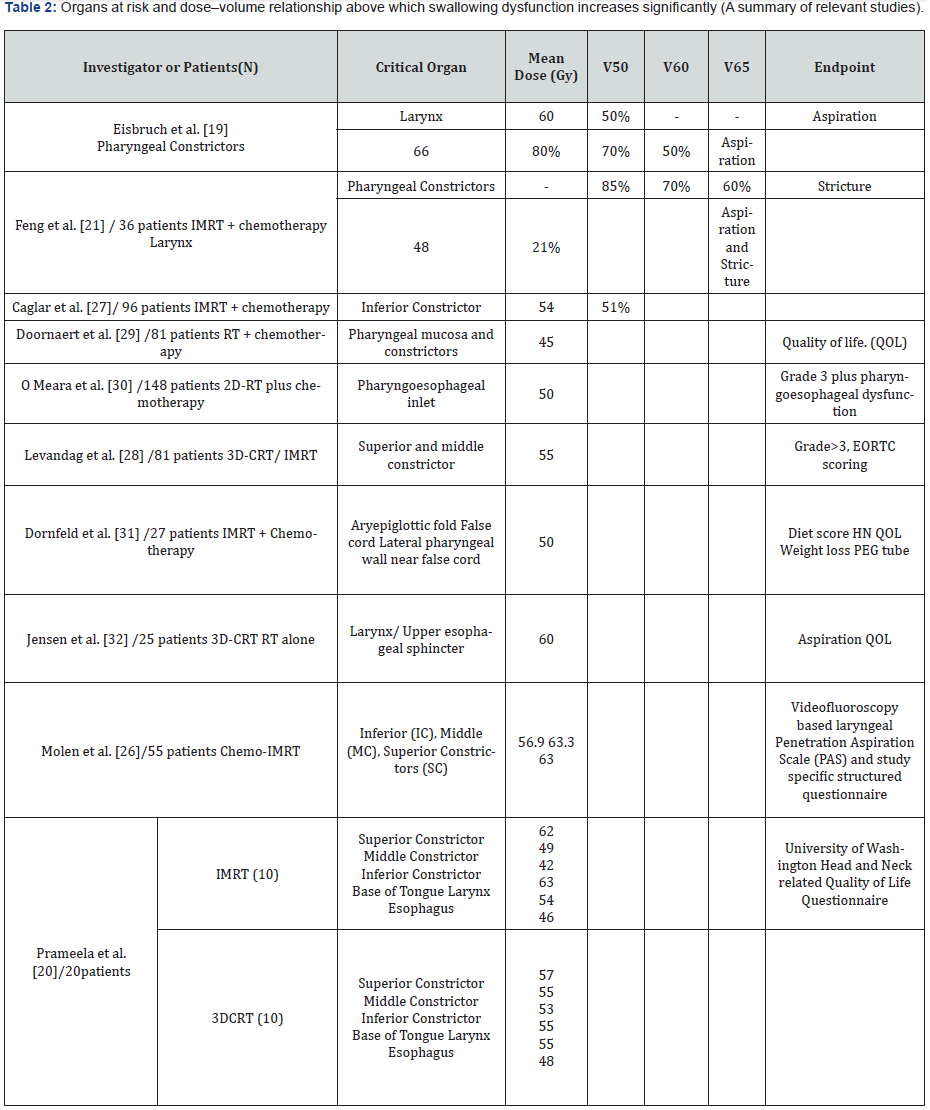
Langendijk et al. has proposed a predictive model for swallowing dysfunction in patients receiving radiotherapy, which includes
a) Host factors such as smoking status during and after treatment, old age, loss of weight from base line
b) Disease factors like site, size and extent of the primary tumour
c) Treatment related factors such as total dose and dose per fraction of radiation, inter-fraction interval, extent of treatment volume and technique of treatment [16].
Lee et al reported the results of a retrospective study of 199 patients treated with chemo radiation. Of 82 patients who underwent swallowing evaluation, 41 patients were found to have a stricture (21% of total). Predictors for stricture formation included twice-per-day radiation, female sex, and hypo pharyngeal primary. All 41 patients underwent dilation for treatment; however, its efficacy was not reported. An interesting side note to this study is the fact that more than half of the strictures occurred below the area that received high-dose radiation. The authors hypothesize that decreased swallowing may result in increased fibrosis [17].
Caudel et. al, assessed the factors associated with long-term dysphagia after definitive radiotherapy for locally advanced head-and-neck cancers. They analyzed cases that used altered fractionation radiotherapy (RT) regimens, as well as concomitant chemotherapy and RT, to intensify therapy for locally advanced head-and-neck cancer dysphagia. A total of 122 patients who had undergone definitive RT for locally advanced head and neck cancer, were taken into analysis. The patient, tumour, and treatment factors were correlated with a composite of 3 objective endpoints as a surrogate for severe long-term dysphagia: percutaneous endoscopic gastrostomy tube dependence at the last follow-up visit; aspiration on a modified barium swallow study or a clinical diagnosis of aspiration pneumonia; or the presence of a pharyngoesophageal stricture. This study found a composite dysphagia outcome occurring in 38.5% of patients. On univariate analysis, the primary site (p = 0.01), use of concurrent chemotherapy (p = 0.01), RT schedule (p = 0.02), and increasing age (p = 0.04) were significantly associated with development of composite long-term dysphagia. The use of concurrent chemotherapy (p = 0.01), primary site (p = 0.02), and increasing age (p = 0.02) remained significant on multivariate analysis. The authors concluded that concurrent chemo radiation for locally advanced head and neck cancer resulted in increased long-term dysphagia. It was also suggested to explore the necessity to use of IMRT to reduce the dose to the uninvolved swallowing structures which are at risk of developing long term dysphagia [18].
Upon seeing the higher incidence of dysphagia in patients receiving chemo radiation, Eisbruch et al, in 2004 conducted a study to identify the anatomic structures whose damage or malfunction could cause late dysphagia and aspiration after intensive chemotherapy and radiotherapy (RT) for head-and-neck cancer, and to explore whether they can be spared by IMRT, without compromising the target. He conducted a 2-arm prospective study (Phase I) trial with 26 patients in one arm receiving RT dose 70 Gy concurrent with gemcitabine. The prospective evaluation of swallowing was done with video fluoroscopy (VF), direct endoscopy, and CT scan showing significant abnormalities. Pre and post-therapy CT scans were reviewed for evidence of post therapy damage to each of these structures, and those demonstrating post therapy changes were deemed dysphagia/aspiration-related structures (DARS). It includes Pharyngeal constrictors, Glotticsupraglottic larynx and oesophagus. The 3-arm comparison of relative sparing of DARS by Standard three-dimensional (3D) RT, standard IMRT (stIMRT), and dysphagia-optimized IMRT (doIMRT) plans in 20 consecutive patients with locally advanced Head and Neck cancer was conducted. The RT dose was 66 Gy and the mean dose to DARS structure was 50 Gy. By comparing with the 3D plans, stIMRT reduced the V50 of the pharyngeal constrictors by 10% on average (range, 0–36%, p < 0.001), and doIMRT reduced these volumes further, by an additional 10% on average (range, 0 -38%; p <0.001). The V50 of the larynx (glottis, supraglottis) was reduced marginally by stIMRT compared with 3D (by 7% on average, range, 0–56%; p=0.054), and doIMRT reduced these volumes by an additional 11%, on average (range, 0–41%; p=0.002). doIMRT reduced laryngeal V50 compared with 3D, by 18% on average (range 0–61%; p =0.001). The maximal DARS doses were not reduced by IMRT because of their partial overlap with the targets. stIMRT and doIMRT did not differ in target doses, parotid gland means dose, spinal cord, or non-specified tissue maximal dose. The only difference is mean dose for DARS is 50Gy. Authors concluded that using DARS Sparing IMRT significantly reduced dose to swallowing structures, resulting in low incidence of aspiration and dysphagia as compared to Standard IMRT [19].
A similar study was done by Prameela et al. comparing 3DCRT and IMRT plans to evaluate the dose to dysphagia aspirationrelated structures and its effect on swallowing. A statistically significant subjective correlation was found to exist between doses to swallowing structures, especially the constrictor muscles, base of tongue, and the larynx. Statistically derived probable mean dose-constraints correlated clinically with swallowing status of patients. IMRT had a statistically significant advantage over 3D CRT, in terms of V30 (p = 0.051), V50 (p = 0.002), V60 (p = 0.002), and D80 (p = 0.023) for the swallowing structures. They concluded that a probable limiting dose for dysphagia was 63 Gy for the superior constrictor and base of tongue, and 55 Gy for the larynx [20].
Feng et al. in 2007 conducted a trial of IMRT aiming to spare the swallowing structures whose dysfunction after chemoradiation is a likely cause of dysphagia and aspiration, without compromising the target doses. This was a prospective, longitudinal study of 36 patients with locally advanced head and neck cancers and definitive chemo-IMRT aiming to spare the swallowing structures: pharyngeal constrictors (PC), glottic and supraglottic larynx (GSL), and oesophagus. Dysphagia endpoints included objective swallowing dysfunction (video fluoroscopy), and both patientreported and observer-rated scores. Significant correlations were observed between video fluoroscopy-based aspirations and the mean doses to the PC and GSL, as well as the partial volumes of these structures receiving 50–65 Gy; the highest correlations were associated with doses to the superior PC (p = 0.005). All patients with aspirations received mean PC doses >60 Gy or PC V65 >50%, and GSL V50 >50%. All 3 patients with strictures had PC V70 >50%. Worsening patient reported liquid swallowing was correlated with mean PC (p = 0.05) and oesophageal (p = 0.02) doses. Only mean PC doses were correlated with worsening patient-reported solid swallowing (p = 0.04) and observer-rated swallowing scores (p = 0.04). Hence, in this study, the authors concluded that the dose–volume-effect relationships provided an initial IMRT optimization goals and motivated further efforts to reduce doses to swallowing structures in order to reduce dysphagia and aspiration [21].
Suen et al. evaluated the clinical correlation of radiation dose to the larynx, pharyngeal constrictor muscles, and parotid glands with chronic dysphagia in head-and-neck cancer patients treated with concurrent chemo irradiation. 30 patients with Stage III-IVB HNSCC involving the oral cavity, oropharynx, larynx, and hypopharynx were treated with definitive concurrent chemo irradiation. Doses to the supraglottic and glottic larynx, pharyngeal constrictor muscles were measured, and dose-volume histograms were formulated. Clinical dysphagia was evaluated utilizing the Common Terminology Criteria for Adverse Events (v.3.0). Univariate analysis was performed, correlating degree of chronic dysphagia with V50, V60, and mean dose to the PCM and larynx. Results showed a statistically significant difference in degree of chronic dysphagia with increasing mean dose to the PCM (p-0.001). V50 and V60 of the PCM were both significantly correlated with degree of dysphagia (p = 0.002, p = 0.004). Hence it was concluded that a positive clinical correlation exists between PCM dose and chronic dysphagia and that it warrants special attention to this structure when designing IMRT plans. The authors also recommend striving to maintain a mean constrictor dose less than or equal to 60 Gy and V60 less than or equal to 60% [22].
Li et al. analysed 39 patients with HNSCC, treated by definitive concurrent chemotherapy and IMRT to a median dose of 70 Gy. A prophylactic gastrostomy tube (GT) was placed, and dysphagia was scored using standardized quality-of-life instruments. Dosevolume histogram (DVH) data incorporating the superior/middle pharyngeal constrictors (SMPC), inferior pharyngeal constrictor (IPC), cricoid pharyngeal inlet (CPI), and cervical oesophagus (CE) were analysed in relation to prolonged GT dependence, dysphagia, and weight loss. At 3 months and 6 months after treatment, 87% and 44% of patients, respectively, were GT dependent. Spearman’s ρ analysis identified statistical correlations (p < 0.05) between prolonged GT dependence and high-grade dysphagia with IPC V65, IPC V60, IPC Dmean, and CPI Dmax. It was concluded that adhering to the following parameters may decrease the risk of prolonged GT dependence and dysphagia: IPC V65 < 15%, IPC V60 < 40%, IPC Dmean < 55 Gy, and CPI Dmax < 60 Gy [23].
Teguh et al. in 2008, undertook studies to assess the relationship between the dose received by the swallowing structures and the dysphagia related quality of life (QoL) in oropharyngeal (OP) cancer and nasopharyngeal (NP) cancers. 85 OP and 47 NP cancer patients treated by radiation therapy were analysed. Various techniques using (IMRT), brachytherapy (BT), or frameless stereotactic radiation/cyberknife (CBK) were used for treatment. The swallowing muscles were delineated, and the mean dose calculated using the original three-dimensional computed tomography–based treatment plans. They noted that for the 81 patients alive with no evidence of disease for at least 1 year, a dose–effect relationship was observed between the dose in the superior constrictor muscle and the ‘‘normalcy of diet’’ (Performance Status Scales) or ‘‘swallowing scale’’ (H&N 35) scores (p < 0.01) [11,24].
The need for contouring DARS and to give a constrain was evident in 2008, but it was Christianen et al in 2011, who provided Computed Tomography (CT)-based delineation guidelines for organs in the head and neck at risk for radiation-induced swallowing dysfunction and termed them Swallowing Organs at Risk (SWOARs) [25].
Later on Peponi et al. in 2011 evaluated the objective and subjective long term swallowing function and the relation of dysphagia to the radiation dose delivered to the critical anatomical structures in head and neck cancer patients treated with IMRT, +/-chemotherapy. It was observed that use of high intensity treatments like IMRT resulted in considerable increase in dysphagia. This could be due to irradiation of critical structures involved in swallowing. As per the study, dose constraints to swallowing structures were 45Gy. PTV primary & Neck nodes - 70Gy and in Post-operative cases (Adjuvant) 60-66Gy. The results showed that sparing the swallowing structures by IMRT seemed effective and relatively safe in terms of avoidance of persistent grade 3/4 late dysphagia and local disease control [12].
Molen et al. conducted a prospective assessment of dysphagia and trismus in chemo-IMRT of head and neck cancer patients in relation to dose-parameters of structures involved in swallowing and mastication in 55 patients, before, 10-weeks after, (N = 49) and 1-year post-treatment (N = 37). Dose-volume parameters for swallowing (IC, MC, and SC), and mastication structures (e.g. masseter) were carried out. Investigation of relationships between dose-parameters and endpoints for swallowing problems (video fluoroscopy-based laryngeal Penetration-Aspiration Scale (PAS), and study-specific structured questionnaire) and limited mouthopening (measurements and questionnaire), considering baseline scores. At 10-weeks, volume of IC receiving V60 Gy and mean dose to IC were significant predictors for PAS. One-year post-treatment reported problems with swallowing solids were significantly related to masseter dose-parameters (mean, V20, V40 and V60) and an inverse relationship (lower dose related to a higher probability) was observed for V60 of the IC. Authors concluded that dose-effect relationships exist for dysphagia and trismus and hence treatment plans should be optimized to avoid these side effects [26].
Caglar et al. conducted a study to evaluate the dose to the larynx that could predict swallowing abnormalities after IMRT. Of the evaluable 96 patients, 32% had clinically significant aspiration and 37% developed a stricture. The radiation dosevolume metrics, including the volume of the larynx receiving >or=50 Gy (p = 0.04 and p = 0.03, respectively) and volume of the inferior constrictor receiving >or=50 Gy (p = 0.05 and p =0.02, respectively) were significantly associated with both aspiration and stricture formation. The authors concluded that the dose given to the larynx and inferior constrictors correlated with aspiration and strictures [27].
Levendag et al. in his study to assess the relationship between the radiation therapy (RT) dose received by the muscular components of the swallowing apparatus and - dysphagia related - quality of life (QoL) in oropharyngeal cancer patients, evaluated 81 patients, treated either with 3DCRT or IMRT, with or without concomitant chemotherapy (CHT); 43 out of these 81 patients were boosted by brachytherapy (BT). 23% experienced severe dysphagia. The SCM, MCM, ICM, the cricopharyngeus muscle and the inlet of the oesophagus, were of paramount importance for swallowing. A significant correlation was observed between the mean dose to the SCM and MCM, and severe dysphagia complaints (univariate analysis). A steep dose-effect relationship, with an increase in the probability of dysphagia of 19% with every additional 10 Gy, was established. The study concluded that a dose-effect relationship between dose and swallowing complaints was observed [28-34].
Looking into the studies which analyzed laryngeal toxicity, the following were considered:
Various predictive models have been developed for the prediction of acute dysphagia following IMRT for head and neck cancer and this was illustrated using EM Lasso technique for model selection [35] Teguh et al also developed a predictive risk model that could be used in selecting patients for hyperbaric oxygen treatment in the prevention or reduction of severe late side-effects caused by radiotherapy. Patients at risk of developing xerostomia and dysphagia was included in this trial [36]. Dosimetric models where brachytherapy can be used to aim to limit the dose to dysphagia/aspiration related structures in patients of oropharyngeal cancers have been described and it demonstrates that effective use of brachytherapy, even as a boost to conventional EBRT can successfully limit dose to DARS [37] Rancati developed radiobiological predictive models using Lyman- Kurcher-Burman and Log equivalent uniform dose model for predicting laryngeal oedema [34]. Nguyen et al conducted a study on the impact of dysphagia on quality of life after treatment of head and neck cancer. It was shown that dysphagia is a significant morbidity and its severity correlated with compromised quality of life, anxiety and depression [38].
A study conducted by Agarwal et al on subjective evaluation of swallowing function and dietary pattern in head and neck cancers treated with chemo radiation showed deterioration of swallowing function in majority of HNSCC patients [39] Another study conducted by Maurer et al. on impact of dysphagia on quality of life after chemo radiotherapy of head and neck cancer also showed dysphagia to be a significant morbidity following chemo radiation to head and neck cancer [40].
In view of the various morbidities related to the radiation induced toxicities like acute and chronic dysphagia, aspiration and its subsequent effect on the quality of life of these patients was much of a concern. Hence, we decided to design this trial to look at the doses received to the DARS and its correlation to dysphagia in patients treated using IMRT Technique for Head and Neck malignancies.
Materials and methods
Study site: Patients registered in the Department of Radiotherapy, Yashoda Hospital, Somajiguda, Hyderabad and fulfilling eligibility criteria were enrolled to this study.
Study population: Data was collected from histologically proven cases of Head and Neck malignancy. Patients planned for treatment with external beam radiotherapy using IMRT techniques along with concurrent chemotherapy was considered for the study.
Study design: A prospective, randomized study
Sample size: A total of 30 cases were included in the study. 15 cases each in Do-IMRT (Experimental RT group) and S-IMRT (Standard RT group) arm. Sample size of this study was determined by previous studies [19-21].
Sample size formula n =Z2pq/d2, where n= desired sample size, Z=standard normal deviate; usually set at 1.96 (or a~2), which correspond to 95% confidence level, p=proportion in the target population estimated to have a particular characteristic. If there is no reasonable estimate, use 50% (i.e 0.5), q=1-p (proportion in the target population not having the characteristics), d= degree of accuracy required, usually set at 0.05 level (Tables 1-6).





Example - If the proportion of a target population with certain characteristics is 0.50, Z statistics is 1.96 & we desire accuracy at 0.05 level, then the sample size is n= (1.962) (0.5) (0.5)/(0.05)2. That is, n=384.
To find a statistical difference between two comparative arms, we should have many patients in each arm (about 400), which is not possible in my study. So according to previous thesis study designs I have selected a total of 30 patients to be included in the study, 15 patients each in Do-IMRT and S-IMRT arm.
Study period: Patients coming to the hospital from November 2016 to October 2017 were recruited to the study.
Patient eligibility criteria:
Inclusion Criteria:
a) Adult patients aged 18 or above, any sex.
b) All histopathologically proven patients of Squamous cell carcinoma of Head and Neck (Nasopharynx, Nasal cavity, Paranasal Sinuses, Oropharynx, Hypopharynx and Supraglottic Larynx) receiving chemoradiation in a definitive setting.
c) Patients who are receiving radiation as primary treatment to a dose of 66-70 Gy in conventional fractionation of 2 Gy/ fraction/ 5 fractions/ week (with concurrent weekly chemotherapy) as the planned treatment.
d) Patients with stage T1-4, N0-3, M0 disease.
e) Patients with Creatinine clearance (≥50 mL/min prior to starting chemotherapy).
f) Patients with good performance status of ECOG 0-1.
g) Patients available to attend long term follow- up.
h) Patients with adequate cognitive ability to complete the MD Anderson Dysphagia Inventory (MDADI) questionnaire.
i) Written informed consent.
Exclusion Criteria:
a) Patients with documented evidence of pre-existing swallowing dysfunction (not related to head and neck cancer).
b) Patients who received previous radiotherapy to the head and neck region.
c) Patients with posterior pharyngeal wall, post-cricoid, or retropharyngeal lymph node involvement.
d) Patients with metastatic disease at presentation.
e) Patients with cancer at any other sites, other than those mentioned in the inclusion criteria.
f) Patients who have undergone tracheostomy or total laryngectomy.
g) Patients not consenting for study
Methodology:
This is a prospective and randomized study, in patients undergoing radical primary chemoradiation for Head and Neck Cancers. An approval of the Ethics Committee and Scientific Review Committee of the institute will be obtained before commencing the study. Written informed consent will be taken from all the participants explaining the nature of study. Suitable patients will be randomized to either S-IMRT or Do-IMRT. Radiotherapy dose and fractionation will be the same in both treatment groups (Tables 7-15).







Work Flow Diagram
Initial Evaluation and Enrollment
a) Full medical history and physical examination.
b) Local examination as an initial clinical assessment of tumour stage.
c) Endoscopic assessment of site, nature, and extent of the disease.
d) Diagnostic workup consisting of hemoglobin, total and differential WBC count, platelet count, renal function tests (Urea, Creatinine, 24hours urinary creatinine clearance), liver function tests (Bilirubin- total, direct and indirect, SGOT, SGPT, Alkaline Phosphatase, Total serum protein, Albumin and Globulin levels),
e) X-ray chest PA view
f) Radiological assessment with a CT scan for site and extent of the disease.
g) Assessment of ECOG performance score.
h) All patients underwent routine dental evaluation by a dentist at our centre prior to RT.
The patient thus evaluated was assigned to one of the treatment arms for further treatment as outlined in the study design (Figure 1).

Registration and randomization
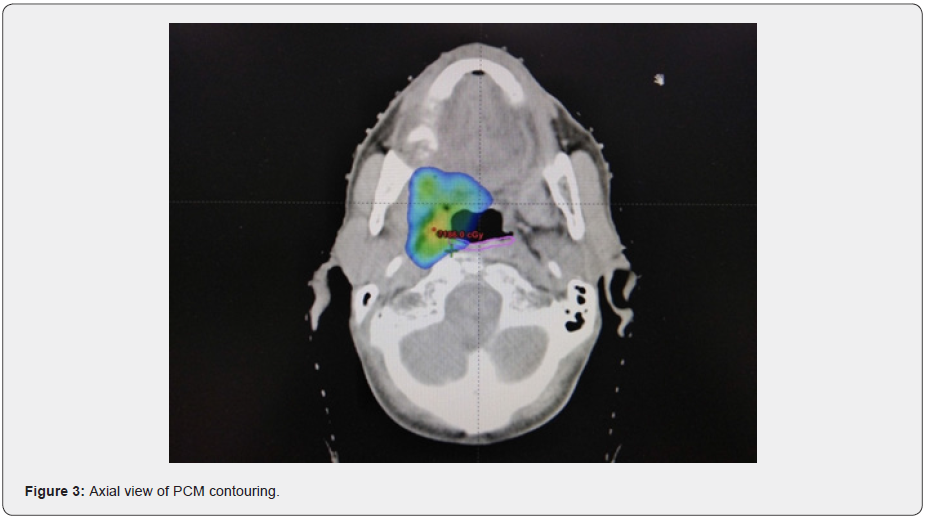
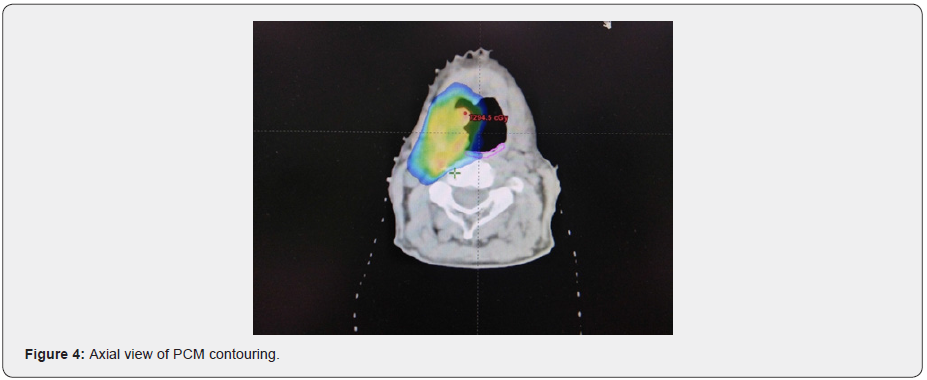
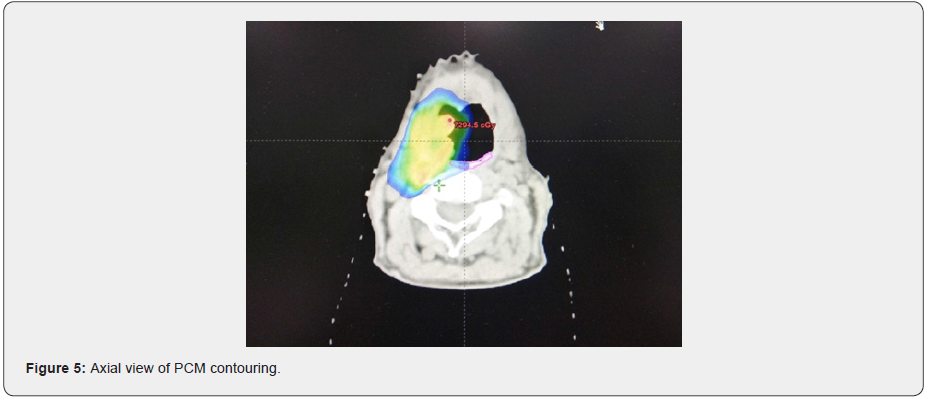
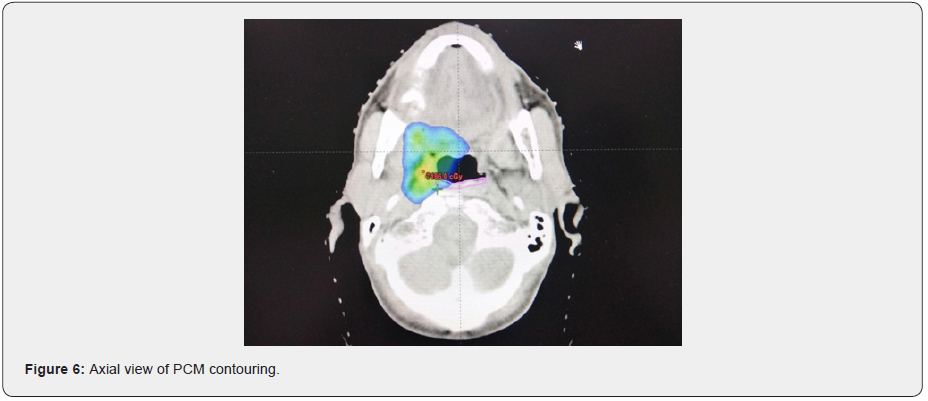
The study has a two-stage entry process of registration and randomization. Randomization occurs only following target outlining, ensuring consistency across both the experimental and standard treatment volumes by avoiding any potential bias that could be introduced by the clinician during delineation. Additionally, patients will be blinded to the treatment allocation to avoid bias during assessments. Patients will be randomized between the two treatment arms on a 1:1 basis using the method of minimization with a random element (Figures 2-6).
Chemotherapy
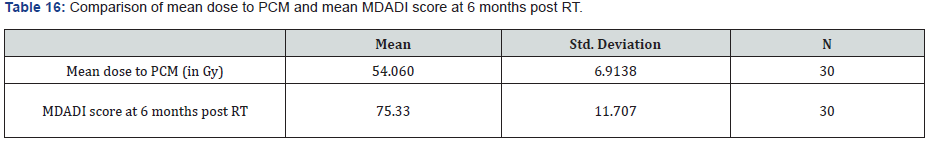
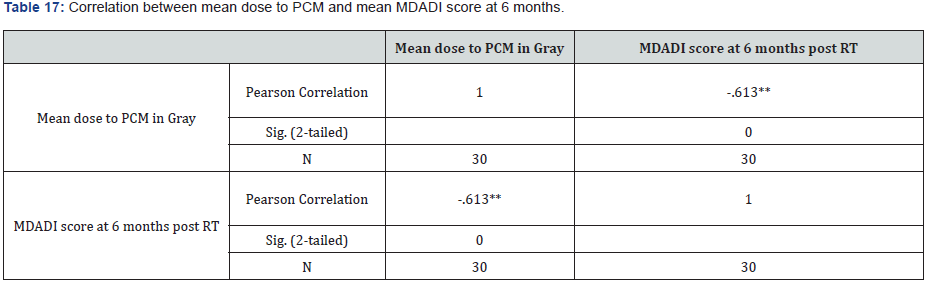
Concomitant chemotherapy is recommended for all patients, using the standard regimen of 40 mg/m2 of Cisplatin every week along with radiotherapy (Tables 16 &17).
**Correlation is significant at the 0.01 level (2-tailed).
Radiotherapy
CT-Simulation
Prior to RT-planning CT-simulation of the patients was taken in supine position with proper immobilization techniques using five clamp thermoplastic cast, head-neck base plate and appropriate headrest. Fiducial markers are used according to laser co-ordinate system for future isocentre reference. Intra-venous contrast according to the body weight was given to all patients for better visualization of tumor and vessels. Serial axial images with a slice thickness of 3mm were obtained and these images were transferred to the EclipseTM (Varian Medical Systems, Palo Alto, CA, USA) Treatment Planning System (TPS), where following image acquisition, the target volume, and critical organs were contoured.
Target Delineation
Target volume delineation DARS has adopted a volumetric approach to define the target volumes. Findings at the time of endoscopy along with pre-therapy imaging were used to aid accurate delineation of the primary tumour. Two clinical target volumes (CTV) will be defined and edited to exclude natural barriers to disease spread. CTV-P will include the primary and nodal gross tumour volume (GTV) with a 1 cm isotropic margin while the prophylactic CTV-LR will include the remainder of the involved subsite and nodal levels at risk of microscopic disease. Corresponding planning target volumes (PTVs) will be grown with 3–5 mm margins, according to the practice of individual centres. CT delineation of nodal levels will follow the recently updated outlining guidelines [41].
Delineation of Organs at Risk
Organs at risk in head and neck malignancies like brainstem, spinal cord, parotid glands (bilateral), submandibular glands, cochlea (bilateral), oral cavity, mandible and temperomandibular joint and brain (carcinoma nasopharynx), optic apparatus (carcinoma nasopharynx, nasal cavity/paranasal tumours) like eyeball, lens, optic chiasma, retina were contoured.
The superior, middle and inferior constrictors will be contoured as one structure (PCM) in the study. Outlining for the PCM is based on the published contouring guidelines defined by Christianen et al. [25]. The experimental Do-IMRT technique aims to spare the PCM lying outside the high dose CTV. For oropharyngeal primaries, mandatory mean dose constraints of <50 Gy is applied to the volume of PCM lying outside CTV-P. Crucially, it is important to note that although the PCM will overlap with the PTVs, there will be no sparing of the constrictor muscle that lie within the PTV-P.
Treatment Planning
Planning objectives will be prioritized in the following order: critical organ constraints (spinal cord and brainstem); PTV-P coverage; constrictor constraints; PTV-LR coverage; parotid gland constraints and other normal tissue constraints. After completion of contouring, the treatment planning was done by the physicist using ECLIPSE™ treatment planning system.
RT Dose and schedule
Patients in both treatment groups were planned to receive 66- 70 Gy in 33-35 fractions to the primary and nodal tumour (PTV-P) and 54-56 Gy in 33-35 fractions (PTV-LR) to the areas considered at risk of harbouring microscopic disease.
RT Technique
Treatment will be delivered by a variety of IMRT techniques. Patients in the S-IMRT group will receive the treatment according to the current standard of radiation planning, whereas PCM irradiation will be reduced by introducing it as an OAR in the treatment planning objectives of patients allotted to the Do- IMRT arm. Treatment verification will include the following as a minimum: orthogonal kilo voltage (kV) isocentre images, or cone beam CT images, taken daily. Any treatment gaps will be managed as per the Royal College of Radiologists guidelines for Category 1 patients, aiming to complete radiotherapy within 6 weeks. Dose constraints as advised by QUANTEC was followed for the OAR structures during IMRT planning. Prior to delivery, a fluence map check and other quality assurance checks were ensured before starting the treatment.
Assessment of toxicity
Assessments Toxicity and response assessments NCI CTCAE v4.0 will be used to assess acute toxicity data that will be collected weekly during radiotherapy, and at 6 weeks after treatment completion. Clinical assessments will be made at 6 weeks, and 3, 6 months after completion of treatment as a minimum. Additional investigations will be requested if clinically indicated. Patients found to have persistent cervical lymphadenopathy will proceed to neck dissection.
Assessment of dysphagia
Subjective assessment of dysphagia was done for each patient using MDADI questionnaire just prior to treatment, 3 months, and 6 months post completion of radiotherapy. The MDADI questionnaire was translated to local language (Telugu) by a translator and re-translation from local language (Telugu) to English was done by another translator and after that both the questionnaires were compared for proper validation. If there is any illiterate patient included in the study, then we will take the help of a close relative who is literate in local language (Telugu) or English for easy understanding of the questionnaire and to decrease bias.
The MDADI is designed to be a valid and reliable instrument for evaluating QOL issues associated with dysphagia. To our knowledge, the MDADI is the first validated and reliable selfadministered questionnaire designed specifically for evaluating the impact of dysphagia on the QOL of head and neck cancer patients. Standardized questionnaires measuring patient’s QOL offer a means for demonstrating treatment impact and for improving medical care.
MDADI questionnaire contains twenty different items and five possible responses to each item like strongly agree, agree, no opinion, disagree, and strongly disagree and scored on a scale of 1 to 5. All the 20 items are grouped into three sub-scales, emotional, functional and physical. One item on the emotional sub-scale (I do not feel self-conscious when I eat) and another on the functional sub-scale (I feel free to go out to eat with my friends, neighbours, and relatives) are scored as 5 points for strongly agree and 1 point for strongly disagree. All other items are scored as 1 point for strongly agree and 5 points for strongly disagree. The first question (global subscale) is scored individually. All other questions regarding each aspect of dysphagia are summed, and a mean score was then calculated. This mean score was multiplied by 20 to obtain a score with a range of 0 (extremely low functioning) to 100 (high functioning). Thus, a higher MDADI score represented better day-to-day functioning and better QOL.
Statistical design
The collected data was analyzed using a standard statistical software package (version 22.0). For statistical analysis, the primary endpoint will be compared between the two groups using a two-sample t-test or a non-parametric test. The primary analysis will be by intention-to-treat, including all patients with 6-month MDADI data. A p-value of <0.05 will be considered statistically significant. Analysis of covariance (ANCOVA) will be used to investigate other patients and clinical factors that could be associated with change in MDADI composite score from baseline to 6 months post- treatment.
Results
A total of 30 patients were enrolled for the study and were randomized after fulfilling the eligibility criteria into two arms, Do-IMRT arm and S-IMRT arm on a 1:1 basis. Overall, 15 patients were included in each arm with no dropouts.
Gender
The male patients accounted for 90% (n=27) of total patient population in the study.
Age
Maximum number of patients in Do-IMRT arm is in the age group of 40-49 years. In S-IMRT arm, 40-49- and 50-59-year age groups contain 5 patients each. The mean age of the study population is 48 years (Figures 7-9).
Site of cancer
When patients are distributed according to the primary site of disease, oropharynx was found to be the most common site in each arm.


Stage of cancer
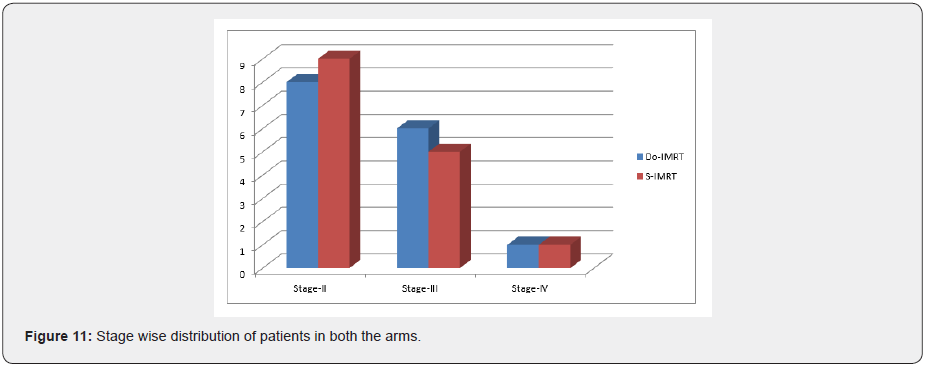
Stage wise distribution of patients showed stage-II patients were higher in both the arms (Figures 10 & 11).
MDADI score Pre-RT
The MDADI scores were used for subjective assessment of dysphagia and associated QOL. The mean MDADI score prior to starting chemo-radiation are 51.2 and 59.8 in Do-IMRT and S-IMRT arms respectively and the p-value was not significant.
MDADI score 3 months Post RT
The mean MDADI scores were found to be 70.67 and 72.13 at three months after completion of chemo-radiation in Do-IMRT and S-IMRT arms, respectively.
MDADI score 6 months Post RT
The mean MDADI scores were again assessed at 6 months after completion of chemo-radiation and were found to be 83.2 and 67.4 in Do-IMRT and S-IMRT arms, respectively.
Comparison of mean MDADI scores
Comparison of trend of MDADI scores in both the arms shows an increasing trend in Do-IMRT arm.
Correlation between mean dose to PCM and mean MDADI score at 6 months
When we tried to find out a correlation between the mean dose to PCM and mean MDADI score at 6 months, we found a negative correlation between the mean dose to PCM and MDADI score at 6 months.
Discussion
Carcinoma of head and neck is more common in men approximately 60-95% cases due to indiscriminate use of tobacco in various forms. Usual time of diagnosis is after 40 years, although most patients are between 50 and 70 years old. Nearly 60% of newly diagnosed patients present with locally advanced disease [42,43].
In our study 90% of patients were male with mean age of 48 years. 12 out of total 30 (40%) patients belong to age group of 40- 49 years. Most of the patients had a good performance status with ECOG score one or less. According to site and stage of the primary disease highest number of patients belong to oropharynx (15 out of 30, 50%) and stage-II (Figures 12-16).


Progress in head and neck cancer (HNC) treatments has improved tumour response and loco-regional control rates. However, despite improved diagnostic and therapeutic approaches, mortality remains high [4,44] Intensification of treatment with chemo-radiotherapy (CRT) or altered fractionation radiotherapy (RT) is associated with improved outcome but causes severe early and late mucosal and pharyngeal toxicities. Oropharyngeal dysphagia is an underestimated symptom in HNC patients [21,45].
Frequent causes of dysphagia in this population include neurological and neuromuscular impairment, and structural and iatrogenic causes. Dysphagia should not be neglected, as it can profoundly diminish the quality of life [46]. The resulting impaired swallowing can cause malnutrition and dehydration and might lead to aspiration pneumonia. Swallowing disorders are often predictable, depending on both tumors associated structures and treatment modalities. In our study, we also witnessed dysphagia as one of the cumbersome side effects apart from xerostomia and mucositis.
Acute and late swallowing dysfunction rates of about 15-63%, and 3-21%, respectively, have been documented [19] Dysphagia optimized IMRT, a step ahead of standard IMRT, where the dose to the DARS could be tailored to be contained within the limits, without compromising the dose to target volumes, has been suggested by Eisbruch et al. and is a promising avenue [19] In the present study also, Do-IMRT patients show a better response about dysphagia and associated QoL.
The dose received by the constrictor muscles has been implicated in post-radiation dysphagia, with the strongest relationship for SCM, as published by pioneers like Feng et al. [21] Consumption of solids was the most important swallowing problem that patients experienced. Though xerostomia is a compounding factor, the dose of radiation does have a significant correlation to the degree of dysphagia. In our study all the patients have received 65Gy in 30 fractions over a period of 6 weeks.


Van der Molen et al. had shown that a mean dose above 63 Gy to the SCM and the MCM resulted in a poor swallowing quality [26]. The same was shown by Feng et al. for the pharyngeal constrictors taken together [21] In our study we have taken a mean dose of 50 Gy to all the three constrictor muscles taken together, in IMRT, stressing that this can be taken as a limiting dose.

Van der Molen et al. had also suggested that the base of the tongue, larynx, and esophagus with the cricopharyngeus muscle, the other structures of DARS, which they had not investigated, also had a significant role in the dysphagia status [26] Our study also restricted to contouring of PCM only, so to see how much it contributes towards dysphagia. A shared dose–volume constraint for the superior-PCM has not yet been clearly defined and how and when it would be safe to spare the PCM is still a matter for debate. Limitation of superior-PCM volume receiving more than 60 Gy represents one of recommended dose–volume limit to reduce incidence and severity of late radiation-related dysphagia.
Quality of life is an important outcome measurement in objectifying the current health status or therapy effects in patients with oropharyngeal dysphagia. In the Maastricht University Medical Center study, the validity and reliability of the Dutch version of the Deglutition Handicap Index (DHI) and the MD Anderson Dysphagia Inventory (MDADI) have been determined for oncology patients with oropharyngeal dysphagia. At Maastricht University Medical Center, 76 consecutive patients were selected and asked to fill in three questionnaires on quality of life related to oropharyngeal dysphagia (the SWAL-QOL, the MDADI, and the DHI) as well as a simple one-item visual analog Dysphagia Severity Scale. None of the quality-of-life questionnaires showed any floor or ceiling effect. The test-retest reliability of the MDADI and the Dysphagia Severity Scale proved to be good [47]. In our study we have taken MDADI questionnaire as the tool for assessment of dysphagia and associated QOL.
Eisbruch et al. [19] were one of the first who reported on the results of a prospective study including 26 patients with locally advanced head and neck cancer treated with radiotherapy and concomitant gemcitabine. Based on the findings of videofluoroscopy, direct endoscopy and computed tomography preand post-treatment, they suggested that malfunction of the pharyngeal constrictor muscles, the supraglottic larynx and the glottic larynx were most likely causing radiation-induced dysphagia and aspiration. After this publication, several other authors reported on the results of studies in which the relationship between the dose distributions to anatomical structures involved in swallowing and post-treatment swallowing dysfunction was analyzed.
The literature review as presented in our study showed several differences in the definition of SWOARs. This may result in subsequent differences in DVH parameters from the same treatment plan. Therefore, the results of the different studies that investigate the relationship between DVH parameters and swallowing function after radiotherapy will be hard to compare. Furthermore, translation of the results of dose–volume–effect relationship studies into clinical practice can only be introduced safely if radiation oncologists use the similar guidelines in clinical practice. Therefore, delineation guidelines are the first prerequisite for unambiguously contouring of SWOARs and for a reliable comparison and interpretation of results from different studies.
As the CT scan currently is the gold standard for target volume and OAR delineation, therefore we decided to define CT imagebased delineation guidelines. However, it should be noted that the visualization of relevant anatomic swallowing structures could be improved by using Magnetic Resonance Imaging (MRI). MRI can help to discriminate the muscles from surrounding tissues in more detail. Therefore, the use of co-registered MRI in conjunction with CT may improve and facilitate the delineation of the pharyngeal muscles. Furthermore, when the tumour extends in one of the SWOARs, or when the tumour or involved lymph nodes alter the normal anatomy, delineation of the SWOARs may be burdensome. Hampered interpretation could also be the case when the CT scan images are blurred due to artifacts. To delineate in a consistent way, the solution would be to delineate the contours in the well perceptible slices and interpolate the delineations in between. In our study we have used MRI fusion with CT for better delineation of target and PCM.
Advanced T-stage (larger tumors) and N-stage and bilateral irradiation of the neck were prognostic factors for feeding tube dependence at 6 months after chemoradiation, it can be hypothesized that the risk of tube feeding dependence is related to the radiation dose distribution in the anatomical structures involved in swallowing, such as the pharyngeal constrictor muscles. Several authors indeed showed dose-volume effect relationships of anatomical structures involved in swallowing and swallowing dysfunction after chemoradiation, such as the pharyngeal constrictor muscles [25,28,47]. We have contoured only the three pharyngeal constrictor muscles as the swallowing related OARs and at the end of 6 months after chemoradiation there is a significant improvement in dysphagia and associated QOL in the Do-IMRT arm as compared to S-IMRT arm.
The group of Eisbruch et al. suggested that the high locoregional control rates have not been compromised by the efforts to spare the parts of the swallowing structures not involved by tumor and not at risk of subclinical disease [19] Studer G et al. in their study of hypopharynx-larynx patient cohort treated with IMRT using midline sparing as far as feasible, local failures were not found related to the midline sparing structures [49] In our study, during 6 months of follow up, we have not found any patient having loco-regional failure.
IMRT using a midline contour to spare swallowing structures outside PTVs is relatively safe and effective in terms of local disease control and avoidance of persistent late dysphagia. The subjective patients’ estimation of late dysphagia was compatible with the objective assessment of swallowing dysfunction [12]
Feng et al. found seventeen percent of the patients with oropharyngeal cancer seen at their institution during the time of their study were not eligible to participate because of gross involvement of the retropharyngeal nodes or the posterior pharyngeal wall which would not allow partial sparing of the PCM. In our study we also excluded the same group of patients [21] Gross involvement of the retropharyngeal nodes is a poor prognostic factor, and the exclusion of these patients may have contributed to the high tumor control rates in our study.
Evaluation of dysphagia and its comparison with other trials are complicated by multiple patient-reported instruments [50] multiple end points for interpretation and summary of VF results [51] poor reproducibility in interpreting the results of VF [52] and low correlations between observer, patient, and objective measures, for which observers tend to underestimate dysphagia [53,54] In our study we have used only the MDADI questionnaire for evaluation and comparison.
Nonetheless, some limitations of this study deserve mention. Video-fluoroscopic evaluation that may have provided a more sensitive measure of the effect of the Do-IMRT on swallowing function was not used in our study. In DARS multi-center randomized trial, in some centers they have objectively assessed the swallowing function by fiberoptic endoscopic evaluation of swallowing (FEES) along with MDADI questionnaire. In this study, only subjective analysis using MDADI questionnaire was done. Fiberoptic endoscopic evaluation needed Intensive Care Unit back up as it included Penetration Aspiration Scoring. Due to limited resources, FEES was not incorporated into this study. We have assessed late dysphagia up to six months only whereas according to literatures, late dysphagia may appear 3-12 months following completion of radiotherapy. In addition, as noted, the small sample size may have limited our ability to detect statistically significant differences in swallowing function as well as accurately identify the magnitude of the differences that were observed.
From clinical practice we know that the reasons for feeding tube placement are not solely related to swallowing problems but may be multifactorial. Nausea due to chemotherapy treatment, changes in taste or saliva production and other factors may also necessitate feeding tube placement. We did not directly take these factors into account in this analysis since it was not possible to obtain information about this for each individual patient.
Conclusion
Dysphagia following chemo-radiation represents a substantial problem and patients identify it as a distressing symptom which adversely affects their day-to-day life. Quality of life is an important outcome measurement in objectifying the current health status or therapy effects in patients with oropharyngeal dysphagia and can lead to malnutrition, dehydration, and aspiration pneumonia. Physicians should be vigilant enough to detect dysphagia during treatment and follow up period by proper clinical and imaging tools.
MDADI questionnaire is a very much useful tool in subjective assessment of dysphagia and associated quality of life, hence can be used routinely for head and neck cancer patients planned for radiation. Dysphagia optimized IMRT, a step ahead of standard IMRT, where the dose to the DARS could be tailored to be contained within the limits, without compromising the dose to target volumes, has been proved to have promising result in improving dysphagia and associated quality of life. So, in all cases of head and neck cancer where swallowing structures are irradiated and late dysphagia is suspected, Do-IMRT technique can be adopted for improvement of dysphagia and quality of life. In Do-IMRT technique, prescribing a dose constraint of 50Gy to the pharyngeal constrictor muscles without compromising the total dose and schedule of radiotherapy in head and neck cancer patients improves dysphagia. Further studies with larger sample size and longer follow up period are required for establishing this observation.
References
- Lutzky VP, Moss DJ, Chin D, Coman WB, Parsons PG, et al. (2008) Biomarkers for Cancers of the Head and Neck. Clinical medicine. Ear, nose and throat. 1: CMENT-S1051.
- D Souza PJ, Chakrabarty J, Sulochana B, Gonsalves J (2013) Quality of life of head and neck cancer patients receiving cancer specific treatments. Journal of Krishna Institute of Medical Sciences University 2(1): 51-57.
- Kulkarni MR (2013) Head and neck cancer burden in India. Int J Head Neck Surg. 4(1): 29-35.
- Pignon JP, Bourhis J, Domenge CO, Designé LL, (2000) Chemotherapy added to locoregional treatment for head and neck squamous-cell carcinoma: three meta-analyses of updated individual data. The Lancet 355(9208): 949-955.
- Pignon JP, Le Maitre A, Maillard E, Bourhis J (2009) Meta-analysis of chemotherapy in head and neck cancer (MACH-NC): an update on 93 randomised trials and 17,346 patients. Radiotherapy and Oncology 92(1): 4-14.
- Leung SW, Lee TF, Chien CY, Chao PJ, Tsai WL, et al. (2011) Health-related quality of life in 640 head and neck cancer survivors after radiotherapy using EORTC QLQ-C30 and QLQ-H&N35 questionnaires. BMC cancer 11(1): 128.
- List MA, Stracks J (2004) Quality of life and late toxicities in head and neck cancer. In: Head and neck cancer. Springer, Boston MA, pp. 331-351.
- Trotti A, Bellm LA, Epstein JB, Frame D, Fuchs HJ, et al. (2003) Mucositis incidence, severity and associated outcomes in patients with head and neck cancer receiving radiotherapy with or without chemotherapy: a systematic literature review. Radiotherapy and oncology 66(3): 253-262.
- Langendijk JA, Doornaert P, Verdonck de Leeuw IM, Leemans CR, Aaronson NK, et al. (2008) Impact of late treatment-related toxicity on quality of life among patients with head and neck cancer treated with radiotherapy. Journal of clinical oncology. 26(22): 3770-3776.
- Jensen SB, Pedersen AM, Vissink A, Andersen E, Brown CG, et al. (2010) A systematic review of salivary gland hypofunction and xerostomia induced by cancer therapies: prevalence, severity and impact on quality of life. Supportive care in cancer 18(8): 1039-1060.
- Teguh DN, Levendag PC, Noever I, Van Rooij P, Voet P, et al. (2008) Treatment techniques and site considerations regarding dysphagia-related quality of life in cancer of the oropharynx and nasopharynx. International Journal of Radiation Oncology Biology Physics 72(4): 1119-1127.
- Peponi E, Glanzmann C, Willi B, Huber G, Studer G (2011) Dysphagia in head and neck cancer patients following intensity modulated radiotherapy (IMRT). Radiation oncology 6(1): 1.
- Bjordal K, Ahlner EM, Hammerlid E, Boysen M, Evensen JF, et al. (2001) A prospective study of quality of life in head and neck cancer patients. Part II: Longitudinal data. The Laryngoscope 111(8): 1440-1452.
- Lin A, Kim HM, Terrell JE, Dawson LA, Ship JA, et al. (2003) Quality of life after parotid-sparing IMRT for head-and-neck cancer: a prospective longitudinal study. International Journal of Radiation Oncology* Biology* Physics 57(1): 61-70.
- Tejpal G, Jai Prakash A, Susovan B, Ghosh Laskar S, Murthy V, et al. (2010) IMRT and IGRT in head and neck cancer: Have we delivered what we promised? Indian journal of surgical oncology 1(2): 166-185.
- Langendijk JA, Doornaert P, Rietveld DH, Verdonck de Leeuw IM, Leemans CR, et al. (2009) A predictive model for swallowing dysfunction after curative radiotherapy in head and neck cancer. Radiotherapy and Oncology 90(2): 189-195.
- Lee WT, Akst LM, Adelstein DJ, Saxton JP, Wood BG, et al. (2006) Risk factors for hypopharyngeal/upper esophageal stricture formation after concurrent chemoradiation. Head & neck 28(9): 808-812.
- Caudell JJ, Schaner PE, Meredith RF, Locher JL, Nabell LM, et al. (2009) Factors associated with long-term dysphagia after definitive radiotherapy for locally advanced head-and-neck cancer. International Journal of Radiation Oncology Biology Physics. 73(2): 410-415.
- Eisbruch A, Schwartz M, Rasch C, Vineberg K, Damen E, et al. (2004) Dysphagia and aspiration after chemoradiotherapy for head-and-neck cancer: which anatomic structures are affected and can they be spared by IMRT? International Journal of Radiation Oncology Biology Physics 60(5): 1425-1439.
- Prameela CG, Ravind R, Nair MH, Mon PR, Sheejamol VS, et al. (2014) P0107 Dose to dysphagia aspiration-related structures and its effect on swallowing: Comparison of 3D CRT and IMRT plans. European Journal of Cancer 50: e38-e39.
- Feng FY, Kim HM, Lyden TH, Haxer MJ, Feng M, et al. (2007) Intensity-modulated radiotherapy of head and neck cancer aiming to reduce dysphagia: early dose–effect relationships for the swallowing structures. International Journal of Radiation OncologyBiology Physics 68(5): 1289-1298.
- Suen AW, Chen PY, Chao K, Galerani AP, Martinez AA (2007) Clinical Correlation of Radiation Dose to Anatomic Structures with Treatment Related Chronic Dysphagia in Definitive Chemoradiotherapy for Head and Neck Cancers. International Journal of Radiation Oncology Biology Physics 69(3): S419-S420.
- Li B, Li D, Lau DH, Farwell DG, Luu Q, et al. (2009) Clinical-dosimetric analysis of measures of dysphagia including gastrostomy-tube dependence among head and neck cancer patients treated definitively by intensity-modulated radiotherapy with concurrent chemotherapy. Radiation Oncology 4(1): 52.
- Teguh DN, Levendag PC, Sewnaik A, Hakkesteegt MM, Noever I, et al. (2008) Results of fiberoptic endoscopic evaluation of swallowing vs. radiation dose in the swallowing muscles after radiotherapy of cancer in the oropharynx. Radiotherapy and oncology. 89(1) :57-63.
- Christianen ME, Langendijk JA, Westerlaan HE, van de Water TA, Bijl HP (2011) Delineation of organs at risk involved in swallowing for radiotherapy treatment planning. Radiotherapy and Oncology 101(3): 394-402.
- Van der Molen L, Heemsbergen WD, De Jong R, Van Rossum MA, Smeele LE, et al. (2013) Dysphagia and trismus after concomitant chemo-Intensity-Modulated Radiation Therapy (chemo-IMRT) in advanced head and neck cancer; dose–effect relationships for swallowing and mastication structures. Radiotherapy and oncology 106(3): 364-369.
- Caglar HB, Tishler RB, Othus M, Burke E, Li Y, et al. (2008) Dose to larynx predicts for swallowing complications after intensity-modulated radiotherapy. International Journal of Radiation Oncology Biology Physics 72(4): 1110-1118.
- Levendag PC, Teguh DN, Voet P, Van der Est H, Noever I, et al. (2007) Dysphagia disorders in patients with cancer of the oropharynx are significantly affected by the radiation therapy dose to the superior and middle constrictor muscle: a dose-effect relationship. Radiotherapy and Oncology 85(1): 64-73.
- Doornaert P, Slotman BJ, Rietveld DH, Leemans CR, Langendijk JA (2007) The mean radiation dose in pharyngheal structures is a strong predictor of acute and persistent swallowing dysfunction and quality of life in head and neck radiotherapy. International Journal of Radiation Oncology Biology Physics. 69(3): S55.
- O Meara EA, Machtay M, Moughan J, McIlvaine J, Galvin JM, et al. (2007) Associations between radiation doses to pharyngeal regions and severe late toxicity in head and neck cancer patients treated with concurrent chemoradiotherapy-an RTOG analysis. International Journal of Radiation Oncology Biology Physics 69(3): S54-S55.
- Dornfeld K, Simmons JR, Karnell L, Karnell M, Funk G, et al. (2007) Radiation doses to structures within and adjacent to the larynx are correlated with long-term diet-and speech-related quality of life. International Journal of Radiation Oncology Biology Physics 68(3): 750-757.
- Jensen K, Lambertsen K, Grau C (2007) Late swallowing dysfunction and dysphagia after radiotherapy for pharynx cancer: frequency, intensity and correlation with dose and volume parameters. Radiotherapy and Oncology 85(1): 74-82.
- Sanguineti G, Adapala P, Endres EJ, Brack C, Fiorino C, et al. (2007) Dosimetric predictors of laryngeal edema. International Journal of Radiation Oncology Biology Physics 68(3): 741-749.
- Rancati T, Schwarz M, Allen AM, Feng F, Popovtzer A, et al. (2010) Radiation dose-volume effects in the larynx and pharynx. International Journal of Radiation Oncology Biology Physics 76(3): S64-S69.
- Duprez F, Werbrouck J, Sabbe N, Boterberg T, Madani I, et al. (2013) A predictive model for dysphagia following IMRT for head and neck cancer: introduction of the EMLasso technique. Radiotherapy and Oncology 107(3): 295-299.
- Teguh DN, Levendag PC, Ghidey W, Van Montfort K, Kwa SL (2013) Risk model and nomogram for dysphagia and xerostomia prediction in head and neck cancer patients treated by radiotherapy and/or chemotherapy. Dysphagia 28(3): 388-394.
- Mohindra P, Bhalavat RL, Vyas S, Jamema SV, Upreti R (2008) Role of brachytherapy in limiting the dose to dysphagia/aspiration related structures in patients of oropharyngeal cancers treated with conventional external beam radiotherapy. Brachytherapy 7(2): 156.
- Nguyen NP, Frank C, Moltz CC, Vos P, Smith HJ, et al. (2005) Impact of dysphagia on quality of life after treatment of head-and-neck cancer. International Journal of Radiation Oncology Biology Physics 61(3): 772-778.
- Agarwal J, Dutta D, Palwe V, Gupta T, Laskar SG, et al. (2010) Prospective subjective evaluation of swallowing function and dietary pattern in head and neck cancers treated with concomitant chemo-radiation. Journal of cancer research and therapeutics. 6(1): 15-21.
- Maurer J, Hipp M, Schäfer C, Kölbl O (2011) Dysphagia. Radiation therapy and oncology 187(11): 744-749.
- Grégoire V, Levendag P, Ang KK, Bernier J, Braaksma M, et al. (2003) CT-based delineation of lymph node levels and related CTVs in the node-negative neck: DAHANCA, EORTC, GORTEC, NCIC, RTOG consensus guidelines. Radiotherapy and oncology 69(3): 227-236.
- Perez CA, Halperin CE (2008) Principles and practice of radiation oncology. Philadelphia: Lippincott Williams & Wilkins.
- Kulkarni MR (2013) Head and neck cancer burden in India. Int J Head Neck Surg 4(1): 29-35.
- Argiris A, Li Y, Forastiere A (2004) Prognostic factors and long‐term survivorship in patients with recurrent or metastatic carcinoma of the head and neck. Cancer 101(10): 2222-2229.
- Russi EG, Corvò R, Merlotti A, Alterio D, Franco P, et al. (2012) Swallowing dysfunction in head and neck cancer patients treated by radiotherapy: review and recommendations of the supportive task group of the Italian Association of Radiation Oncology. Cancer treatment reviews. 38(8): 1033-1049.
- Pikus L, Levine MS, Yang YX, Rubesin SE, Katzka DA, et al. (2003) Videofluoroscopic studies of swallowing dysfunction and the relative risk of pneumonia. American Journal of Roentgenology 180(6): 1613-1616.
- Speyer R, Heijnen BJ, Baijens LW, Vrijenhoef FH, Otters EF, et al. (2011) Quality of life in oncological patients with oropharyngeal dysphagia: validity and reliability of the Dutch version of the MD Anderson Dysphagia Inventory and the Deglutition Handicap Index. Dysphagia 26(4): 407-414.
- Bhide SA, Gulliford S, Kazi R, El Hariry I, Newbold K, et al. (2009) Correlation between dose to the pharyngeal constrictors and patient quality of life and late dysphagia following chemo-IMRT for head and neck cancer. Radiotherapy and Oncology 93(3): 539-544.
- Studer G, Peponi E, Kloeck S, Dossenbach T, Huber G, et al. (2010) Surviving hypopharynx–larynx carcinoma in the era of IMRT. International Journal of Radiation Oncology Biology Physics 77(5): 1391-1396.
- Belafsky PC, Mouadeb DA, Rees CJ, Pryor JC, Postma GN, et al. (2008) Validity and reliability of the Eating Assessment Tool (EAT-10). Annals of Otology, Rhinology & Laryngology 117(12) :919-924.
- McHorney CA, Bricker DE, Kramer AE, Rosenbek JC, Robbins J, et al. (2000) The SWAL-QOL outcomes tool for oropharyngeal dysphagia in adults: I. Conceptual foundation and item development. Dysphagia 15(3): 115-121.
- Stoeckli SJ, Huisman TA, Seifert BA, Martin Harris BJ (2003) Interrater reliability of videofluoroscopic swallow evaluation. Dysphagia 18(1): 53-57.
- Gluck I, Feng FY, Lyden T, Haxer M, Worden F, et al. (2010) Evaluating and reporting dysphagia in trials of chemoirradiation for head-and-neck cancer. International Journal of Radiation Oncology Biology Physics 77(3): 727-733.
- Jensen K, Jensen AB, Grau C (2006) The relationship between observer-based toxicity scoring and patient assessed symptom severity after treatment for head and neck cancer. A correlative cross-sectional study of the DAHANCA toxicity scoring system and the EORTC quality of life questionnaires. Radiotherapy and Oncology 78(3): 298-305.






























