An Outcome of Localized HER2 Positive Breast Cancer Treated with Adjuvant Trastuzumab in Clinical Practice: Retrospective Observational Study
Musaab Raji Albayati1*, Haider Saadoon Alhilfi2, Khalid Obiad Almohammadawi2 and Saad Muhsin Althaalibi3
1Department of Medical Oncology, AI-habobi Hospital, Iraq
2Department of Medicine, University of Misan, Iraq
3Department of Internal Medicine, Al-Sadder Teaching Hospital, Iraq
Submission: September 26, 2017; Published: October 12, 2017
*Corresponding author: Musaab Raji Albayati, Department of Medical Oncology, Al-habobi Hospital, Ministry of Health/ Environment, Thi-Qar, Iraq, Tel: +9647733962400; Fax: +9647706707661; Email: ahmeds201258@yahoo.com
How to cite this article: Musaab R A, Haider S A, Khalid O A, Saad M A. An Outcome of Localized HER2 Positive Breast Cancer Treated with Adjuvant Trastuzumab in Clinical Practice: Retrospective Observational Study. Canc Therapy & Oncol Int J. 2017; 7(3): 555712. DOI: 10.19080/CTOIJ.2017.07.555712
Abstract
Background: HER2 positive breast cancer is well known to be associated with poor prognosis. However, the prognosis of these patients has markedly improved since the introduction of trastuzumab for the treatment of HER2 positive breast cancer in the adjuvant and metastatic setting. The aim of this study was to determine the rate and pattern of recurrences in patients with HER2 positive breast cancer treated with adjuvant trastuzumab.
Methods: Retrospective observational study of 118 patients with HER2 positive breast cancer treated with adjuvant trastuzumab for one year in one center between May 2012 and January 2017.
Results: After a median follow-up of 26.7 months, 29 cases of disease recurrence reported yielding a 24.58% recurrence rate. Most relapses occurred in the second year of treatment with a median time for developing relapse of 18 months. About 62.71% of patients with primary tumor ≥ 5 cm, 79.65% of patients with ≥ 4 involved axillary lymph nodes, and 72.41% of hormone receptor negative tumors have experienced the relapse. Large tumor size, number of involved axillary lymph nodes, and hormone receptor status were strongly associated with risk of relapse with significant P values. The most common site of the first recurrence was the brain (34.48%) followed by the liver (24.14%j.
Conclusion: Recurrence rates in our patients with HER2 positive disease treated with trastuzumab seems to be higher than the reported rates in clinical trials. This difference in results may, in part, be explained by the difference in pathological stage between this cohort and the appropriately selected patients in clinical trials. However, a larger study with centrally reviewed receptor status is needed to confirm these results.
Keywords: Breast cancer; Trastuzumab; HER2; PR; ER; Retrospective observational study
Abbreviations: A: Anthracyclines; AT: Anthracyclines and Taxanes; CISH: Chromogenic in Situ Hybridization; ER: Estrogen receptor; HER2: Human Epidermal Growth Factor Receptor 2; IHC: Immuno Histo Chemistry; PR: Progesterone Receptor; RR: Recurrence Rate; T: Tumor size
Introduction
Breast cancer is a heterogeneous disease comprised of several molecular subtypes based on the expression of estrogen receptor (ERj, progesterone receptor (PRj, and human epidermal growth factor 2-neu (HER2j receptor [1]. Over expression of HER2 receptor is well known to be associated with a more aggressive course of breast cancer, although, the molecular subtype of breast cancer may be more important for prognosis than the HER2 status [2]. In the United States, HER2 positive breast cancer constitutes about 15% of all cases of breast cancer [3]. Population based studies have confirmed the unfavorable prognosis of HER2 positive breast cancer in the (Pre-Trastuzumabj era, however, these studies are limited by the small number of patients and short follow up [4,5]. In the control arm of the TEACH trial which included 1260 patients with HER2 positive breast cancer who have received placebo, the disease free survival was 75% at 5 years and 61% at 10 years [6].
Trastuzumab, a humanized monoclonal antibody with specificity for the extracellular domain of HER-2 was approved by the US Food and Drug Administration for the adjuvant treatment of early HER2 positive breast cancer [7]. Four randomized clinical trials of adjuvant trastuzumab in early breast cancer have shown a reduction of the risk of recurrence by about 50% and a 30% reduction in mortality [8-11]. In a Meta-analysis of five of the largest trials of adjuvant trastuzumab, the rates of recurrence were 8.5% in patients who have received adjuvant trastuzumab [12]. In the HERA trial, one of the largest trials of adjuvant trastuzumab, the rates of recurrence were 7.5% at 1 year [8], and, 13% at 2 years [13]. Outside clinical trials settings, two retrospective observational studies in Italy shows that the recurrence rate at 3 years was 11% in patients who have received adjuvant trastuzumab [14,15]. In this study, we aimed to study the recurrence rate and types of recurrences in Iraqi patients with localized HER2 positive breast cancer treated with adjuvant trastuzumab and to compare these results with the published data.
Patients and Methods
Study Design and Setting
A retrospective observational study of patients with HER2-positive breast cancer who have received trastuzumab between May 2012 and June 2017 in Thi-Qar medical oncology department. One hundred and eighteen patients with HER2- positive breast cancer have received adjuvant trastuzumab during the study period.
Participants
Trastuzumab was given every 21 days at a dose of 8 mg/kg for the first dose and 6 mg/kg for subsequent doses for 17 cycles. 79 patients received trastuzumab after completing chemotherapy, and, 39 patients started trastuzumab concomitant with chemotherapy.
Immunohistochemistry Parameters
Estrogen receptor (ER), progesterone receptor (PR), and HER2 over expression were determined by Immunohistochemistry. In 19 patients, HER2 amplification was determined by CISH technique because of equivocal results on IHC. Interpretation of the results of IHC and CISH were performed by different pathologists and in different laboratories, a central review were not done because of the retrospective nature of the study.
Data Collection
Data were collected manually by review of the medical records of each patient. Duration of follow up was calculated from the day of surgical treatment of breast cancer, while the time of recurrence was calculated from the day of start of trastuzumab until diagnosis of relapse.
Results
During the study period, 135 patients diagnosed with HER2 positive breast cancer. Seventeen patients were excluded from the study because of short follow up (less than 6 months) or missing pathological data or refusal of trastuzumab treatment. From the 118 patients included in the study, 104 patients completed one year of trastuzumab, while; only 14 patients discontinued the drug before completing one year (median treatment duration of 7.2 months) because of cardiac toxicity, disease progression and/ or non-compliance. Regarding to the baseline characteristics of the patients. All patients were females. The median age of patients in the study was 47 (Range 25-84) years. About60.17% of patients was less than 50 years of age. Only 5.93% of patients have primary tumor less than 2 cm. About 79.65% of the study population has node positive disease, while ten patients; the nodal status was unknown because of neoadjuvant treatment or lack of axillary staging. All patients have received chemotherapy with 85.59% received anthracyclines (Table 1).

After a median follow up of 26.7 months, 29 cases of disease recurrence were reported suggesting an overall recurrence rate of 24.58%. None of the patients with tumors less than 2 cm have relapsed, while, about 62.07% of relapses occurred in patients with T3 or T4 tumors. Most relapses occurred in patients with node positive disease with about 93.10% of cases in patients with four or more lymph nodes involved. About 72.41% of recurrences reported in patients with hormone receptor negative disease and the remainder were in hormone positive disease. Most relapses occurred in the second year of treatment, and, to a less extent in the third year, with a median time for developing relapse of 18 months. The most common site of first relapse was the brain 34.48% followed by the liver 24.14%, with about 17.24% of patients has isolated loco regional relapses without distant metastasis (Table 2).

By comparing twenty-nine patients who relapsed with eighty-nine disease free non-relapsed patients, the larger tumor size pT2 and more, node positive disease and hormone receptor negative status were associated with increased risk of relapse and reach a significance differences. The age, adjuvant radiotherapy and the schedule of trastuzumab therapy (sequential vs. concomitant) did not seem to influence the risk of recurrence. The effect of the type of chemotherapy on the risk of recurrence was not analyzed because of the small number of patients in the study, and most patients have received anthracyclines.
Discussion
This study is the first study to be done on Iraqi patients who have received adjuvant trastuzumab, the recurrence rate was found to be 24.58% after a median follow up of 26.7 months. This figure seems to be higher than the reported rates of recurrence in patients in the trastuzumab arms in the major adjuvant trastuzumab trials, namely the HERA trial [13], the BCIRG 006 trial [10], and the joint analysis of the B31 and N9831 trials [9]. Moreover, the reported rates of recurrence are even higher than the reported rates in patients in the no trastuzumab arms in the four trials. One of the major limitations of this study is that determination of receptor status was not central, and, performed in daily practice by IHC staining and not by genomic studies. IHC based surrogates use a reduced set of biomarkers and gives an incomplete approximation to genomically defined breast cancer subtypes [16]. A larger study with central review of receptor status, preferably by genomic studies, is needed to support the results of this study.
Although it is difficult to attribute this difference in the results to a single factor, differences in the sample size and the pathological characteristics between patients in clinical trials and this cohort seems to be very important. Included larger numbers of patients with high risk features for recurrence (large tumor size, four or more involved axillary lymph nodes, and, hormone receptor negative tumorsj, and most of the relapses have occurred in patients with more advanced stages of breast cancer. Our results confirms previous reports which have suggested that relapses in trastuzumab treated patients in clinical practice are usually seen in patients with more advanced stages of the disease consistent with the observations of decreased dependence of advanced tumors on the HER2 signaling pathway [17] (Figure 1).
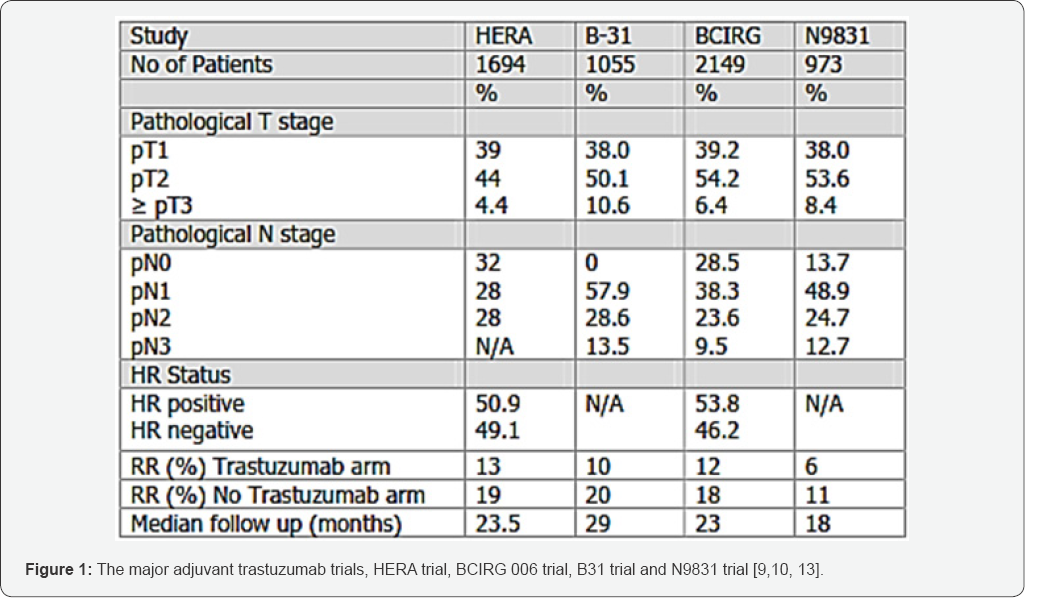
The Outcome of HER2 positive breast cancer not receiving trastuzumab therapy strongly depends on hormone receptor expression. In one study, the mean hazard of recurrence in years 1 to 5 in HER2 positive HR negative compared to HR positive patients is 9 vs. 5%/year (P = 0.002) and the authors concluded that this observation must be taken into account in health care settings with limited access to anti HER2 therapy [6]. In the observational RETROHER study, there was no effect of trastuzumab on recurrence free survival in patients with both ER and PR positive staining in > 50% of tumor cells [15]. In this study, HR negative tumors were associated with statistically significant higher risk of relapse compared with HR positive tumors supporting previous observations.
The most relapses in patients treated with adjuvant trastuzumab have occurred in the second year, which is after end of treatment by months. This is consistent with observations in clinical trials that the maximum activity of trastuzumab is seen in the first year and few months after the end of treatment [13], likely due to antibody wash out. Regarding noticed a high incidence of brain metastases in patients receiving trastuzumab. These findings are in line with the results of two meta-analyses of adjuvant trastuzumab trials which showed that the incidence of CNS metastases as first site of disease recurrence in HER2- positive patients receiving trastuzumab was higher than patients who did not receive trastuzumab [18,19], which may be explained by the inability of the drug to penetrate the blood brain barrier and, hence, preventing brain metastasis [20,21].
Conclusion
Recurrence rates in Iraqi patients with HER2 positive breast cancer treated with trastuzumab it is higher than the reported rates in clinical trials. Lack of effective breast cancer screening programs in Iraq and other factors may account for late diagnosis of breast cancer with increased risk of recurrence which may partly explain the difference in results. Certain subgroups of HER2 positive patients, such as those with small tumors or hormone receptor positive tumors may be at low risk of recurrence and derive small benefit from trastuzumab. This observation has an economic impact given the high cost of one year of adjuvant trastuzumab treatment. Finally, adjuvant trastuzumab treatment increase the risk of brain metastasis and this needs to be addressed in future drug developments.
Acknowledgement
We would like to thank Dr. Rasha Khalil Al-Saad, Assistant Lecturer at the Faculty of Medicine, Misan University and Dr Ahmed Salih Al-shewered from the Misan Radiation Oncology Centre for their help.
Conflict of Interest
There is no any financial interest or any conflict of interest exists.
References
- Santa-Maria CA, Gradishar WJ (2015) Changing Treatment Paradigms in Metastatic Breast Cancer: Lessons Learned. JAMA Oncol 1(4): 528534.
- Prat A, Carey LA, Adamo B, Vidal M, Tabernero J (2014j Molecular features and survival outcomes of the intrinsic subtypes within HER2- positive breast cancer. J Natl Cancer Inst 106(8j.
- Howlader N, Altekruse SF, Li CI, Vivien W Chen, Christina A Clarke (2014) US incidence of breast cancer subtypes defined by joint hormone receptor and HER2 status. J Natl Cancer Inst 106(5j.
- Jian Xue, Zefei Jiang, Fan Qi, ShuanghongLv, Shaohua Zhang, Tao Wang, et al. (2014j Risk of Trastuzumab-Related Cardiotoxicity in Early Breast Cancer Patients: A Prospective Observational Study. J Breast Cancer 17(4j: 363-369.
- Palmieri C, Shah D, Krell J, Gojis O, Hogben K, et al. (2011j Management and outcome of HER2-positive early breast cancer treated with or without trastuzumab in the adjuvant trastuzumab era. Clin Breast Cancer 11(2j: 93-102.
- Lee JS, Son B, Ahn SH, (2012j The Predictive Value of Serum HER2/ neu for Response to Anthracycline Based and Trastuzumab-Based Neoadjuvant Chemotherapy. J Breast Cancer 15(2j: 189-196.
- National Cancer Institute. FDA approval for trastuzumab. National Institutes of Health, USA.
- Piccart-Gebhart MJ, Procter M, Leyland-Jones B, Aron Goldhirsch, Michael Untch, et al. (2005j Trastuzumab after adjuvant chemotherapy in HER2-positive breast cancer. N Engl J Med 353: 1659-1672.
- Romond E, Perez E, Bryant J, Vera J Suman, Charles E Geyer, et al. (2005j Trastuzumab plus adjuvant chemotherapy for operable HER2- positive breast cancer. N Engl J Med 353: 1673-1684.
- Jitawatanarat P, O'Connor TL, Kossoff EB, Levine EG, Chittawatanarat K, et al. (2014j Safety and Tolerability of Docetaxel, Cyclophosphamide, and Trastuzumab Compared to Standard Trastuzumab-Based Chemotherapy Regimens for Early-Stage Human Epidermal Growth Factor Receptor 2-Positive Breast Cancer. J Breast Cancer 17(4j: 356362.
- Lee YR, Huh SJ, Lee DH, Yoon HH, Seol Y, et al. (2011j Phase II Study of Vinorelbine Plus Trastuzumab in HER2 Overexpressing Metastatic Breast Cancer Pretreated with Anthracyclines and Taxanes. J Breast Cancer 14(2j: 140-146.
- Viani GA, Afonso SL, Stefano EJ (2007j Adjuvant trastuzumab in the treatment of her-2-positive early breast cancer: a meta-analysis of published randomized trials. BMC Cancer 8(7j: 153.
- Smith I, Procter M, Gelber RD, Guillaume S, Feyereislova A (2007j 2-year follow-up of trastuzumab after adjuvant chemotherapy in HER2-positive breast cancer: a randomized controlled trial. Lancet 369(9555j: 29-36.
- Campiglio M, Bufalino R, Sasso M, Ferri E, Casalini P, et al. (2013j Effect of adjuvant trastuzumab treatment in conventional clinical setting: an observational retrospective multicenter Italian study. Breast Cancer Res Treat 141(1}: 101-110.
- Vici P, Pizzuti L, Natoli C, Luca Moscetti, Lucia Mentuccia, et al. (2014j Outcomes of HER2-positive early breast cancer patients in the pre-trastuzumab and trastuzumab eras: a real-world multicenter observational analysis. The RETROHER study. Breast Cancer Res Treat 147(3j: 599-607.
- Goldhirsch A, Wood WC, Coates AS, Gelber RD, Thurlimann B, et al. (2011j Strategies for subtypes--dealing with the diversity of breast cancer: highlights of the St. Gallen International Expert Consensus on the Primary Therapy of Early Breast Cancer 2011. Ann Oncol 22(8j: 1736-1747.
- Sharma SV, Settleman J (2007j Oncogene addiction: setting the stage for molecularly targeted cancer therapy. Genes Dev 21(24j: 32143231.
- Olson EM, Abdel-Rasoul M, Maly J, Wu CS, Lin NU (2013j Incidence and risk of central nervous system metastases as site of first recurrence in patients with HER2-positive breast cancer treated with adjuvant trastuzumab. Ann Oncol 24(6j: 1526-1533.
- Bria E, Cuppone F, Fornier M, Nistico C, Carlini P, et al. (2008j Cardiotoxicity and incidence of brain metastases after adjuvant trastuzumab for early breast cancer: the dark side of the moon? A meta-analysis of the randomized trials. Breast Cancer Res Treat 109(2j: 231-239.
- Pestalozzi B, Brignoli S (2000j Traztuzumab in CSF. J Clin Oncol 18(11j: 2350-2351.
- Azim HA, Azim HA (2012j Systemic treatment of brain metastases in HER2-positive breast cancer: current status and future directions. Future Oncol 8(2j: 135-144.






























