Removal of Lead (II) Ion from Aqueous Solutions Using Black Walnut (Juglan Nigra) Seed Husk
OS Lawal1*, OS Amuda2*, OO Rabiu3 and KO Adebowale3
1Department of Chemical Sciences, Olabisi Onabanjo University, Nigeria
2Department of Chemistry, Ladoke Akintola University of Technology, Nigeria
3Department of Chemistry, University of Ibadan, Nigeria
Submission: April 18, 2017; Published: September 15, 2017
*Corresponding author: OS Lawal, Department of Chemical Sciences Olabisi Onabanjo University, P.M.B 2002, ΔG0-Iwoye, Ogun State Nigeria, Tel: + Email: laidelawal2@yahoo.com
OS Amuda, Department of Chemistry, Ladoke Akintola University of Technology, Ogbomoso, Oyo State, Nigeria, Tel: +; Email: os@lautch.edu.ng
How to cite this article: OS Lawal, OS Amuda, OO Rabiu, KO Adebowale. Removal of Lead (II) Ion from Aqueous Solutions Using Black Walnut (Juglan Nigra) Seed Husk. Curr Trends Biomedical Eng & Biosci. 2017; 9(1): 555755. DOI: 10.19080/CTBEB.2017.09.555755
Abstract
The biosorption characteristics of Pb (II) ions from aqueous solution using black walnut (Juglan nigra) seed husks (WSH) biomass were investigated using batch techniques. The effects of pH, contact time, initial metal ion concentration, and temperature were studied. The Langmuir, the Freundlich and the Temkin isotherms were used to analyze the equilibrium data. It was found that adsorption of Pb (II) ions onto WSH, was best described by Freundlich adsorption model. Biosorption kinetics data were fitted using pseudo-first-order and pseudo-second-order models, and it was observed that the kinetics data fitted better into the pseudo-second order model. Thermodynamic parameters such as standard Gibbs free energy (ΔG0), standard enthalpy (ΔH0) and standard entropy (ΔS0) were evaluated. The result showed that biosorption of lead (II) ions onto WSH was spontaneous and endothermic in nature. The FTIR study showed that the following functional groups: O-H, C=O, C-O, C-H and N-H were involved in binding Pb(II) ions to the biomass.
Keywords: Walnut biomass; Adsorption; Pb(II) Removal; Pollution
Introduction
Environmental pollution by metal ions in industrial waste waters is one of the most important causes of contamination to humans, as they can accumulate throughout the food chain, and environment, due to their great persistence and high toxicity. The removal of heavy metal from contaminated waste waters is generally accomplished by conventional methods such as chemical precipitation, membrane separations, evaporation, resin ion-exchange, electro winning and reverse osmosis. However, these conventional methods of removing heavy metals from effluents have major disadvantages such as high energy requirements, incomplete metal removal and large quantity of toxic waste sludge. Moreover, these technologies are most suitable in situations where the concentrations of heavy metal ions are relatively high [1]. To compare with conventional processes, new methods must be economically viable as well as successful in the removal of environmental contaminant [2].
Biosorbents are alternatives to conventional methods. The term 'biosorbent’ includes the usage of dead biomass (agricultural by-products such as fibre, peat and wool) as well as living plants and bacteria as sorbents [2]. Processing of agricultural products often yield by-products which are produced in an excess of 320,000,000kg/year [3] and are considered to be of low value. Agricultural by-products usually are composed of lignin and cellulose as major constituents and may also include other polar functional groups of lignin, which includes alcohols, aldehydes, ketones, carboxylic, phenolic and ether groups. These groups have the ability to bind heavy metals by the replacement of hydrogen ions for metal ions in aqueous solution [4].WSH is a by-product of black walnut production. Walnuts (genus Juglans) are plants in the family Juglandaceae. They are deciduous trees with wide distribution across the globe. Walnut is grown mainly for its edible nuts, oil and timber production. Medicinal uses have also been reported for the nuts of walnut [5]. These uses of walnut have widely enhanced its cultivation particularly in the tropic zone area. However, the hard husk of walnut is often seen as waste material after the nut is extracted. In areas where walnut oil is used, the husk often constitute environmental nuisance due to large quantity of walnut fruits needed for the walnut oil production. The objective of this study therefore was to investigate the possible use of a waste material, WSH as an alternative biosorbent material for removal of Pb (II) ions from aqueous solutions. The dynamic behavior of the adsorption was investigated on the effect of the pH of the solution, initial metal ion concentration, contact time, and temperature.
Material and Methods
Preparation of biomass
Walnut fruits were obtained in large quantity from a market in Ibadan, Nigeria. The fruits were soaked and rinsed with deionized water to remove any physical and weakly bound materials on the outer-shell. The fruits were cooked to enhance easy breaking and removal of the fruits to collect enough WSH to be used as biosorbent for the research studies. The WSH were then oven-dry, at 60 °C for 24h. The dried WSH were ground and then sieved using a 150 size mesh screen. The sieved WSH was then packed in polythene bags (2g each) until when required for the experiments.
Fourier-transform infrared spectroscopy of WSH
The IR spectra of the free and metal-adsorbed biosorbents were run as KBr pellets on impact 410 Nicolet FTIR spectrometer in the frequency range 4000-500cm-1. Finely ground biomass (10mg) was mixed with KBr (500mg) and the two were ground together to prepare a translucent sample disk.
Preparation of Pb (II) solutions
Pb(NO3)2, NaOH and HNO3 used are analytical grades purchased from Merck Schuchardt, Germany. Stock Pb (II) solution (1000 mg/L) was prepared by dissolving Pb(NO3)2 (1.6g) in deionized water (100mL) and diluting quantitatively to 1000mL using deionized water. Pb(II) solutions of different concentrations were prepared by adequate dilution of the stock solution with deionized water. The pH of the solution was adjusted with HNO3 and NaOH solutions for pH studies. All the glassware used were overnight immersed in 10% (v/v) nitric acid and rinsed several times with deionized water before drying.
Effect of pH on adsorption of Pb (II) ions
The influence of pH on the biosorption process was investigated by adjusting the Pb(II) solution to a desired value within the range 1.0 to 8.0 with HNO3 or NaOH. The metal ion solution (25mL; 100mg/L) was then contacted with a fixed quantity of WSH (0.5g, dry weight) in a boiling tube. The mixture was left in a water bath to maintain the constant temperature of 28 °C for 24h with constant shaking at 75 rpm. The biosorbent was then removed from the mixture by filtration through a Whatmann 32 filter paper. The concentration of the metal ions remaining in solution, were then determined using Atomic Absorption Spectrophotometer (Buck 200A Analytical Model) at 283.2nm. The experiments were performed in triplicates. The percentage removal of Pb(II) ions by the WSH was calculated from the following equation:
Biosorption(%)=(C_o-C_e)/C_o X 100 (1)
Where, Co and Ce are initial and equilibrium metal ion concentrations (mg/L) in aqueous solution.
Effect of initial metal ion concentration on adsorption of Pb (II) ions
Batch adsorption studies were carried out using initial metal ion concentrations of 25, 50, 100, 150, 200 and 400 mg/L in order to test for the effect of initial metal ion concentration on adsorption of Pb (II) ions. A fixed quantity of WSH (0.5g) was packed into boiling tubes, and the desired metal ion solution (25mL) was added. The mixture was left in a water bath while maintaining constant temperature of 28 °C for 1h with constant shaking at 75 rpm. The biosorbent was then removed from the mixture by filtering through a Whatmann 32 filter paper. The concentration of the metal ion remaining in solution was then determined using AAS.
The amount of metal ions adsorbed by the biosorbent was then calculated as follows:
q_e (mg/g)=(C_o-C_e )V/1000W (2)
Where qe is the amount of metal ion adsorbed on the biosorbent in (mg/g)
V is the volume of metal ion solution used (mL) and
W is the weight of the biosorbent used (g)
The data obtained were then used to test the fitness of the sorption process to Langmuir, Freundlich and Temkin isotherms.
Effect of contact time on adsorption of Pb (II) ions
The adsorption of Pb (II) ions on to WSH was studied at various time intervals. A constant concentration of 100mg/L was used. A fixed quantity of the biosorbent (0.5g) was weighed into various boiling tubes, and the metal ion solution (25mL) was added into each boiling tube and its content. The biosorbent was then filtered at different time intervals of 10, 20, 30, 45, 60, 74, 90, 120, 150 and 180min. The filtrates were then analyzed with AAS, to determine the amount of metal ion remaining in solution. All experiments were performed in triplicates. The data obtained were then used to determine the values of k1 and k2 in the Langergren and Ho's pseudo first order and pseudo second order equations respectively.
Thermodynamic studies
For thermodynamics study, experiments were carried out at 35, 40, 50, 60 and 700Cusing initial metal ion concentration of 100mg/L. Each experiment was carried out for 1 h with triplicate determinations. The filtrates were decanted and analyzed with AAS to obtain the concentration of the metal ions remaining in solution.
Results and Discussion
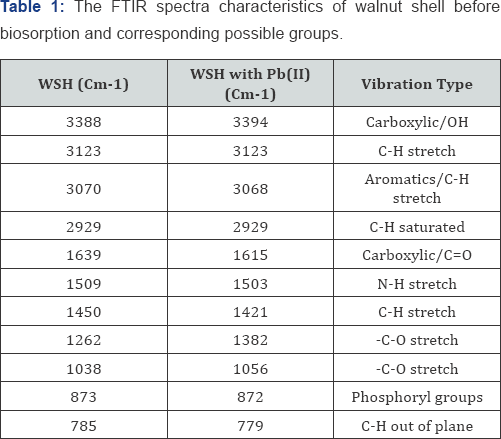
The details of the assignment of bands due to functional groups present on the surface of the biomass before and after biosorption are given in Table 1. Different adsorption mechanisms including complexation, ion exchange, and electrostatic attraction may be involved in the biosorption process and these processes depend on the functional groups on the surface of the biomass. In the present study, the broad peak around 3388cm-1 was assigned to the presence of -OH or -NH group on WSH surface, 1038cm-1 shows C-O groups, 1639cm-1 shows C=O, the peak at 2911cm-1 is due to C-H vibration of CH3 groups, 3159cm-1 and 3029cm-1 show the vibration of the aromatics. O-H, C=O, C-O, C-H and N-H were identified to be involved in Pb (II) ions binding to the WSH surface due to the shift in the spectra band after adsorption.
The effect of pH on the adsorption of Pb (II) ions
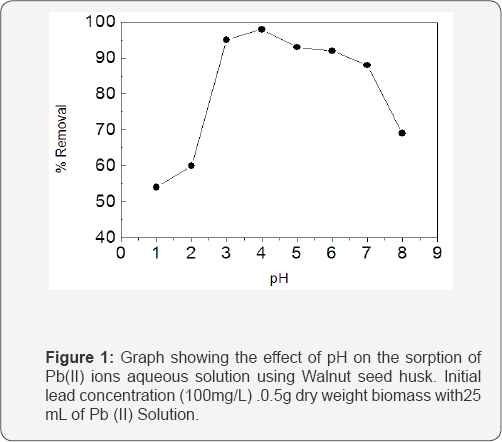
One of the most important factors affecting biosorption of metal ions is the acidity of the solution. The acidity of the medium affects the competition between hydrogen ions and metal ions for the active sites on the biosorbent surface [6]. The solution pH affects the cell wall metal binding sites and the metal ion chemistry in water. The effect of initial pH on biosorption is shown in Figure 1. In the current study, pH was varied from 1-8. The minimal adsorption capacity (54%) was at pH1 and the trend increased progressively until it reached 98% at pH 4. It was observed that the adsorption capacity decreased progressively above pH4. As revealed by the FTIR study, the WSH surface contains acidic functional groups. It is reasonable that at lower pH (pH <V2.0), the dissociation of the acidic functional groups like carboxylic acids on the surface of the adsorbent did not take place. However, when the pH increased, the acidic functional groups were deprotonated. The deprotonation of the acidic functional groups increased the negative charges on the adsorbent. This means that below pH3 the overall surface charge on WSH was less negative when compared with surface charge on WSH at higher pH. The increase in the number of negative charges on the WSH surface facilitated the adsorption of the metals ions but reduced on further increase of pH above 4 due to the hydrolysis of lead ions. These observations are consistent with previous studies on the biosorption of Pb(II) ions untoground and NaOH-treated cereals Low et al. [7] moringa oleifera bark and coir [7-10].
Effect of initial metal ion concentration on the adsorption of Pb (II) ions
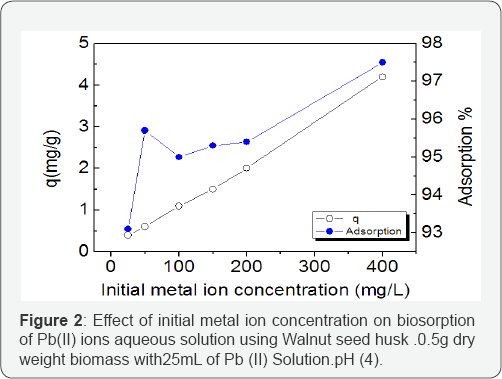
The initial metal ion concentration plays an important role in the biosorption process. The biosorption characteristics of WSH, in the removal of Pb(II) ions, increased with increasing metal ion concentration (Figure 2). This implies that the removal of Pb (II) ions from its solution by WSH at higher concentration will most likely be favorable. Similar observations were reported by Kilic et al. & Denga et al. [11,12]. It is reasonable that the increase in initial metal ion concentrations enables the mass transfer of Pb(II) ions between the aqueous and solid phase and this facilitated a higher probability of collision between metal ions and sorbents. An improved probability of collision between metal ions and sorbents led to higher metal adsorption.
Effect of contact timeon the adsorption of Pb (II) ions
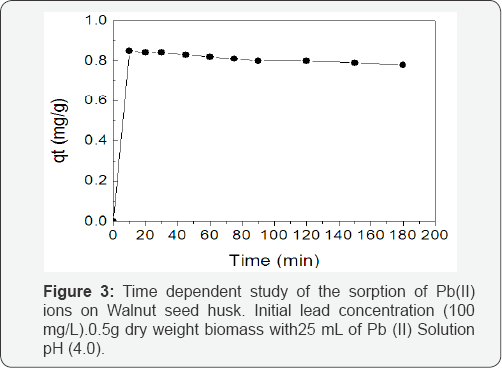
The time variation Figure 3 for the adsorption Pb (II) ions unto WSH showed that more than 80% of Pb(II) ions were adsorbed within the first 10min, but sorption continued up to 3h. It was observed that the sorption process reached equilibrium after 60min when 96% of Pb (II) ions have been absorbed by the WSH biomass. The observed fast biosorption kinetics is consistent with the biosorption of metal involving non-energy mediated reactions, where metal removal from solutions is due purely to physico-chemical interactions between the biomass and the metal solution. Similar observations have been reported for the biosorption of lead from solution using rice husks [13,14].
Effect of biosorbent dosage on the adsorption of Pb (II) ions
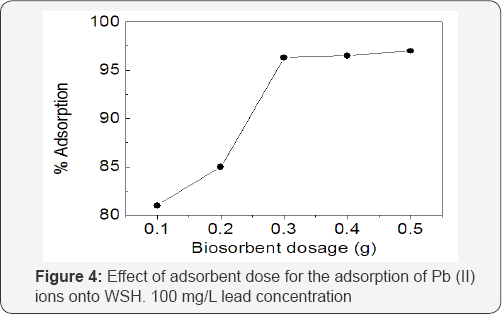
The effect of biosorbent dosage on the adsorption Pb (II) ions is depicted in Figure 4. The trend revealed a progressive increase in the amount of lead adsorbed as adsorbent dosage increased from 0.1 to 0.5g. The percentage of lead removed increased from 81 to 96 %. It is reasonable that increasing the biosorbent dose increased the surface area, thus providing increase in the available active sites for the adsorption. Similar trend was also observed for lead removal using Moringa oleiferal bark [8].
Adsorption isotherms
Langmuir, Freundlich and Temkin adsorption isotherms were applied to describe the relationship between the adsorbed amount of lead and its equilibrium concentration in solution.
Langmuir model: Langmuir model suggests monolayer sorption on a homogeneous surface without interaction between the sorbed molecules. It relates the coverage of molecules on a solid surface to concentration of a medium above the solid surface at fixed temperature. It is represented in the lineralized form as shown below [15].
1/q_e =1/(q_m K_L ) [1/C_e ]+1/q_m (3)
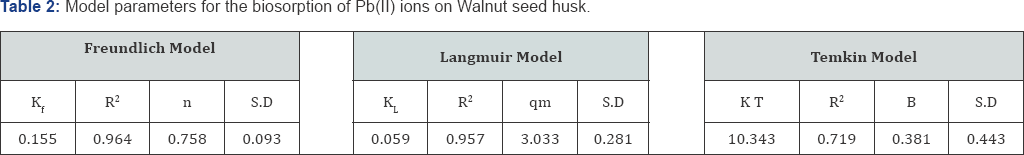
where, qe is the heavy metal adsorbed on the biosorbent (mg/g dry weight), Ce is the final concentration of metal (mg/L) in the solution, qm is the maximum possible amount of metallic ion adsorbed per unit weight of adsorbent; KL is the equilibrium constant related to the affinity of the binding sites for the metals. The adsorption constants (qm and KL) were obtained by plotting 1/qe against 1/Ce Figure 5 giving a straight line with a slope of (1/ KLqm) and an intercept of (1/ qm).

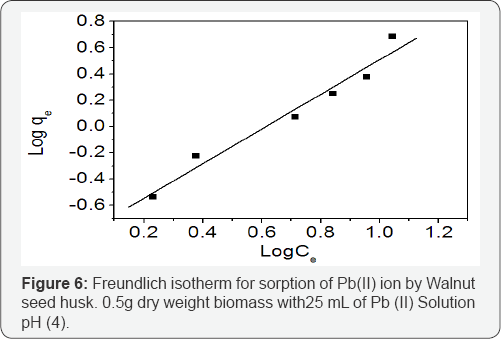
Freundlich models: The Freundlich isotherm model assumes that the uptake ofmetal ions occurs on a heterogeneous surface by multilayer adsorption and that the amount of adsorbate adsorbed increases infinitely with an increase in concentration [16]. This model isempirical in nature and it further assumes that the stronger bindingsites are occupied first and that the binding strength decreases with increasing degree of site occupation (Khambhaty et al, 2009) [17]. It is represented in the lineralized form as shown below;
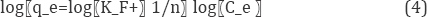
The adsorption constants (1/n and kf) were obtained, by plotting log qe against log Ce Figure 6, from the slope and the intercept respectively.
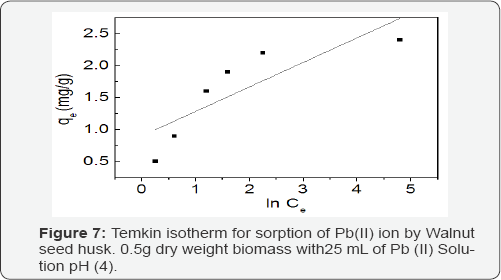
Temkin model: Temkin isotherm can be expressed in the linearized form as:
qe = B lnKT+ B lnCe (5)
A plot of qe versus lnCe Figure 7 enables the determination of the isotherm constants B and KT from the slope and the intercept, respectively. Temkin constants are given in Table 2. KT is the equilibrium binding constant corresponding to the maximum binding energy and constant B is related to the heat of adsorption [18].
The equilibrium data, in Table 2, shows the values of regression coefficients R2 between the sorbate and sorbent systems for Langmuir, Freundlich and Temkin models. The best description of the adsorption process of Pb (II) ions was observed in Freundlich model based on the regression coefficients R2=0.964 compared with 0.957 and 0.719 for Langmuir and Temkin isotherms respectively. Possibly the adsorption process began with the initial monolayer coverage on the outer surface of the adsorbent, after which the sorption progressed to the multilayer dimensions.
Kinetics of adsorption
One of the major characteristics to define the efficiency of sorption is its kinetics. Different biosorbents conform to different models. Here in our investigations, the Lagergren first-order and pseudo-second-order models were used to test the adsorption kinetics data with a view to investigating the mechanism of biosorption. The Lagergren rate equation is the most widely used model for the sorption of a solute from a liquid solution and the first-order, rate expression of Lagergren is given as [19].
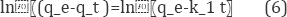
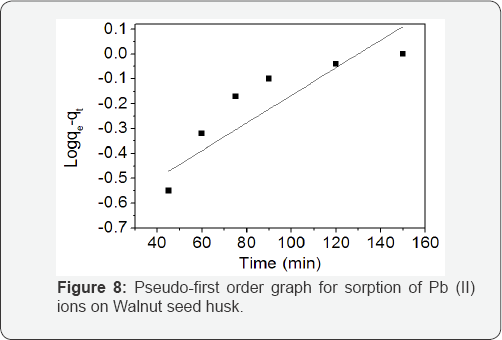
where qe and qt (mg/g) are the amounts of lead adsorbed on the WSH at equilibrium and time t, and K1 is the rate constant of first-order kinetic. The slope and intercept of the plot of log (qe - qt) versus t for WSH was used to determine the values of qe and K1 (Figure 8).
The pseudo-second-order kinetic model McKay & Ho [20] in its integrated and linearized form has been used and is given as:

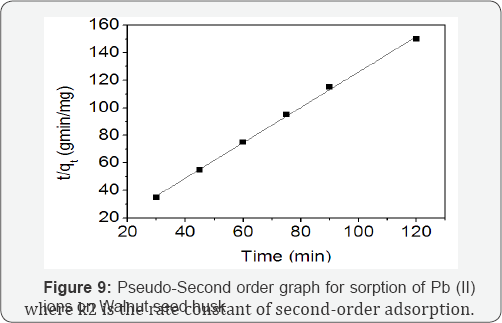
The plot t/qt versus t gave qe and K2 Figure 9 which were determined from slope and intercept of the plot, respectively.
The pseudo-first-order kinetic model provide a poor description of the data for the biosorption of Pb(II) ions, this is evident from their correlation coefficient (R2) values in (Table 3). In previous reports where the pseudo-first-order kinetic model disagrees with experimental values, this discrepancy was attributed to a time lag which probably caused the presence of the boundary layer or external resistance controlling at the beginning of the sorption process [21]. The pseudo-second- order kinetic model is more likely to predict the kinetic behaviour of the biosorption with chemical sorption being Table 3: Comparison of Pseudo-first and second-order biosorption of the rate-controlling step [19,21]. The linear plot of t/qt versus t for the pseudo-second-order model for the biosorption of Pb(II) ions onto WSH is presented in Figure 9. It is clear from Table 3 that the R2 value for the pseudo-second-order model is high. This result suggests that the biosorption of Pb(II) ions onto WSH follows the pseudo-second-order kinetic model well. Several authors that studied sorption of divalent metals on heterogeneous sorbents had reported that most metal sorption kinetic follow pseudo-second-order mechanism [22,23].

Thermodynamics study
The result obtained from varying temperature within the range 308-343 K showed progressive increase in metal uptake as temperature increased (Figure 10). Similar increase in metal uptake with increasing temperature had been reported earlier [11,24,25]. The increase in adsorption with increasing temperature indicated endothermic nature of the adsorption process which may be attributed to either the increase in the number of active surface sites available for sorption on the adsorbent or due to the decrease in the boundary layer thickness surrounding the sorbent, so that the mass transfer resistanc[26].
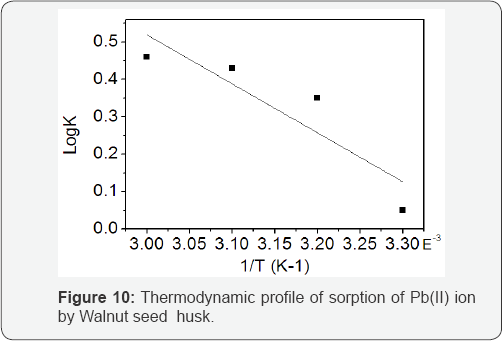
In order to describe the thermodynamic behaviour of the biosorption of Pb(II) ions onto WSH, thermodynamics parameters including the change in free energy (ΔG0), enthalpy (ΔH0) and entropy (ΔS0) were calculated from the following equations:
ΔG0 = -RT ln K (8)
where, T is temperature in Kelvin (K), R is ideal gas constant having value as 8.314 Jmol-1K-1 and K is thermodynamic equilibrium constant. The relationship between Gibbs free energy change, entropy change (ΔS0) and enthalpy change
(ΔH0) can be expressed as:

From the two equations above, we obtain:
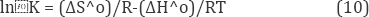
According to this equation, the ΔH0 and ΔS0 can be calculated from the slope and intercept of plot of ln K versus I/T Figure 10 respectively [27].
The thermodynamic equilibrium constant was determined as follows:
K = Ca/Ce (11)
where, Ca is amount (mg) of the adsorbate adsorbed per litre, and Ce is the equilibrium concentration of solution in mg/L. The negative ΔG0 values indicate thermodynamic feasibility and spontaneity of the biosorption process. The increase in the magnitude of ΔG0 values for Pb(II) biosorption with increase in temperature shows an increase in its feasibility. The slight decrease in the value of AG0 in the biosorption of Pb (II) ions by WSH at 343K may be due to gradual damage of active binding sites in the biomass [28] or the tendency to desorb metal ions from the interface to the solution [29]. The ΔH0 value for Pb(II) ions was calculated to be (25.35 kJmol-1), indicating that the binding of Pb(II) ions onto WSH was endothermic. This enthalpy change agrees with previous works reported in the literature [25,30,31]. The magnitude of ΔH0 value gives an indication of the type of adsorption, which can either be physical or chemical. The heat of adsorption, ranging from 2.1-20.9 kJmol-1, is said to be physical adsorption, and the activation energy for chemical adsorption is of the same magnitude as the heat of chemical reactions, 20.9-418.4kJmol-1 [32]. ΔH0 value obtained in the biosorption experiment indicates that the binding of Pb(II) ions to the WSH is by chemical adsorption. Also, the ΔS0 was observed to be positive (86.1kJmol-1K-1), indicating the increasing randomness at the solid/liquid interface during biosorption. A comparison of the maximum adsorption capacities for lead adsorption to different adsorbents is presented in (Table 4).
Conclusion
The study concerns the use of walnut seed husk for the removal of heavy metal, Pb from the environment. The results obtained indicate that the pH, contact time, temperature and initial concentrations, affect the uptake of the metal ions by the biosorbent. It was observed that the adsorption equilibrium data fitted best into the Freundlich model compared with the Langmuir and Temkin isotherms. The pseudo-second-order kinetic model agrees with the biosorption of Pb(II) onto WSH. The negative value of AG0 indicate the spontaneity of the biosorption process and the positive values of ΔH0 and ΔS0also showed the endothermic nature and irreversibility of Pb(II) ion biosorption. Results obtained from this study showed that WSH could be useful at removing Pb(II) ions from the aqueous solution.
The justificationsfor our reports are:
i. WSH is waste material that constitutes environmental nuisance in areas where the seed is processed for its oil, hence finding a technical application for WSH becomes reasonable.
ii. Since it is a waste material, then it is very cheap.
iii. Like most other biomaterials, it is eco-friendly and nontoxic material for the removal of lead from aqueous solution.
iv. Unlike other biomaterials such rice husk which can be processed into animal feed or sawdust which can be used as building material after processing, WSH has no other competing utilization to the best of our knowledge.
Therefore the strength of this study is that a waste material, WSH can be used to address an important environmental problem.
References
- Kaewsarn P (2002) Biosorption of copper (II) from aqueous solutions by pre-treated biomass of marine algae Padina sp. Chemosphere 47(10): 1081-1085.
- Sonia AD, Tobin MT (1999) Uptake of chromium cations and anions by milled peat Resour Conserv Recycl 27(1-2): 151-156.
- Wing RE (1996) Starch Citrate: preparation and Ion-Exchange properties. Starch-Starke 48(7-8): 275-279.
- Ofomaja AE, Yuh-Shan Ho (2007) Effect of pH on cadmium biosorption by coconut copra meal. J Hazard Mater 139(2): 356-362.
- Chauhan N, Wang KC, Wegiel J, Malik MN (2004) Walnut Extract Inhibits the Fibrillization of Amyloid Beta-Protein, and also Defibrillizes its Preformed Fibrils. Curr Alzheimer Res 1(3): 183-188.
- Lodeiro P, Barriada JL, Herrero R, Sastre de Vicente ME (2006) The marine macroalga Cystoseira baccata as biosorbent for cadmium(II) and lead(II) removal: kinetic and equilibrium studies. Environ Poll 142(2): 264-273.
- Low KS, Lee CK, Liew SC (2000) Sorption of cadmium and lead from aqueous solutions by spent grain. Process Biochem 36(1-2): 59-64.
- Reddy DHK, Seshaiah K, Reddy AVR, Rao MM, Wang MC (2010) Biosorption of Pb2+ from aqueous solutions by Moringa Oleifera bark:Equilibrium and kinetic studies. J Hazard Mater 174(1-3): 831-838.
- Conrad K, Hans C, Hansen B (2007) Sorption of zinc and lead on coir, Bioresour Technol 98(1): 89-97.
- Quek SY, Al Duri B, Wase DAJ, Forster CF (1998) Coir as biosorbent of copper and lead. Process Safety Environ Prot 76(1): 50-54.
- Kilic M, Keskin ME, Mazlum S, Mazlum N (2008) Effect of conditioning for Pb(II) and Hg(II) biosorption on waste activated sludge. Chem Eng Proc 47(1): 31-40.
- Denga L, Sua Y, Sua H, Wang X, Zhua X (2007) Sorption and desorption of lead (II) from wastewater by green algae Cladophora fascicularis. J Hazard Mater 143(1-2): 220-225.
- Khalid N, Ahmad S, Kiani SN, Ahmed J (1998) Removal of lead from aqueous solutions using rice husk. Sep Sci Technol 33(15): 2349-2362.
- King P, Rakesh N, Beenalahari S, Prasanna Kumar Y, Prasad VSRK (2007) Removal of lead from aqueous solution using Syzygium cumini L.: Equilibrium and kinetic studies. J Hazard Mater 142(1-2): 340-347.
- Langmuir I (1918) The adsorption of gases on plane surfaces of glass, mica and platinum. J Am Chem Soc 40(9): 1361-1403.
- Freundlich H (1907) Ueber die Adsorption in Leousun-gen Z. Physic Chem 57: 385-470.
- Khambhaty Y, Mody K, Basha S, Jha B (2009) Kinetics, equilibrium and thermodynamic studies on biosorption of hexavalent chromium by dead fungal biomass of marine Aspergillus niger. Chem Eng J 145(3): 489-495.
- Seker A, Shahwan T, Eroglu AE, Yilmaz S, Demirel Z et al. (2008) Equilibrium, thermodynamic and kinetic studies for the biosorption of aqueous lead(II), cadmium(II) and nickel(II) ions on Spirulinaplatensis. J Hazard Mater 154(1-3): 973-980.
- Ho YS, McKay G (1999) The sorption of lead(II) ions on peat response to comment. Wat Res 33(2): 578-584.
- McKay G, Ho YS (1999) Pseudo-second-order model for sorption processes. Process Biochem 34(5): 451-465.
- Unlu N, Ersoz M (2006) Adsorption characteristics of heavy metal ions onto a low cost biopolymeric sorbent from aqueous solution. J Hazard Mater 136(2): 272-280.
- Reddad Z, Gerente C, Andres Y, LeCloirec P (2002) Adsorption of several metal ions onto a low-cost biosorbent. Kinetic and equilibrium studies. Environ Sci Technol 36(9): 2067-2073.
- Ho YS (2004) Comment on cadmium removal from aqueous solutions by chitin, kinetic and equilibrium studies. Water Res 38(12): 29622964.
- Bahadir T, Bakan G, Altas L, Buyukgungor H (2007) The investigation of lead removal by biosorption: An application at storage battery industry wastewaters. Enzy Microbiol Technol 41(1-2): 98-102.
- Senthilkumar R, Vijayaraghavan K, Thilakavathi M, Iyer PVR, Velan M (2007) Application of seaweeds for the removal of lead from aqueous solution. Biochem Eng J 33(3): 211-216.
- Meena AK, Mishra GK, Rai PK, Rajgopal C, Nagar PN (2005) Removal of heavy metal ions from aqueous solutions using carbon aerogel as an adsorbent. J Hazard Mater 122(1-2): 161-170.
- Sarin V, Pant KK (2006) Removal of Chromium from industrial waste by using eucalyptus bark. Bioresour Technol 97(1): 15-20.
- Ozer A, Ozer D (2003) Comparative study of the biosorption of Pb(II), Ni(II) and Cr(VI) ions onto S. cerevisiae: determination of biosorption heats J Hazard Mater 100(1-3): 219-229.
- Saltali K, Sari A, Aydin M (2007) Removal of ammonium ion 31. Tewari N, Vasudevan P, Guha BK, (2005) Study on biosorption of Cr(VI) from aqueous solution by natural Turkish (Yildizeli) zeolite for by Mucor hiemalis. Biochem Eng J 23(2): 185-192.
- Ho YS, Chiang TH, Hsueh YM (2005) Removal of basic dye from pp. 566-575.
- Tewari N, Vasudevan P, Guha BK, (2005) Study on biosorption of Cr(VI) by Mucor hiemalis. Biochem Eng J 23(2): 185-192.
- Treybal RE (1980) Treybal RE (1980) Mass-Transfer Operations, McGraw Hill, Singapore,pp. 185-192.






























