Circulating Microvesicles in Regenerative Angiogenesis
DroŻdŻ A and StępieńE*
Department of Medical Physics, Jagiellonian University, Poland
Submission: April 04, 2017; Published: April 10, 2017
*Corresponding author: Ewa Stępień, Zakład Fizyki Medycznej, Instytut Fizyki im. Mariana Smoluchowskiego, Uniwersytet Jagielloński, ul. S. Lojasiewicza 11, 30-348 Krakow, Polska, Poland, Tel: +48 12 664 47 62; Email;e.stepien@uj.edu.pl
How to cite this article: DroŻdŻ A, Stępień E. Circulating Microvesicles in Regenerative Angiogenesis. Curr Trends Biomedical Eng & Biosci. 2017; 3(1): 555602. DOI: 10.19080/CTBEB.2017.03.555602
Abstract
Extracellular microvesicles (EVs) released by different cells have been shown to facilitate the transfer of several pro-angiogenic as well as anti-antigenic factors that can regulate gene expression in endothelial cells. Our recent study has demonstrated that the profiles of cytokines and angiogenic-active factors in EVs are significantly changed in patients with diabetes that can act both pro-and anti-proliferative. We suggest that those factors may facilitate vascular regeneration and angiogenesis.
Keywords:Angiogenesis; Ectosomes; Exosomes; Microparticles; MicroRNA; Regeneration; Vessels
Abbreviations: CAD: Coronary Artery Disease; Cryo-TEM: Cryogenic Transmission Electron Microscopy; DAT-Mscs: Type 2 Diabeticadipose Tissue-Derived Mesenchymal Stem Cells; EC:Ectosomes; Evs: Extracellular Vesicles; Ex: Exosomes; MVB: Multivesicular Bodies; MSC: Mesenchymal Stem Cells; NAT-Mscs: Non-Diabetic Adipose Tissue-Derived Mesenchymal Stem Cells; TEM: Transmission Electron Microscopy
What are Extracellular Microvesicles
Extracellular microvesicles (EVs) are cell-derived membrane fragments of granules generated by most eukaryotic cells. Most commonly used transmission electron microscopy visualization allowed to determine their diameter between 50 nm and 1000 nm. Despite the small size, their presence in blood was firstly reported using flow cytometry method in late 60's by Wolf, who suggested that the lipid-rich particles originated from the osmophilic platelet granules ("platelet dust") have a thrombotic potential [1]. Previously, George E. Palade's transmission electron microscopy (TEM) studies on segregation of ferritin showed a number of membrane structure budded to the capillary lumen involved in protein transport [2]. In the second half of the 70's - the last century they were alongside reported in cancer cell lines [3]. It has been commonly accepted that EVs can be produced in two alternative ways: 1. by budding or peeling of cell membrane fragments erasing as cell protrusions by a calcium-depending mechanism - this population of EVs is called ectosomes (Ect); 2. by a fusion of multivesicular bodies (MVB) with the cell membrane and subsequent exocytosis-exosomes (Ex) [4]. Generally, this classification is based on the release mechanism criteria, while the size, density and cellular origin determine their taxonomy: Ect represent rather heterogeneous population of vesicles whose size ranges from 0,1 to 1 |im in diameter, Ex are smaller in size ranging 50-100nm [5] (Figure 1).
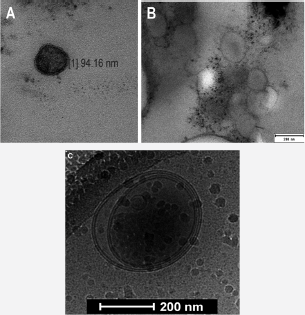
Microscopically, EVs consist of cell components including a lipid bilayer, cytoplasm and ribosomal fragments. On their surface a number of phospholipids and transmembrane proteins can be detected, which can mark the cell origin and regulate their biological activity [6].
Circulating EVs are Abundant in Body Fluids
EVs are commonly present in a wide spectrum of body fluids such as blood (plasma), urine, saliva, breast milk, amniotic fluid, cerebrospinal fluid or semen. In different cardiovascular diseases including: coronary artery disease (CAD), myocardial infarction, stroke cardiomyopathy, hypertension and diabetes and thrombosis, EVs are treated as potential biomarkers of ongoing pathophysiological process or hyperactivation [6,7]. Despite the number lot of clinical evidences about the involvement of EVs in physiological and pathophysiological processes, the role of EVs is not well established, and the mechanisms regulating EVs formation are still unrevealed and there are extensively investigated on in vivo and in vitro.
Biological Role of EVs
Recent studies have confirmed that EVs act as conveyors, which can horizontally transfer biologically active molecules(proteins, microRNA) participating in cell adhesion, chemotaxis and angiogenesis [6,8-10]. Our preliminary data showed that EVs isolated from patient plasma have both pro- angiogenic and anti-angiogenic characteristics in terms of their microRNA profile [8].
Potential Therapeutic Application of EVs in Regenerative Medicine
EVs from mesenchymal stem cells (MSC) showed the ability to alter the cell phenotype and function. For example, EVs derived from non-diabetic adipose tissue-derived mesenchymal stem cells (nAT-MSCs) improved migration and wound healing ability of diabetic MSC in vitro [11]. However, type 2 diabetic adipose tissue-derived mesenchymal stem cells (dAT-MSCs) increased cell aggregation and adhesion in vitro and impaired wound healing in vivo. Nevertheless, the mechanism how EVs from nAT-MSCs acts on cells remains unknown.
Conclusion
The contribution of EVs in the regenerative angiogenesis is depending on their microRNA cargo and their protein profile. The significant role of EVs as novel mediators, potential biomarkers and therapeutic targets in angiogenesis should be explored.
Acknowledgement
This study was supported by the Polish National Science Centre (NCN) the grant OPUS 4 for Ewa St^pien (2012/07/B/ NZ5/02510). The authors are grateful to Dr Eng. Olga Woznicka from Department of Cell Biology and Imaging Institute of Zoology, Jagiellonian University for her kind help in TEM imaging and to Dr Marcin Libera Centre of Polymer and Carbon Materials, Polish Academy of Sciences (Zabrze, Poland) for his help in Cryo-TEM study.
References
- Wolf P (1967) The nature and significance of platelet products in human plasma. Br J Haematol 13(3): 269-288.
- Farquhar MG, Palade GE (1960) Segregation of ferritin in glomerular protein absorption droplets. J Biophys Biochem Cytol 7(2): 297-304.
- Trams EG, Lauter CJ, Salem N Jr, Heine U (1981) Exfoliation of membrane ecto-enzymes in the form of micro-vesicles. Biochim Biophys Acta 645(1): 63-70.
- Cocucci E, Meldolesi J (2015) Ectosomes and exosomes: shedding the confusion between extracellular vesicles. Trends Cell Biol 25(6): 364¬372.
- Kim DK, Lee J, Kim SR, Choi DS, Yoon YJ, et al. (2015) EVpedia: a community web portal for extracellular vesicles research. Bioinformatics 31(6): 933-939.
- Tokarz A, Szuścik I, Kusnierz-Cabala B, Kapusta M, Konkolewska M, et al. (2015) Extracellular vesicles participate in the transport of cytokines and angiogenic factors in diabetic patients with ocular complications. Folia Med Cracov 55(4): 35-48.
- StępieńE, Enguita FJ, Targosz-Korecka M, Nalepa A, Tylko G, et al. (2015) Analysis of miRNA cargo in plasma microvesicles in human plasma EuroMedLab Paris. ClinChem Lab Med 53(Special Suppl): S1- S1450.
- StępieńE, Enguita FJ, Targosz-Korecka M, Nalepa A, Tylko G, et al. (2015) Analysis of miRNA cargo in plasma microvesicles in human plasma EuroMedLab Paris. ClinChem Lab Med 53(Special Suppl): S1- S1450.
- Alexandru N, Badila E, Weiss E, Cochior D, St^pien E, et al. (2016) Vascular complications in diabetes: Microparticles and microparticle associated microRNAs as active players. BiochemBiophys Res Commun 472(1): 1-10.
- Kasprzyk J, StępieńE, Piekoszewski W (2017) Application of nano- LC-MALDI-TOF/TOF-MS for proteomic analysis of microvesicles. Clin Biochem 50(4-5): 241-243.
- Trinh NT, Yamashita T, Tu TC, Kato T, Ohneda K, et al. (2016) Microvesicles enhance the mobility of human diabetic adipose tissue- derived mesenchymal stem cells in vitro and improve wound healing in vivo. Biochem Biophys Res Commun 473(4): 1111-1118.






























