Clock Genes, Chronodisruption, Nutrition and Obesity
Francisco J Sánchez Muniz* and Cristina Simón Martín
Nutrition Department, Universidad Complutense, Spain
Submission: July 24, 2017; Published: July 31, 2017
*Corresponding author: Francisco J Sanchez Muniz, Nutrition Department, Pharmacy School, Universidad Complutense, Spain, Tel: 34 913-941828; Fax: 34 913-941-810; Email: frasan@ucm.es
How to cite this article: Francisco J S M, Cristina S M. Clock Genes, Chronodisruption, Nutrition and Obesity. Curre Res Diabetes & Obes J. 2017; 3(2): 555607. DOI:10.19080/CRDOJ.2017.03.555607
Abstract
The existence of biological clocks has been demonstrated in all living beings. Such clocks control the physio-metabolic activities of cells, organs and systems to warrantee efficacy in the process to obtain energy and metabolize nutrients under a homeostatic point of view. These ordered activities are known as circadian rhythms, occurring approximately every 24h, and depend on the activity of groups of neurons (oscillators) negatively interrelated. The oscillatory activity is related to gene expressions that implicate rhythms in the mRNA and protein production. However, alterations in such synchronism is frequently found because of many of human activities are performed in "unexpected” environmental conditions. Thus, light intensity and environmental temperature are normally rather constant; in addition light/darkness cycles are modified by working/leisure times, travels, meals and activities braking the ticking of the biological clocks producing chronodisruption. The aim of this mini review is to give some central information on most common chronodisruption aspects and their relationships with dietary habits and obesity. After a short and concise introduction explaining central aspects of biology clocks and disruptors, the role of some prevalent dietary behaviors (e.g. frequent snaking, late meals and large food consumption) acting at the levels of CLOCK genes are described and discussed. This mini review far from to be exhaustive pretends to open discussion on the most accepted information, ending suggesting some future research to understand the role and importance of CLOCK genes in obesity.
Keywords: Chronodisruption; CLOCK genes; Dietary habits; Obesity
Abbreviations: CD: Chronodisruption; Cry: Cryptochrome; CCGs: Clock Controlled Genes; PER: Clock Genes Perio; SCN: Suprachiasmatic Nucleus
Introduction
Most living organisms have developed since millennium mechanisms to improve response to daily or annually environmental changes. Thus, physiometabolic changes induced by light/dark, low/warm/high temperatures, could be more predictable during the season/day time, and consequently metabolic processes could be prepared in advance (e.g. migrations, reproduction, breeding) to improve their efficacy. The circadian system (from Latin circa that means approximately and diem meaning one day) has the aptitude to organize the internal temporal order of physiological processes according to predictable environmental cyclic signals. The circadian system is organized in a similar manner to a clock that ticking controls activities to optimize energy and nutrient utilization. It is accepted the existence of clocks in the different organs and systems of mammals controlling their activities that, in turn, are controlled by a master clock [1]. The central clock is located in the suprachiasmatic nucleus (SCN) of the hypothalamus where two groups of various neurons (ca., 20,000 each)-the ventrolateral and the dorsomedial areas-are negatively interrelated. The light/ dark information analyzed in the retina arrives the SCN via the retino hypothalamic tract, inducing at a cellular level changes in the gene expression ofthose antagonic neuronal groups [1]. Thus, it can be accepted that circadiam system and, thus, rhythmicity is controlled by oscillatory -positive/negative-mechanisms that at cellular levels implicate daily control of the mRNA and proteins. Although each neuron is able to act as an independent oscillator within a cell-autonomous period, each neuronal group acts as an oscillator that is synchronized to produce a circa 24h common periodicity [2,3]. GABAergic interneurons are responsible to inhibit one oscillator when the antagonic one is active and vice versa [4]. Thus, the ventrolateral area is the responsible for light synchronisation, while output regulation is mediated by dorsomedial area [5,6]. The BMAL1 and CLOCK transcription factors constitute a heterodimer, which activates the expression of the clock genes Perio (PER), Cryptochromes (CRY), REVERB, and ROR, as well as other clock controlled genes (CCGs) by binding to E-box augmentation elements [7]. PER and CRY dimerize and inhibit their own expression by translocation into the nucleus and also acting as repressor of the CLOCK/BMAL1 heterodimer with a delay of several hours [8]. A second loop also regulated by CLOCK: BMAL1 heterodimers activates the transcription of Rev-erb and Ror genes. Latter, REV-ERB and ROR transcription factors compete to bind response elements present on the Bmall promoter [1]. Thus, REV-ERB proteins repress Bmall transcription [9], while ROR proteins activate it [10] (Figure 1). In addition it has been described that ROR proteins are implicated in rhythmic Bmal1 expression [11] and that PER is lysed by the proteasome regulating the oscillatory loops. As the cellular processes implicated in the fine control of the master clock, and in turn, in that of the different organ and tissue clocks is out from the aim of this mini review we will not insist more in these molecular aspects, although the reading of recent published reviews in chronicity is highly recommended.

Circadian system
In humans the circadian system is formed by a structural frame, ordered in a hierarchical manner, which is responsible for generating this rhythm and its synchronization with the environment. It consists of three main elements: circadian clocks, imputs tracks and outputs tracks [12].
Circadian clocks: As previously commented the central clock is located in the SCN of the hypothalamus. Its existence has been known since 1972, "resetting" daily by signals of light/darkness that come from the retina, through the optical pathways. Changes in the light/dark relationship are the main input to the SCN but there are other information produced by the interaction of the living being with the surrounding environment, such as propioceptive, thermoactive, acoustic signals that reach the SCN or its connections with other brain areas contributing to the adjustment of this central clock. In addition, the schedule of meals and exercise seem to influence mainly the activity of other clocks located in different organs of the body. Thus, since 2001, it is known that the alarm central harmonizes the action of peripheral clocks located in organs and tissues such as lung, heart, liver, pancreas, kidney and adipose tissue, among others, through the activity of the vegetative nervous system and the secretion of hormones [12,13]. In addition the SCN has important connections with the adenohypophysis and pineal gland participating among other functions in the cortisol and melatonin production controls [13].
Inputs tracks: Our existence and behavior clearly depend on the signals from the environment and from or body organs. These signals have central importance in the circadian rhythms as the "Clock" must be adjusted periodically through the synchronizers or zeitgebers (in German, "giver") which fluctuate rhythmically and integrate together these tracks [13].
Output tracks: The master "Clock" temporary transmit signals to the rest of the effectors by sending information to those brain areas involved in the regulation of body temperature, patterns of behavior and sleep-wake cycle, to neuroendocrine axes and peripheral organs. The SCN controls humoral mediators (e.g. melatonin and cortisol), sends nerve projections and physical signs such as the pace of body temperature. From all mediators, the best known is the melatonin hormone, which is involved in the regulation of sleep and circadian rhythmicity, its synthesis subjects to the SCN activity, the inhibitory action of light, with minimum and maximum levels at day and night, respectively [13]. With regard to the timing light, the pineal gland is responsible for regulating the sensitivity on the part of the SCN to various signals responsible for encoding the lighting of the environment [14]. That means, the lack of light would reduce the inputs to the system by inducing a lower production of melatonin, which in turn is an sleep inducer, through implementing circuit inhibitors modulated by genes. Unlike, the presence of light energy would reduce the production of melatonin, which would delay or inhibit the induction of neurons involved in the initiation and maintenance of sleep. Therefore, melatonin, mainly produced by the pineal, makes the different circadian oscillators located in the SCN act similarly to the object to operate as a circadian clock [14].
Chronodisruption
The alteration on the rhythmicity in any of the central or peripheral oscillators, the unstable wrong phase relationship between them, the uncoupling between inputs, pacemarkers and outpouts induces chronodisruption. Chronodisruption (CD) is every day more prevalent and favored by environmental factors that in turn are conditioned by the present living styles. Thus, working in rooms illuminated by artificial light, the almost permanent exposition to bright light at night (working with machines, computers, tablets) or dim lights during daytime, chronic and/or social jet-lag and shift-work can induce circadian system disruption [15]. However, other input alterations may also result in CD. Thus, warm and stable temperatures, irregular sleep time, low physical activity and frequent meals or constant snacking work as disruptors [16,17]. Ageing is a source of CD since it affects the circadian system and in some way can be related with the modification of some biomarkers involved in obesity. Three main factors are implicated in the frequent CD observed in aged people: a) decreasing inputs (e.g. reducing light reception and blue light transmission) [18], and those other receptor signals (e.g. noice, odors); b) necrosis and/ or apoptosis and loss of functionality of oscillators and their antagonists in the central clock [19,20] as well as of pineal and adenohypophysis neuroendocrine cells [21-23]; c) the reduction of day-night contrast due to losses in the rhythm amplitude [24,25].
Chronodisruption, obesity and metabolic syndrome
CD is associated to a predisposition to obesity, metabolic syndrome, cardiovascular diseases, cognitive and affective impairments, sleep disorders, premature ageing, prostatic, mammary and colorectal cancer and, in general, higher mortality [1,26,27]. This mini review will mainly focus on obesity, given the existing scientific evidence between obesity and CD, which contributes to negatively affect many functions involved in the metabolic regulation of lipids and carbohydrates as well as in the response to insulin [28].
Obesity is a chronic disease of multifactorial origin in which multiple factors of genetic, hormonal, metabolic, social and cultural type influence and, in a coordinated and interactive way, provoke an imbalance between energy income and expenditure, leading to a fat mass and body weight gain. Obesity is characterized by an increase and content of adipose tissue, which causes morphological and metabolic alterations together with increase in several comorbidities [29]. Currently, a nearly unstoppable incidence and prevalence growth of obesity has been reported, representing a very important public health problem, and may lead to different degrees of insulin resistance, steatosis, metabolic syndrome and increased cardiovascular morbidity and mortality [12,29]. As already mentioned, a number of factors are involved in the etiology of obesity. Normally, obesity is treated modifying eating behavior and physical activity; however, in order to optimize results the impact and interaction of life style with genes should be considered (Figure 2 & Table 1). In relation to the topic of this mini review, two major factor groups can be defined.
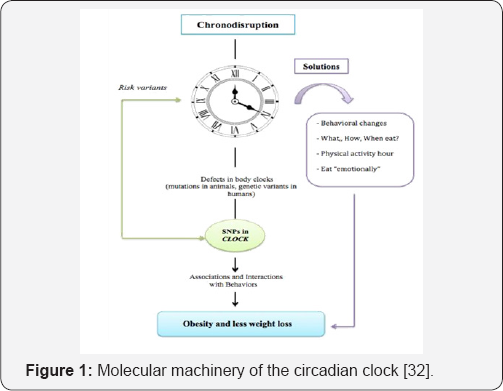
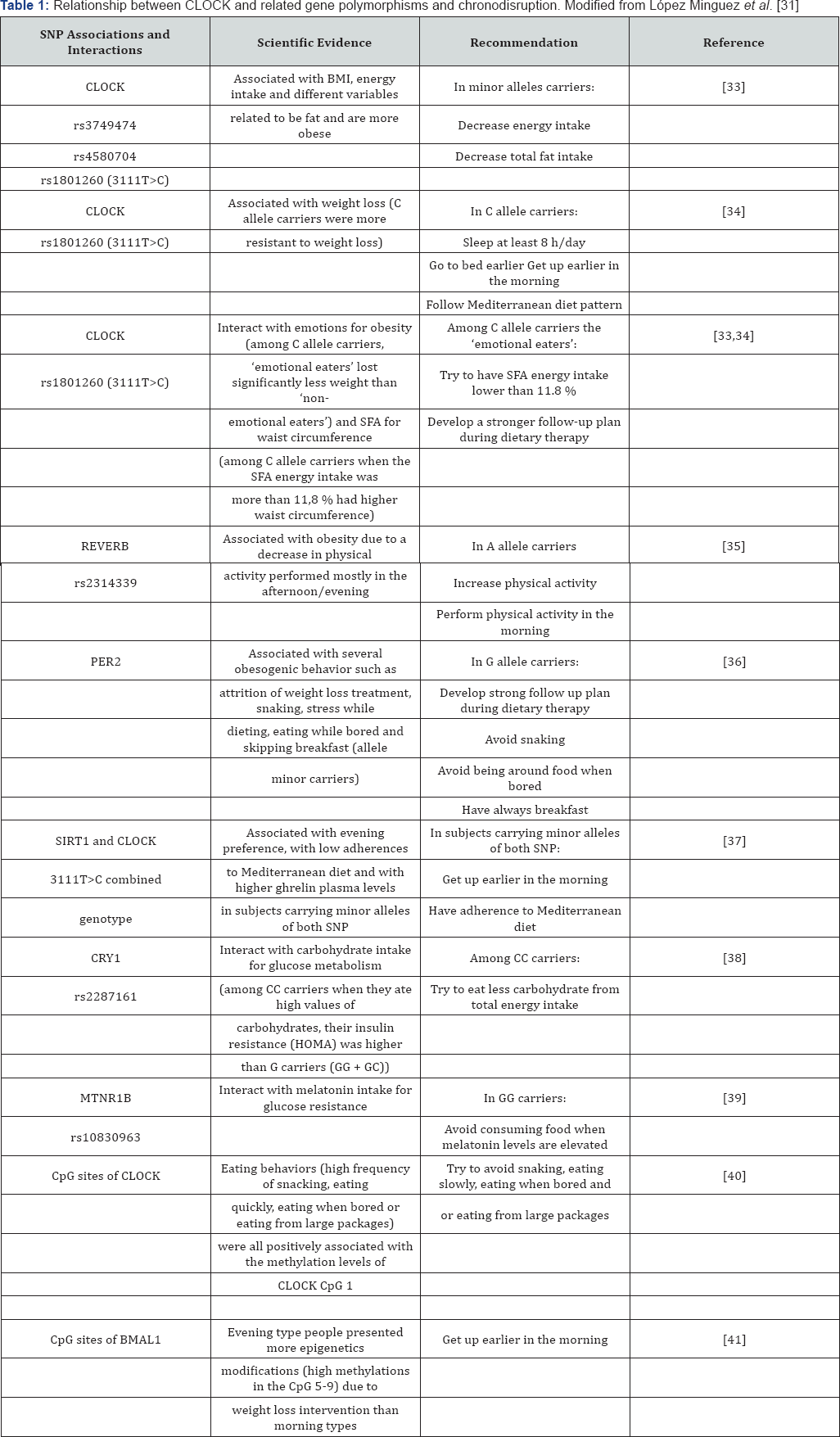
Endogenous factors: The Clock, Bmal1 and Per2 proteins play a very important role in this circadian functioning, so some failure in their synthesis and / or structure or in their associated CLOCK genes can induce CD. Polymorphisms of a single nucleotide (SNPs) are very common in a specific position of the genome and they are responsible, at less in part, of the inter individual differences in the vulnerability to certain diseases [28]. Although information is growing up very quick we have selected some SNPs in the CLOCK genes whose importance on obesity and metabolic markers has been reported. (Table 1) includes general information about a selection of the most important gene polymorphisms implicated in the CD-obesity relationship.
Clock: SNPs rs3749474, rs4580704 and rs1801260 were associated with variables related to obesity, energy intake and BMI; SNP rs1801260 with weight loss, sleeping less, poor adherence to the Mediterranean diet. They carriers fit better within the evening type, and interact with saturated fatty acid intake for waist circumference and emotions. Also SNP rs4580704 interacted with monounsaturated fatty acid intake for blood glucose [31]. New evidences at epigenetic level of CpG sites of CLOCK gene suggest association with erratic eating behaviors (eating quickly and/or hudge amounts, snacking and eating when bored) [31].
Other genes: PER2 SNP rs2304672 was associated with snacking, eating when bored, stress, etc; SIRT1 with CLOCK (3111 T>C combined genotype) were related to adherence to Mediterranean diet; REVERB rs2314339 appeared associated with physical activity, and CRY1 rs2287161 interacted with carbohydrate intake [31]. Epigenetic modifications at the level of CpG sites of BMAL1 have been reported to be associated with weight loss intervention [31].
Exogenous factors: Both the physical activity and the meal schedules, among others, act as relevant synchronizers. Thus, it is clear that it is not only important what and how we eat but when we do it. In this regard, it has been found that a regular meal schedule contributes to the maintenance of the internal temporal order of the circadian system [13]. In addition, the response to energy dietary restriction differs in some individual with respect to others depending on the degree of CD. Therefore, to this aim, it has been proposed an index that based in some CD markers helps predicting weight loss [28]. Various determinations were made throughout the day, being the most predictive variables body temperature, blood pressure and the secretion of certain hormones such as melatonin and cortisol, since they present more precise rhythms [28,30]. From these, the so-called CD index was calculated permitting to classify patients with or without CD. Corbalan-Tutau et al. observed that the weighted mean of the first four factors explained the 53.8% of the total variance. These authors proposed the value 40.3 as CD index cut-off point as the area of the COR (Operational Characteristic of the Receiver) curve was 0.95, being 40.3 the optimal cut-off point as this value corresponded to the point that shows the best sensitivity along with the best specificity [30]. Therefore, individuals with moderate obesity who present a value for the already commented CD index above 40.3 will have CD. By the way, women with high CD score displayed higher BMI, waist perimeter, fasting glucose, total cholesterol and triglyceride concentrations and also higher systolic and diastolic blood pressure values; thus, were at risk for metabolic syndrome [28]. In agreement with others [31] and taking into account all already commented facts, some general nutritional and lifestyle suggestions should be taken into consideration in order to palliate the growing incidence and prevalence of obesity:
- Sleep during the night and be active during the day. Sleep in total darkness when possible.
- Avoid intense light exposition during night time.
- Make exercise during the morning.
- Avoid eating at night hours.
- Have adherence to Mediterranean diet
- Eat the main meal of the day before 15:00hrs.
Conclusion
Light/dark cycles are central in the control of the circadian system and that of several metabolic and physiological aspects in humans. However, environmental factors have drastically changed the daily inputs arriving to the master clock favoring its disruption and that of the other pacemarkers. Among them, eating and sleeping times, light intensity exposition, noice, social jet-lag and shift works have been considered crutial in the understanding of CD. Nonetheless, taking into account the existence of interactions between way of living and presence of some CLOCK polymorphisms, active research is demanded to improve knowledge in such interaction. This effort should be performed to find out mechanisms and interactions occurring at critical life periods (fetal, lactation, childhood, aging) and also addressed in at risk people (e.g. over weighted, with type 1 obesity and familial antecedents) in order improve our knowledge and to decrease the growing tendency of both obesity and CD.
References
- Terzibasi Tozzini E, Martinez Nicolas A, Lucas Sánchez A (2017) The clock is ticking. Ageing of the circadian system: From physiology to cell cycle. Semin Cell Dev Biol.
- Webb AB, Angelo N, Huettner JE, Herzog ED (2009) Intrinsic, nondeterministic circadian rhythm generation in identified mammalian neurons. Proc Natl Acad Sci USA 106(38): 16493-16498.
- Welsh DK, Logothetis DE, Meister M, Reppert SM (1995) Individual neurons dissociated from rat suprachiasmatic nucleus express independently phased circadian firing rhythms. Neuron 14(4): 697706.
- Vujovic N, Gooley JJ, Jhou TC, Saper CB (2015) Projections from the subparaventricular zone define four channels of output from the circadian timing system. J Comp Neurol 52: 2714-2737.
- Hattar S, Liao HW, Takao M, Berson DM, Yau KW (2002) Melanopsin- containing retinal ganglion cells: architecture, projections, and intrinsic photosensitivity. Science 29: 1065-1070.
- Moore RY, Speh JC, Leak RK (2002) Suprachiasmatic nucleus organization. Cell Tissue Res 309: 89-98.
- Reppert SM, Weaver DR (2002) Coordination of circadian timing in mammals. Nature 418: 935-941.
- Vanselow JT, Kramer A (2010) Posttranslational regulation of circadian clocks, In: U Albrecht (Ed.), Protein Reviews. Volume 12. The Circadian Clock, Springer Science, New York, pp. 79-104.
- Preitner N, Damiola F, Lopez-Molina L, Zakany J, Duboule D, et al.
- Guillaumond F, Dardente H, Giguere V, Cermakian N (2005) Differential control of Bmal1 circadian transcription by REV-ERB and ROR nuclear receptors. J Biol Rhythms 20: 391-403.
- Emery P, Reppert SM (2004) A rhythmic Ror. Neuron 43: 443-446.
- Corella D, Coltell O, Ordovás JM (2016) Genetic and epigenetic of obesity. An Real Acad Farm 82 (Special issue): 129-136.
- Garaulet M (2015) La cronobiología, la alimentación y la salud. Nutr y Sal 27(8): 101-121.
- Aguilar Roblero R, Guadarrama P, Mercado C, Chávez JL (2004) El núcleo supraquiasmático y la glándula pineal en la regulación de los ritmos circadianos en roedores. In: J Velazquez Moctezuma (Ed.), Temas selectos de neurociencias III. Universidad Autonómica de México, México, pp. 321-330.
- Rajaratnam SM, Arendt J (2001) Health in a 24h-society. Lancet 358: 999-1005.
- Erren TC, Reiter RJ (2009) Light hygiene: time to make preventive use of insights-old and new-into the nexus of the drug light, melatonin, clocks, chronodisruption and public health. Med Hypotheses 73: 537541.
- Martinez Nicolas A, Madrid JA, Rol MA (2014) Day-night contrast as source of health for the human circadian system. Chronobiol Int 31: 382-393.
- Turner PL, Mainster MA (2008) Circadian photoreception: ageing and the eye's important role in systemic health. Br J Ophthalmol 92: 14391444.
- Tsukahara S, Tanaka S, Ishida K, Hoshi N, Kitagawa H (2005) Age- related change and its sex differences in histoarchitecture of the hypothalamic suprachiasmatic nucleus of F344/N rats. Exp Gerontol 40: 147-155.
- Nakamura TJ, Nakamura W, Tokuda IT, Ishikawa T, Kudo T, Colwellet CS, Block GD (2015) Age-related changes in the circadian system unmasked by constant conditions. eNeuro 2(4).
- Myers BL, Badia P (1995) Changes in circadian rhythms and sleep quality with aging: mechanisms and intervertions. Neurosci Biobehav Rev 19: 553-571.
- Kunz D, Schmitz S, Mahlberg R, Mohr A, Stöter C, Wolf KJ, Herrmann WM (1999) A new concept for melatonin deficit: on pineal calcification and melatonin excretion. Neuropsychopharmacology 21: 765-772.
- Schmid HA, Requintina PJ, Oxenkrug GF, Sturner W (1994) Calcium, calcification and melatonin biosynthesis in the human pineal gland: a postmortem study into age-related factors. J Pineal Res 16: 178-183.
- Huang YL, Liu RY, Wang QS, Van Someren EJ, Xu H, Zhou JN (2002) Age- related associated difference in circadian sleep-wake and rest-activity rhythms. Physiol Behav 76: 597-603.
- Van Someren EJW, Swaab DF, Colenda CC, Cohen W, McCall WV Rosenquist PB (1999) Bright light therapy: improved sensitivity to its effects on rest-activity rhythms in Alzheimer patients by application of non-parametric methods. Chronobiol Int 16(4): 505-518.
- Davis S, Mirick DK (2006) circadian disruption, shift work and the risk of cancer: a summary of the evidence and studies in Seattle. Cancer Causes Control 17(4): 539-545.
- Schernhammer ES, Laden F, Speizer FE, Willett WC, Hunter DJ, et al.
- Garaulet M, Gómez-Abellán P (2016) Clock genes. Circadian rhythms and predisposition to obesity. An Real Acad Farm 82 (Special issue): 44-54.
- Sánchez Muniz FJ (2016) Obesity: a very serious public health problem. An Real Acad Farm. 82(Special issue): 6-26.
- Corbalán-Tutau MD, Gómez-Abellán P, Madrid JA, Canteras M, Ordovás JM, Garaulet M (2015) Toward a chronobiological characterization of obesity and metabolic syndrome in clinical practice. Clin Nutr 34: 477483
- López-Minguez J, Gómez-Abellán P, Garaulet M (2016) Circadian rhythms, food timing and obesity. Proc Nutr Soc 75(4): 501-511.
- Gómez-Abellán P, Madrid JA, Ordovás JM, Garaulet M (2012) Aspectos cronobiológicos de la obesidad y el síndrome metabólico. Endocrinol Nutr 59(1): 50-61.
- Garaulet M, Lee YC, Shen J, Parnel LD, Arnett DK, et al. (2009) CLOCK genetic variation and metabolic syndrome risk: modulation by monounsaturated fatty acids. Am J Clin Nutr 90(6): 1466-1475.
- López-Guimera G, Dashti HS, Smith CE, Sánchez Carracedo D Ordovás JM, Garaulet M (2014) CLOCK 3111 T/C SNP interacts with emotional eating behavior for weight-loss in a Mediterranean population. PLoS One 9(6): e99152.
- Garaulet M, Smith CE, Gomez Abellán P, Ordovás Montañés M, Lee YC, et al. (2014) REV-ERB-ALPHA circadian gene variant associates with obesity in two independent populations: Mediterranean and North American. Mol Nutr Food Res 58: 821-829.
- Garaulet M, Corbalan Tutau MD, Madrid JA, et al. (2010) PERIOD2 variants are associated with abdominal obesity, psycho-behavioral factors, and attrition in the dietary treatment of obesity. J Am Diet Assoc 110: 917-921.
- Garaulet M, Corbalán Tutau MD, Madrid JA, Baraza JC, Parnell LD, et al. (2012) SIRT1 and CLOCK 3111T> C combined genotype is associated with evening preference and weight loss resistance in a behavioral therapy treatment for obesity. Int J Obes (Lond) 36: 1436-1441.
- Dashti HS, Smith CE, Lee YC, Parnell LD, Lai CQ, et al. (2014) CRY1 circadian gene variant interacts with carbohydrate intake for insulin resistance in two independent populations: Mediterranean and North American. Chronobiol Int 31(5): 660-667.
- Garaulet M, Gómez-Abellán P, Rubio-Sastre P, Madrid JA, Saxena R, Scheer FA (2015) Common type 2 diabetes risk variant in MTNR1B worsens the deleterious effect of melatonin on glucose tolerance in humans. Metabolism 64(12) : 1650-1657.
- Milagro FI, Gómez-Abellán P, Campión J, Martínez JA, Ordovás JM, et al. (2012) CLOCK, PER2 and BMAL1 DNA methylation: Association with obesity and metabolic syndrome characteristics and monounsaturated fat intake. J Biol Rhythms 29(9): 1180-1194.
- Samblas M, Milagro FI, Gómez-Abellán P, Martínez JA, Garaulet M, et al. (2016) Methylation on the Circadian Gene BMAL1 is Associated with the effects of a weight loss intervention on serum lipid levels. J Biol Rhythms 31(3): 308-317.






























