Protein-Losing Enteropathy - Diagnostic Challenge, Unusual Pathologies and Literature Review
Tirzah de Mendonça Lopes Secundo¹*, Virgínia Lúcia Ribeiro Cabral¹, Cirstiane Kibune Nagasako², Rita Barbosa Carvalho³, Carolina Oliveira Santos¹, Marcello Imbrizi Rabello¹ and Natalie Cavalcanti Mareco Silva¹
1Gastroenterology Unit, State University of Campinas, Brasil
2Endoscopy Unit, State University of Campinas, Brasil
3Pathologic Anatomy Unit, State University of Campinas, Brasil
Submission: May 07, 2021; Published: June 01, 2021
*Corresponding author: Tirzah de Mendonça Lopes Secundo, Gastroenterology Unit, State University of Campinas- Unicamp. Campinas, São Paulo, Brasil
How to cite this article: Tirzah d M L S, Virgínia L R C, Cirstiane K N, Rita B C, Carolina O S, et al. Protein-Losing Enteropathy - Diagnostic Challenge, Unusual Pathologies and Literature Review. Adv Res Gastroentero Hepatol, 2021; 17(1): 555953. DOI: 10.19080/ARGH.2021.17.555953.
Abstract
Introduction: Protein-losing enteropathy (PLE) is a rare condition in clinical practice that can be present in a variety of diseases, including those primarily from the gastrointestinal tract (GIT) or not. The mechanisms possibly involved in its pathogenesis are: erosive and non-erosive mucosal disease, increased central venous pressure and mesenteric lymphatic obstruction. This diagnosis must be considered in patients with severe hypoalbuminemia in which proteinuria, liver disease and malnutrition were excluded.
Aim: Present unusual causes of PLE, its clinical management and literature review. Methods: retrospective study, in which we describe seven cases of PLE with uncommon etiologies and its treatment approaches.
Conclusion: The prognosis of these patients depends mainly on the primary disease and its specific treatment. However, in situations in which the etiology cannot be defined, protein loss can be stopped with approaches based on its possible physiopathological mechanisms.
Keywords: Protein-losing enteropathy; Physiopathological mechanisms; Hypoalbuminemia; Serum proteins; Gastrointestinal tract
Introduction
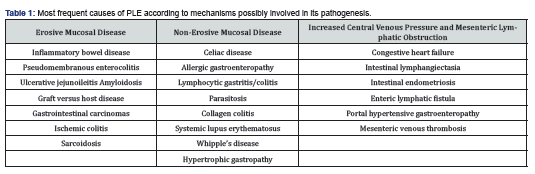
Protein-losing enteropathy (PLE) is a rare condition in clinical practice [1]. It is characterized by excessive loss of serum proteins in the gastrointestinal tract, leading to hypoalbuminemia and generalized edema, among other clinical manifestations [2]. Several mechanisms are involved in its process, including erosive and non-erosive mucosal disease and increased central venous pressure or mesenteric lymphatic obstruction [3,4] (Table 1).
As protein loss is not selective, serum albumin, globulin, fibrinogen, lipoproteins, alpha-1 antitrypsin, transferrin and ceruloplasmin are decreased. Lymphopenia is also observed in cases in which lymphangiectasia is the mechanism involved in the protein loss. Despite the humoral and cellular deficiency, an increased susceptibility to opportunistic infections is not common [5,6]. This diagnosis must be considered in patients with severe hypoalbuminemia in which proteinuria, liver disease and malnutrition have been excluded [6,7]. Alpha 1-antitrypsin is a glycoprotein with molecular weight similar to albumin, also synthesized by the liver. In gastrointestinal tract, alpha-1-antitrypsin is resistant to proteolysis, so it is not absorbed neither secreted. Under physiological conditions, its concentration in feces is low. Thus, its faecal excretion dosage is useful in the diagnosis of PLE [4,8].
99mTc-labeled human serum albumin scan can detect protein loss into the gut and thus confirm suspected protein losing enteropathy and provide anatomical imaging to confirm the precise site of protein loss [4,5,8-10]. Losing enteropathy protein treatment consists primarily in the management of primary disease and its complications, such as malnutrition. Based on knowledge of the involved mechanism in intestinal protein loss, some therapeutic alternatives have been proposed, although more studies are still needed to set them as specific treatment [4].
Heparin, in vitro studies, has shown increased lymphangiogenesis mediated by growth factor in addition to an antiinflammatory effect. It is believed that, based on these effects, it can restore the integrity of the basement membrane and selective permeability in the gut. The exact mechanism is still unclear, and only a few patients have shown good response to its use [4,11]. In the same way, corticosteroids don´t have the mechanism by which can modify LPE evolution well defined. It is often observed a response in the first six months of treatment, with long-term relapse [4,7]. Some authors believe it is more frequently in patients who are in maintenance treatment with corticosteroid alone than in those who are on combined therapy with immunomodulator agents [5].
Octreotide is also an option. Its use has been described in association with corticosteroids in cases of refractory PLE secondary to amyloidosis [12]. Its mechanism of action is not fully understood, and more research is needed for this setting [13].
Aim
Present rare causes of protein-losing enteropathy, its management and literature review.
Case Reports and Discussion
Protein-losing enteropathy by erosive mucosal disease.
Case 1
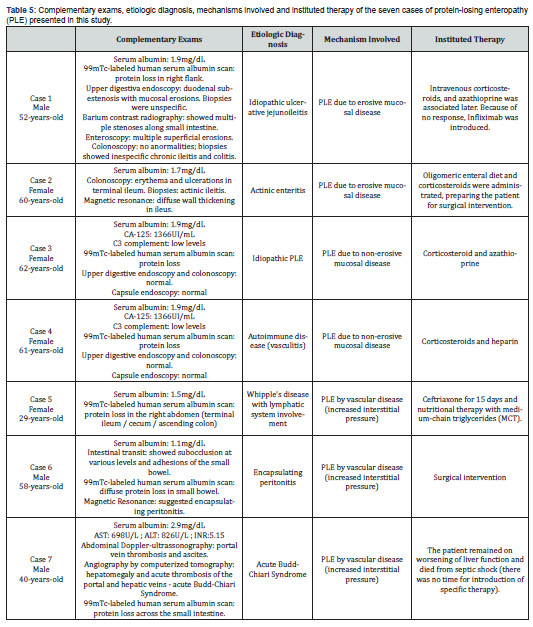
Male, 52 years old, referred to Gastroenterology from Nephrology service due to anasarca frame, malnutrition, hypoalbuminemia (serum albumin: 1,9mg/dL). He had no diarrhea, and renal causes for the symptoms had been discarded. Protein-losing enteropathy was confirmed by 99mTc-labeled human serum albumin scan that showed protein loss in the topography of the right flank (Figure 1). Endoscopy showed duodenal subestenosis and local mucosal erosions. Anatomopathologic study of these lesions was unspecific. Barium contrast radiography showed multiple stenoses along small intestine. Enteroscopy showed multiple superficial erosions, and stenosis dilatation was performed. Colonoscopy was macroscopically normal, and biopsies showed in specific chronic ileitis and colitis (Table 2). Corticosteroid therapy was started, and clinical improvement was observed. But the patient gave up treatment, returning after worsening of edema and malnutrition. He was admitted for a possible bowel resection, but because of diffuse intestinal protein loss at that moment, the surgery could not be realized. Intravenous corticosteroid therapy was initiated, and azathioprine prescribed later. As there was no response, Infliximab was initiated. There was a significant clinical worsening, pyoarthritis and pneumonia been diagnosed. Despite antibiotic therapy and nutritional support, the patient died.


Case 2
Female, 60 years old, started four years ago a clinical condition including diarrhea, crampy abdominal pain, swelling in limbs and weight loss (10kg in six months). She had a previous history of cervical neoplasia 10 years ago and underwent radiotherapy at the time. She developed severe malnutrition frame (30Kg; BMI 11), an important muscle weakness and lower limbs and abdominal wall swelling. She was hospitalized at the time, and laboratory tests showed albumin 1.7 mg/dL, prealbumin 12mg/dL, Hb 8.0, K 2.1mg/dL and Mg 0.89mg/dL, with normal ESR and CRP 2mg/dL. Colonoscopy showed erythema and ulcerations in the terminal ileum, and pathologic study showed actinic ileitis. Resonance revealed diffuse wall thickening in ileal loop (Table 2). Oligomeric enteral diet and corticosteroids were started. She was discharged with serum albumin level and clinical improvement ((alb 3.8mg/dL; weight = 34.5Kg). The patient presented intestinal subocclusion and was hospitalized for nutritional support and surgical intervention. Bowel resection target in three areas of ileal stenosis was performed. She progressed well, in good nutritional status. pneumonia been diagnosed. Despite antibiotic therapy and nutritional support, the patient died.
Discussion
Protein-losing enteropathy originating in the gut can be classified into erosive and non-erosive. Ulcerative colitis and Crohn's disease are the classic examples of the erosive form. The mechanism includes an increased fluid passage rich in proteins through the damaged epithelium, and the degree of protein loss is relating directly to the lesion mucosa [3].
Idiopathic ulcerative jejunum ileitis is a rare disease, which etiology is not well known [14,15]. Its presentation ranges from mucosal inflammation to involvement of deeper layers, causing ulceration and stenosis. It is an unusual and exclusion diagnosis, since over 50% of patients with ulcerative jejunum ileitis are diagnosed as Crohn's disease, celiac disease, lymphoma or infectious diseases [16]. Although little thing is known about its pathophysiology, it is believed to be the participation of T lymphocytes, with extensive mucosal infiltration, and prednisone, azathioprine and biological being therapy options for these pacientes [16,17]. Our patient, although had receive immunosuppression therapy and heparin, did not provide satisfactory answers and evolved to death due to infectious complications, probably due to the immunosuppression associated with severe malnutrition.
Actinic enteritis is a complication of pelvic radiation therapy used to treat cancer with an estimated incidence ranging 3-15% [18], mainly affecting the ileum, rectum and sigmoid [19]. The pathophysiology includes damage to epithelial cells and connective tissue by radiation, leading to obliterans endarteritis and tissue ischemia. The consequences range from mucosal ulceration to necrosis of the intestinal wall, and the symptoms may arise during the first 5 to 10 years, but up to 30 years later [18-21]. In case 2, the patient presented ulcerations of the intestinal mucosa in the distal ileum and substenosis areas as manifestations of enteritis actinic. She achieved good performance with the use of corticosteroids and nutritional therapy, being submitted to bowel resection.
Protein-losing enteropathy by non-erosive mucosal disease
Case 3
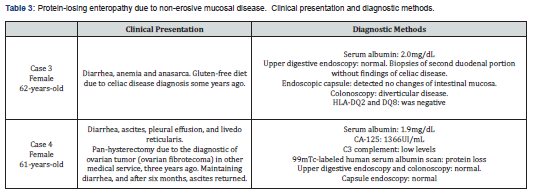
Female, 62 years old, was admitted for investigation of diarrhea and anasarca. She had previous episode of acute pulmonary edema due to cardiac tamponade and diagnosis of celiac disease at that time. She displayed anemia, serum alb 2.0 mg/dL, prealbumin 17.6mg/dL and increased PCR. During hospitalization, she evolved with deep vein thrombosis in the left arm, receiving anticoagulation therapy. It was started empirical treatment with corticosteroids, with improvement in diarrhea, and gluten-free diet was maintained. Colonoscopy showed only diverticular disease of the colon, and upper endoscopy with biopsy of the second duodenal portion didn´t show suggestive findings of celiac disease. Endoscopic capsule detected no changes of intestinal mucosa. Laboratory tests showed albumin 3.6 mg/dL, and genetic screening of HLA-DQ2 and DQ8 was negative (Table 3). There was clinical worsening with the decrease in corticosteroid dose, progressing to anasarca, diarrhea return and serum albumin of 1.2 mg/dL. After increasing dose of prednisone to 30 mg day, there was a significant improvement, reaching serum albumin of 4.0 mg/dL. Nowadays, the patient is in use of Azathioprine 150mg/day alone, remaining asymptomatic, with no ascites and normal serum albumin for a year.
Case 4
Female, 61 years old, presenting with diarrhea for three years associated to ascites, weakness and weight loss. At that time, it was diagnosed an ovarian tumor. She was submitted to pan-hysterectomy, and pathological results in left ovarian fibrotecoma. She progressed well, although maintaining diarrhea. After six months, because of returned ascites frame, she underwent exploratory laparotomy with biopsies of intestinal serous and peritoneum, and study of ascites: all negative for malignancy. She was than referred to our clinic for research, presenting with ascites, pleural effusion, emaciated, with livedo reticularis and keeping interspersed diarrhea with constipation. Laboratory tests showed alb 1.9 mg/dL, gamma globulin 0.58mg/dL, VHS 103/mm³, increased CRP, CA-125 1366UI/mL and C3 below the lower limit of normal. After liver and kidney causes for anasarca were excluding, protein-losing enteropathy was confirmed through 99mTc-labeled human serum albumin scan. Upper and lower endoscopy showed no findings that justify the frame. The capsule endoscopy also showed no mucosal lesion, and autoantibodies tests were negative (Table 3). Steroid therapy was started. During follow-up, she developed deep vein thrombosis in the left arm, and anticoagulation with heparin was started. During its use, along with steroids, the patient evolved with clinical and laboratory improvement. The patient keeps on using of corticosteroids, with significant reduction in CA-125 and improvement in complement serum level.
Discussion
Even without mucosal erosions, conditions that lead to loss of surface epithelial cells may cause loss of proteins in excess in the gastrointestinal tract. Among them, the most common is Menetrier's disease, wherein the enlargement of intercellular junctions leads to an increased mucosal permeability [3]. Other non-erosive causes of LPE are eosinophilic gastroenteritis, microscopic colitis, celiac disease, among others [9]. These are clinical conditions that, although do not occur frequently, the inflammatory process leads to increased vascular permeability.
When associated with autoimmune diseases, a hypothesis for its mechanism is that there must be a direct relationship with vasculitis, leading to increased permeability of protein trough the intestinal vessels. Another hypothesis is that inflammatory mediators may cause vasodilation, with a consequent increase in vascular permeability [5]. Ong et al. [7] reported a case of a patient with LPE, increased CA-125 and low levels of C3, as well as ascites and edema in the lower limbs as the initial presentation of systemic lupus erythematosus. In case 4, although the investigation has not been completed, the presence of livedo reticularis associated to high CA-125 levels and low levels of complement, with improvement of these parameters after the use of corticosteroids, suggest that it must be an autoimmune process, consistent with the hypothesis of vasculitis as a mechanism of protein loss in this case.
Besides been a demonstration of a variety of diseases, LPE may occur as an idiopathic disease [5], as in case 3, in which even after thorough investigation, an underlying disorder were not diagnosed. The treatment of LPE, as said before, primarily consists on resolution of underlying disease. However, even when the primary cause of protein loss is not identified, steroid therapy is an option, as seen in cases 3 and 4.
Protein-losing enteropathy by vascular disease (increased interstitial pressure)
Case 5
Female, 29 years old, diagnosed with Whipple's disease with enteric and lymph node involvement. The diagnosis was confirmed by upper endoscopy with duodenal biopsy and inguinal lymph node. Besides in use of sulfamethoxazole-trimethoprim since the last year, she kept diarrhea board and anasarca, associated with hypoalbuminemia (1.5mg/dL) and decreased levels of immunoglobulins. Scintigraphy with 99mTc-labeled human albumin showed protein loss in the right abdomen (terminal ileum / cecum / ascending colon). In addition to the protein loss, had elevated inflammatory markers (Table 4). Sulfamethoxazole-trimethoprim was suspended and a new treatment with ceftriaxone was started and maintained for 15 days, in addition to nutritional therapy with medium-chain triglycerides (MCT). There was improvement in bowel habits and gradual reduction of edema associated with the use of diuretic.
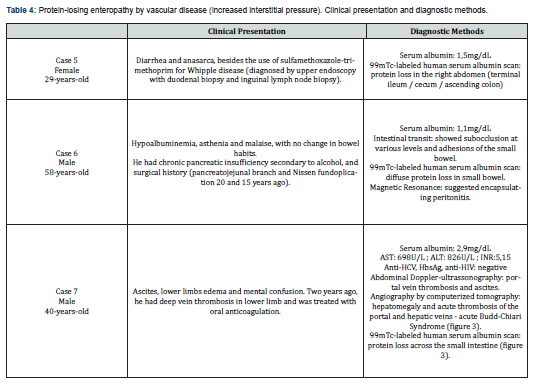
Case 6
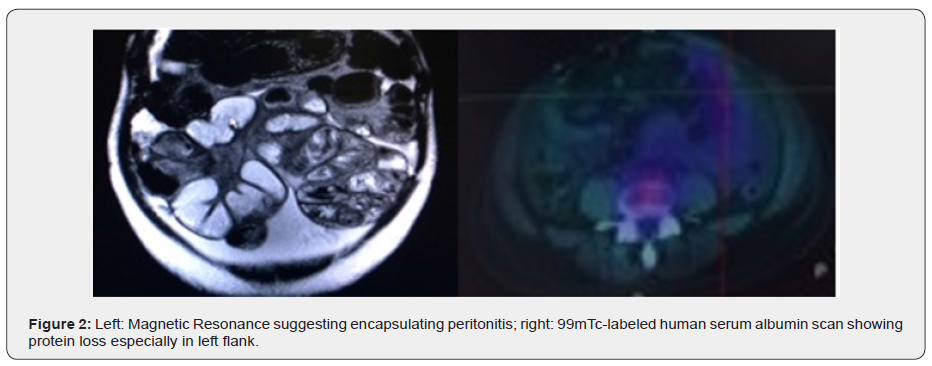
Male, 58 years-old, referred to our service because of hypoalbuminemia (serum albumin 1.15mg/dL). He had a history of chronic pancreatic insufficiency secondary to alcohol, diabetes mellitus, hypertension, and surgical history pancreatojejunal branch in 1992 and Nissen fundoplication in 1999. He complained of asthenia and malaise nonspecific, with no change in bowel habits. On physical examination, the patient presented lower limb edema without ascites. Intestinal transit showed subocclusion at various levels and adhesions of the small bowel. Due to the hypothesis of protein-losing enteropathy, scintigraphy with 99mTc-labeled human albumin was performed, and diffuse protein loss in small bowel was confirmed, in addition to diffuse bowel distension with air-fluid levels, suggesting a subocclusive bowel frame. Magnetic Resonance suggested the possibility of encapsulating peritonitis (Figure 2, Table 4). He was submitted to surgery, and pathology of the surgical specimen showed vascularized fibrous connective tissue fragments with diffuse infiltration of polymorphonuclear organization and hyaline fibrosis in the wall, confirming the diagnosis. Postoperatively return, the patient was without asthenia and edema, with serum albumin 3.1mg/dL.
Case 7
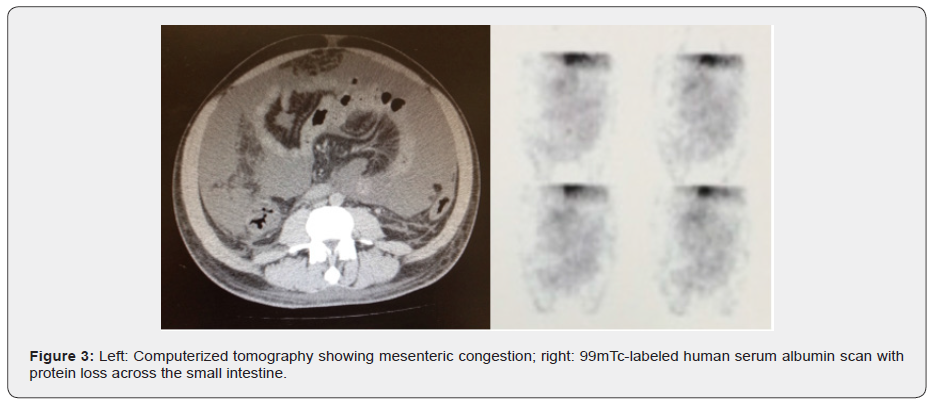
Male, 40 years-old, previously healthy, presented ascites, oliguria, lower limbs edema and mental confusion. He had a history of deep vein thrombosis in the right lower limb two years ago and received oral anticoagulation at the time. Laboratory tests showed: alb 2.9 mg/dL, BT 5.73 mg/dL, ALT 698 U/L, AST 826 U/L, INR 5.15, and serological tests for virus hepatitis B and C and HIV were negative. Abdominal USG-Doppler showed portal vein thrombosis and ascites. Angiography by computerized tomography showed hepatomegaly with acute thrombosis of the portal and hepatic veins, consistent with the diagnosis of acute Budd-Chiari Syndrome. A 99mTc-labeled human serum albumin scan was performed (Figure 3), which showed an important protein loss across the small intestine, compatible with protein-losing enteropathy of vascular origin (Table 4). The patient remained on worsening of liver function and died from septic shock.
Discussion
Physiologically, when proteins pass through the gastrointestinal mucosa, reaching its light, new proteins are synthesized in a compensatory manner. In normal situations, enteric protein loss accounts for only 1 to 2% of the proteinaceous shell, with albumin losing accounting for less than 10% of the total catabolism. In patients with PLE, this enteric albumin loss can reach 60% of the total albumin body reserve [1]. One of the first reports of PLE was a patient with congestive heart failure. The mechanism includes lymphatic obstruction due to increased central venous pressure (CVP) in these patients. A high pressure in lymphatic vessels would cause stroke of rich protein content into gastrointestinal tract light [3].
Although more commonly associated with congestive heart failure, any situation that leads to increased CVP may be associated with PLE by vascular disease as a complication. For example, constrictive pericarditis, non-cardiac valve diseases or other conditions as described above in cases 5, 6 and 7. Whipple's disease may be associated with hypoalbuminemia, usually secondary to intestinal malabsorption and malnutrition. However, the ascites is found only in 5% of cases in which there is a lymphatic system involvement [22-26]. However, although studies reveal up to 66% of cases with lymphadenopathy, PLE as a manifestation of Whipple's disease is not common. Case 5 exemplifies this situation, presenting with hypoalbuminemia secondary to PLE (confirmed by scintigraphy with labeled albumin).
Encapsulating peritonitis, first described in 1907 by O'Wtschinnikow [27], can be classified as idiopathic or secondary, the latter one being more common. While most commonly related to peritoneal dialysis, any inflammatory process, whether infectious or not, can lead to this complication. Its most frequent manifestation consists in abdominal pain associated with bowel subocclusion [27,28], being PLE an unusual presentation, as is the case 6. The surgical interventions led to a chronic inflammation of the peritoneum, forming a fibrous capsule surrounding the bowel. This imprisonment led to a persistent and ongoing inflammatory process, damaging the intestinal mucosa, increasing its permeability, but mostly, this process also caused a lymphatic obstruction, contributing to the PLE presented by this patient. In case 7, what was observed was an increased pressure venous secondary to portal and hepatic veins thrombosis, leading to a sub-acute increased lymphatic pressure. In this situation, treatment options would be the clearing of the portal system through TIPS or liver transplantation. Both were unfortunately not possible (Table 5).
Conclusion
Protein-losing enteropathy is a clinical condition that can complicate a variety of gastrointestinal and systemic diseases, with pathophysiological mechanism still poorly elucidated. The most commonly described causes in literature are inflammatory bowel disease, systemic lupus erythematosus and Fontan operation.
Here we describe a variety of uncommon etiologies of this disease and treatment approaches. It has been shown that the prognosis of these patients depends mainly on the primary disease and its specific treatment. But in some situations, where it is not possible to define the associated disease, protein loss can be stopped with measures based on its possible pathophysiological mechanisms.
References
- Furtado AK, Cabral VLR, Santos TN, Mansour E, Nagasako CK, et al. (2012) Giardia infection: Protein-losing enteropathy in an adult with immunodeficiency. World Journal of Gastroenterology 18(19): 2430-2433.
- Greenwald DA (2014) Gastroenteropatia perdedora de proteí In: Feldman, M, Friedman, LS, Brandt, LJ (Edt.), Sleisenger e Fordtran Gastroenterologia e Doenças do Fígado [tradução Alcir Costa Fernandes Filho et al]. Elsevier, Rio de Janeiro, Brazil, 1: 445-451.
- Umar, SB, Dibaise, JK (2010) Protein-Losing Enteropathy: Case Illustrations and Clinical Review. The American Journal of Gastroenterology 105(1): 43-49.
- Meadows, J, Jenkins K (2011) Protein-losing enteropathy: integrating a new disease paradigm into recommendations for prevention and treatment. Cardiology in the Young 21(4): 363-377.
- AL-Mogairen SM (2011) Lupus protein-losing enteropathy (LUPLE): A systematic review. Rheumatol Int 31(8): 995-1001.
- Gornisiewicz M, Rodriguez M, Smith JK, Saag K, Alarco GS (2001) Grand Rounds from International Lupus Centres Protein-losing enteropathy in a young African-American woman with abdominal pain, diarrhea and hydronephrosis. Lupus 10(12): 835-840.
- Ong CS, Cheah TE, Jasmin R, Yahya F, Sockalingam S (2013) Painless ascites and elevated CA125: initial presentation of lupus-associated protein-losing enteropathy. Lupus 22(11): 1174-1177.
- Gursu HA, Varan B, Erdogan I (2014) Use of oral budesonide in the management of protein-losing enteropathy due to restrictive cardiomyopathy. Cardiology in the Young 24: 764-766.
- F O A A Carneiro, L R Sampaio, L A R Brandão, L L B C Braga, F A C Rocha (2012) Protein-losing enteropathy as initial manifestation of systemic lupus erythematosus. Lupus 21(4): 445-448.
- Kuo-Chih Tseng, Bor-Shyang Sheu, Jenq-Chang Lee, Hong-Ming Tsai, Nan-Tsing Chiu (2005) Application of Technetium-99m-Labeled Human Serum Albumin Scan to Assist Surgical Treatment of Protein-Losing Enteropathy in Cronkhite-Canada Syndrome: Report of A Case. The American Society of Colon and Rectal Surgeons 48(4): 870-873.
- M Facchini, I Guldenschuh, J Turina, R Jenni, C Schalcher, C H Attenhofer Jost (2000) Resolution of protein-losing enteropathy with standar high molecular heparin and urokinase after Fontan repair in a patient with tricuspid atresia. J Cardiovasc Surg 41: 567-570.
- Tomohisa Fushimi, Yasuhumi Takahashi, Yuichiro Kashima, Kazuhiro Fukushima, Wataru Ishii, et al. (2005) Severe protein-losing enteropathy with intractable diarrhea due to systemic AA amyloidosis, successfully treated with corticosteroid and octreotid. Amyloid 12(1): 48-53.
- Jin-Kyeong Shin, Young-Hee Jung, Myoung-Nam Bae, In-Woon Baek, Ki-Jo Kim, et al. (2013) Successful treatment of protein-losing enteropathy due to AA amyloidosis with octreotide in a patient with rheumatoid arthritis. Mod Rheumatol 23(2): 406-411.
- Ferrante, M, Penninckx, F, De Hertach, G (2006) Protein-losing enteropathy in Crohn´s Disease. Acta Gastroenterol Belg 69: 384-389.
- Hyung Hun Kim, You Sun Kim, Kyung Sun Ok, Soo Hyung Ryu, Jung Hwan Lee, et al. (2010) Chronic Non-granulomatous Ulcerative Jejunoileitis Assessed by Wireless Capsule Endoscopy. Korean J Gastroenterol 56(6): 382-386.
- Seven G, Assaad A, Biehl T, Kozarek RA (2012) Use of anti-tumor necrosis factor-alpha monoclonal antibody for ulcerative jejunoileitis. World J Gastroenterol 18(36): 5135-5137.
- Enns R, Lay T, Bridges R (1997) Use of azathioprine for nongranulomatous ulcerative jejunoileitis. Can J Gastroenterol 11(6): 503-506.
- Diniz LMM, Silva MT, Lima MVM, Misson NR, Trocoli-Couto PHF (2009) Intestinal obstruction secondary to radiation enteritis. Rev Med Minas Gerais 19(2 Supl 3): S27-S31.
- Wang J, Yao D, Zhang S, Mao Q, Li Y, et al. (2014) Laparoscopic surgery for radiation enteritis. Journal of Surgical Research 194(2): 415-419.
- Harb AH, Fadel CA, Sharara AI (2014) Radiation Enteritis. Curr Gastroenterol Rep 16(5): 383.
- Magro PMH (2014) Oclusión intestinal secundaria A enteritis por radiación. Reporte de caso. Revista de Gastroenterología de Mé
- Dobbins WO (1987). Whipple's disease. Springfield, Illinois, USA.
- Kelly JJ, Weisinger BB (1963) The arthritis of Whipple´s disease. Arthritis Rheum 6: 615-332.
- Fleming JL, Wiesner RH (1988) Whipple’s disease: clinical, biochemical and histopathologic features and assessment of treatment in 29 patients. Mayo Clin Proc 63(6): 539-551.
- Maizel H, Ruffin JM, Dobbins WO (1993) Whipple’s disease: a review of 19 patients from one hospital and a review of the literature since 1950. 1970. Medicine (Baltimore) 72(5): 343-355.
- Vital-Durand D, Lecomte C, Cathébras P (1997) Whipple disease: clinical review of 52 cases. Medicine (Baltimore) 76(3): 170-184.
- Tannoury JN, Abboud BN (2012) Idiopathic sclerosing encapsulating peritonitis: abdominal cocoon. World J Gastroenterol 18(17): 1999-2004.
- Spence R, Gillespie S, Loughrey M, Gardiner K (2013) Encapsulating peritoneal sclerosis - A 5 year experience. Ulster Med J 82(1): 11-15.






























