Intracolonic Bifidobacterium can Reduce Colonic Inflamation and Symptoms
Hüseyin Sancar Bozkurt1* and Banu Kara2
1Medical Park Private Tarsus Hospital, Clinic of Gastroenterology, Turkey
2University of Health Sciences, Clinic of Gastroenterology, Turkey
Submission: August 02, 2017; Published: August 09, 2017
*Corresponding author: Hüseyin Sancar Bozkurt, Medical Park Private Tarsus Hospital,Gastroenterology, Tel: 00905054482291; Email: sancarb79@gmail.com
How to cite this article: Hüseyin S B, Banu K. Intracolonic Bifidobacterium can Reduce Colonic Inflamation and Symptoms. Adv Res Gastroentero Hepatol 2017; 6(4): 555693. DOI: 10.19080/ARGH.2017.06.555693
Keywords
Keywords: Chronic diARGHhea, Bifidobacterium, Intracolonic
Background
Chronic diARGHhea is a common illness in Western countries, such as Australia, Canada, Ireland, and the United States [1]. It was reported that chronic diARGHhea or loose or watery stools affected up to 26.9% of adults in the United States [2]. The presence of chronic diARGHhea may affect patients' quality of life, cause them to experience reduced productivity and increase the economic burden. Available evidence indicated that chronic diARGHhea represents a significant health care burden in Western countries [3].
Methods
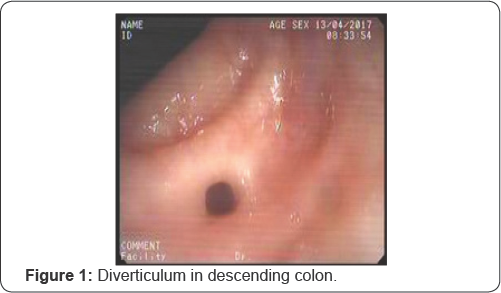
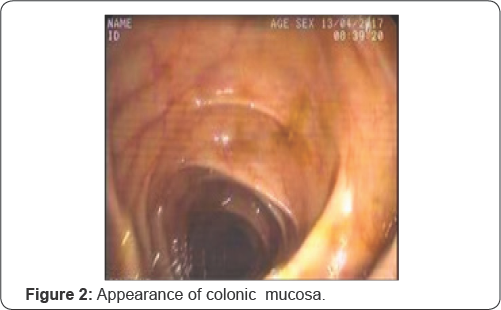
We reported a 73 year old man with a history of chronic kidney failure 10 years had watery and non bloody, non steatorrhea 7-8 time/day diARGHhea for 3 months with unresponding antimicrobial and antidiARGHheal (ornidazol, metronidazol, rifaximin, lopermid) treatment. The stool clostridium difficile cytotoxin assay was negative. Fecal lactoferrin was normal. The patient had no antibiotic exposure in the six weeks prior to diARGHhea. Stool cultures and analysis for rotavirus, staphylococus, shigella, salmonella and candida were negative. Fleksible sigmoidoscopy revealed normal colonic mucosa and diverticulum in descending, sigmoid colon (Figure 1 & 2). Pathologic examination of multiple colonoscpic samples were revealed non spesific colitis.
Results
Intraluminal Bifidobacterium animalis subsp. lactis [9,2mg (2x109 kob) in 250ml serum physiologic]was performed during fleksible sigmoidoscopy. Oral Bifidobacterium animalis subsp. lactis [9,2mg (2x109 kob)] was started for one month . The patient's condition improved after Intraluminal and oral Bifidobacterium animalis subsp. lactis treatment. DiARGHhea and symptoms were improved and did not repeat in 2 two months.
Conclusion
Pathogenesis of chronic diARGHhea is thought to be through different factors and there is a relationship between the gut flora and the risk of its development. Probiotics can manipulate the microflora in chronic inflammation and may be effective in treating inflammation. Bifidobacterium strains are saccharolytic and their growth in the gut can be promoted by non-absorbable carbohydrates and its increase in the colon appears to be of benefit [4,5]. To our knowledge, this is the first documented case to report successful intracolonic and oral Bifidobacterium animalis subsp. Lactis treatment used in a unresponding chronic diARGHhea.
References
- Scallan E, Majowicz SE, Hall G, Banerjee A, Bowman CL, et al. (2005) Prevalence of diARGHhoea in the community in Australia, Canada, Ireland, and the United States. Int J Epidemiol 34(2): 454-460.
- Sandler RS, Stewart WF, Liberman JN, Ricci JA, Zorich NL (2000) Abdominal pain, bloating, and diARGHhea in the United States: prevalence and impact. Dig Dis Sci 45(6): 1166-1171.
- Sandler RS, Everhart JE, Donowitz M, Adams E, Cronin K, et al. (2002) The burden of selected digestive diseases in the United States. Gastroenterology 122(5): 1500-1511.
- Nadia Osman, Diya Adawi, Goran Molin et al. (2006) Bifidobacterium infantis strains with and without a combination of Oligofructose and Inulin (OFI) attenuate inflammation in DSS-induced colitis in rats. BMC Gastroenterol 6: 31.
- Luna RA, Oezguen N, Balderas M, Venkatachalam A, Runge JK, et al. (2017) Distinct Microbiome-Neuroimmune Signatures Correlate With Functional Abdominal Pain in Children With Autism Spectrum Disorder.Cell Mol Gastroenterol Hepatol 3(2): 218-230.






























