Effect of Bifidobacterium Longum Bb536 Plus Lactoferrin in the Treatment of Irritable Bowel Syndrome. A Double Blind Clinical Trial
Ivano Biviano, Sara Rossi, Daria Piacentino, Valeria Alvino, Enrico S Corazziari, Danilo Badiali* and Gastroenterology A
Department of Internal Medicine and Medical Speciality, Univerisity La Sapienza, Italy
Submission: June 27, 2017; Published: July 27, 2017
*Corresponding author: Danilo Badiali, Department of Internal Medicine and Medical Speciality, Univerisity La Sapienza, Italy, Tel: 00390649978384; Email: danilo.badiali@uniroma1.it
How to cite this article: Ivano B, Sara R, Daria P, Valeria A, Enrico S C,et al. Effect of Bifidobacterium Longum Bb536 Plus Lactoferrin in the Treatment of Irritable Bowel Syndrome. A Double Blind Clinical Trial. Adv Res Gastroentero Hepatol 2017; 6(4): 555691. DOI: 10.19080/ARGH.2017.06.555691
Abstract
Background: Presence of Bifidobacteria in patients with Irritable Bowel Syndrome (IBS) is decreased and their use as probiotics has been indicated to re-establish eubiosis in this condition. Bifidobacterium Longum BB-536 (BB-536) has favorable direct effect on epithelial adherence, reinforcement of tight junctions, stimulation of IgA production and of cell-mediated immunity, anti gram-negative and pathogenic microbe action. Lactoferrin acts as prebiotic for bifidobacteria (bifidogenic effect) and has antinflammatory, antioxidant, antibacterial, and antiviral activity.
Aim: An exploratory study with primary aim to evaluate in IBS patients the effectiveness of Bifidobacterium Longum BB-536+Lactoferrin on abdominal pain, bloating and on the severity of IBS (IBS-SS).
Protocol: Rome III IBS patients of both genders (age 18-65 yrs) were consecutively recruited at a tertiary referral center. Patients, after registering their symptoms for one basal run-in week on a first daily diary card, were randomly and blindly allocated to treatment with active drug, a capsule containing BB-536 3 x 109 UFC+Lactoferrin 50 mg or an identical placebo for 2 weeks. During the treatment period patients registered symptoms on a second daily diary card. IBS-SS was reported at the end of the run-in period and of the two weeks of treatment. Self-perceived intensity of abdominal pain and bloating was reported on a visual analogue scale (VAS), ranging 1-100mm during the week of the run-in period and the last 10 days of the treatment.
Results: Thirty-five patients were recruited; one patient dropped out after the first daily diary card completion, thus 34 final outpatients completed the study trial (18, F15 mean age 44.7±13.1 yrs, in the BB 536+Lactoferrin arm and 16, F11 mean age 46.6±14.4yrs, in the placebo arm). Demography, BMI, and IBS-SS at basal period did not differ between the two groups. In comparison to basal run-in period the IBS-SS improved significantly (p<0.007) after BB536+Lactoferrin and not significantly after placebo. In comparison to placebo abdominal pain and bloating VAS severity improved significantly after BB536+Lac (p<0.007; p<0.03).
Conclusion: IBS patients benefit of two-week treatment with Bifidobacterium Longum BB536 enriched with Lactoferrin. The main benefit is on the reduction of abdominal pain severity. Results of this exploratory double-blind trial are promising but confirmation with a long term, larger and more differentiated population is desirable
Keywords: Abdominal Bloating; Abdominal Pain; Bifidobacterium Longum BB-536; Irritable Bowel Syndrome; Lactoferrin Running Head Bifidobacterium Longum BB536 plus Lactoferrin in IBS
Background
Several studies have reported that the Intestinal microbiota of IBS patients is altered and differs from healthy people. Such condition of dysbiosis, associated with physical or psychological stress, may increase the intestinal bARGHier permeability, hypersensitize the afferent fibers of the enteric nervous system, and thus contributing to the clinical manifestations of the syndrome. Bifidobacteria are normally present in the human intestine where they exert a fundamental role. Presence of Bifidobacteria in patients with Irritable Bowel Syndrome (IBS) is decreased and their use as probiotics has been indicated to re-establish eubiosis in this condition. Previous Japanese studies have reported the efficacy of the probiotic Bifidobacterium Longum BB-536 to normalize bowel frequency and stool consistency in young adult [1,2] and elderly subjects [3]. Bifidobacterium Longum BB536 has favorable direct effect on epithelial adherence [4], reinforcement of tight junctions [5], stimulation of IgA production [6] and of cell-mediated immunity [7], anti gram-negative and pathogenic microbe action. A recent controlled clinical study has shown the efficacy of BB536, combined with two other Bifidobacteria, on IBS symptoms in pediatric patients [8]. Lactoferrin is a glycoprotein, present in mother's milk and in several other exocrine secretions as saliva tears, bile, gastric and pancreatic. It is released from neutrophils during the inflammation process. Lactoferrin has antinflammatory, antioxidant, antibacterial, and antiviral activity [9-11], in addition to the high resistance of its peptide components to degradation effect of gastroenteric proteases, it reaches the colon where it electively exerts a bifidogenic effect favoring the development of Bifidobacteria [11,12]. To our knowledge no clinical controlled studies have been performed to assess the efficacy of BB536 and lactoferrin on symptoms of IBS adult patients. AIM Primary aim of this preliminary study is to evaluate the effectiveness of Bifidobacterium Longum BB 536+Lactoferrin on abdominal pain, bloating and the global IBS symptomatology in IBS patients.
Protocol and Methods
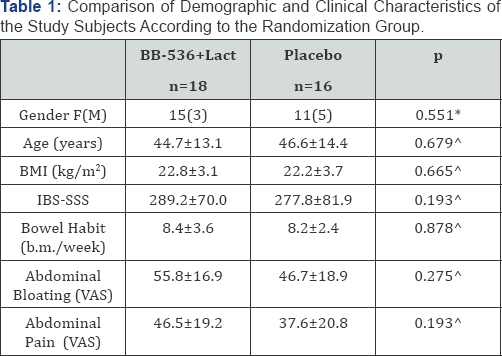
Outpatients of a tertiary referral centers presenting with abdominal pain and bowel alterations matching the were consecutively assessed. Italian speaking patients matching the Rome III IBS [13], and with no structural alterations, systemic diseases, psychiatric disorders, cognitive impairment, history of drug or excessive alcohol consumption, and BMI>30 were recruited. After formal acceptance of informed consent patients were asked not to assume any drug for one basal week period during which they registered their symptoms on a first daily diary card. At the end of this run-in wash-out period, patients were randomly and blindly allocated to treatment with active drug, a capsule containing BB536 3 x 109 UFC+Lactoferrin 50mg or identical placebo for 2 weeks. During the treatment period patients registered symptoms on a second daily diary card. Irritable Self-perceived intensity of abdominal pain and bloating was reported on a visual analogue scale (VAS), ranging 1-100mm during the week of the run-in period and the last 10 days of the treatment. In addition patients filled in the Bowel Syndrome Symptom Severity Scale (IBS-SS) questionnaire at the end of the run-in period and of the two weeks of treatment. IBS-SS score assesses severity of symptoms being 0-75 low-absent and >300 severe [14]. Descriptive continuous variables are expressed as mean (m) and standard deviation (sd). The comparison between different study periods are performed using the Mann-Whitney U-test for continuous variables and X2 test f°r dichotomic variables. A value of p<0.05 is considered as the limit for statistical significance (Table 1).
Results
Thirty-five patients were recruited; one patient dropped out after first daily diary card completion, thus 34 final outpatients completed the study trial (18, F15 mean age 44.7±13.1yrs, in the BB 536+Lactoferrin arm and 16, F11 mean age 46.6±14.4yrs, in the placebo arm). Demography, BMI, and IBS-SS score at basal period did not differ between the two groups (Table 1). In comparison to basal run-in period the reduction of IBS-SS score was 50(p<0.007) after BB536+Lac and 38 (n.s.) after placebo. Compared to placebo, pain and bloating VAS was significantly reduced after BB536+Lac (Figure 1). DiARGHhea-, Constipation-, Mixed- and Unspecified- predominant IBS subtypes did not have different symptomatology response.

Discussion
The main result of this exploratory study indicates that BB536 plus Lactoferrin combination induces an overall improvement of IBS symptomatology, more specifically of abdominal pain severity. It is of note that patients benefit of this improvement in the short period of two-week treatment. It can be hypothesized that such a rapid efficacy response can be due to the specific properties of BB536. In comparison to other bifidobacteria, BB536 has greater epithelial adhesivity and a long term proliferation activity after the ingestion [4,15]. BB536 has been shown to normalize the dysbiotic condition induced by antibiotic treatment [16,17], and It is, thus, likely that this probiotic plays a relevant role also in normalizing the altered microbiota present in IBS patients. In addition BB536 has been shown to reinforce the epithelial bARGHier and to decrease intestinal permeability, increasing occludin and zonulin production of the tight junctions [18]. Lactoferrin can contribute to obtain a more effective and rapid improvement of IBS symptoms for its antinflammatory and antioxidant actions as well as for its bifidogenic potential of enhancing vitality and proliferation of BB536 in the intestinal lumen. The favorable effect of BB536 plus Lactoferrin combination on pain severity may derive from its antinflammatory action on the mucosal immune system and thus a lesser afferent nerve fiber activation. Such antinflammatory action of BB536 in reducing the stimulation of the mucosal immune cells is obtained both directly and indirectly, reinforcing the epithelial bARGHier and has been previously demonstrated in active ulcerative colitis patients [18]. Undoubtedly, the strength of the present study is the randomized, double blind parallel group protocol of the trial. Nonetheless the results of this study must be viewed as preliminary due to the limited number of patients and for its short duration. In addition these observations have been made in selected patients of a tertiary referral center and may not be representative of the larger and unselected IBS population.
In conclusion, IBS patients appear to benefit of a two-week treatment with Bifidobacterium Longum BB536 enriched with Lactoferrin. The main benefit is on the reduction of abdominal pain severity. Results of this exploratory double-blind trial are promising but confirmation with a long term, larger and more differentiated population is desirable.
References
- Ogata T, Nakamura T, Anjtsu K, Yaeshima T, Takahashi S, et al. (1997) Effect of Bifidobacterium longum BB536 administration on the intestinal environment, defecation frequency and fecal characteristics of human volunteers. Bioscience Microflora 16(2): 53-58.
- Yaeshima T, Takahashi S, Ogura A (2001) Effect of non-fermented milk containing Bifidobacterium longum BB536 on the defecation frequency and fecal characteristics in healthy adults. J Nutr Food 4: 1-6.
- Seki M, Igarashi M, Fukuda Y (1978) The effect of Bifidobacterium cultured milk on the regularity among an aged group. J Jpn Soc Nutri Food Sci 4: 379-387.
- Tomada T, Nakano Y, Kageyama T (1986) The variation and adherence of the species of bifidobacterium in intestine during oral administration of bifidobacterium. Medicine and Biology 113: 128-138.
- Takeda Y, Nakase H, Namba K, Inoue S, Ueno S, et al. (2009) Upregulation of T-bet and tight junction molecules by Bifidobacteriumlongum improves colonic inflammation of ulcerative colitis. Inflamm Bowel Dis 15(11): 1617-1618.
- Gill HS, Rutherfurd KJ, Cross ML (2001) Dietary probiotic supplementation enhances natural killer cell activity in the elderly: an investigation of age-related immunological changes. J Clin Immunol 21(4): 264-271.
- Nagao F, Nakayama M, Muto T, Okumura K (2000) Effects of a fermented milk drink containing Lactobacillus casei strain Shirota on the immune system in healthy human subjects. Biosci Biotechnol Biochem 64(12): 2706-2708.
- Giannetti E, Maglione M (2017) Alessandrellathe variation and adherence of the species of bifidobacterium in intestine during oral administration of bifidobacterium A, et al A Mixture of 3 Bifidobacteria Decreases Abdominal Pain and Improves the Quality of Life in Children With Irritable Bowel Syndrome: A Multicenter, Randomized, DoubleBlind, Placebo-Controlled, Crossover Trial. J Clin Gastroenterol 51: e5- e10.
- Jenssen H, Hancock RE (2009) Antimicrobial properties of lactoferrin. Biochimie 91(1): 19-29.
- Goldman IL, Deikin AV, Sadchikova ER (2010) Human Lactoferrin Can Be Alternative to Antibiotics. Proceedings of the World Medical Conference, pp. 27-38.
- Saito H, Miyakawa H, Ishibashi N, Tamura Y, Hayasawa H, et al. (1996) Effect or iron-free and metal-bound forms of lactoferrin on the growth of bifidobacteria, E. coliand S. aureus. Bioscience Microflora 15(1): 1-7.
- Rahman MU, Kim WS, Tanaka T, (2006) Lactoferrin effects on the growth of bifidobacteria. FFI Journal 9: 763-70.
- Drossman DA, Corazziari E, Delvaux (2006) Rome III: The functional gastrointestinal disorders. Degnon Associates Inc., McLean, USA, pp. 963-969.
- Francis CY, Morris J, Whorwell PJ (1997) The irritable bowel severity scoring system: a simple method of monitoring irritable bowel syndrome and its progress. Aliment Pharmacol Ther 11(2): 95-402.
- Crociani J, Grill JP, Huppert M, Ballongue J (1995) Adhesion of different bifidobacteria strains to human enterocyte-like Caco-2 cells and comparison with in vivo study. Lett Appl Microbiol 21(3): 146-148.
- Colombel JF, Cortot A, Neut C, Ramond C (1987) Yoghurt with Bifidobacterium longum reduces erythromycin-induced gastrointestinal effects. Lancet 2(8549): 43.
- Orrhage K, Sjostedt S, Nord CE (2000) Effect of supplements with lactic acid bacteria and oligofructose on the intestinal microflora during administrationof cefpodoxime proxetil. J Antimicrob Chemother 46(4): 603-612.
- Tamaki H, Nakase H, Inoue S, Kawanami C, Itani T, et al. (2016) Efficacy of probiotic treatment with Bifidobacterium longum 536 for induction of remission in active ulcerative colitis: A randomized, double-blinded, placebo-controlled multicenter trial. Dig Endosc 28(1): 67-74.






























