Does Early Caffeine Therapy Effect Patent Ductus Arteriosus Outcomes in Very Low Birth Weight Infants?
April Jones1*, Ekta Patel2, Kyle Carey3, Christina Jreisat4, Richard Kampanatkosol5 and Christine Sajous6
1Neonatology Fellow, Neonatology, Loyola University Medical Center, USA
2 Pediatric Resident, Pediatrics, Loyola University Medical Center, USA
3Statistician, Clinical research office, Loyola University Chicago Health Sciences Division, USA
4 Medical student, Stritch School of Medicine, USA
5 Neonatologist, Neonatology, Advocate Christ Medical Center, USA
6Neonatologist, Neonatology, Loyola University Medical Center, USA
Submission: February 08, 2017; Published: April 18, 2017
*Corresponding author: April Jones, Neonatology Fellow, Neonatology, Loyola University Medical Center, 2160 South 1st Ave, Maywood, IL 60153-3328, USA.
How to cite this article: April J, Ekta P, Kyle C, Christina J, Richard K et al . Does Early Caffeine Therapy Effect Patent Ductus Arteriosus Outcomes in Very Low Birth Weight Infants?. Acad J Ped Neonatol. 2017; 4(3): 555696. DOI: 10.19080/AJPN.2017.04.555696
Abstract
Objective:To determine if early versus late use of caffeine is associated with a reduction in the incidence of clinically significant patent ductus arteriosus requiring treatment in VLBW infants
Study Design: Retrospective review of 493 neonates admitted to the NICU with a birth weight ≤1500grams treated with early (≤3 days of life) or late (>3 days of life) caffeine. Patient outcomes were compared using Student’s t-tests, Wilcoxon’s rank-sum tests, chi-squared tests and multivariable logistic regression.
Result: Infants who received early caffeine had lower odds of patent ductus arteriosus (P<0.001) and lower odds of medical treatment or surgical ligation (P<0.001). The early caffeine group also had lower odds of being diagnosed with bronchopulmonary dysplasia (P=0.02) or severe (grade 3-4) intraventricular hemorrhage (P<0.001). No difference was seen in the odds of retinopathy of prematurity, necrotizing enterocolitis or spontaneous intestinal perforation.
Conclusion: Infants who received early caffeine had decreased odds of patent ductus arteriosus requiring medical treatment and/or surgical ligation.
Keywords: Patent ductus arteriosus; Caffeine therapy; Very low birth weight infant; Bronchopulmonary dysplasia; Neonatal Outcomes
Abbrevations: PDA: Patent Ductus Arteriosus; BPD: Bronchopulmonary Dysplasia; IVH: Intraventricular Hemorrhage; NEC: Necrotizing Enterocolitis; ROP: Retinopathy Of Prematurity; ELBW: Extremely Low Birth Weight Infants; IV: Intravenous; VLBW: Very Low Birth Weight; CAP: Caffeine For Apnea Of Prematurity; DOL: Day Of Life; SIP: Spontaneous Intestinal Perforation; Echo: Echocardiogram; BMP: Basic Metabolic Panel; CBC: Complete Metabolic Panel; PMA: Post-Menstrual Age; Wga: Weeks Gestational Age; IQR: Interquartile Range; PGE2: Prostaglandin 2; COX: Cyclooxygenase; G: Grams; SD: Standard Deviation
Introduction
In the developing fetus, the ductus arteriosus is a vessel which sends blood from the pulmonary artery to the proximal descending aorta, thereby shunting blood from the right ventricle away from the fluid filled fetal lungs. In the first 3 days of life the ductus arteriosus is a physiologic shunt found in both healthy term and preterm infants [1]. However, if the ductus remains open, it now becomes a patent ductus arteriosus (PDA) and can have varying degrees of clinical consequence depending on the degree of left to right shunting [2]. The incidence of persistent PDA in term infants is 57 in 100,000 live births. Whereas, a persistent PDA can be found in 1 in every 3 preterm infants with a birth weight between 500 and 1500 grams [1,3-5]. The persistence of a PDA has been historically associated with increased neonatal morbidity and mortality [6,2].
A PDA with significant left to right shunting in preterm infants has been associated with increased risk of bronchopulmonary dysplasia (BPD), intraventricular hemorrhage (IVH), necrotizing enterocolitis (NEC) and retinopathy of prematurity (ROP) [7]. However, the indications for treatment of a PDA remain controversial [8]. Spontaneous permanent closure of the PDA occurs in 34% of extremely low birth weight (ELBW) neonates 2 to 6 days postnatal [9]. Of those PDAs that do not close, 60-70% need either medical treatment and/or surgical ligation [10]. Treatment is intended to prevent increased morbidity and mortality.
There are 4 major forms of PDA treatment. These include no intervention, conservative medical management, intravenous (IV) prostaglandin inhibitors and surgical ligation. Historically, waiting for spontaneous closure in preterm infants was reported to be associated with an increased frequency of BPD in survivors [11]. Recent research questions this notion [12,13]. Conservative medical management includes fluid restriction and diuretics [13- 15]. IV prostaglandin inhibitors have significant risks of morbidity and mortality. These include risk of intestinal perforation [16,17] and acute renal insufficiency [3,18]. Surgical ligation is usually reserved for PDAs refractory to medical management or infants too unstable to be treated with IV prostaglandin inhibitors [7]. Given the adverse outcomes associated with clinically significant PDAs and the risk of PDA treatment, therapies targeted at reducing PDAs in VLBW infants would be of significant value.
The Caffeine for Apnea of Prematurity (CAP) trial published in 2006 was a randomized, placebo controlled, multicenter trial of caffeine studying short and long term efficacy and safety of methylxanthine (caffeine) treatment for apnea of prematurity in very low birth weight (VLBW) infants [19]. Apnea of prematurity is a common diagnosis in preterm infants with approximately 84% of neonates less than 1000 grams at birth and 25% of neonates less than 2500 grams experiencing apnea during the newborn period [20-22]. The percent of VLBW infants exposed to caffeine therapy increased from 43% in 1999 to 73% in 2010 [23]. In the CAP trial, infants allocated to receive caffeine were found to have lower incidence of BPD when compared with control infants. Unexpectedly, in post hoc analysis, infants assigned to the caffeine group were also significantly less likely to undergo therapy to close a PDA than were the infants in the control group [19]. Further studies done by Dobson & Patel et al. [5] also had similar secondary outcomes, showing PDA requiring treatment was less frequent in the early versus late caffeine therapy group in VLBW infants [2,3,5]. Lodha et al. [20] retrospectively found early prophylactic use of caffeine was associated with a reduction in the rates of BPD and PDA. Each of these studies concluded a possible association between early caffeine and decreased incidence of PDA.
Given its therapeutic advantages, cost effectiveness and low side effect profile [24]. Optimizing the use of caffeine has been widely studied in VLBW infants. We performed a single center, retrospective review of VLBW (≤1500grams) infants receiving early caffeine (≤DOL 3) versus late caffeine (>DOL 3) to compare the incidence of clinically significant PDA requiring medical management and/or surgical ligation. Our primary hypothesis was VLBW infants who received early caffeine treatment would have a decreased incidence of clinically significant PDA, and therefore require less medical management and/or surgical ligation compared to infants who received late caffeine. Our secondary outcomes included evaluating early versus late caffeine treatment and the incidence of BPD, NEC, and spontaneous intestinal perforation (SIP), IVH and ROP.
Subjects and Methods
Patient population
This study is a retrospective chart review of neonates admitted to the neonatal intensive care unit at Loyola University Medical Center in Maywood, Illinois from June 2008 through June 2015. A list of infants meeting criteria was generated using Filemaker Pro dataset and individual charts were reviewed using electronic medical records. Infants were eligible for evaluation of primary and secondary outcomes if they met the following inclusion criteria:
1) birth weight ≤1500grams (VLBW) and
2) treatment with caffeine during hospitalization.
Exclusion criteria included death prior to discharge, major congenital anomalies, incomplete charting and no caffeine use during hospitalization. This study was approved by Loyola University Medical Center Institutional Review Board.
Definitions
Patients were divided into 2 groups (early versus late) based upon day of initiation of caffeine therapy. Early caffeine therapy defined as DOL 3 or before and late caffeine therapy defined as DOL 4 or later. We selected DOL 3 as the cut off based on the median day of caffeine initiation in the CAP trial [19]. A loading dose of 20mg/kg of caffeine followed by 8-10mg/kg daily dose was initiated at the discretion of the attending neonatologists based on clinical indications.
Our primary outcomes were incidence of PDA, incidence of PDA requiring medical management, incidence of PDA requiring both medical and surgical management and surgical ligation alone. Since an echocardiogram (echo) was obtained only if the infant showed clinically significant symptoms attributed to the PDA, not all VLBW infants had an echo during hospitalization. For any echo read with a range for PDA size, the larger size was used for consistency. PDA was deemed clinically significant only if treatment was necessary for closure either medically and/or surgically. Medical management included use of either indomethacin or ibuprofen. Treatment included 3 doses given 24hours apart unless side effects limited dosing. During the study period, our unit consisted of the same attending neonatologists who used the same strategy for PDA management and caffeine initiation. However, in April of 2015, based on current research data, caffeine was initiated on admission in all VLBW infants. Routine screening with complete blood count (CBC) for thrombocytopenia and basic metabolic panel (BMP) for renal insufficiency were done on all patients prior to treatment. Surgical ligation was done in the NICU by a cardiovascular surgeon.
BPD was defined as the requirement of any supplemental oxygen at postmenstrual age (PMA) of 36 weeks or discharge with oxygen. Additionally, we identified infants of mothers who received prenatal dexamethasone and postnatal steroid use of hydrocortisone and/or dexamethasone. All infants ≤32 weeks gestational age (wga) received head ultrasounds, read by attending radiologists, at 7 days and 28 days to screen for IVH and eye exams, performed by an ophthalmologist, for evaluation of ROP.NEC and SIP diagnosis was made by attending neonatologist and treated appropriately.
Statistical meta-analysis
Patient characteristics and outcomes were compared between the early and late caffeine treatment groups. Continuous variables were analyzed using Student’s t-tests, or Wilcoxon’s rank-sum tests when appropriate. Categorical variables were analyzed using chi-squared tests.
Multivariable logistic regression was used to model much relevant morbidity by timing of caffeine therapy following preterm birth. Multivariable models controlled for potential confounding effects of gestational age on PDA. Gender was not found to be a potential confounder.
Results
From June 2008 through June 2015, there were 661 infants weighing ≤1500grams at birth admitted to Loyola University Medical Center NICU, of which 493 met criteria. Of the 168 infants excluded, 24% died before hospital discharge, 18% were transferred to other institutions, 10% had congenital anomalies, 39% received no caffeine (10% of total patients reviewed) and 9%had poor documentation (Figure 1).

Patient characteristics
We divided the cohort into 2 groups based on initiation of caffeine. There were more infants in the early caffeine group (75% vs 25%) and they were significantly more mature (28wga vs 26wga, P<0.0001) and larger (1063g vs 810g, P<0.001) than the late caffeine group. Of the 368 infants in the early caffeine group, the median day of caffeine initiation was DOL 1 (IQR 0,1). Of the 125 infants in the late caffeine group, the median day of caffeine initiation was DOL 19 (IQR 7,28). The mean duration of caffeine therapy was similar between both groups (45 days vs 44 days). Infants discharged home with caffeine included 20 in the early caffeine group and 6 in the late caffeine group. There were no significant differences in out born infants (P=0.08) or race (P=0.64), but there were significant differences in sex (52% vs 65% males, P=0.02) and antenatal steroid use (90% vs 82%, P<0.01) (Table 1).

g: grams, SD: standard deviation, DOL: day of life, IQR: Interquartile range. Frequency and incidence are provided for each outcome stratified by caffeine therapy group. Continuous variables were analyzed using Student’s t-tests, or Wilcoxon’s rank-sum tests when appropriate. Categorical variables were analyzed using chi-squared tests
Neonatal outcomes
Infants who received early caffeine therapy had significantly decreased odds of having PDA and PDA requiring medical management and/or surgical ligation (P<0.0001). Infants in both groups received their first echo on a mean DOL 6 (early group) and DOL 4 (late group) (P=0.0005). Of the early group infants, 36% versus 10% of the late group had no echo (P<0.0001). Of the infants who did not receive an echo in the early group, 35% were ≥29wga and 65% ranged from 26-28wga. This is in contrast to the late group where all infants who did not receive echo were ≥29wga (median 29 vs 31, P=0.003). More infants and more immature infantsreceivedno echo in the early caffeine group than the late caffeine group (P<0.0001). This suggested lower incidence of clinically significant PDA in the early group (Table 2).
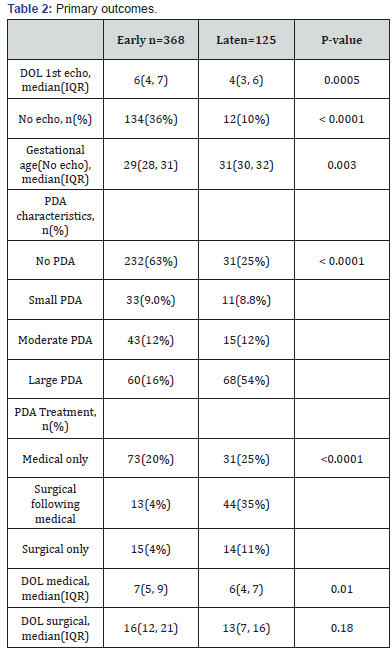
IQR: interquartile range, PDA: patent ductus arteriosus, DOL: day of life. Frequency and incidence are provided for each outcome stratified by caffeine therapy group. Continuous variables were analyzed using Student’s t-tests, or Wilcoxon’s rank-sum tests when appropriate. Categorical variables were analyzed using chi-squared tests.
Infants who received early caffeine were significantly more likely to have no PDA than those in the late caffeine group (63% vs 25%, P<0.0001). Significantly more infants in the late group had largePDAs versus those in the early caffeine group (54% vs16%, P<0.0001). After controlling for gestational age, timing of caffeine therapy was found to be significantly associated with PDA (P<0.0001), with infants given caffeine late having 2.5 times higher odds of having PDA than infants given caffeine early (Table 3). Incidence of small and moderate PDA was essentially the same between the two groups, with small PDA being 9% vs 8% and the moderate PDA being 11% vs 12% (early vs late).The median DOL of medical treatment for PDA was similar between the two groups at 7 days for the early group and 6 days for the late group (P=0.01). Surgical ligation occurred at a median DOL 16 and 13 for the early and late groups respectively (P=0.18) (Table 2).
After controlling for gestational age, timing of caffeine therapy was found to be significantly associated with medically treated PDA (P<0.0001). Infants given caffeine late had a 2.8 times higher odds of receiving medical management for PDA than infants given early caffeine (Table 3). Infants who received early caffeine were significantly less likely to receive surgical ligation (8% vs 46%, P<0.0001). Infants given caffeine late had 4.9 times higher odds of receiving surgical ligation for PDA (P<0.0001) (Table 3).
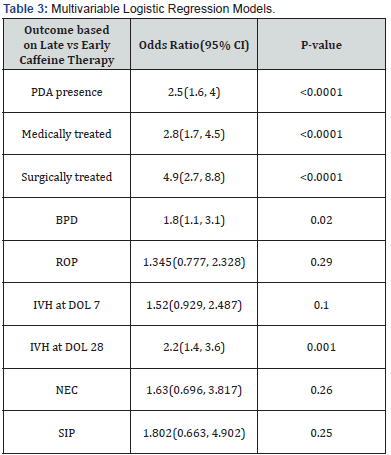
PDA: patent ductus arteriosus, BPD: bronchopulmonary dysplasia, ROP: retinopathy of prematurity, IVH: intraventricular hemorrhage, NEC: necrotizing enterocolitis, SIP: spontaneous intestinal perforation Multivariable logistic regression was used to control potential confounding effects of gestational age. Gender was not found to be to be a potential confounder.
We also evaluated common neonatal morbidities (Table 4). Infants in the early caffeine group received less mean days of mechanical ventilation (1day vs 25days, P<0.0001). Postnatal steroid use was significantly lower in the early caffeine group (9% versus 57%, P<0.0001). 66% of infants in the late group compared to only 31% in the early group were diagnosed with BPD. After controlling for gestational age, timing of caffeine therapy was found to be significantly associated with BPD (P<0.0001), with infants given caffeine late having 1.8 times higher odds of having BPD than infants given caffeine early (Table 3).
After controlling for gestational age, timing of caffeine therapy was not associated with severe IVH (grade 3-4) at DOL 7 (P=0.10) but was found to be significantly associated with severe IVH at DOL 28 (P=0.001), with infants given caffeine late having 2.2 times higher odds of having severe IVH at DOL 28 (Table 3).
After controlling for gestational age, timing of caffeine therapy was found to not be associated with ROP (P=0.29), SIP (P=0.25) or NEC (P=0.26) (Table 4).

IQR: Interquartile range, BPD: bronchopulmonary dysplasia, IVH: intraventricular hemorrhage, DOL: day of life, ROP: retinopathy of prematurity, SIP: spontaneous intestinal perforation, NEC: necrotizing enterocolitis Frequency and incidence of secondary outcomes stratified by caffeine therapy group. Continuous variables were analyzed using Student’s t-tests, or Wilcoxon’s rank-sum tests when appropriate. Categorical variables were analyzed using chi-squared tests.
Discussion
For years, caffeine has been widely used in VLBW infants as a safe [25] and cost effective medication [24] for the prevention and treatment of apnea of prematurity [20]. However, caffeine may have beneficial effects outside of this routine treatment. Since the publication of the CAP trial in 2006 [19], there has been increased interest on whether early initiation of caffeine is beneficial in preterm infants to reduce common morbidities. Several studies have began to look into these associations [5,20,23]. However, more research is needed to conclusively say caffeine should routinely be started prophylactic ally in VLBW infants.
The goal of this study was to determine if there was an association between early versus late caffeine therapy and PDA outcomes in VLBW infants. In our study, infants weighing ≤1500grams at birth who received early caffeine therapy had significantly lower odds of PDA as well as medical management and surgical ligation. As secondary outcomes, infants also had significantly lower odds of BPD and severe (grade 3-4) IVH at 28 days. All these findings remained after controlling for gestational age. Our findings were consistent with those of Lodha et al. [20] who also found reduced rates of BPD and PDA in preterm infants with early initiation of caffeine [20]. Patel et al. [5] & Dobson et al. [23] also found infants receiving early caffeine had reduced rates of BPD and secondarily reduced rates of PDA.
The exact mechanism by which caffeine would reduce the incidence of PDA, BPD and other common morbidities in VLBW infants is unknown. Suggested mechanisms of reduction in PDA incidence have included increased cardiac output and improved cardiac function [26], altered fluid balance through its diuretic effect via tubular adenosine A1 receptors [24,25,27,28]. And effects on signaling pathways involved in ductal constriction [29,30] Fiebich et al. [31] showed in rat microglia cells that caffeine inhibited PGE2 release through inhibition of COX2 therefore altering PDA closure [31]. Caffeine has been shown to improve pulmonary compliance, airway resistance, minute ventilation and respiratory muscle contractility. These benefits likely facilitate earlier weaning from mechanical ventilation and improved pulmonary mechanics [32-35]. The reduced incidence of ROP is also likely due to the reduced need for ventilation and exposure to supplemental oxygen [23]. Reduction in IVH may be due to decreased cerebral blood flow [36,37].
There were limitations in our single-center retrospective study. The primary limitation was the discrepancy in the number of infants that were in the early versus late groups (75% vs 25%). There was also a difference in gestational age (28wga vs 26wga) amongst the early and late caffeine groups. However, we attempted to account for this difference in the statistical analysis. Infants of higher gestational age are less likely to have a PDA requiring treatment. It is not routine in our institution to screen VLBW infants with an echo without clinical evidence of PDA, thus it is possible PDAs were undiagnosed in both groups. We did not include chorioamnionitis which could play a role in the persistence of the PDA. We also did not account for sepsis, which could have significant effects on the closure and re-opening of PDAs. We found a statistically significant difference between the early and late caffeine groups and antenatal steroid use (90% vs 82%). Glucocorticoids have been shown to change the sensitivity of the ductus arteriosus to PGE2 [38]. This may explain the reduced incidence of PDA in VLBW infants whose mothers received antenatal Glucocorticoids treatment.
Although our study was done in a single institution, our investigation benefited from a large cohort of 493 infants. Our unit is unique in we have had the same attending neonatologists throughout the study period with very little variation in management practices of PDA and initiation of caffeine. Additionally, our findings are in line with outcomes found in previous retrospective studies done on early initiation of caffeine. This which further enhances the association of early caffeine on reducing PDA. Further research may provide us with a better idea of the path physiologic function of caffeine and the most appropriate timing for initiation in VLBW infants to optimize all its therapeutic effects.
Conclusion
Ethical approval
Our results demonstrate early initiation of caffeine on or before DOL 3 was associated with decreased odds of PDA and need for medical treatment and/or surgical ligation. Given caffeine has been shown to prevent BPD in preterm infants, initiation of caffeine on or before DOL 3 may have additional benefits on reducing PDA. However, randomized control studies are warranted to conclusively support routine initiation of early caffeine therapy in VLBW infants.
Acknowledgement
We would like to acknowledge Dr. Stephanie Kleithermes, William Adams and Dr. Cara Joyce for their input on the bio statistical methods used in this analysis.
References
- Skinner J (2001) Diagnosis of patent ductus arteriosus. Semin Neonatol 6(1): 49-61.
- Hamrick SEG, Hansmann G (2010) Patent Ductus Arteriosus of the Preterm Infant. Pediatrics 125(5): 1020-1030.
- Hoffman JI, Kaplan S (2002) The incidence of congenital heart disease. J Am Coll Cardiol 39(12): 1890-1900.
- (1993) The Vermont-Oxford Trials Network: very low birth weight outcomes for 1990. Pediatrics 91(3): 540-545.
- Patel RM, Leong T, Carlton DP, Vyas-Read S (2013) Early caffeine therapy and clinical outcomes in extremely preterm infants. J Perinatol 33(2): 134-140
- Stoll BJ, Hansen NI, Bell EF, Shankaran S, Laptook AR, et al. (2010) Neonatal outcomes of extremely preterm infants from the NICHD Neonatal research Network. Pediatrics 126(3): 443-456.
- 7. Brooks JM, Travadi JN, Patole SK, Doherty DA, Simmer K (2005) Is surgical ligation of patent ductus arteriosus necessary? The Western Australian experience of conservative management. Arch Dis Child Fetal Neonatal Ed 90(3): 235-236.
- Sung SI, Chang YS, Chun JY, Yoon SA, Yoo HS, et al (2016) Mandatory Closure Versus Nonintervention for Patent Ductus Arteriosus in Very Preterm Infants. J Pediatr 177(10): 66-71.
- Koch J, Hensley G, Roy L, Brown S, Ramaciotti C, et al (2006) Prevalence of spontaneous closure of the ductus arteriosus in neonates at a birth weight of 1000 grams or less. Pediatrics 117(4): 1113-1121.
- Clyman RI (2000) Ibuprofen and patent ductus arteriosus. N Engl J Med 343 (10): 728-730
- Lelong Tissier MC, Benoit A, Dehan M, Vial M, Ropert JC, et al. (1979) Conservative management of patent ductus arteriosusin preterm infants. Intensive Care Med 5(1): 33-36.
- Kugelman A, Durand M (2011) A comprehensive approach to the prevention of bronchopulmonary dysplasia. Pediatr Pulmonol 46(8): 1153-1165.
- Farstad T, Bratlid D, Medbo S, Markestad T, Norwegian Extreme Prematurity Study Group (2011) Bronchopulmonary dysplasia prevalence, severity and predictive factors in a national cohort of extremely premature infants. Acta Paediatr 100(1): 53-58
- Stephens BE, Gargus RA, Walden RV, Mance M, Nye J, et al. (2008) Fluid regimens in the first week of life may increase risk of patent ductus arteriosus in extremely low birth weight infants. J Perinatol 28(11): 123-128.
- Bell EF, Acarregui MJ (2014) Restricted versus liberal water intake for preventing morbidity and mortality in preterm infants. Cochrane Database Syst Rev 3: CD000503.
- Watterberg KL, Gerdes JS, Cole CH, Aucott SW, Thilo EH, et al. (2004) Prophylaxis of early adrenal insufficiency to prevent bronchopulmonary dysplasia: a multicenter trial. Pediatrics 114(6): 1649-1657.
- Attridge JT, Clark R, Walker MW, Gordon PV (2006) New insights into spontaneous intestinal perforation using a national data set: (1) SIP is associated with early indomethacin exposure. J Perinatol 26(2): 93-99.
- Van Overmeire B, Smets K, Lecoutere D, Van de Broek H, Weyler J, et al. (2000) A comparison of ibuprofen and indomethacin for closure of patent ductus arteriosus. N Engl J Med 343(10): 674-681.
- Schmidt B, Roberts RS, Davis P, Doyle LW, Barrington KJ, et al. (2006) Caffeine Therapy for Apnea of Prematurity. N Engl J Med 354(20): 2112-2121.
- Lodha A, Seshia M, McMillan D, Barrington K, Yang J, et al. (2015) Association of early caffeine administration and neonatal outcomes in very preterm neonates. JAMA Pediatr 169(1): 33-38.
- Alden ER, Mandelkorn T, Woodrum DE, Wennberg RP, Parks CR, et al. (1972) Morbidity and mortality of infants weighing less than 1000 grams in an intensive care nursery. Pediatrics 50(1): 40-49.
- Bhatia J (2000) Current options in the management of apnea of prematurity. Clin Pediatr 39(6): 327-336.
- Dobson NR, Patel RM, Smith PB, Kuehn D, Clark J, et al. (2014) Trends in caffeine use and associate between clinical outcomes and timing of therapy in very low birth weight infants. J Pediatr 164(5): 992-998.
- Nobile S, Carnielli VP (2015) Caffeine for preterm infants: current indications and uncertainties. Acta Biomed 86(1): 32-35
- Spitzer AR (2012) Evidence based methylxanthine use in the NICU. Clin Perinatol 39(1): 137-148
- Soloveychik V, Bin-Nun A, Ionchev A, Sriram S, Meadow W (2009) Acute hemodynamic effects of caffeine administration in premature infants. J Perinatal 29(3): 205-208.
- Gillot I, Gouyon JB, Guignard JP (1990) Renal effects of caffeine in preterm infants. Biol Neonate 58(3): 133-136.
- Bauer J, Maier K, Linderkamp O, Hentschel R (2001) Effect of caffeine on oxygen consumption and metabolic rate in very low birth weight infants with idiopathic apnea. Pediatrics 107(4): 660-663.
- Blaustein MP, Golovina VA, Song H, Choate J, Lencesova L, et al. (2002) Organization of Ca2+ stores in vascular smooth muscle: functional implications. Novartis Found Symp 246: 125-137, discussion 137-41, 221-227.
- Whorton AR, Collawn JB, Montgomery ME, Yound SL, Kent RS (1985) Arachidonic acid metabolism in cultured aortic endothelial cells: effect of cAMP and 3-isobutyl-1-methylxanthine. Biochem Pharmacol 34(1): 119-123.
- Fiebich BL, Lieb K, Hull M, Aicher B, Van Ryn J, et al. (2000) Effects of caffeine and paracetamol alone or in combination with acetylsalicylic acid on prostaglandin E2 synthesis in rat microglial cells. Neuropharmacology 39(11): 2205-2213
- Davis JM, Bhutani VK, Stefano JL, Fox WW, Spitzer AR (1989) Changes in pulmonary mechanics following caffeine administration in infants with BPD. Pediatr Pulmono 6(1): 49-52.
- Yoder B, Thomson M, Coalson J (2005) Lung function in immature baboons with respiratory distress syndrome receiving early caffeine therapy: a pilot study. Acta Paediatr 94(1): 92-98.
- Supinski GS, Deal EC, Kelsen SG (1984) The effects of caffeine and theophylline on diaphragm contractility. Am Rev Respir Dis 130(3): 429-433.
- Aranda JV, Turmen T, Davis J, Trippenbach T, Grondin D, et al. (1983) Effect of caffeine on control of breathing in infantile apnea. J Pediatr 103(6): 975-978.
- Hoecker C, Nelle M, Poeschl J, Beedgen B, Linderkamp O (2002) Caffeine impairs cerebral and intestinal blood flow velocity in preterm infants. Pediatrics 109(5): 784-787
- Tracy MB, Klimek J, Hinder M, Ponnampalam G, Tracy SK (2010) Does caffeine impair cerebral oxygenation and blood flow velocity in preterm infants? Acta Paediatr 99(9): 1319-1323.
- Clyman RI, Mauray F, Roman C, Heymann MA, Ballard PL, et al. (1981) Effects of antenatal glucocorticoid administration on ductus arteriosus of preterm lambs. Am J Physiol 241(3): 415-420.






























